13 September 2020: Database Analysis
Nomogram for Predicting Breast Cancer-Specific Mortality of Elderly Women with Breast Cancer
Xunxi Lu12ABCDEF, Xiaoguang Li1ADEFG, Hong Ling12DFG, Yue Gong12ACD, Linwei Guo12ACE, Min He12DF, Hefen Sun12DG, Xin Hu12EFG*DOI: 10.12659/MSM.925210
Med Sci Monit 2020; 26:e925210
Abstract
BACKGROUND: The objectives of this study were to evaluate the cumulative incidence of breast cancer-specific death (BCSD) and other cause-specific death in elderly patients with breast cancer (BC) and to develop an individualized nomogram for estimating BCSD.
MATERIAL AND METHODS: Data were retrieved from the Surveillance, Epidemiology, and End Results program. A total of 25 241 patients older than 65 years with stage I–III BC diagnosed between 2004 and 2008 was included in the study cohort. We used the cumulative incidence function (CIF) to describe the cause-specific mortality and Gray’s test to compare the differences in CIF among the groups. Fine and Gray’s proportional subdistribution hazard model was applied to validate the independent prognostic factors, upon which the competing-risks nomogram and web-based calculator was built. The performance of the nomogram was assessed with the C-indexes and calibration plot diagrams.
RESULTS: After data screening, 25 241 cases were included for statistical analysis. In the training cohort, the 5-, 8-, and 10-year cumulative incidence of BCSD was 5.7, 8.1, and 9.1%, respectively. Ten independent prognostic factors associated with BCSD were identified. The C-index of the nomogram was 0.818 (0.804–0.831) in the training cohort and 0.808 (0.783–0.833) in the validation cohort. Calibration plot diagrams showed near-ideal consistency between the predicted probabilities and actual observations.
CONCLUSIONS: We built a reliable dynamic nomogram for predicting BCSD in elderly patients, and this individualized predictive tool is favorable for risk classification and complex personalized treatment decision making in clinical practice.
Keywords: Breast Neoplasms, Mortality, nomograms, SEER Program, Aged, 80 and over, Incidence
Background
Breast cancer (BC) is the most common cancer and was the leading cause of cancer death in females worldwide in 2018, accounting for an estimated 24.2% of new cancer diagnoses and 15.0% of cancer deaths [1]. In the United States, there were 317 468 new cases in 2011–2015, of which approximately 43.2% were in women 65 years or older [2]. The probability of developing invasive BC among women ages >70 has increased to 1 in 15, compared with 1 in 52 among women ages <49 in 2018 [3]. With the world population aging [4], the elderly will continue to be a large component of BC patients.
BC in older women, compared with their younger counterparts, has distinctive biological and clinical characteristics. Syed et al. [5] reported that a high expression of estrogen receptor (ER), progesterone receptor (PR), BCL2, and MUC1, along with a low expression of human epidermal growth factor receptor-2 (HER-2), epidermal growth factor receptor, Ki-67, and p53, is more frequently observed in the tumor biology of the elderly. According to the San Antonio Breast Cancer Database and the Surveillance, Epidemiology, and End Results (SEER) program of the National Cancer Institute, the proportion of ER-positive patients in the 65-year-old group is 4–8% more than those in the 55- to 64-year-old group [6], who are good candidates for endocrine therapy.
Increasing age, however, confers high risks for loss of function and numerous chronic health problems, such as ischemic heart disease, diabetes, hypertension, and arthritis [7]. Inevitably, physiological decline signifies a reduced tolerance to treatments. Additionally, corresponding to the lack of prospective studies for the elderly with BC [8–10], the evidence and guidelines for treatment in this population are insufficient. Given all these complexities, clinicians are challenged with proposing reasonable treatment strategies for this population.
Consequently, it is of great significance to develop a model for evaluating disease risk on a personalized level. Nomograms constructed on the basis of prognostic factors are increasingly becoming widely applied to quantify the likelihood of the specific events of interest [11–14]. Likewise, this kind of convenient and practical tool can be used to predict the mortality resulting from BC for elderly patients, potentially facilitating the screening of individuals in need of positive treatment strategies and intensive caring intervention. In this study, a nomogram was built by competing-risks analysis based on data from the SEER database, and after validation, the nomogram manifested an excellent predictive ability.
Material and and Methods
STUDY POPULATION:
Data for this retrospective study were obtained from the SEER program, which consists of 18 population-based cancer registries, for the period 1973–2015 [15]. To ensure integrated data and adequate follow-up time, the clinicopathological information of patients diagnosed between 2004 and 2008 was collected from SEER*Stat Version 8.3.5 (http://www.seer.cancer.gov/seerstat). The inclusion criteria to identify eligible patients were as follows: 1) female; 2) age 65 years or older at diagnosis; 3) BC as the first and only primary malignancy; 4) diagnosis confirmed by positive histology other than by autopsy or death certificate only; 5) unilateral BC; 6) breast-adjusted American Joint Committee on Cancer (AJCC) 6th edition (1998+) stage I–III; 7) surgical treatment with either breast-conserving surgery (BCS) or mastectomy; and 8) active follow-up. Simultaneously, patients diagnosed with inflammatory BC or Paget’s disease were excluded, as were those with missing information on marital status, race, histological grade, tumor (T) and nearby lymph node (N) stage, ER and PR status, and survival data. After a detailed screening, a total of 25 241 patients was eventually enrolled in our study. Data were analyzed according to the following clinicopathological characteristics: age (65–75 or 75+ years), marital status (unmarried or married), race (white, black, or others), histological type (invasive ductal carcinoma [IDC], invasive lobular carcinoma, or others), histological grade (I, II, or III/IV), laterality (left or right), ER and PR status (positive or negative), T stage (T1, T2, T3, or T4), N stage (N0, N1, N2, or N3), and surgery (BCS or mastectomy).
STATISTICAL ANALYSIS:
To establish and validate a competing-risks nomogram, the eligible patients were divided randomly into a training cohort (20 798) for building the model and a validation cohort (4443) for evaluating model performance. Breast cancer-specific death (BCSD) was measured as the time from the date of diagnosis to death attributed to BC, the date of the last follow-up or December 31, 2015 (if date of last contact was after 2015). Other cause-specific death (OCSD) was defined as competing outcomes. Cumulative incidence function (CIF) was applied to describe the probability of BCSD and OCSD grouped by age, marital status, race, histological type, histological grade, laterality, T and N stage, ER and PR status, and surgery; the differences of cause-specific deaths in CIF among the categorical groups were estimated with Gray’s test [16].
Variables with P<0.05 in the univariate analysis were entered into a multivariate competing-risks survival analysis via the proportional subdistribution hazard model by Fine and Gary [17]. Subsequently, we constructed a competing-risks nomogram on the basis of the model we developed to predict the probability of mortality due to BC at 5, 8, and 10 years. To evaluate the model performance, the nomogram was subjected to validation both internally and externally with a 1000-resampling bootstrap method. The concordance index (C-index) value was used to quantify the discrimination performance [18], and calibration curves were plotted to visually assess the calibration, which compared the nomogram-predicted probabilities with the observed marginal cumulative incidence. The C-index ranges from 0.5 to 1.0, indicating random chance to a perfectly precise discrimination. For a well-calibrated model, the dots in the calibration curve should be located close to a 45° diagonal line. Finally, we used the “shiny” and “DNbuilder” packages to generate a web-based BCSD calculator, which can dynamically predict cancer-specific death rates (https://www.shiny apps.io/).
All statistical analyses were performed using SPSS software, version 23 (IBM Corporation, Armonk, NY, USA) and R version 3.5.2 software (Institute for Statistics and Mathematics, Vienna, Austria; http://www.r-project.org/), with the R packages cmprsk [19], rms [20], and mstate [21] for developing the model and nomogram and the package pec [22] for evaluating model performance. All P-values were two sided and P<0.05 was considered statistically significant.
Results
PATIENT’S BASELINE CHARACTERISTICS:
A total of 25 241 eligible patients (20 798 patients in the training cohort and 4443 patients in the validation cohort) with histologically confirmed invasive BC was included in this study. The demographic and baseline characteristics of these patients are shown in Table 1. In the entire population, the majority of patients were married (85.4%) and white (86.7%). Of all the histological types, most cases were of infiltrating ductal carcinoma (72.6%). Of the patients treated with surgery, 64.8% underwent BCS, whereas 35.2% received mastectomy.
UNIVARIATE AND MULTIVARIATE ANALYSIS:
The median follow-up time was 98 (interquartile range 84–117) months. In total, 7480 patients (29.6%) died during the follow-up period, of whom 2271 patients (9.0%) died from BC and 5209 patients (20.6%) died from other causes. For the training cohort, the 5-, 8-, and 10-year cumulative incidences of BCSD stratified by clinicopathological parameters are shown in Table 2, and those of OCSD are displayed in Supplementary Table 1. The 5-, 8-, and 10-year estimates of the crude cumulative incidence of BCSD were 5.7, 8.1, and 9.1%, respectively, and that of OCSD were 9.1, 17.3, and 23.0%, respectively, which were almost twice as much as that of BCSD. The cumulative incidences of BCSD were strongly associated with all the variables (P<0.001), except laterality (P=0.170), and OCSD differed significantly between those with different ages (P<0.001), marital status (P<0.001), race (P<0.001), T stage (P<0.001), and surgery (P<0.001) in the univariate analysis. The differences of BCSD and OCSD discovered by Gray’s test in all categorical variables were presented visually in Figure 1 and Supplementary Figure 1, respectively.
Multivariate analysis further validated the following variables used for building the BCSD model: age, marital status, race, histological type, histological grade, ER and PR status, T and N stage, and surgery. Coefficients and subdistribution hazard ratios (sdHRs) from the multivariable analysis for building the model are presented in Table 3. Both age and marital status were significant independent predictors for BCSD, with a sdHR of 1.43 (1.30–1.58) and 0.73 (0.65–0.82) for patients older than 75 years and who were married, respectively. Patients with grade III/IV or II were more likely to die of BC than those with grade I. Moreover, positive ER and PR predicted a lower incidence of BCSD, with sdHR values of 0.33 (0.20–0.37) and 0.38 (0.35–0.42), respectively. Notably, advanced T and N stages led to a higher likelihood of BCSD. Compared with patients who underwent BCS, patients who underwent mastectomy had an increase in the probability of BCSD, with a sdHR of 2.81 (2.56–3.10).
CONSTRUCTION AND VALIDATION OF THE COMPETING-RISKS NOMOGRAM:
All of the validated factors were incorporated to develop the competing-risks nomogram for predicting the 5-, 8-, and 10-year probability of BCSD by calculating the sum of the point values corresponding to each patient’s characteristics. Figure 2 shows that the N stage was the strongest contributor to BCSD, followed by the T stage and histological grade. The model showed a great discriminative ability in both the internal and external calibrations, with C-indexes of 0.818 (0.804–0.831) and 0.808 (0.783–0.833), respectively. The calibration plots in Figure 3 indicated that the nomogram was well calibrated because the predicted probability of mortality and the actual observations showed near-ideal consistency. According to these results, we built a web-based calculator (https://bcsd.shinyapps.io/DynNomapp/) to predict the BCSD of the elderly patients on the basis of the nomogram. As shown in Figure 4, the dynamic nomogram predicted the mortality of patients according to their clinical characteristics. For example, the 8-year BCSD was approximately 47.0% (95% confidence interval 39.0–58.0%) for patients ages 65–74 years, married, black race, IDC, grade II, with T3, N2 disease, ER negative, PR negative, with mastectomy.
Discussion
With the rapid development of precision medicine, clinicians need to develop individualized treatment and follow-up strategies for patients, which requires more reliable and convenient assessment models. Nomograms integrate both clinical and demographic characteristics into a comprehensive model for predicting the long-term survival of patients. Furthermore, web-based calculators based on the nomogram improve the approachability of the predictive model. In this study, we identified 10 predictors for BCSD by means of a competing-risk analysis that included age, marital status, race, histological type, histological grade, ER and PR status, T and N stage, and surgery, and then established a novel dynamic nomogram. Therefore, clinicians could evaluate a patient’s BCSD much more effectively and then create personalized treatment strategies and follow-up plans. To the best of our knowledge, this is the first nomogram constructed for predicting BCSD in elderly patients.
Competing-risks events pervasively exist in clinical studies for elderly patients, a frail population commonly accompanied by various chronic diseases, and the occurrence of death due to other causes may arise before and preclude that of the event of interest. Considering this, the Kaplan-Meier method and Cox proportional hazards regression model, which are usually used in survival analysis, were not appropriate for the competing-risks framework because the use of the Kaplan-Meier survival function leads to crude estimates of incidence biased upward, regardless of whether the competing events are independent of one another [23,24]. To address this problem, Gray’s test and the proportional subdistribution hazard model are recommended for statistical analysis in the presence of competing risks.
Bastiaannet et al. [25] proved that the percentage of patients above age 75 who die from their BC is less than 50% and the percentage of deaths due to other causes increases with age. Similarly, our results showed that the incidence of OCSD is higher than that of BCSD at any time point for patients over 65 years in all categories, except for those with T3 and T4 stage and N2 and N3 stage. As Supplementary Table 1 showed, the factors associated with OCSD are age, marriage, race, T stage, and surgery. Of interest, there are six other factors including N stage that showed no statistical significance with OCSD in our research. OCSD is literally defined as the death caused by reasons other than breast cancer. It is certain that the clinicopathologic factors of breast cancer, such as N stage, have less influence on OCSD than BCSD. As the follow-up continued, the percentage gain of OCSD was much greater than that of BCSD. As previously mentioned, it is necessary to consider competing-risk events when evaluating a patient’s disease-specific mortality.
In our study, we found that increasing age was a strongly independent predictive factor for an elevated probability of BCSD. This finding was consistent with the findings from a prospective study of postmenopausal women [26], in which the HRs for patients ages 65–74 years and ≥75 years were 1.12 (95% CI, 0.94–1.34) and 1.66 (95% CI, 1.34–2.06), respectively, with patients ages <65 years as the reference. On one hand, age added a layer of biological complexity beyond BC molecular subtypes and classic pathological and clinical variables [27]. On the other hand, compared with their younger counterparts, older patients are prone to nonstandard treatment due to a lower tolerance to surgery, chemotherapy, and radiotherapy [28,29].
Multiple clinical trials have proved that BCS+radiotherapy (RT) has an equal or better effect than mastectomy [30–32]. However, in our study, we found that surgery is an independent prognostic factor, unlike previous studies. For this result, we think there are two reasons: One is that we didn’t include RT data. We dropped the treatment data because of the lack of RT information. This may lead to bias of surgery result. The other is that the choice of treatment options are influenced by the patient’s wishes and their physical condition, especially for elderly patients. For example, some early-stage patients with a poor physical condition may refuse RT considering its side effects. Surgery is an independent factor in our results, but it is influenced by certain factors in reality. Actually, among four main therapies including surgery, chemotherapy, endocrine therapy, and RT, endocrine therapy is offered for treatment strategies more frequently. ER and PR positivity is more common in elderly patients than in younger patients [6,33], indicating that the elderly are more likely to be sensitive to endocrine therapy. Additionally, endocrine therapy offers great benefit to elderly patients considering its equivalent efficacy to chemotherapy with a low risk of toxicities if appropriately used [34]. Although the conventional treatment for breast cancer is surgical resection, several prospective clinical trials have indicated much better outcomes for older patients with small, HR-positive tumors who receive tamoxifen alone than surgery [35]. The information above is consistent with our finding that ER and PR were protectors against BCSD. However, because of the limited access to the SEER database, information on endocrine treatment can’t be acquired. This hinders us from further exploration.
Before applying treatment to patients, it’s necessary to build an effective and convenient tool to identify high-risk patients who need intensive treatment and clinical care. Though the TNM staging system for BC is an important prognostic predictor, the fact that certain significant prognostic factors such as age are not included cannot be ignored and may result in bias to some extent when evaluating a patient’s survival. This nomogram incorporates not only parameters from the international AJCC but also some individual demographic and pathological characteristics and presents a prognostic numerical value. Thus, it provides enhanced comprehensiveness and convenience. Nevertheless, unavailable data on some characteristics regarding geriatric assessment is not included in the models, such as comorbidities, physical function status, mental health, and social support, which may make a difference in prognosis and clinical decision making [9,36]. This important part in optimal treatment tailoring must be considered when applying our nomogram.
There were certain limitations in our study that should be mentioned. First, adjuvant therapies such as chemotherapy and RT are not included in this study because of their incomplete and ambiguous data; for example, the categories “no treatment” and “unknown if patients received treatment” can’t be distinguished for RT, and because of the large possibility of undertreatment in adjuvant therapies for elderly patients, bias may be involved in the statistical analysis if they were selected as candidate factors in the model, so we excluded this information even if it made a difference in prognosis. Unfortunately, several significant characteristics and follow-up information associated with prognosis were also not provided by the SEER database, including HER-2 status, Ki-67 positivity, tumor progression, and subsequent metastasis, which affects the effectiveness of our model certainly. Second, nearly half of the patients were excluded because of a lack of specific data. As a result, selection bias could affect our findings because of this underlying preference. Third, this was a single data-set study. To enhance the persuasiveness of the model, investigation from other centers or databases is needed for model validation. Finally, subject to retrospective methodology, the nomogram is supposed to be confirmed and supplemented via further prospective studies before clinical application.
Conclusions
In conclusion, we estimated the cumulative incidence of BCSD and OCSD in elderly patients diagnosed with BC on the basis of a large population-based cohort. A web-based dynamic nomogram predicting the 5-, 8-, and 10-year incidence of BCSD was built on the basis of 10 independent prognostic factors identified by a competing-risks analysis. The model performed excellently, and we hope that it can help clinicians evaluate patient risk of BCSD more effectively and propose individualized treatment strategies.
Figures
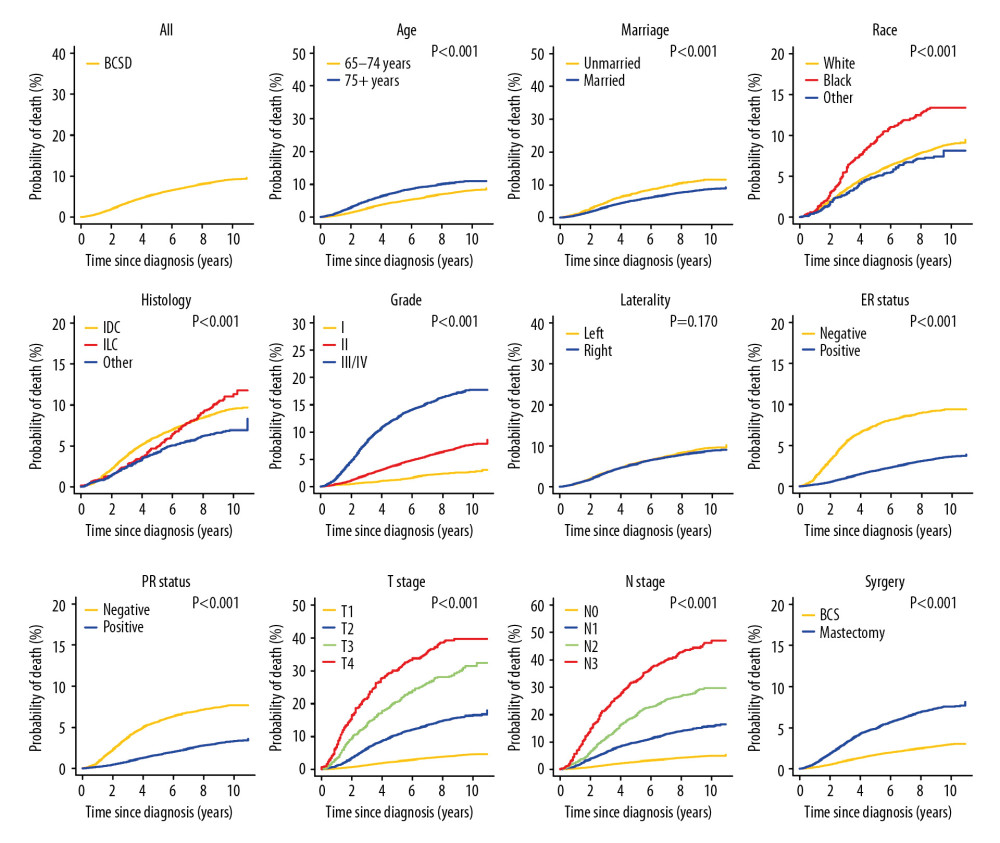 Figure 1. Cumulative incidence functions curves of breast cancer-specific death (BCSD) by patients’ characteristic.
Figure 1. Cumulative incidence functions curves of breast cancer-specific death (BCSD) by patients’ characteristic. 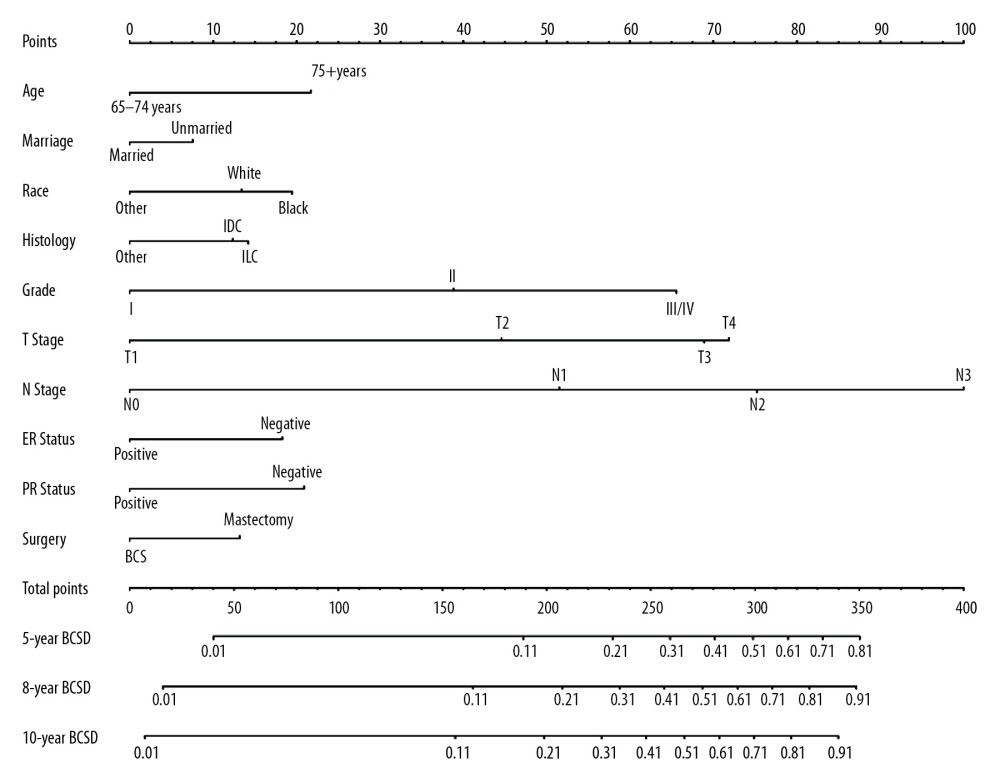 Figure 2. Competing-risks nomogram for predicting 5-, 8- and 10-year probabilities of breast cancer-specific death (BCSD) in elderly patients with breast cancer.
Figure 2. Competing-risks nomogram for predicting 5-, 8- and 10-year probabilities of breast cancer-specific death (BCSD) in elderly patients with breast cancer. 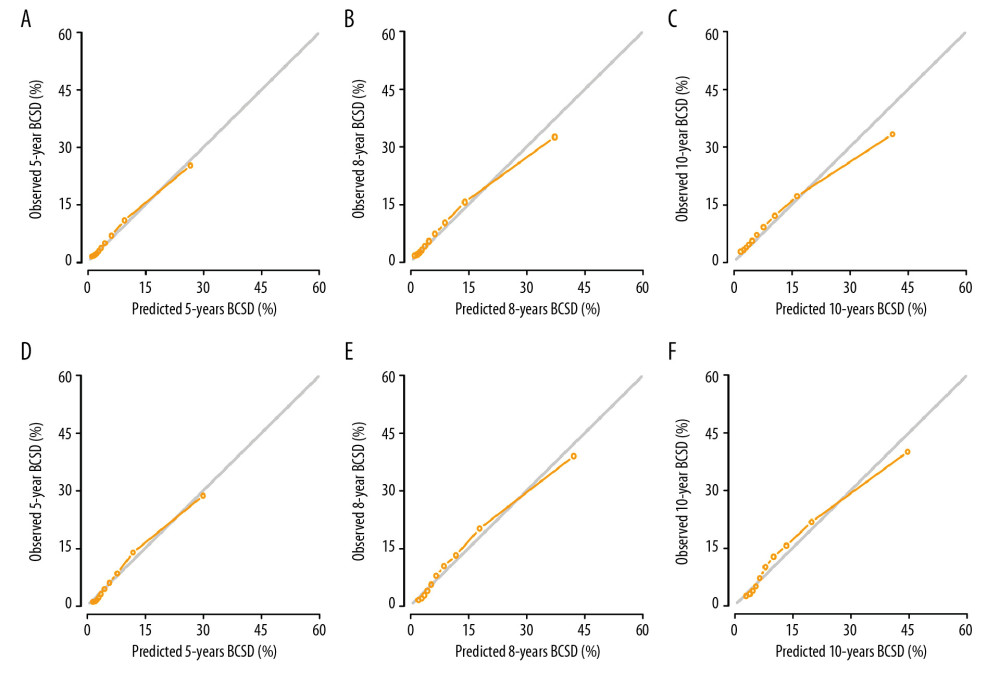 Figure 3. Internal calibration curves for (A) 5-, (B) 8-, and (C) 10-year breast cancer-specific death (BCSD) and external calibration curves for (D) 5-, (E) 8-, and (F) 10-year BCSD.
Figure 3. Internal calibration curves for (A) 5-, (B) 8-, and (C) 10-year breast cancer-specific death (BCSD) and external calibration curves for (D) 5-, (E) 8-, and (F) 10-year BCSD. 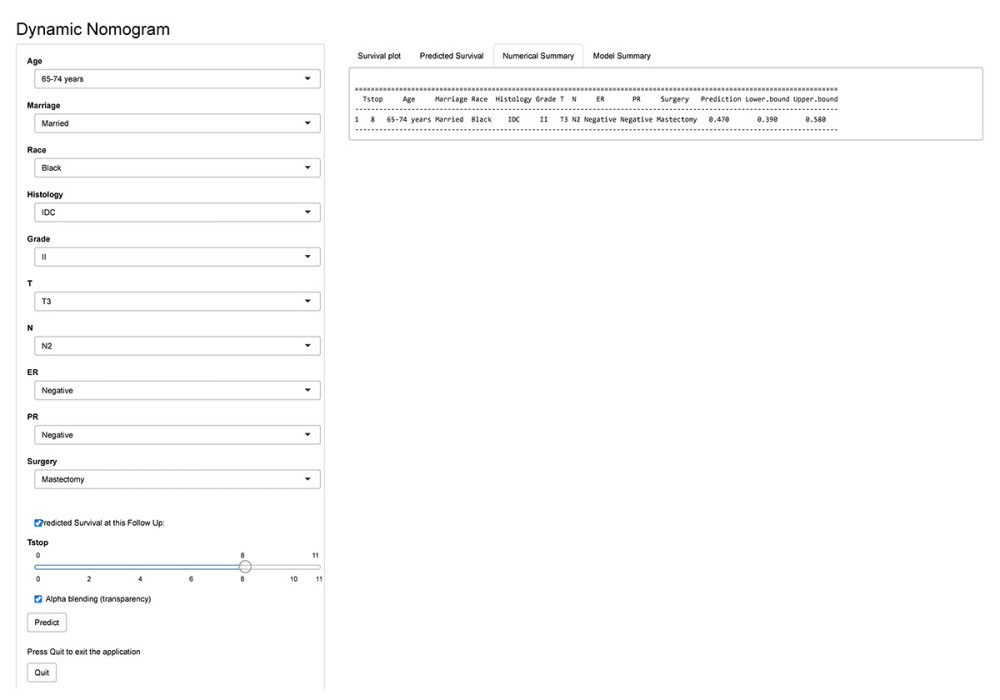 Figure 4. A patient age 65–74 years, married, black race, invasive ductal carcinoma (IDC, grade II, with tumor (T)3, nearby lymph node (N)2 disease, estrogen receptor (ER)-negative, progesterone (PR)-negative with mastectomy was evaluated using the web-based calculator.
Figure 4. A patient age 65–74 years, married, black race, invasive ductal carcinoma (IDC, grade II, with tumor (T)3, nearby lymph node (N)2 disease, estrogen receptor (ER)-negative, progesterone (PR)-negative with mastectomy was evaluated using the web-based calculator. Tables
Table 1. Patients’ demographics and baseline characteristics.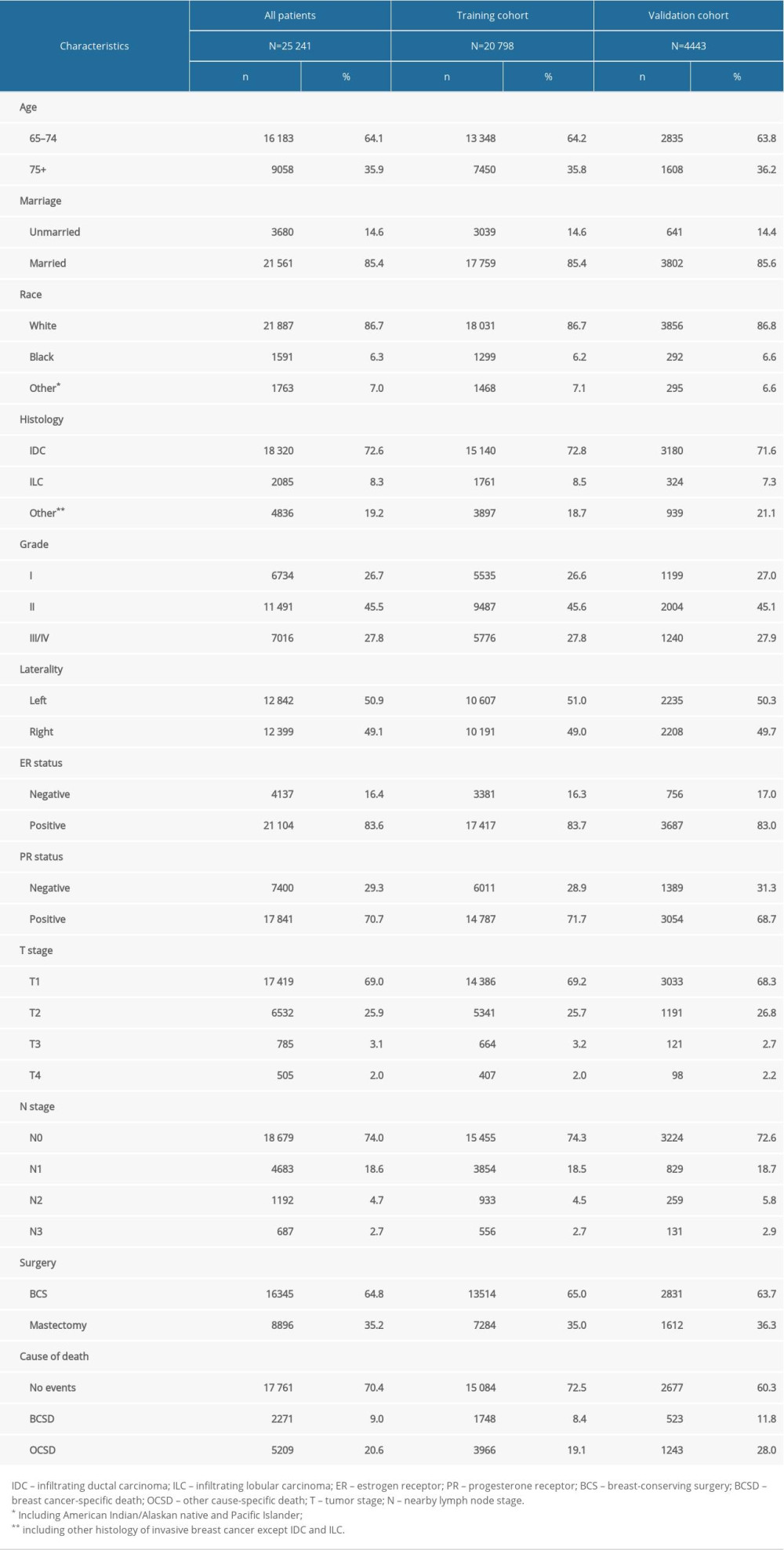 Table 2. 5-, 8-, 10-Year cumulative incidences of BCSD among patients with breast cancer in the training cohort.
Table 2. 5-, 8-, 10-Year cumulative incidences of BCSD among patients with breast cancer in the training cohort.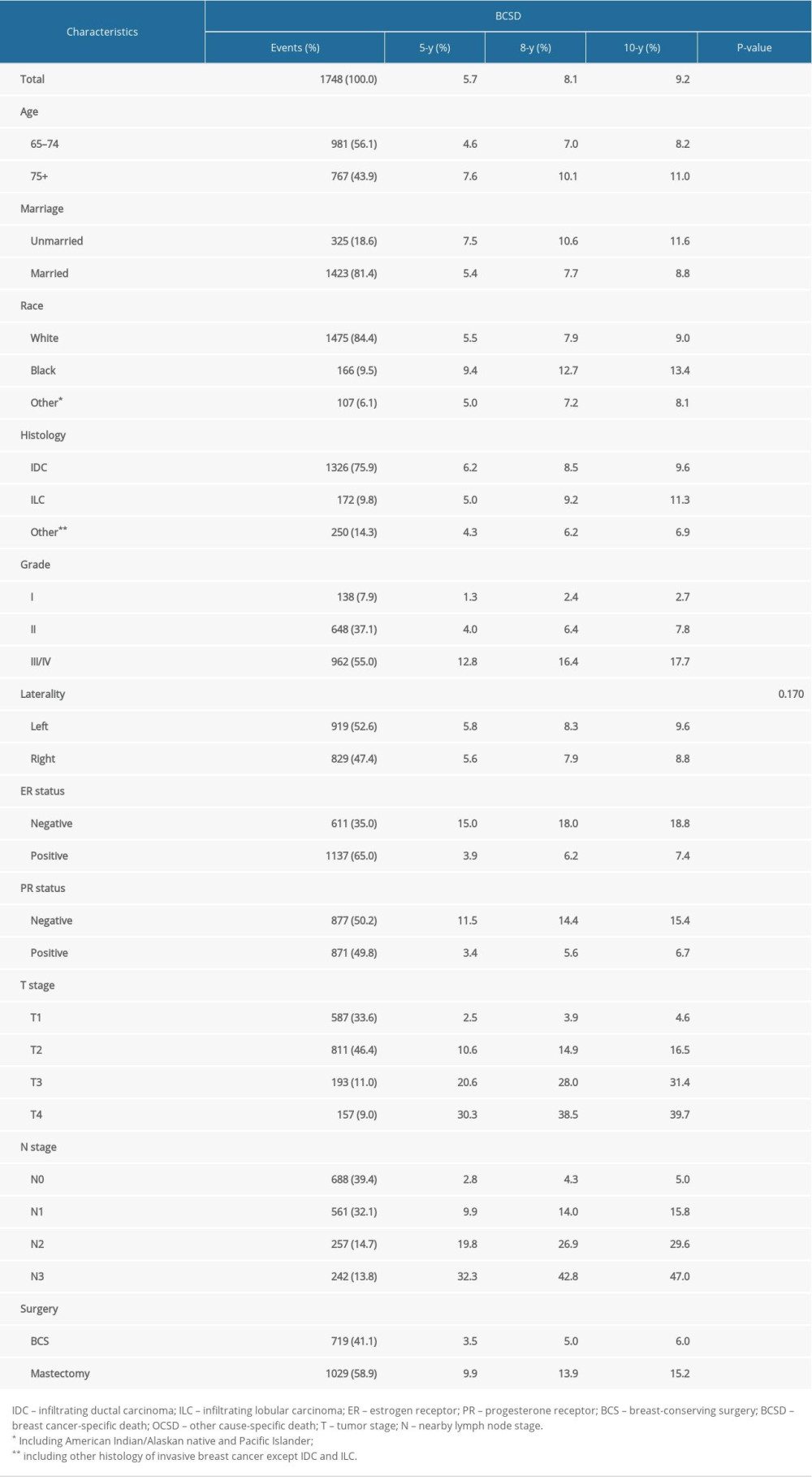 Table 3. Proportional subdistribution hazard model of probabilities of BCSD for patients in the training cohort.
Table 3. Proportional subdistribution hazard model of probabilities of BCSD for patients in the training cohort.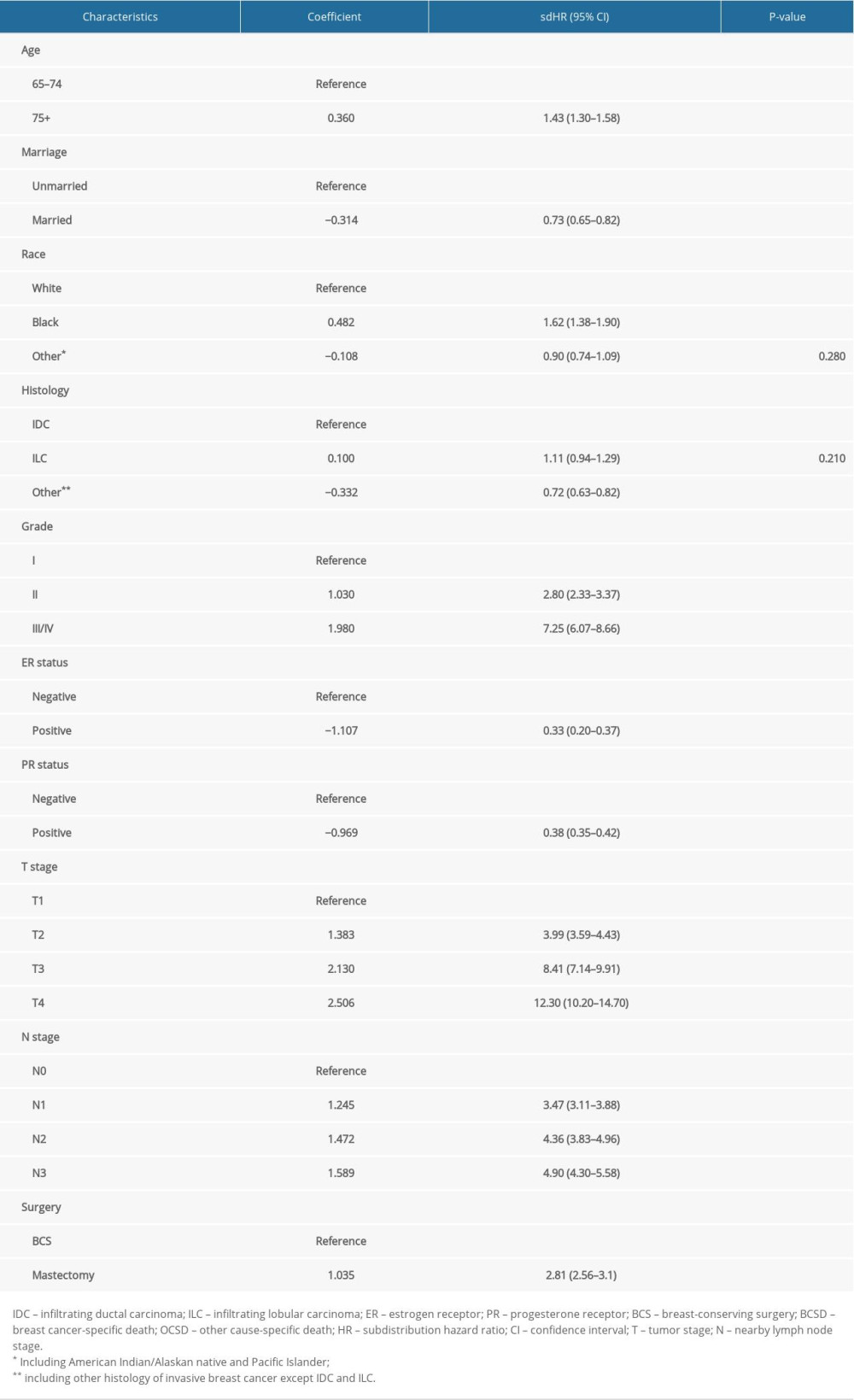 Supplementary Table 1. 5-, 8-, 10-Year cumulative incidences of OCSD among patients with breast cancer in the training cohort.
Supplementary Table 1. 5-, 8-, 10-Year cumulative incidences of OCSD among patients with breast cancer in the training cohort.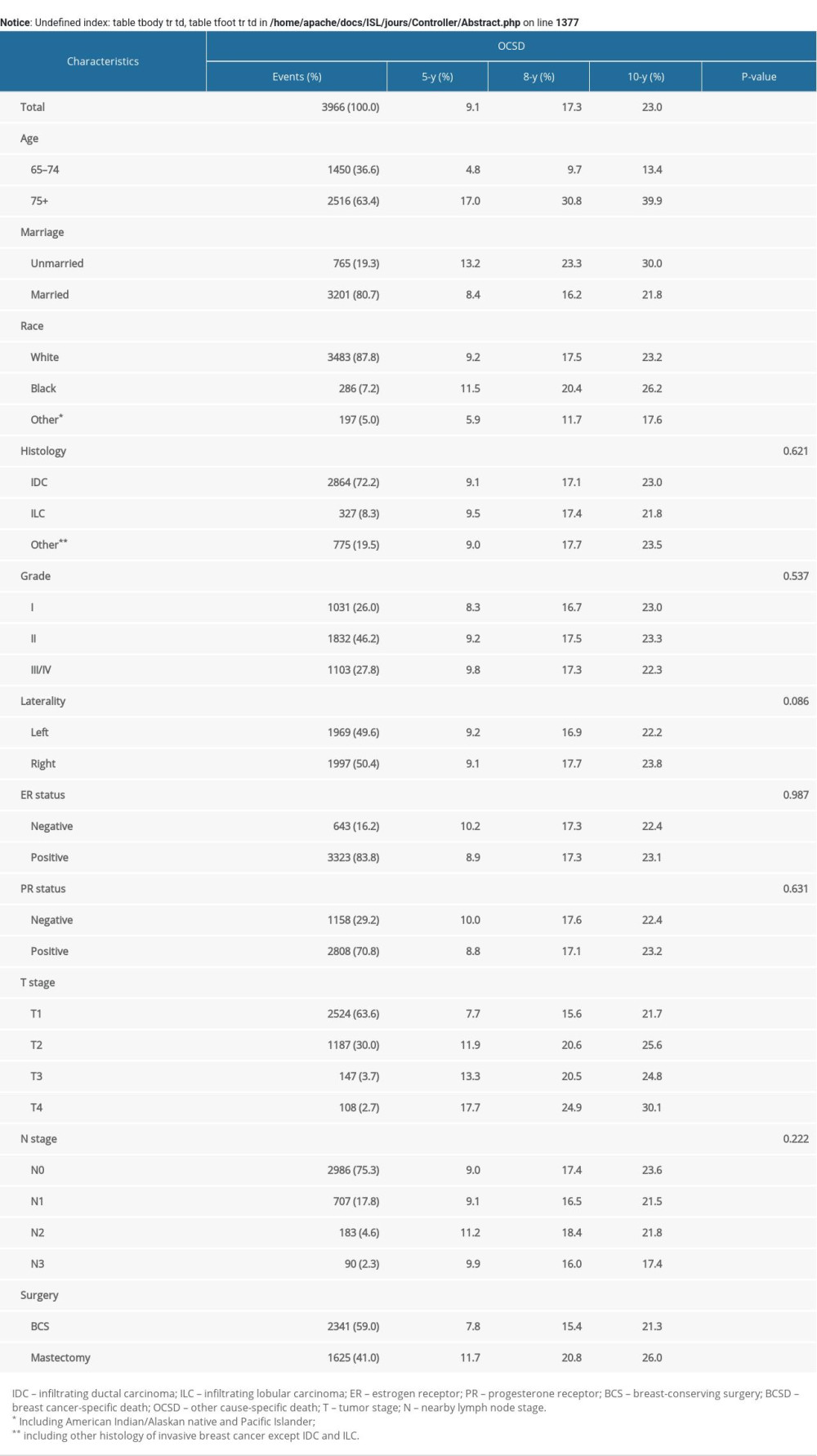
References
1. Bray F, Ferlay J, Soerjomataram I, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2018; 68(6); 394-424
2. Noone AM, Krapcho M, Miller D: SEER Cancer Statistics Review (CSR) 1975–2015, 2018
3. Siegel RL, Miller KD, Jemal A, Cancer statistics, 2018: Cancer J Clin, 2018; 68(1); 7-30
4. Suzman R, Beard JR, Boerma T, Chatterji S, Health in an ageing world – What do we know?: Lancet, 2015; 385(9967); 484-86
5. Syed BM, Green AR, Paish EC, Biology of primary breast cancer in older women treated by surgery: With correlation with long-term clinical outcome and comparison with their younger counterparts: Br J Cancer, 2013; 108(5); 1042-51
6. Diab SG, Elledge RM, Clark GM, Tumor characteristics and clinical outcome of elderly women with breast cancer: J Natl Cancer Inst, 2000; 92(7); 550-56
7. Williams GR, Mackenzie A, Magnuson A, Comorbidity in older adults with cancer: J Geriatr Oncol, 2016; 7(4); 249-57
8. Murthy VH, Krumholz HM, Gross CP, Participation in cancer clinical trials: Race-, sex-, and age-based disparities: J Am Med Assoc, 2004; 291(22); 2720-26
9. Pallis AG, Fortpied C, Wedding U, EORTC elderly task force position paper: Approach to the older cancer patient: Eur J Cancer, 2010; 46(9); 1502-13
10. Zulman DM, Sussman JB, Chen X, Examining the evidence: A systematic review of the inclusion and analysis of older adults in randomized controlled trials: J Gen Int Med, 2011; 26(7); 783-90
11. Albert JM, Liu DD, Shen Y, Nomogram to predict the benefit of radiation for older patients with breast cancer treated with conservative surgery: J Clin Oncol, 2012; 30(23); 2837-43
12. Brockman JA, Alanee S, Vickers AJ, Nomogram predicting prostate cancer-specific mortality for men with biochemical recurrence after radical prostatectomy: Eur Urol, 2015; 67(6); 1160-67
13. Fakhry C, Zhang Q, Nguyen-Tân PF, Development and validation of nomograms predictive of overall and progression-free survival in patients with oropharyngeal cancer: J Clin Oncol, 2017; 35(36); 4057-65
14. Liang W, Zhang L, Jiang G, Development and validation of a nomogram for predicting survival in patients with resected non-small-cell lung cancer: J Clin Oncol, 2015; 33(8); 861-69
15. Fisher B, Anderson S, Bryant J, Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer: N Engl J Med, 2002; 347(16); 1233-41
16. Gray RJ, A class of $K$-sample tests for comparing the cumulative incidence of a competing risk: Ann Stat, 1988; 16(3); 1141-54
17. Fine JP, Gray RJ, A proportional hazards model for the subdistribution of a competing risk: J Am Stat Assoc, 1999; 94(446); 496-509
18. Wolbers M, Koller MT, Witteman JC, Steyerberg EW, Prognostic models with competing risks: Methods and application to coronary risk prediction: Epidemiology, 2009; 20(4); 555-61
19. : cmprsk:Subdistribution analysis of competing risks. R package version 2.2-7, 2014 [computer program]. https://CRAN.R-project.org/package=cmprsk
20. : rms:Regression modeling strategies. R package version 5.1-2, 2018 [computer program]. https://CRAN.R-project.org/package=rms
21. : mstate:Data preparation, estimation and prediction in multi-state models. R package version 0.2.11, 2018 [computer program]. https://CRAN.R-project.org/package=mstate
22. : pec:Prediction error curves for risk prediction models in survival analysis, 2018 [computer program]. https://CRAN.R-project.org/package=pec
23. Austin PC, Lee DS, Fine JP, Introduction to the analysis of survival data in the presence of competing risks: Circulation, 2016; 133(6); 601-9
24. Satagopan JM, Ben-Porat L, Berwick M, A note on competing risks in survival data analysis: Br J Cancer, 2004; 91(7); 1229-35
25. Bastiaannet E, Liefers GJ, de Craen AJ, Breast cancer in elderly compared to younger patients in the Netherlands: Stage at diagnosis, treatment and survival in 127,805 unselected patients: Breast Cancer Res Tr, 2010; 124(3); 801-7
26. van de Water W, Markopoulos C, van de Velde CJ, Association between age at diagnosis and disease-specific mortality among postmenopausal women with hormone receptor-positive breast cancer: J Am Med Assoc, 2012; 307(6); 590-97
27. Azim HA, Nguyen B, Brohée S, Genomic aberrations in young and elderly breast cancer patients: BMC Med, 2015; 13; 266
28. Owusu C, Lash TL, Silliman RA, Effect of undertreatment on the disparity in age-related breast cancer-specific survival among older women: Breast Cancer Res Tr, 2007; 102(2); 227-36
29. Tahir M, Robinson T, Stotter A, How not to neglect the care of elderly breast cancer patients?: Breast, 2011; 20(4); 293-96
30. Fisher B, Anderson S, Bryant J, Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer: N Engl J Med, 2002; 347(16); 1233-41
31. Veronesi U, Cascinelli N, Mariani L, Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer: N Engl J Med, 2002; 347(16); 1227-32
32. Arriagada R, Lê MG, Rochard F, Contesso G, Conservative treatment versus mastectomy in early breast cancer: Patterns of failure with 15 years of follow-up data. Institut Gustave-Roussy Breast Cancer Group: J Clin Oncol, 1996; 14(5); 1558-64
33. Anderson WF, Katki HA, Rosenberg PS, Incidence of breast cancer in the United States: Current and future trends: J Natl Cancer Inst, 2011; 103(18); 1397-402
34. Lee SY, Seo JH, Current strategies of endocrine therapy in elderly patients with breast cancer: BioMed Res Int, 2018; 2018 6074808
35. Hind D, Wyld L, Reed MW: Br J Cancer, 2007; 96(7); 1025-29
36. Caillet P, Laurent M, Bastuji-Garin S, Optimal management of elderly cancer patients: Usefulness of the comprehensive geriatric assessment: Clin Interv Aging, 2014; 9; 1645-60
Figures
 Figure 1. Cumulative incidence functions curves of breast cancer-specific death (BCSD) by patients’ characteristic.
Figure 1. Cumulative incidence functions curves of breast cancer-specific death (BCSD) by patients’ characteristic. Figure 2. Competing-risks nomogram for predicting 5-, 8- and 10-year probabilities of breast cancer-specific death (BCSD) in elderly patients with breast cancer.
Figure 2. Competing-risks nomogram for predicting 5-, 8- and 10-year probabilities of breast cancer-specific death (BCSD) in elderly patients with breast cancer. Figure 3. Internal calibration curves for (A) 5-, (B) 8-, and (C) 10-year breast cancer-specific death (BCSD) and external calibration curves for (D) 5-, (E) 8-, and (F) 10-year BCSD.
Figure 3. Internal calibration curves for (A) 5-, (B) 8-, and (C) 10-year breast cancer-specific death (BCSD) and external calibration curves for (D) 5-, (E) 8-, and (F) 10-year BCSD. Figure 4. A patient age 65–74 years, married, black race, invasive ductal carcinoma (IDC, grade II, with tumor (T)3, nearby lymph node (N)2 disease, estrogen receptor (ER)-negative, progesterone (PR)-negative with mastectomy was evaluated using the web-based calculator.
Figure 4. A patient age 65–74 years, married, black race, invasive ductal carcinoma (IDC, grade II, with tumor (T)3, nearby lymph node (N)2 disease, estrogen receptor (ER)-negative, progesterone (PR)-negative with mastectomy was evaluated using the web-based calculator. Tables
 Table 1. Patients’ demographics and baseline characteristics.
Table 1. Patients’ demographics and baseline characteristics. Table 2. 5-, 8-, 10-Year cumulative incidences of BCSD among patients with breast cancer in the training cohort.
Table 2. 5-, 8-, 10-Year cumulative incidences of BCSD among patients with breast cancer in the training cohort. Table 3. Proportional subdistribution hazard model of probabilities of BCSD for patients in the training cohort.
Table 3. Proportional subdistribution hazard model of probabilities of BCSD for patients in the training cohort. Table 1. Patients’ demographics and baseline characteristics.
Table 1. Patients’ demographics and baseline characteristics. Table 2. 5-, 8-, 10-Year cumulative incidences of BCSD among patients with breast cancer in the training cohort.
Table 2. 5-, 8-, 10-Year cumulative incidences of BCSD among patients with breast cancer in the training cohort. Table 3. Proportional subdistribution hazard model of probabilities of BCSD for patients in the training cohort.
Table 3. Proportional subdistribution hazard model of probabilities of BCSD for patients in the training cohort. Supplementary Table 1. 5-, 8-, 10-Year cumulative incidences of OCSD among patients with breast cancer in the training cohort.
Supplementary Table 1. 5-, 8-, 10-Year cumulative incidences of OCSD among patients with breast cancer in the training cohort. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








