11 February 2021: Clinical Research
Risk Factors of Coronavirus Disease 2019-Related Mortality and Optimal Treatment Regimens: A Retrospective Study
Yuehong Wang1BC, Shuang Yao2CDEF, Xiaoling Liu3BDEF, Yinghao Cao4ABCEF, Yaling Wang5BF*, Mao Xie6AEFDOI: 10.12659/MSM.926751
Med Sci Monit 2021; 27:e926751
Abstract
BACKGROUND: Coronavirus disease 2019 (COVID-19) is spreading rapidly worldwide, and scientists are trying to find a way to overcome the disease. We explored the risk factors that influence patient outcomes, including treatment regimens, which can provide a reference for further treatment.
MATERIAL AND METHODS: A retrospective cohort study analysis was performed using data from 97 patients with COVID-19 who visited Wuhan Union Hospital from February 2020 to March 2020. We collected data on demographics, comorbidities, clinical manifestations, laboratory tests, treatment methods, outcomes, and complications. Patients were divided into a recovered group and a deceased group. We compared the differences between the 2 groups and analyzed risk factors influencing the treatment effect.
RESULTS: Seventy-six patients recovered and 21 died. The average age and body mass index (BMI) of the deceased group were significantly higher than those of the recovered group (69.81±6.80 years vs 60.79±11.28 years, P<0.001 and 24.95±3.14 kg/m² vs 23.09±2.97 kg/m², P=0.014, respectively). The combination of antiviral drugs and supportive therapy appears to be associated with the lowest mortality (P<0.05). Multivariate Cox regression analysis revealed that age, BMI, H-CRP, shock, and acute respiratory distress syndrome (ARDS) were independent risk factors for patients with COVID-19 (P<0.05).
CONCLUSIONS: Elderly patients and those with a high BMI, as well as patients who experience shock and ARDS, may have a higher risk of death from COVID-19. The combination of antiviral drugs and supportive therapy appears to be associated with lower mortality, although further research is needed.
Keywords: Aftercare, COVID-19, Decision Support Techniques, SARS Virus, Age Factors, Antiviral Agents, COVID-19, Drug Therapy, Combination, Drugs, Chinese Herbal, Hospital Mortality, Risk Factors, SARS-CoV-2, Shock, gamma-Globulins
Background
Coronavirus disease 19 (COVID-19) is caused by the novel coronavirus severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and has evolved into a global pandemic since its original outbreak in Wuhan, China in 2019. COVID-19 can be transmitted from person to person with high infectivity and has high mortality and a low cure rate, bringing a heavy burden and great challenge to the global public health system. COVID-19 can cause a range of respiratory symptoms, which vary in severity. In some patients, their condition can deteriorate rapidly in a short period of time. It is now believed that inflammation is the main cause of this deterioration, and viral replication affects the inflammatory process [1].
Many factors may affect the prognosis of patients with COVID-19, including clinical characteristics, degree of severity, laboratory test results, and treatment options. Among these, the treatment plan plays a particularly important role in the patient’s prognosis. In the face of aggressive COVID-19, countries are striving to explore and optimize treatment options, and a variety of drugs are undergoing clinical trials. A variety of vaccines have entered phase III clinical trials, and the safety and efficacy of the vaccines are being evaluated. Although many are pinning their hopes on a vaccine to fight COVID-19, antiviral treatment is still essential. SARS-CoV-2 invades the host cell using angiotensin-converting enzyme 2 as a receptor; therefore, antiviral drugs are favored. In addition, based on the treatment for SARS, convalescent plasma is used. The technique uses antibodies from the blood plasma or serum of people who have recovered from COVID-19 infection to boost the immunity of newly-infected patients and those at risk of contracting the disease. These antibodies contained in the blood’s serum have the ability to bind to and neutralize SARS-CoV-2, the virus that causes COVID-19. Gamma globulin, traditional Chinese medicine, immunomodulatory drugs, and glucocorticoids have also been widely used for the treatment of COVID-19. Although some drugs have shown efficacy, more randomized trials are still needed [2]. Previous studies have found that early intervention in patients with new COVID-19 pneumonia significantly improves the prognosis of patients.
In this study, we aimed to identify factors that affect the mortality of patients with COVID-19 by analyzing the clinical characteristics, laboratory test results, and treatment regimens of patients who either recovered from or died of COVID-19, so as to effectively intervene early and improve the cure rate of patients.
Material and Methods
PATIENTS:
A retrospective cohort study analysis was performed on 116 patients with COVID-19 and a positive SARS-CoV-2 test who were admitted to Wuhan Union Hospital from February 2020 to March 2020. All the patients met the diagnostic and typing criteria in the “Diagnosis and Treatment Plan for Novel Coronavirus Pneumonia (trial version 8)” issued by the National Health Commission [3]. Nineteen patients were excluded for the following reasons: (1) age less than 18 years or more than 85 years; (2) pregnant; (3) lack of complete data. The Ethics Committee of the Union Hospital of Tongji Medical College of Huazhong University of Science and Technology approved the study, and it was conducted according to the Helsinki Declaration. Each patient gave informed consent and signed a written informed consent form.
OBSERVATION INDEX AND GROUPING:
Data on patient demographics, comorbidities, clinical manifestations, laboratory tests, treatment methods, outcomes, and complications were collected. According to the outcome after treatment, the patients were divided into the recovered group or the deceased group.
DEFINITIONS:
The standard for recovery was disappearance of clinical symptoms and 2 negative nucleic acid tests [4]. The endpoint of this study was overall survival, which was defined as the length of time from the date of diagnosis to the date of death or discharge from the hospital. Acute respiratory distress syndrome (ARDS) and shock were defined in accordance with the WHO interim guidance on the novel coronavirus [5]. Acute kidney injury was determined according to the highest serum creatinine and urea level criteria [6]. Secondary bacterial pneumonia was defined in accordance with the clinical symptoms or signs of nosocomial pneumonia or bacteremia, using lung imaging studies and bacterial culture results of respiratory secretions. Acute cardiac injury was diagnosed if the serum levels of cardiac biomarkers were higher than the 99th percentile reference limit or if ECG and echocardiography showed new abnormalities [7].
DATA ANALYSIS:
Continuous variables that conformed to a normal distribution were expressed as mean±standard deviation; otherwise, variables were expressed as the median and interquartile range. We used the
Results
COMPARISON OF DEMOGRAPHIC INFORMATION BETWEEN THE 2 GROUPS:
There were 76 patients in the recovery group and 21 patients in the deceased group, and the detailed flow diagram of the patient selection process is shown in Figure 1. Both groups had a history of living in Wuhan’s epidemic area before the onset of disease, and there was no statistically significant difference in contact with COVID-19-positive patients. The average age and BMI of the patients in the deceased group were significantly higher than those in the recovered group (69.81±6.80 years vs 60.79±11.28 years, P<0.001 and 24.95±3.14 kg/m2 vs 23.09±2.97 kg/m2, P=0.014, respectively). No significant differences were found in sex, smoking status, alcohol consumption, or comorbidities (hypertension, cardiovascular disease, lung disease, chronic hepatorenal diseases, and tumor history) between the 2 groups (P>0.05 for all) (Table 1).
COMPARISON OF CLINICAL SYMPTOMS AND LABORATORY TEST RESULTS BETWEEN THE 2 GROUPS:
The proportion of patients with fever in the recovered group was lower than that in the deceased group (61.84% vs 66.67%), but the difference was not statistically significant (P=0.801). No significant differences were found in the incidence or disease severity of other common clinical symptoms between the 2 groups, including dry cough, expectoration, nasal congestion, headache, weakness, myalgia, chest tightness, dyspnea, grasp, and gastrointestinal symptoms (P>0.05 for all) (Table 2). The disease severity in this study was defined as mild, normal, severe, or critical, according to “Diagnosis and Treatment Plan for Novel Coronavirus Pneumonia (trial version 8)” [3].
Compared with patients in the recovered group, patients in the deceased group were more likely to have different degrees of increase in white blood cell counts (9.70 [7.81–13.85]×109/L vs 5.98 [4.28–9.06]×109/L), neutrophil counts (8.92 [7.08–12.73]×109/L vs 4.79 [3.28–7.965]×109/L), prothrombin time (15.6 [13–17.4] s vs 13.15 [12.4–14.55] s), activated partial thromboplastin time (41.5 [37.7–43.9] s vs 34.05 [32.25–38.45] s), glutamic-oxaloacetic transaminase (42 [25–66] u/L vs 27 [20–43.5] u/L), direct bilirubin (5.16 [3.04–8.15] umol/L vs 2.69 [1.88–4.13] umol/L), creatinine (83 [64–95] umol/L vs 65.5 [54.5–75.5] umol/L), blood urea nitrogen (7.37 [6.12–9.69] mmol/L vs 5.88 [4.61–7.45] mmol/L), and hypersensitive C–reactive protein (88.88 [59.63–99.67] mg/L vs 35.6 [21.53–82.39] mg/L) (P<0.05 for all). There was no significant difference in other laboratory indexes between the 2 groups (P>0.05 for all) (Table 3).
COMPARISON OF TREATMENT BETWEEN THE 2 GROUPS:
The rate of combination antiviral therapy was lower in the deceased group than in the recovered group (47.62% vs 81.58%, P=0.004). Additionally, the rate of other supportive treatment in the recovered group was significantly higher than that in the deceased group (63.16% vs 38.1%, P=0.048). However, the rates of anti-infection therapy and gamma globulin therapy were significantly higher in the deceased group (90.48% vs 28.95, P<0.001 and 90.48% vs 39.72, P<0.001, respectively). There was no statistically significant difference in the administration of hormone therapy or single-agent antiviral therapy between the 2 groups (P>0.05) (Table 4).
Figure 1 depicts the Kaplan-Meier survival curves of the patients. The survival rates of patients who received more than 1 antiviral therapy and other supportive therapy were higher than those of patients who received single-agent antiviral therapy and no other supportive therapy (P<0.05) (Figure 2A, 2D). By contrast, the survival rates of patients who received gamma globulin therapy and more than 1 anti-infection therapy were lower than those of patients who received no gamma globulin therapy and only a single anti-infection therapy (P<0.05) (Figure 2B, 2C).
COMPARISON OF COMPLICATIONS BETWEEN THE 2 GROUPS:
The rate of extremely severe cases in the deceased group was higher than that in the recovered group (76.19% vs 13.16%, P<0.001). Compared with patients in the recovered group, patients in the deceased group were more likely to develop shock (0% vs 19.05%, P=0.002), ARDS (19.74% vs 90.48%, P<0.001), kidney injury (3.95% vs 90.48, P<0.001), acute heart injury (3.95% vs 76.19%, P<0.001), and secondary bacterial pneumonia (17.11% vs 66.67%, P<0.001) (Table 5).
RISK FACTORS AFFECTING THE OUTCOME OF TREATMENT:
The univariate Cox analysis showed that age (HR: 1.092, 95% CI: 1.036–1.150, P=0.001), BMI (HR: 1.336, 95% CI: 1.112–1.607, P=0.002), neutrophil count (HR: 1.141, 95% CI: 1.068–1.219, P<0.001), prothrombin time (HR: 1.213, 95% CI: 1.045–1.407, P=0.011), total bilirubin (HR: 1.044, 95% CI: 1.015–1.073, P=0.002), direct bilirubin (HR: 1.125, 95% CI: 1.050–1.206, P=0.001), blood urea nitrogen (HR: 1.074, 95% CI: 1.025–1.125, P=0.003), hypersensitive C-reactive protein (HR: 1.008, 95% CI: 1.002–1.013, P=0.004); extremely severe disease (HR: 13.395, 95% CI: 1.773–101.175, P=0.012), shock (HR: 11.726, 95% CI: 3.373–40.760, P=0.001), ARDS (HR: 11.22, 95% CI: 2.573–48.915, P=0.013), kidney injury (HR: 5.233, 95% CI: 1.823–15.016, P=0.002), and acute heart injury (HR: 4.533, 95% CI: 1.600–12.840, P=0.004) were the risk factors affecting the clinical outcome. Multivariate Cox regression analysis showed that age (HR: 1.319, 95% CI: 1.115–1.561, P=0.001), BMI (HR: 1.344, 95% CI: 1.014–1.783, P=0.008), prothrombin time (HR: 0.489, 95% CI: 0.271–0.884, P=0.018), H-CRP (HR: 1.014, 95% CI: 1.003–1.025, P<0.001), shock (HR: 34.713, 95% CI: 2.596–464.133, P=0.007), and ARDS (HR: 46.252, 95% CI: 1.504–1422.171, P=0.028) were independent risk factors affecting the treatment effect of patients with COVID-19 (Table 6).
Discussion
In this study, we analyzed the risk factors of mortality in patients with COVID-19. We found no significant difference in clinical symptoms such as cough, fever, fatigue, chest tightness, diarrhea, and dyspnea between patients who died and those who recovered. However, a variety of factors affected the mortality of patients with COVID-19, especially older age, higher BMI, lower prothrombin time, higher bilirubin, higher H-CRP, disease severity, comorbidities, and treatment options. Further, the mortality rate was lower in patients receiving combined antiviral therapies than in those receiving single-agent therapies. Patients who received supportive treatment had a lower mortality rate than those who did not receive supportive treatment. However, gamma globulin or antibiotics did not significantly improve the mortality rate of patients with COVID-19.
It has been reported that MERS and SARS are important independent predictors of death in old age, and recent studies have also shown that patients over the age of 65 years and with complications such as hypertension have a higher risk of death [8]. Previous studies have shown that the risk factors related to the development of ARDS and progression from ARDS to death included neutrophilia and organ and coagulation dysfunction [9].
In addition, previous studies have shown that total bilirubin, prothrombin time, and H-CRP were significantly higher in COVID-19 patients who died than in those who recovered, and the increase in these indicators may be related to acute lung injury [10]. Our research findings are consistent with this. According to previous reports, older patients have higher COVID-19-related mortality rates. Older patients may have a stronger innate response to viral infection than younger patients, resulting in insufficient inhibition of viral replication and longer proinflammatory responses, which may lead to poor prognosis [11, 12]. Obesity was an important factor that causes significant changes in bronchoalveolar lavage fluid and blood. Higher BMI results in higher expression levels of CD147-related genes in immune cells, but lower expression levels in barrier cells, which may affect the development and progression of COVID-19 [13]. In our study, the average age and BMI in the deceased group were significantly higher than those in the recovered group, which suggests that a high BMI and older age may be risk factors for mortality. Shock and ARDS are serious complications in COVID-19 patients that may be directly caused by SARS-CoV-2 infection or by the low immune response in elderly patients [14]. This is consistent with our finding that patients in the deceased group were older and the incidence of complications was higher. However, further studies are needed to investigate the pathogenesis of sudden shock and ARDS caused by COVID-19.
The level and duration of viral replication are significant factors in assessing the risk of transmission and making patient isolation decisions. Therefore, the rational use of antiviral drugs is vital. However, there is no clear treatment for COVID-19 at present. Previous studies have shown that arbidol monotherapy is superior to lopinavir/litonavir for COVID-19 [3]. Further, Deng et al reported that the combination of arbidol and lopinavir/litonavir led to faster viral clearance than lopinavir/litonavir alone [15]. In addition to antiviral drugs, some antibiotics have also been shown to be effective for the treatment of COVID-19. Azithromycin is a broad-spectrum macrolidene antibiotic. In vitro studies have suggested that azithromycin has antiviral activity, and its mechanism is similar to that of chloroquine or hydroxychloroquine [16]. In clinical studies, azithromycin combined with chloroquine or hydroxychloroquine reduced the viral load of patients with COVID-19 and increased the recovery rate [17,18]. In severe influenza, prolonged viral shedding is associated with mortality, and delayed antiviral treatment is an independent risk factor for prolonged viral shedding [19,20]. Similarly, effective antiviral treatment may improve the prognosis of COVID-19. To improve the recovery rate, the focus should be on starting antiviral therapy early, which would reduce the peak viral load and thus reduce the degree of associated immunopathological damage [21].
There are some limitations in our study. First, the sample size was not large enough because this study was performed in a single center, which may have led to selection bias. Therefore, the mortality rate in our study may not reflect the true mortality rate of COVID-19. Second, the analysis indexes included in this study were not comprehensive enough, and there may be information bias. Third, it was a retrospective design with inherent limitations in study design, and some missing variables may have affected the results of our study. Finally, the lack of effective antiviral drugs in the early stage of the disease may also have led to poor clinical outcomes in some patients. Large-scale clinical studies with more clinical features and treatment performed in multiple centers are needed to validate our conclusions.
Conclusions
Age, BMI, prothrombin, H-CRP, shock, and ARDS are independent risk factors affecting the treatment effect of patients with COVID-19. The combination of antiviral drugs and supportive therapy appears to be associated with lower mortality, but further research is needed.
Figures
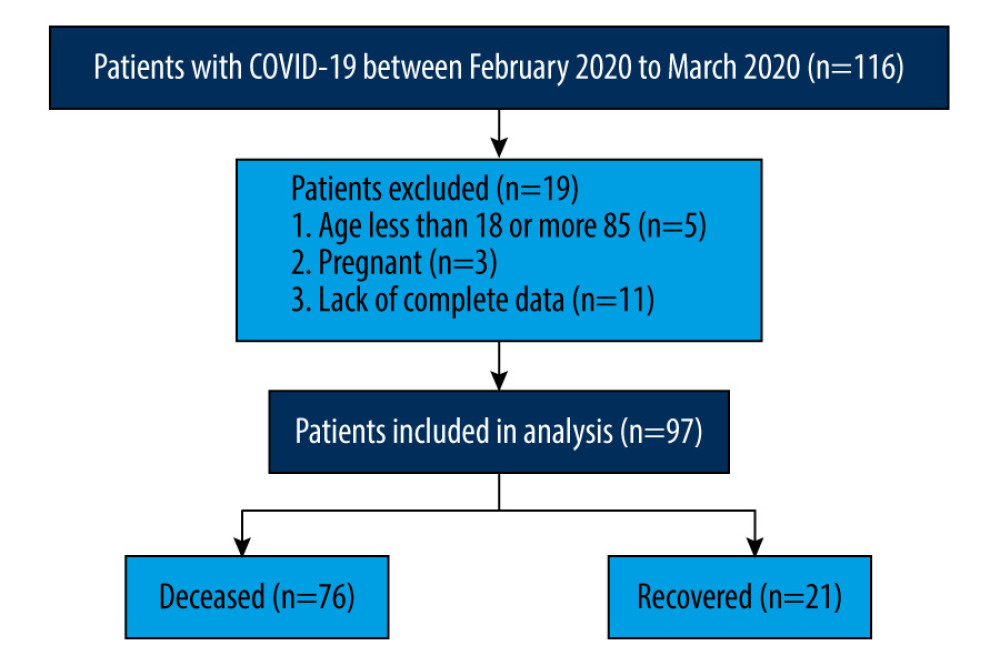 Figure 1. Flow chart of patient selection.
Figure 1. Flow chart of patient selection. 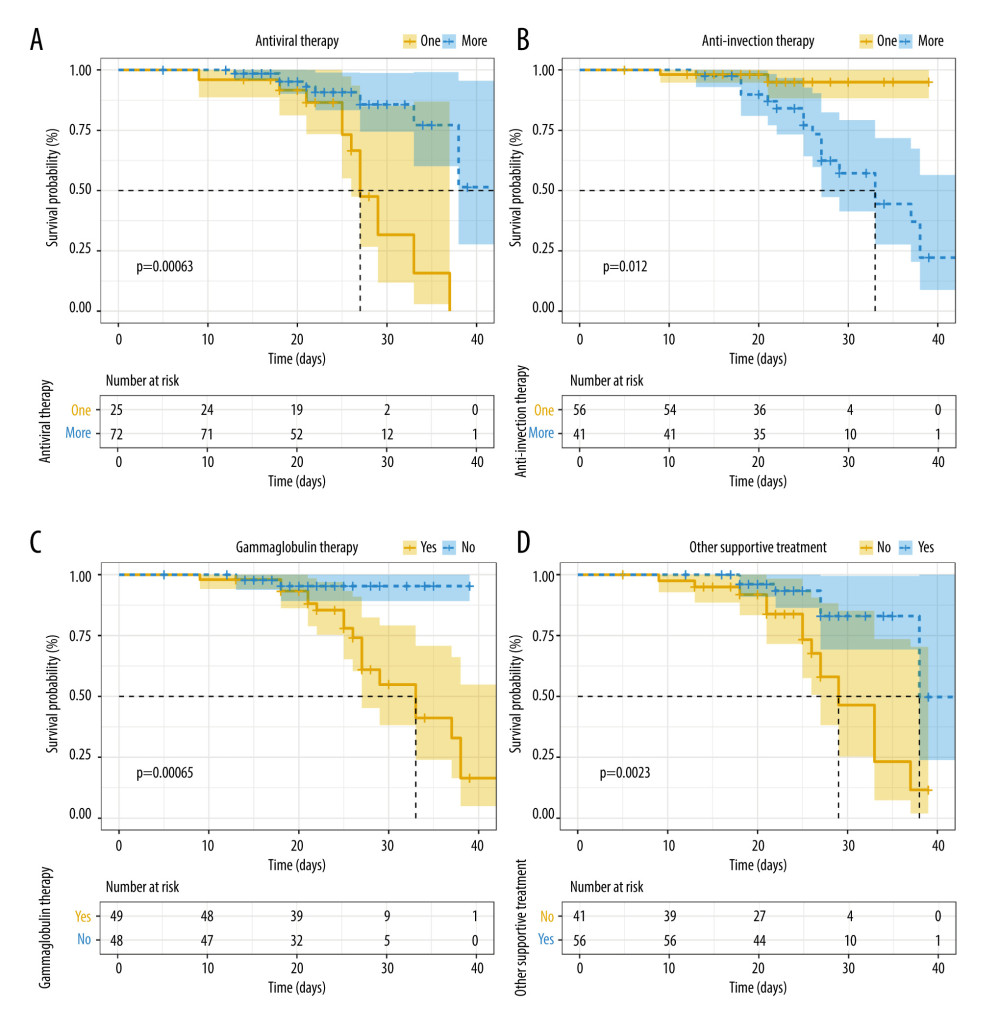 Figure 2. Kaplan-Meier survival curves of patients with COVID-19. (A) Survival curve of patients receiving single-agent antiviral therapy and those receiving combination antiviral therapy (P=0.004). (B) Survival curve of patients receiving single-agent anti-infection therapy and those receiving combination anti-infection therapy (P<0.001). (C) Overall survival for all patients based on administration of gamma globulin therapy (P<0.001). (D) Overall survival for all patients based on receiving other supportive treatment (Chinese medicine or other) (P=0.048).
Figure 2. Kaplan-Meier survival curves of patients with COVID-19. (A) Survival curve of patients receiving single-agent antiviral therapy and those receiving combination antiviral therapy (P=0.004). (B) Survival curve of patients receiving single-agent anti-infection therapy and those receiving combination anti-infection therapy (P<0.001). (C) Overall survival for all patients based on administration of gamma globulin therapy (P<0.001). (D) Overall survival for all patients based on receiving other supportive treatment (Chinese medicine or other) (P=0.048). Tables
Table 1. Demographic and comorbidity characteristics of the COVID-19 patients in the recovered and deceased groups.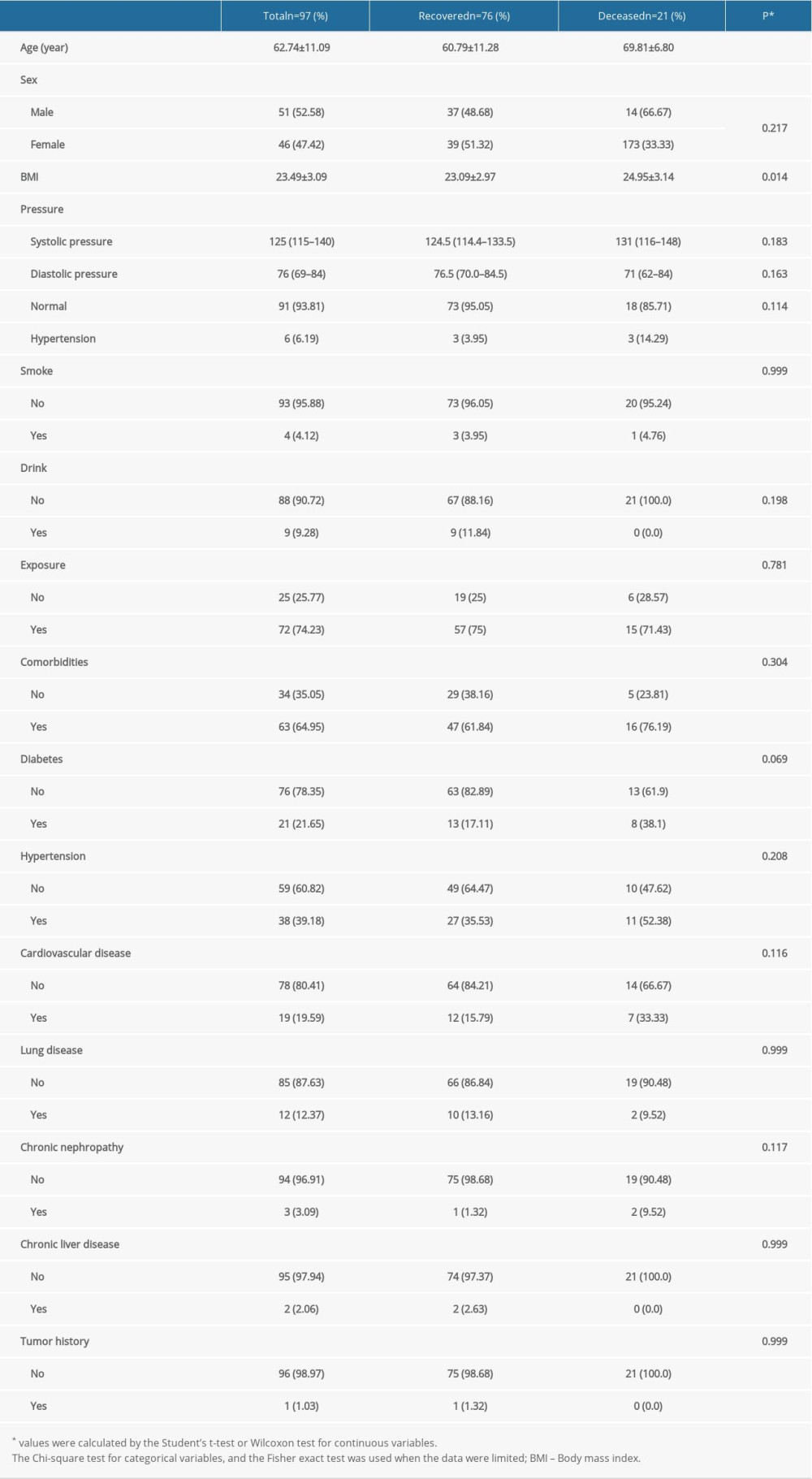 Table 2. The Clinical symptoms characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 2. The Clinical symptoms characteristics of the COVID-19 patients in the recovered and deceased groups.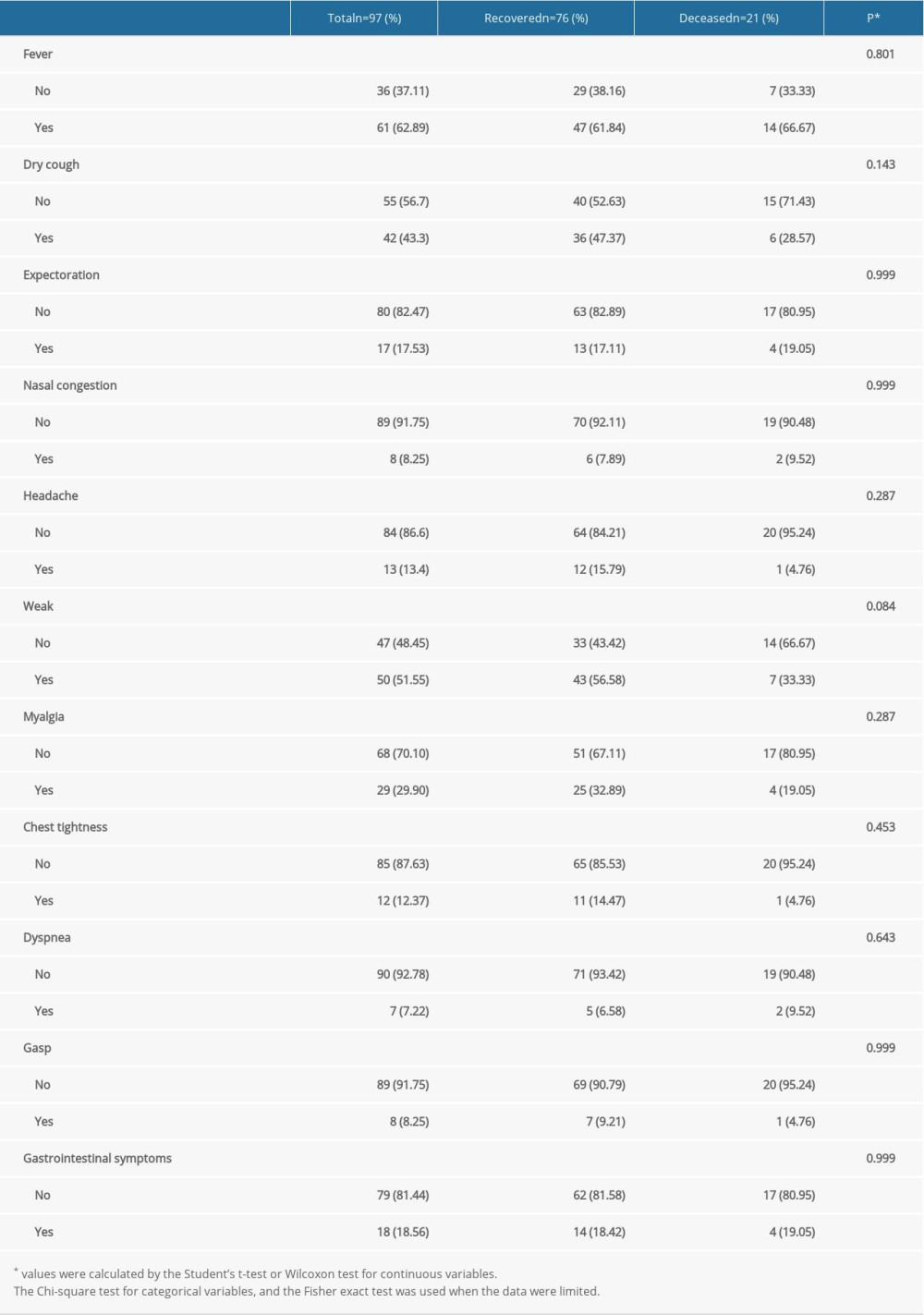 Table 3. The laboratory characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 3. The laboratory characteristics of the COVID-19 patients in the recovered and deceased groups.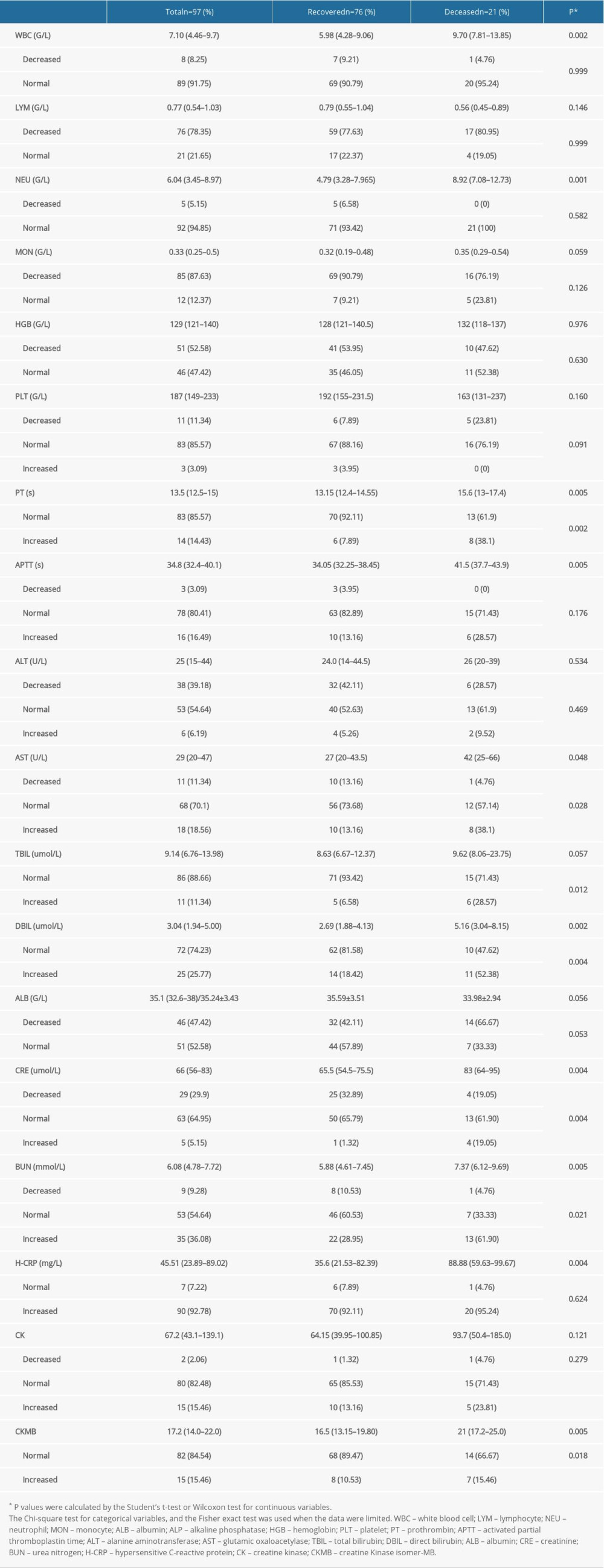 Table 4. The clinical treatment of the COVID-19 patients in the recovered and deceased groups.
Table 4. The clinical treatment of the COVID-19 patients in the recovered and deceased groups.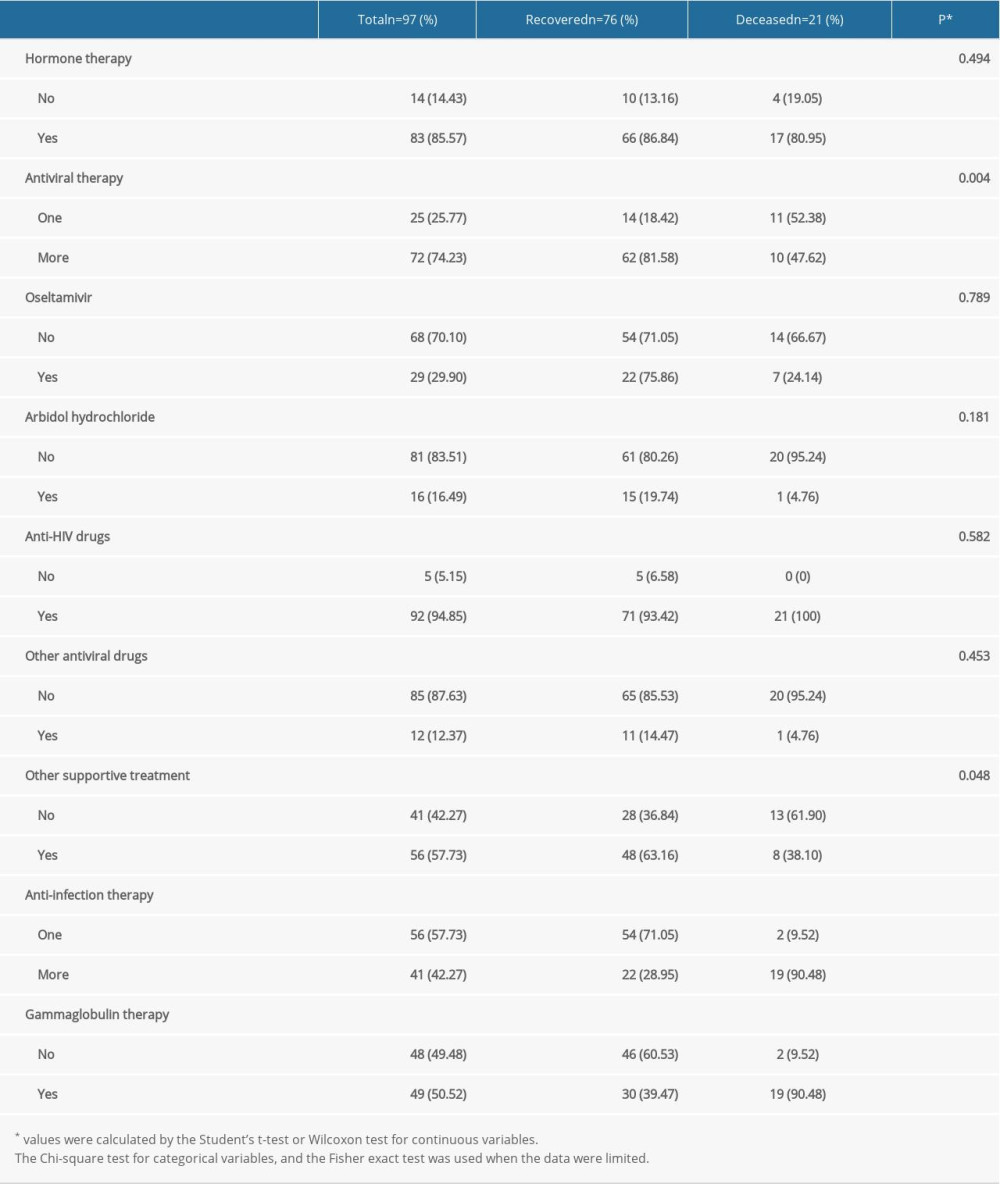 Table 5. The complications characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 5. The complications characteristics of the COVID-19 patients in the recovered and deceased groups.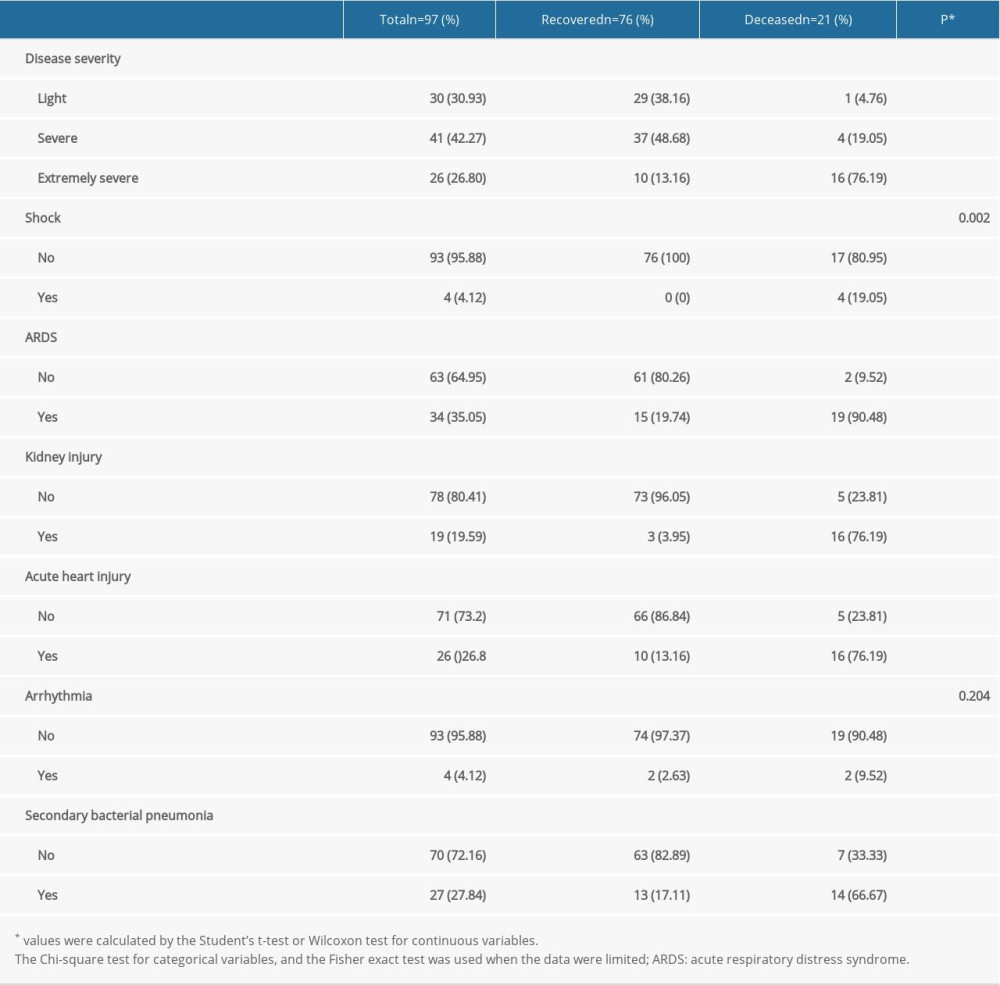 Table 6. The factors associated with survival of COVID-19 patients by univariate and multivariate cox regression analysis.
Table 6. The factors associated with survival of COVID-19 patients by univariate and multivariate cox regression analysis.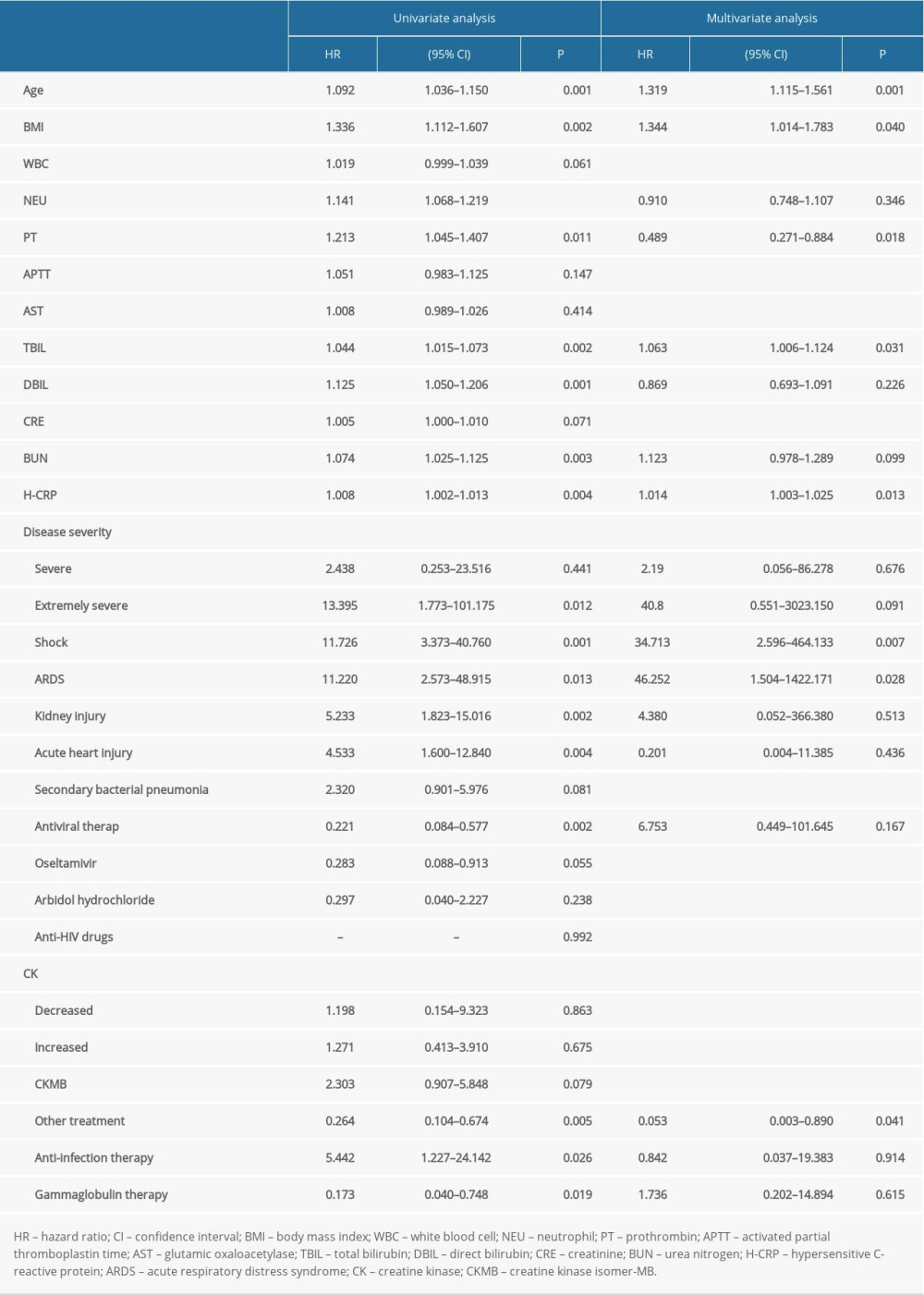
References
1. Chen Y, Liu Q, Guo D, Emerging coronaviruses: Genome structure, replication, and pathogenesis: J Med Virol, 2020; 92; 418-23
2. Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB, Pharmacologic treatments for coronavirus disease 2019 (COVID-19): A review: JAMA, 2020; 323(18); 1824-36
3. Zhu Z, Lu Z, Xu T, Arbidol monotherapy is superior to lopinavir/ritonavir in treating COVID-19: J Infect, 2020; 81(1); e21-23
4. NHCO C: Diagnosis and treatment plan for novel coronavirus pneumonia (trial version 6), 2020 http.//www.nhc.gov.cn/yzygj/s7653p/202002/8334a8326dd94d329df351d7da8aefc2/files/b218cfeb1bc54639af227f922bf6b817
5. World Health Organization: Novel coronavirus – China, 2020 http://www.who.int/csr/don/12-january-2020-novel-coronavirus-china/en/
6. Jha V, Arici M, Collins AJ, Understanding kidney care needs and implementation strategies in low- and middle-income countries: Conclusions from a “Kidney Disease: Improving Global Outcomes” (KDIGO) Controversies Conference: Kidney Int, 2016; 90; 1164-74
7. Huang C, Wang Y, Li X, Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China: Lancet, 2020; 395; 497-506
8. Jordan RE, Adab P, Cheng KK, COVID-19: Risk factors for severe disease and death: BMJ, 2020; 368; m1198
9. Zheng Z, Peng F, Xu B, Risk factors of critical & mortal COVID-19 cases. A systematic literature review and meta-analysis: J Infection, 2020; 81; e16
10. Chen T, Wu D, Chen H, Clinical characteristics of 113 deceased patients with coronavirus disease 2019: Retrospective study: BMJ, 2020; 368; m1091
11. Goronzy JJ, Fang F, Cavanagh MM, Naive T cell maintenance and function in human aging: J Immunol, 2015; 194; 4073-80
12. Anderson RM, Heesterbeek H, Klinkenberg D, Hollingsworth TD, How will country-based mitigation measures influence the course of the COVID-19 epidemic?: Lancet, 2020; 395; 931-34
13. Radzikowska U, Ding M, Tan G, Distribution of ACE2, CD147, CD26, and other SARS-CoV-2 associated molecules in tissues and immune cells in health and in asthma, COPD, obesity, hypertension, and COVID-19 risk factors: Allergy, 2020; 75(11); 2829-45
14. Wu C, Chen X, Cai Y, Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China: JAMA Intern Med, 2020; 180; 934-43
15. Deng L, Li C, Zeng Q, Arbidol combined with LPV/r versus LPV/r alone against Corona Virus Disease 2019: A retrospective cohort study: J Infect, 2020; 81(1); e1-e5
16. Damle B, Vourvahis M, Wang E, Clinical pharmacology perspectives on the antiviral activity of azithromycin and use in COVID-19: Clin Pharmacol Ther, 2020; 108(2); 201-11
17. Gautret P, Lagier J, Parola P, Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial: Int J Antimicrob Agents, 2020; 56(1); 105949
18. Gautret P, Lagier J, Parola P, Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: A pilot observational study: Travel Med Infect Dis, 2020; 34; 101663
19. Khoury J, Szwarcwort M, Kra-Oz Z, Duration of viral shedding and factors associated with prolonged shedding among inpatients with influenza treated with oseltamivir: A prospective cohort study: Eur J Clin Microbiol, 2018; 37(2); 319-23
20. Xu K, Chen Y, Yuan J, Factors associated with prolonged viral RNA shedding in patients with coronavirus disease 2019 (COVID-19): Clin Infect Dis, 2020; 71(15); 799-806
21. Xu Z, Shi L, Wang Y, Pathological findings of COVID-19 associated with acute respiratory distress syndrome: Lancet Respir Med, 2020; 8; 420-22
Figures
 Figure 1. Flow chart of patient selection.
Figure 1. Flow chart of patient selection. Figure 2. Kaplan-Meier survival curves of patients with COVID-19. (A) Survival curve of patients receiving single-agent antiviral therapy and those receiving combination antiviral therapy (P=0.004). (B) Survival curve of patients receiving single-agent anti-infection therapy and those receiving combination anti-infection therapy (P<0.001). (C) Overall survival for all patients based on administration of gamma globulin therapy (P<0.001). (D) Overall survival for all patients based on receiving other supportive treatment (Chinese medicine or other) (P=0.048).
Figure 2. Kaplan-Meier survival curves of patients with COVID-19. (A) Survival curve of patients receiving single-agent antiviral therapy and those receiving combination antiviral therapy (P=0.004). (B) Survival curve of patients receiving single-agent anti-infection therapy and those receiving combination anti-infection therapy (P<0.001). (C) Overall survival for all patients based on administration of gamma globulin therapy (P<0.001). (D) Overall survival for all patients based on receiving other supportive treatment (Chinese medicine or other) (P=0.048). Tables
 Table 1. Demographic and comorbidity characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 1. Demographic and comorbidity characteristics of the COVID-19 patients in the recovered and deceased groups. Table 2. The Clinical symptoms characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 2. The Clinical symptoms characteristics of the COVID-19 patients in the recovered and deceased groups. Table 3. The laboratory characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 3. The laboratory characteristics of the COVID-19 patients in the recovered and deceased groups. Table 4. The clinical treatment of the COVID-19 patients in the recovered and deceased groups.
Table 4. The clinical treatment of the COVID-19 patients in the recovered and deceased groups. Table 5. The complications characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 5. The complications characteristics of the COVID-19 patients in the recovered and deceased groups. Table 6. The factors associated with survival of COVID-19 patients by univariate and multivariate cox regression analysis.
Table 6. The factors associated with survival of COVID-19 patients by univariate and multivariate cox regression analysis. Table 1. Demographic and comorbidity characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 1. Demographic and comorbidity characteristics of the COVID-19 patients in the recovered and deceased groups. Table 2. The Clinical symptoms characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 2. The Clinical symptoms characteristics of the COVID-19 patients in the recovered and deceased groups. Table 3. The laboratory characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 3. The laboratory characteristics of the COVID-19 patients in the recovered and deceased groups. Table 4. The clinical treatment of the COVID-19 patients in the recovered and deceased groups.
Table 4. The clinical treatment of the COVID-19 patients in the recovered and deceased groups. Table 5. The complications characteristics of the COVID-19 patients in the recovered and deceased groups.
Table 5. The complications characteristics of the COVID-19 patients in the recovered and deceased groups. Table 6. The factors associated with survival of COVID-19 patients by univariate and multivariate cox regression analysis.
Table 6. The factors associated with survival of COVID-19 patients by univariate and multivariate cox regression analysis. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








