20 October 2020: Lab/In Vitro Research
Verteporfin Promotes the Apoptosis and Inhibits the Proliferation, Migration, and Invasion of Cervical Cancer Cells by Downregulating SULT2B1 Expression
Lijun Yin1BCDEF, Guilin Chen2AF*DOI: 10.12659/MSM.926780
Med Sci Monit 2020; 26:e926780
Abstract
BACKGROUND: Cervical cancer threatens women’s health worldwide. Verteporfin (VP), a small-molecule YAP1 inhibitor, inhibits cancer cell growth. This study investigated whether VP could inhibit the proliferation and promote the apoptosis of cervical cancer cells by decreasing SULT2B1 expression.
MATERIAL AND METHODS: Normal and cancerous cervical cell proliferation after VP treatment was detected by CCK-8 assay. HeLa cell migration, invasion, and apoptosis after VP treatment and transfection were analyzed by wound healing assay, transwell assay, and TUNEL assay, respectively. The expression of related proteins was determined by western blot analysis. Western blot and RT-qPCR analysis detected mRNA and protein expression of SULT2B1.
RESULTS: Different VP concentrations (0.5, 1, 2, and 5 μM) inhibited the viability of HeLa cells and had no obvious effect on H8 cells. Therefore, 5 μM VP was selected for subsequent experiments. VP inhibited the proliferation, migration, and invasion of HeLa cells and promoted their apoptosis. Bcl-2 expression decreased, and expression of Bax, caspase-3, and caspase-9 in VP-treated HeLa cells increased. SULT2B1 expression increased in cervical cancer cells compared with normal cervical cells. Furthermore, SULT2B1 expression increased in HeLa cells and VP suppressed SULT2B1 expression. SULT2B1 overexpression reduced the inhibiting effect of VP on the proliferation, migration, and apoptosis of HeLa cells, and reduced VP effect on apoptosis of HeLa cells. SULT2B1 overexpression upregulated the Bcl-2 expression and downregulated the expression of Bax, caspase-3, and caspase-9 in VP-treated HeLa cells.
CONCLUSIONS: VP inhibited the proliferation, migration, and invasion and promoted apoptosis of cervical cancer cells by decreasing SULT2B1 expression.
Keywords: Apoptosis Regulatory Proteins, Hela Cells, Sulfotransferases, Verteporfin
Background
Cervical cancer is the fourth most common type of malignant tumor, threatening the health of women worldwide. According to the most recent global annual report on cancer, there were approximately 569 847 new cases of cervical cancer worldwide in 2018, and the number of deaths due to cervical cancer was 311 365 [1]. A study of 38 countries across 5 continents found that while the rates of cervical cancer were falling in developed countries, they were stable and even rising in economically poor regions [2]. The latest national cancer statistics released by the National Cancer Center of China in 2017 showed that among the top 10 malignant tumors in women, cervical cancer accounted for 6.25% of cases, ranking sixth; and deaths due to cervical cancer accounted for approximately 3.96% of all deaths from malignant tumors in women, ranking eighth. From the top 10 malignant cancers reported, the proportion of rural and urban cervical cancer cases was 6.34% and 2.68%, respectively. [3]. Therefore, there is an urgent need to find effective methods to treat cervical cancer.
Verteporfin (VP), a second-generation porphyrin photosensitizer, can be activated by light irradiation (wavelength 689 nm) and used in photodynamic therapy for macular degeneration. VP is a small-molecule yes-associated protein 1 (YAP1) inhibitor that induces the conformation changes of YAP1 and its TEA domain transcription factor (TEAD)-YAP complex [4]. Liu et al. [5] demonstrated that VP, as an inhibitor of YAP1/TEAD interaction, inhibited the YAP1-induced liver overgrowth in murine models. YAP1 is a potent oncogene, which is amplified in various human cancers and it is related to the Hippo tumor suppressor pathway [6–9]. YAP is a transcriptional coactivator that performs transcriptional functions by binding to the end-effector (transcription factor TEADs) of the Hippo pathway [10]. Deletion of TEADs in cells can inhibit YAP-dependent gene regulation and significantly reduce the YAP-dependent proliferation of cells [11]. The TEAD-YAP complex can jointly exert the activity of transcription factors and activate important genes such as cell cycle-related and anti-apoptotic genes to promote cell growth and proliferation and inhibit cell apoptosis [12,13]. VP has a strong inhibitory effect on the growth and proliferation of cancer cells [14–16]. Liao et al. [17] reported that VP exerted inhibiting effects on the proliferation and cell cycle of papillary thyroid cancer cells, partially by suppressing the ERK1/2 signaling pathway. Moreover, VP suppressed the tumor growth of gastric carcinomas
In the Search Tool for the Retrieval of Interacting Genes/Proteins (STRING) database, we found that YAP combines with SULT2B1. The SULT2B1 gene encodes 2 isoforms, SULT2B1a and SULT2B1b, which are generated by alternate splicing of the first exon [21]. SULT2B1b, a member of the SULT2 family, is expressed in multiple normal tissues including prostate, skin, breast, intestine, lung, and placenta [22,23]. In hepatocarcinoma, SULT2B1b expression was increased in tumors compared with in adjacent tissues. SULT2B1b interference suppressed cell growth, induced cell cycle arrest and apoptosis of Hepa1–6 cells, and suppressed tumor growth in nude mouse xenografts [24]. Hu et al. [25] demonstrated that SULT2B1b expression is increased in colorectal carcinoma (CRC) and CRC-derived cell lines, and SULT2B1b overexpression promotes the growth and invasion of CRC cells. SULT2B1b is significantly overexpressed in gastric cancer (GC) and SULT2B1b overexpression promoted the angiogenesis and tumor growth in a GC mice model [26]. The UALCAN database indicates that SULT2B1 expression is also upregulated in cervical cancer. Thus, we speculated that the knock-down of SULT2B1 could inhibit the development of cervical cancer.
The present study aimed to investigate whether VP could inhibit the proliferation, migration, and invasion of cervical cancer cells and promote their apoptosis by decreasing SULT2B1 expression.
Material and Methods
CELL CULTURE:
Cervical cancer cell lines (HeLa, C33A, SiHa, Caski, and MS751) were purchased from American Type Culture Collection (Rockville, MD, USA). H8 cells were obtained from Tong Pai Bio-tech Co, Ltd (Shanghai, China). HCC94 cells were obtained from the cell bank of the Chinese Academy of Sciences (Shanghai, China). All cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) (Gibco; Thermo Fisher Scientific, Inc, Waltham, MA, USA) containing 10% fetal bovine serum (FBS) (Pan Life Sciences) in a 37°C and 5% CO2 incubator. The cell culture was performed as previously described [27].
CCK-8 ASSAY:
The wells of a 96-well plate were seeded with 1×104 cervical cancer cells and treated with different concentrations of VP (0.5, 1, 2, and 5 μM) for 24, 48, and 72 h. To each group, 10 μL CCK-8 solution (Dojindo, Kumamoto, Japan) was added in the dark at 24, 48, and 72 h. After conventional incubation for 2 h, the optical density value at a wavelength of 450 nm was detected by the multifunctional enzyme marker, indicating the viability of the cells. The cell proliferation assay was performed as previously described [27].
CLONE FORMATION ASSAY:
Cervical cancer cells in the logarithmic growth stage were seeded in a 6-well plate (1×105 cells/well) and treated with VP. After 14 d, 70% ethanol (Sigma-Aldrich, Shanghai, China) was added to fix the cells, which were then stained with 0.5% crystal violet (Beyotime, Haimen, China) for 30 min. After washing with phosphate-buffered saline (PBS) (Sigma-Aldrich), the number of colonies was recorded.
WOUND HEALING ASSAY:
Cervical cancer cells were inoculated in a 6-well plate (1×105 cells/well) for culture. When cell fusion reached 80%, the cells were scraped with a pipette tip. After washing with PBS for 3 times, the 6-well plate was put into an incubator for further culture with VP for 24 h, and was then taken out for observation under a microscope.
TRANSWELL ASSAY:
Matrigel (50 μL) was added to a transwell assay chamber and solidified at 37°C for 3 h. Serum-free medium and complete medium were added to the upper and lower chambers. Then, cells were inoculated in the upper chamber and cultured with VP for 48 h. The cells that did not pass through the membrane were removed, and the remaining cells were fixed with paraformaldehyde (Sigma-Aldrich). After crystal violet (10 g/L) staining, the cells were observed and photographed under a microscope.
WESTERN BLOT ANALYSIS:
The total cell proteins were extracted with radioimmunoprecipitation assay (RIPA) lysis buffer (Beyotime Institute of Biotechnology, Shanghai, China) and quantitatively analyzed by a bicinchoninic acid (BCA) assay kit (Pierce, Appleton, WI, USA). Each well was loaded with 30 μg protein. Each group of proteins was separated by 100 g/L sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE), and the separated proteins were transferred to a polyvinylidene fluoride (PVDF) membrane through semi-dry membrane transfer. An amount of 50 g/L of dried skimmed milk powder was used to seal the PVDF membrane at room temperature for 2 h and then the following primary antibodies were added to the membrane for culture at 4°C overnight: MMP2 (ab92536, Abcam, UK), MMP9 (ab76003, Abcam, UK), Bcl-2 (ab32124, Abcam, UK), Bax (ab32503, Abcam, UK), caspase-3 (ab13847, Abcam, UK), pro-caspase-3 (ab32499, Abcam, UK), caspase-9 (ab32539, Abcam, UK), pro-caspase-9 (ab138412, Abcam, UK), and SULT2B1 (ab254616, Abcam, UK). The following day, the PVDF membrane was washed with PBS solution at least 3 times. The rabbit horseradish peroxidase-linked IgG second antibody was added to the PVDF membrane, which was then incubated at room temperature for 2 h. The protein bands were exposed, developed with enhanced chemiluminescent solution (Sigma-Aldrich), and quantified with Image J software. GAPDH was used as internal reference. The western blot analysis was performed as previously described [28].
TUNEL ASSAY:
Cervical cancer cells were inoculated into a 24-well plate (5×104 cells/well) and cultured with VP for 48 h. Then, the HeLa cells were washed with PBS after the supernatant was discarded. Next, HeLa cells were fixed with 4% paraformaldehyde for 20 min and washed with PBS for 3 times. Then, the HeLa cells were treated with 70% ethanol at 4°C for overnight. The next day, the HeLa cells were washed once with PBS. Using a TUNEL apoptosis kit according to the manufacturer’s instructions, apoptosis was observed by a microscope.
QUANTITATIVE REVERSE TRANSCRIPTION POLYMERASE CHAIN REACTION ANALYSIS:
Cervical cancer cells were lysed with lysate and total RNA was extracted by the Trizol method. Then, RNA was reverse-transcribed to cDNA with a PrimeScript reverse transcription reagent kit (Takara Biotechnology Co, Ltd, Beijing, China). Next, quantitative reverse transcription polymerase chain reaction (RT-qPCR) was performed by a TaqMan Universal PCR Master Mix kit (Invitrogen; Thermo Fisher Scientific, Inc). The PCR reaction procedure was conducted as follows: pre-denaturation at 95°C for 30 s, 95°C for 5 s, and 60°C for 30 s, for a total of 40 cycles. The result of RT-qPCR analysis was calculated by the 2–ΔΔCt method with the reference gene GAPDH. The RT-qPCR analysis was performed as previously described [28].
CELL TRANSFECTION:
Cervical cancer cells in the logarithmic growth stage were taken and 1×105 cells/well were inoculated into a 6-well plate. FBS was replaced with serum-free medium after the cells were adhered to the wall. Plasmid transfection (OE-NC and OE-SULT2B1) was performed according to the instructions of the Lipofectamine 2000 kit (Invitrogen; Thermo Fisher Scientific, Inc) for 2 h and transfected cells were cultured in the medium for an additional 24 h. OE-NC plasmid was used as negative control. OE-SULT2B1 plasmid was used to overexpress SULT2B1.
STATISTICAL ANALYSIS:
SPSS version 21.0 was used to analyze the experimental data which were presented as mean±standard deviation (SD). The
Results
EFFECT OF VP ON THE PROLIFERATION OF CERVICAL CANCER CELLS:
At each time point (24, 48, and 72 h), the proliferation of HeLa cells was decreased with the increasing concentrations of VP (0.5, 1, 2, and 5 μM) (Figure 1A). However, the proliferation of H8 cells was not obviously changed with the increasing concentrations of VP at each time point (Figure 1B). Therefore, 5 μM VP inhibited the proliferation of HeLa cells but did not affect the proliferation of H8 cells. As shown in Figure 1C, the clone formation of the HeLa cells was suppressed with 5 μM of VP treatment compared with no suppression in the control group.
VP INHIBITS THE MIGRATION AND INVASION OF CERVICAL CANCER CELLS:
VP treatment inhibited the migration (Figure 2A, 2C) and invasion (Figure 2B, 2D) of HeLa cells compared to the control group. As shown in Figure 2E, the expression of MMP2 and MMP9 was decreased in the VP group compared with that of the control group.
VP PROMOTES THE APOPTOSIS OF CERVICAL CANCER CELLS:
As shown in Figure 3A, the apoptosis of HeLa cells was enhanced after VP treatment. Accordingly, the expression of Bcl-2 was decreased while the expression of Bax, caspase-3, and caspase-9 in the VP-treated HeLa cells was increased (Figure 3B, 3C).
VP SUPPRESSED SULT2B1 EXPRESSION IN CERVICAL CANCER CELLS:
The mRNA and protein expression of SULT2B1 in cervical cancer cells was increased compared with that of the H8 group, and mRNA and protein expression of SULT2B1 in HeLa and HCC94 cells was the highest of the cervical cancer cells. Because HeLa cells were used in the above experiment, they were selected for the subsequent experiments (Figure 4A, 4B). As shown in Figure 4C and 4D, SULT2B1 expression in VP-treated HeLa cells was decreased.
SULT2B1 OVEREXPRESSION WEAKENED THE INHIBITING EFFECT OF VP ON THE PROLIFERATION OF CERVICAL CANCER CELLS:
After HeLa cells were transfected with OE-NC, the mRNA and protein expression of SULT2B1 in the OE-NC transfected HeLa cells was not obviously changed compared with that of the control group. However, the mRNA and protein expression of SULT2B1 was increased in HeLa cells that were transfected with OE-SULT2B1 (Figure 5A, 5B). As shown in Figure 5C, the results indicated that VP strongly suppressed the viability of HeLa cells, while SULT2B1 overexpression improved the viability of HeLa cells. Further, the clone formation ability of HeLa cells was decreased after VP treatment and was enhanced by SULT2B1 overexpression (Figure 5D).
SULT2B1 OVEREXPRESSION WEAKENED THE INHIBITING EFFECT OF VP ON THE MIGRATION AND INVASION OF CERVICAL CANCER CELLS:
The migration (Figure 6A, 6B) and invasion (Figure 6C, 6D) of HeLa cells were significantly decreased after VP treatment, while SULT2B1 overexpression reduced the inhibiting effect of VP on the migration and invasion of HeLa cells. As shown in Figure 6E, VP decreased the expression of MMP2 and MMP9, which was improved by SULT2B1 overexpression.
SULT2B1 OVEREXPRESSION WEAKENED THE EFFECT OF VP ON APOPTOSIS:
As shown in Figure 7A, VP promoted the apoptosis of HeLa cells, and SULT2B1 overexpression reduced the effect of VP on the apoptosis of HeLa cells. As shown in Figure 7B, VP decreased Bcl-2 expression and increased the expression of Bax, caspase-3, and caspase-9 in HeLa cells. However, SULT2B1 overexpression reversed the expression of Bcl-2, Bax, caspase-3, and caspase-9 present in VP-treated HeLa cells.
Discussion
VP is a phenolic porphyrin derivative photosensitizer used in the clinical treatment of neovascular macular degeneration [29]. In 2000, it was approved by the FDA for the clinical treatment of age-related macular disease with choroid angiogenesis. VP could be used as a non-photosensitive activator to treat a variety of diseases [30]. VP has the ability to kill tumor cells and has a good antitumor effect [31]. Jiang et al. [32] demonstrated that VP suppressed the proliferation, invasion, and migration of MDA-MB-231 cells by decreasing YAP expression. VP also induced apoptosis and inhibited the proliferation, migration, and invasion of uveal melanoma cells [33]. The present study indicated that VP suppressed the migration and invasion of HeLa cells while promoting their apoptosis.
VP is also an inhibitor of YAP. Studies have shown that the expression of YAP1 in cervical precancerous lesions and different clinical stages of cervical cancer had correlations with this disease [34,35]. Hui et al. [36] verified the role of YAP1 in cervical cancer and indicated that YAP1 interference could suppress the proliferation and invasion of HeLa cells. SULT2B1 was found to combine with YAP1 based on our search in the STRING database. Research shows that human SULTs contain 4 family members: SULT1, SULT2, SULT4, and SULT6, which also contain 13 subfamilies [37]. SULT2B1 is commonly studied in hormone-sensitive tissues, such as the uterus, breast, ovary, and prostate [23,38]. It was found that the level of SULT2B1 in endometrial carcinoma tissues was significantly higher than that in paracancerous tissues [39]. Fu et al. [40] reported that the expression of enzymes is related to steroid and estrogen metabolism in breast cell lines, demonstrating that SULT2B1 is not expressed in normal breast epithelial cells, but is expressed in estrogen receptor-positive cells. Researchers have screened genes from biopsy samples of breast cancer and found that the expression level of SULT2B1 in breast cancer with positive estrogen receptors is significantly higher than that in normal breast tissue and breast cancer with negative estrogen receptors [41,42]. The inhibition of SULT2B1b hinders cell growth and promotes the cell cycle arrest and apoptosis of Hepa1–6 cells [24]. SULT2B1b overexpression promotes the growth and invasion of CRC cells and enhances the angiogenesis and tumor growth in GC mouse models [25,26]. In the present study, we found that the expression level of SULT2B1 in cervical cancer cells was increased compared with that of normal cervical cells. Furthermore, VP could also inhibit the SULT2B1 expression in HeLa cells. However, SULT2B1 overexpression reduced the inhibiting effect of VP on the proliferation, migration, and invasion of HeLa cells and the promotion effect of VP on the apoptosis of these cells.
Conclusions
In conclusion, in this study, VP inhibited the proliferation, migration, and invasion of HeLa cells and promoted their apoptosis by decreasing SULT2B1 expression. SULT2B1 overexpression improved the proliferation, migration, and invasion abilities of VP-treated HeLa cells and suppressed their apoptosis. This novel finding provides a potential treatment strategy for cervical cancer.
Figures
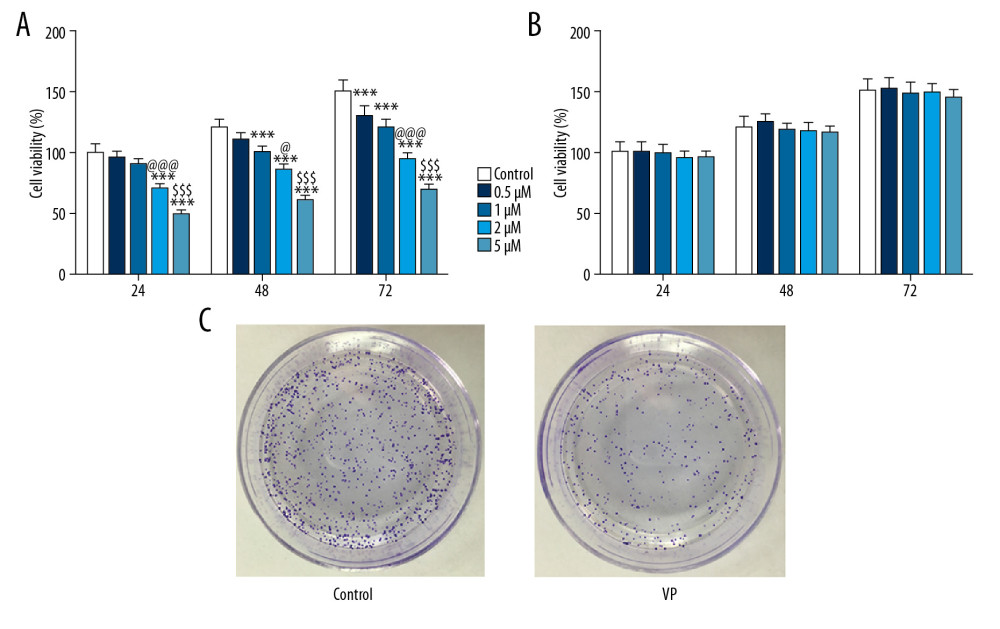 Figure 1. Effect of verteporfin (VP) on the proliferation of cervical cancer cells. (A) The viability of HeLa cells treated with different concentrations (0.5, 1, 2, and 5 μM) of VP was detected by CCK-8 assay. *** P<0.001 vs. Control group. @ P<0.05 and @@@ P<0.001 vs. 1 μM group. $$$ P<0.001 vs. 2 μM group. (B) The viability of H8 cells treated with different concentrations (0.5, 1, 2, and 5 μM) of VP was detected by CCK-8 assay. (C) The clone formation ability of HeLa cells treated with 5 μM VP was analyzed by clone formation assay.
Figure 1. Effect of verteporfin (VP) on the proliferation of cervical cancer cells. (A) The viability of HeLa cells treated with different concentrations (0.5, 1, 2, and 5 μM) of VP was detected by CCK-8 assay. *** P<0.001 vs. Control group. @ P<0.05 and @@@ P<0.001 vs. 1 μM group. $$$ P<0.001 vs. 2 μM group. (B) The viability of H8 cells treated with different concentrations (0.5, 1, 2, and 5 μM) of VP was detected by CCK-8 assay. (C) The clone formation ability of HeLa cells treated with 5 μM VP was analyzed by clone formation assay. 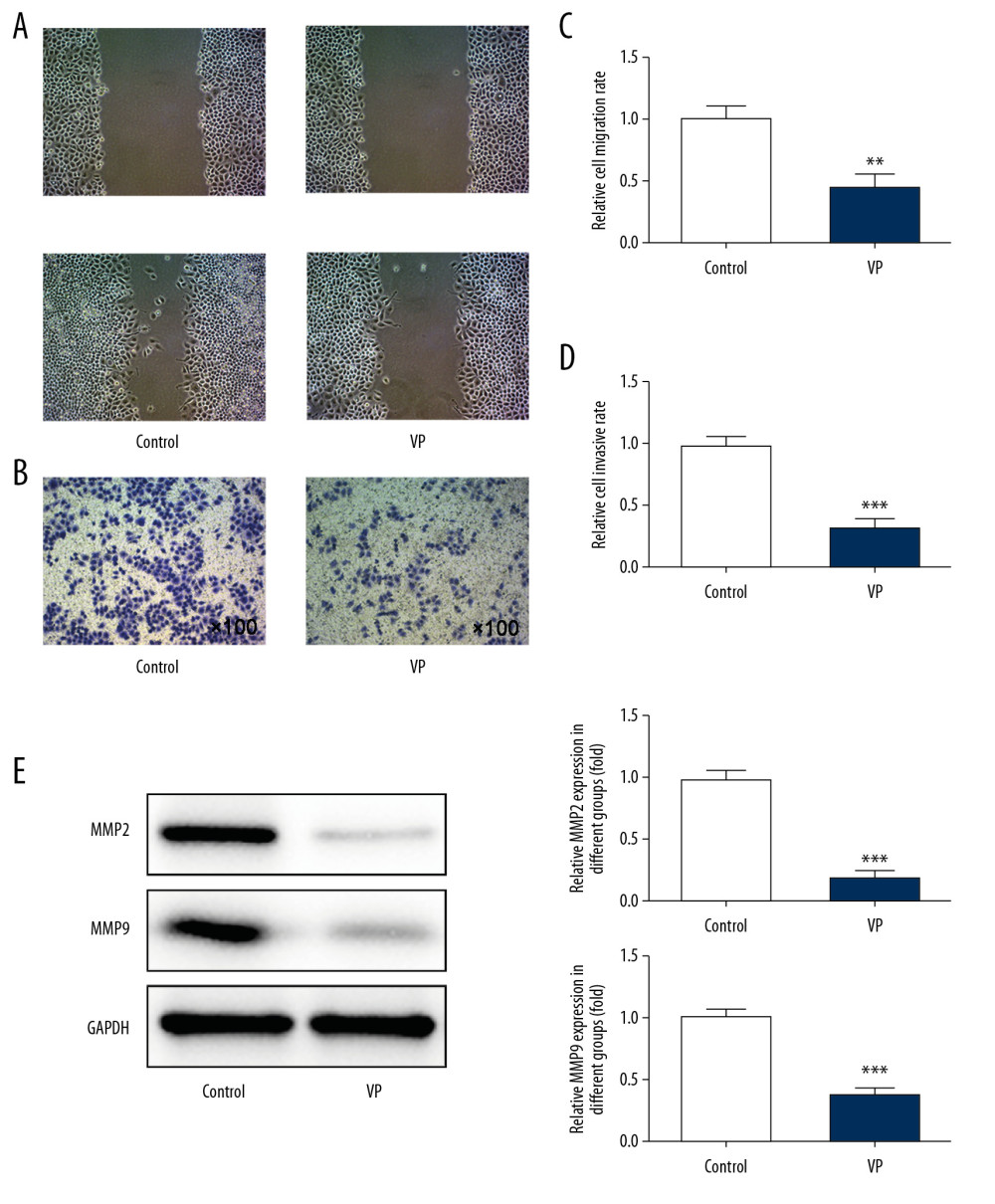 Figure 2. Verteporfin (VP) inhibited the migration and invasion of cervical cancer cells. (A, C) The migration of HeLa cells treated with 5 μM VP was analyzed by wound healing assay. ** P<0.01 vs. Control group. (B, D) The migration of HeLa cells treated with 5 μM VP was analyzed by transwell assay. *** P<0.001 vs. Control group. (E) The expression of MMP2 and MMP9 in HeLa cells treated with 5 μM VP was determined by western blot analysis. *** P<0.001 vs. Control group.
Figure 2. Verteporfin (VP) inhibited the migration and invasion of cervical cancer cells. (A, C) The migration of HeLa cells treated with 5 μM VP was analyzed by wound healing assay. ** P<0.01 vs. Control group. (B, D) The migration of HeLa cells treated with 5 μM VP was analyzed by transwell assay. *** P<0.001 vs. Control group. (E) The expression of MMP2 and MMP9 in HeLa cells treated with 5 μM VP was determined by western blot analysis. *** P<0.001 vs. Control group. 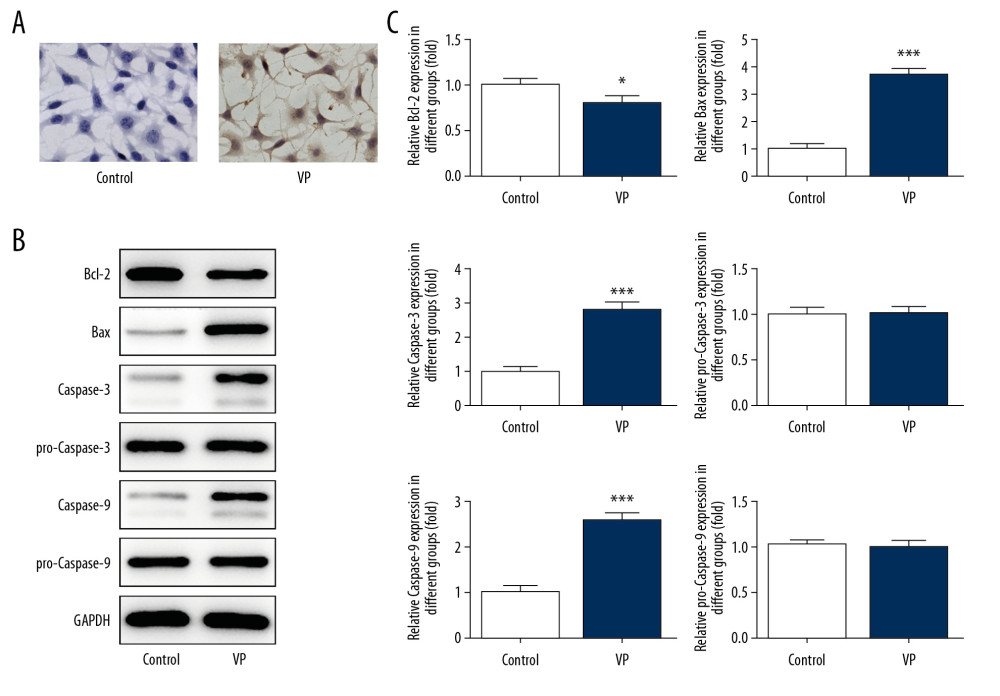 Figure 3. Verteporfin (VP) promoted the apoptosis of cervical cancer cells. (A) The apoptosis of HeLa cells treated with 5 μM VP was detected by TUNEL assay. (B, C) The expression of Bcl-2, Bax, caspase-3, pro-caspase-3, caspase-9, and pro-caspase-9 in HeLa cells treated with 5 μM VP was determined by western blot analysis. * P<0.05 and *** P<0.001 vs. Control group.
Figure 3. Verteporfin (VP) promoted the apoptosis of cervical cancer cells. (A) The apoptosis of HeLa cells treated with 5 μM VP was detected by TUNEL assay. (B, C) The expression of Bcl-2, Bax, caspase-3, pro-caspase-3, caspase-9, and pro-caspase-9 in HeLa cells treated with 5 μM VP was determined by western blot analysis. * P<0.05 and *** P<0.001 vs. Control group. 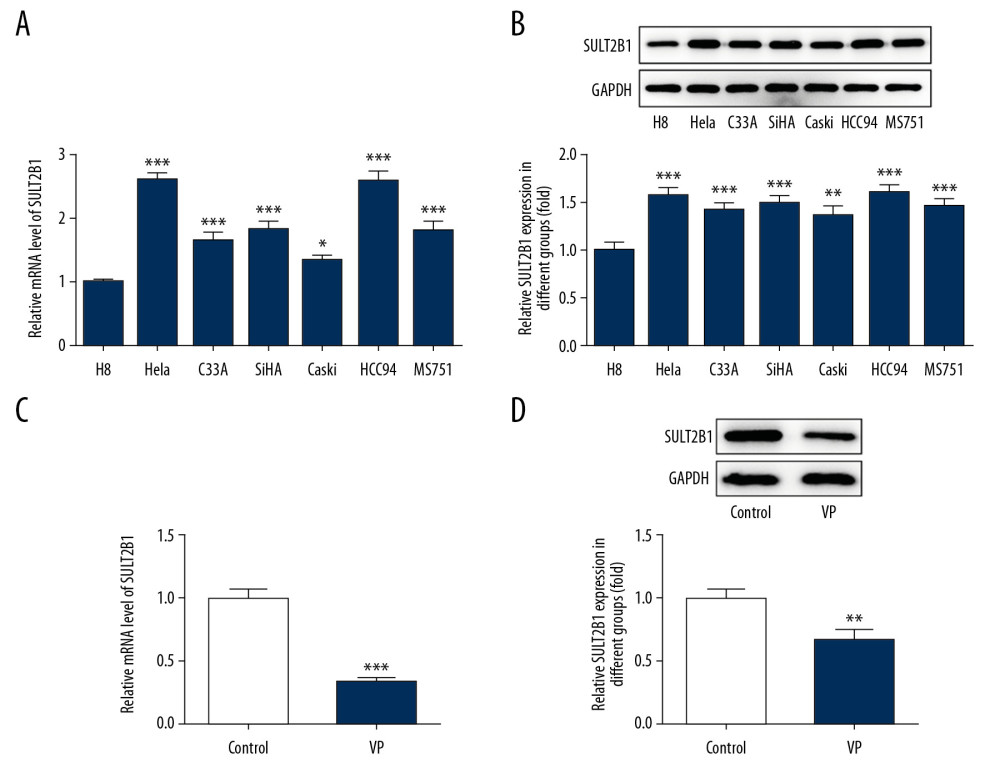 Figure 4. Verteporfin (VP) suppressed the SULT2B1 expression in cervical cancer cells. (A) The mRNA expression of SULT2B1 in normal cervical cells and cervical cancer cells was detected by RT-qPCR analysis. * P<0.05 and *** P<0.001 vs. H8 group. (B) The protein expression of SULT2B1 in normal cervical cells and cervical cancer cells was detected by western blot analysis. ** P<0.01 and *** P<0.001 vs. H8 group. (C) The mRNA expression of SULT2B1 in HeLa cells treated with 5 μM VP was detected by RT-qPCR analysis. *** P<0.001 vs. Control group. (D) The protein expression of SULT2B1 in HeLa cells treated with 5 μM VP was detected by western blot analysis. ** P<0.01 vs. Control group.
Figure 4. Verteporfin (VP) suppressed the SULT2B1 expression in cervical cancer cells. (A) The mRNA expression of SULT2B1 in normal cervical cells and cervical cancer cells was detected by RT-qPCR analysis. * P<0.05 and *** P<0.001 vs. H8 group. (B) The protein expression of SULT2B1 in normal cervical cells and cervical cancer cells was detected by western blot analysis. ** P<0.01 and *** P<0.001 vs. H8 group. (C) The mRNA expression of SULT2B1 in HeLa cells treated with 5 μM VP was detected by RT-qPCR analysis. *** P<0.001 vs. Control group. (D) The protein expression of SULT2B1 in HeLa cells treated with 5 μM VP was detected by western blot analysis. ** P<0.01 vs. Control group. 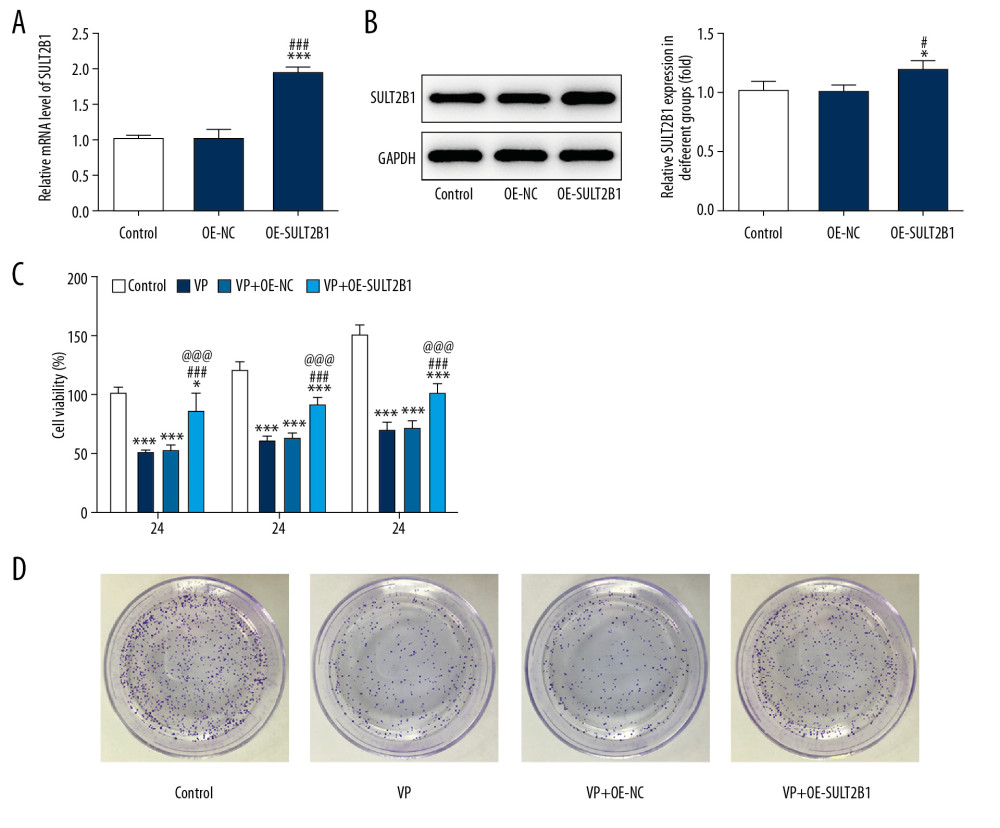 Figure 5. SULT2B1 overexpression weakened the inhibiting effect of verteporfin (VP) on the proliferation of cervical cancer cells. (A) The mRNA expression of SULT2B1 in HeLa cells after transfection was detected by RT-qPCR analysis. *** P<0.001 vs. Control group. ### P<0.001 vs. OE-NC group. (B) The protein expression of SULT2B1 in HeLa cells after transfection was detected by western blot analysis. * P<0.05 vs. Control group. # P<0.05 vs. OE-NC group. (C) The cell viability of HeLa cells after transfection and VP treatment was analyzed by CCK-8 assay. * P<0.05 and *** P<0.001 vs. Control group. ### P<0.001 vs. VP group. @@@ P<0.001 vs. VP+OE-NC group. (D) The clone formation ability of HeLa cells after transfection and VP treatment was determined by clone formation assay.
Figure 5. SULT2B1 overexpression weakened the inhibiting effect of verteporfin (VP) on the proliferation of cervical cancer cells. (A) The mRNA expression of SULT2B1 in HeLa cells after transfection was detected by RT-qPCR analysis. *** P<0.001 vs. Control group. ### P<0.001 vs. OE-NC group. (B) The protein expression of SULT2B1 in HeLa cells after transfection was detected by western blot analysis. * P<0.05 vs. Control group. # P<0.05 vs. OE-NC group. (C) The cell viability of HeLa cells after transfection and VP treatment was analyzed by CCK-8 assay. * P<0.05 and *** P<0.001 vs. Control group. ### P<0.001 vs. VP group. @@@ P<0.001 vs. VP+OE-NC group. (D) The clone formation ability of HeLa cells after transfection and VP treatment was determined by clone formation assay. 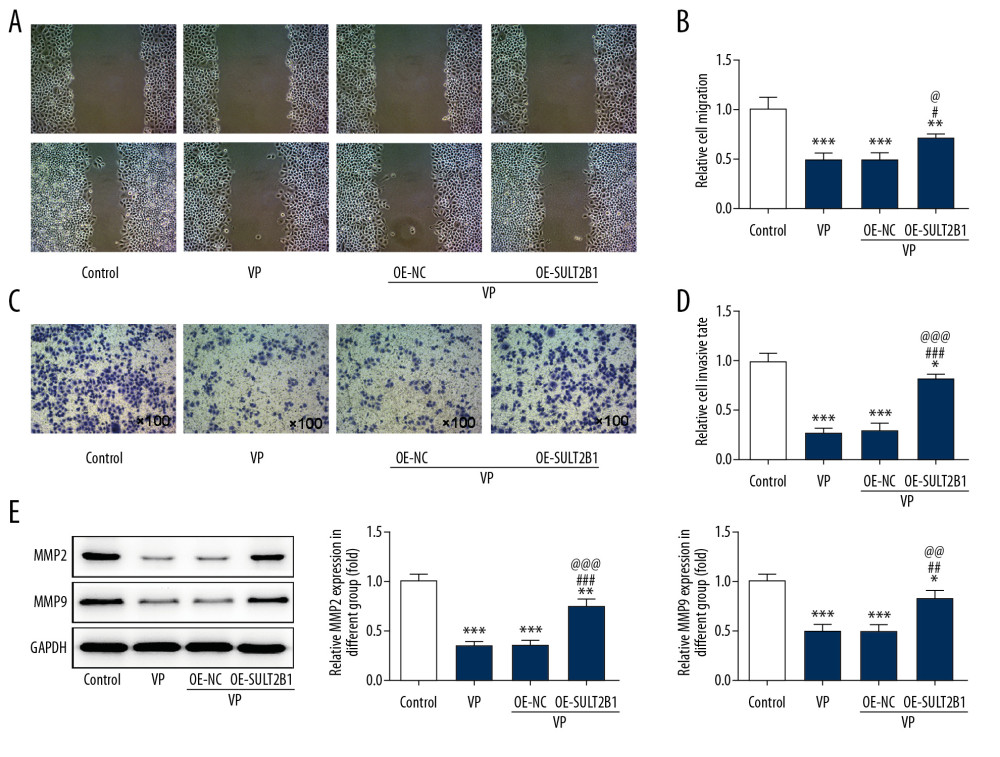 Figure 6. SULT2B1 overexpression weakened the inhibiting effect of verteporfin (VP) on the migration and invasion of cervical cancer cells. (A, B) The migration of HeLa cells after transfection and VP treatment was analyzed by wound healing assay. ** P<0.01 and *** P<0.001 vs. Control group. # P<0.05 vs. VP group. @ P<0.05 vs. OE-NC group. (C, D) The migration of HeLa cells after transfection and VP treatment was analyzed by transwell assay. * P<0.05 and *** P<0.001 vs. Control group. ### P<0.001 vs. VP group. @@@ P<0.001 vs. OE-NC group. (E) The expression of MMP2 and MMP9 in HeLa cells after transfection and VP treatment was determined by western blot analysis. * P<0.05, ** P<0.01 and *** P<0.001 vs. Control group. ## P<0.01 and ### P<0.001 vs. VP group. @@ P<0.01 and @@@ P<0.001 vs. OE-NC group.
Figure 6. SULT2B1 overexpression weakened the inhibiting effect of verteporfin (VP) on the migration and invasion of cervical cancer cells. (A, B) The migration of HeLa cells after transfection and VP treatment was analyzed by wound healing assay. ** P<0.01 and *** P<0.001 vs. Control group. # P<0.05 vs. VP group. @ P<0.05 vs. OE-NC group. (C, D) The migration of HeLa cells after transfection and VP treatment was analyzed by transwell assay. * P<0.05 and *** P<0.001 vs. Control group. ### P<0.001 vs. VP group. @@@ P<0.001 vs. OE-NC group. (E) The expression of MMP2 and MMP9 in HeLa cells after transfection and VP treatment was determined by western blot analysis. * P<0.05, ** P<0.01 and *** P<0.001 vs. Control group. ## P<0.01 and ### P<0.001 vs. VP group. @@ P<0.01 and @@@ P<0.001 vs. OE-NC group. 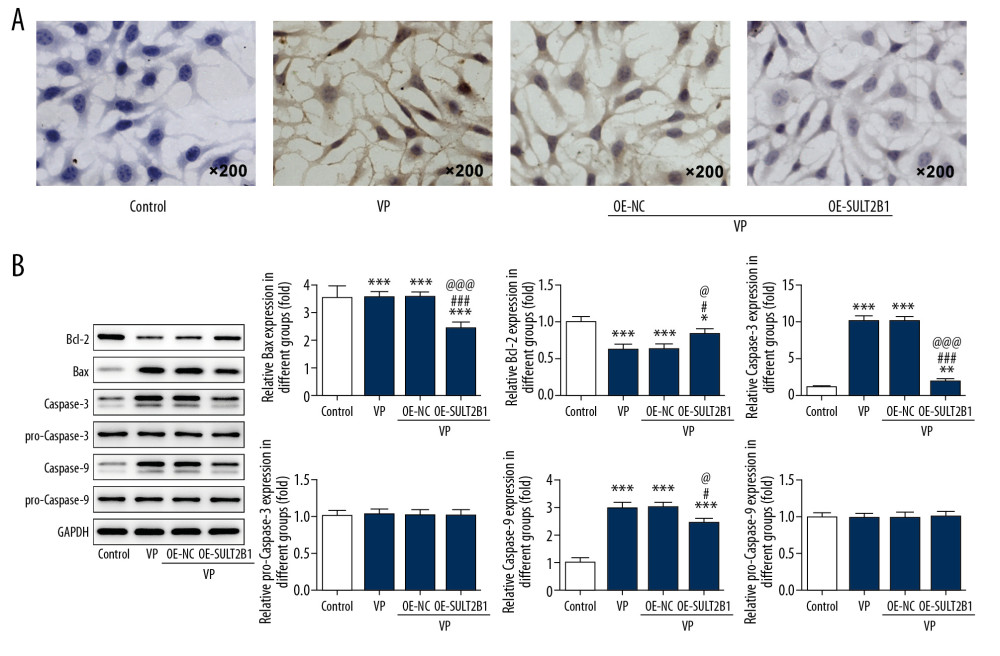 Figure 7. SULT2B1 overexpression weakened the promotion effect of verteporfin (VP) on the apoptosis of cervical cancer cells. (A) The apoptosis of HeLa cells after transfection and VP treatment was detected by TUNEL assay. (B) The expression of Bcl-2, Bax, caspase-3, pro-caspase-3, caspase-9 and pro-caspase-9 in HeLa cells after transfection and VP treatment was determined by western blot analysis. * P<0.05, ** P<0.01 and *** P<0.001 vs. Control group. # P<0.05 and ### P<0.001 vs. VP group. @ P<0.05 and @@@ P<0.001 vs. OE-NC group.
Figure 7. SULT2B1 overexpression weakened the promotion effect of verteporfin (VP) on the apoptosis of cervical cancer cells. (A) The apoptosis of HeLa cells after transfection and VP treatment was detected by TUNEL assay. (B) The expression of Bcl-2, Bax, caspase-3, pro-caspase-3, caspase-9 and pro-caspase-9 in HeLa cells after transfection and VP treatment was determined by western blot analysis. * P<0.05, ** P<0.01 and *** P<0.001 vs. Control group. # P<0.05 and ### P<0.001 vs. VP group. @ P<0.05 and @@@ P<0.001 vs. OE-NC group. References
1. Bray F, Ferlay J, Soerjomataram I, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2018; 68; 394-424
2. Vaccarella S, Lortet-Tieulent J, Plummer M, Worldwide trends in cervical cancer incidence: Impact of screening against changes in disease risk factors: Eur J Cancer, 2013; 49; 3262-73
3. Siegel R, Miller K, Jemal A, Cancer Statistics, 2017: Cancer J Clin, 2017; 67; 7-30
4. Lee K-W, Lee S, Kim S-B, Significant association of oncogene YAP1 with poor prognosis and cetuximab resistance in colorectal cancer patients: Clin Cancer Res, 2014; 21; 357-64
5. Liu-Chittenden Y, Huang B, Shim J, Genetic and pharmacological disruption of the TEAD-YAP complex suppresses the oncogenic activity of YAP: Genes Dev, 2012; 26; 1300-5
6. Zhao L, Lou G, Li A, Liu Y, lncRNA MALAT1 modulates cancer stem cell properties of liver cancer cells by regulating YAP1 expression via miR-375 sponging: Mol Med Rep, 2020; 22; 1449-57
7. Li S, Zhu H, Chen H, Glucose promotes epithelial-mesenchymal transitions in bladder cancer by regulating the functions of YAP1 and TAZ: J Cell Mol Med, 2020 [Online ahead of print]
8. Liu Z, Yao YY, Huang SS, LINC00662 promotes gastric cancer cell growth by modulating the Hippo-YAP1 pathway: Biochem Biophys Res Commun, 2018; 505; 843-49
9. Pan Y, Tong J, Lung R, RASAL2 promotes tumor progression through LATS2/YAP1 axis of hippo signaling pathway in colorectal cancer: Mol Cancer, 2018; 17; 102
10. Schlegelmilch K, Mohseni M, Kirak O, Yap1 acts downstream of α-catenin to control epidermal proliferation: Cell, 2011; 144; 782-95
11. Zhao B, Ye X, Yu J, TEAD mediates YAP-dependent gene induction and growth control: Genes Dev, 2008; 22; 1962-71
12. Ota M, Sasaki H, Mammalian Tead proteins regulate cell proliferation and contact inhibition as transcriptional mediators of Hippo signaling: Development (Cambridge, England), 2008; 135; 405-69
13. Thompson BJ, Cohen SM, The Hippo pathway regulates the bantam microRNA to control cell proliferation and apoptosis in Drosophila: Cell, 2006; 126; 767-74
14. Perra A, Kowalik MA, Ghiso E, YAP activation is an early event and a potential therapeutic target in liver cancer development: J Hepatol, 2014; 61; 1088-96
15. Huggett MT, Jermyn M, Gillams A, Phase I/II study of verteporfin photodynamic therapy in locally advanced pancreatic cancer: Br J Cancer, 2014; 110; 1698-704
16. Brodowska K, Al-Moujahed A, Marmalidou A: Exp Eye Res, 2014; 124; 67-73
17. Liao T, Wei WJ, Wen D, Verteporfin inhibits papillary thyroid cancer cells proliferation and cell cycle through ERK1/2 signaling pathway: J Cancer, 2018; 9; 1329-36
18. Julie G, Silvia M-C, Lornella S, Verteporfin targeting YAP1/TAZ-TEAD transcriptional activity inhibits the tumorigenic properties of gastric cancer stem cells: Int J Cancer, 2020; 146; 2255-67
19. Dong L, Lin F, Wu WJ, Verteporfin inhibits YAP-induced bladder cancer cell growth and invasion via Hippo signaling pathway: Int J Med Sci, 2018; 15; 645-52
20. Liu K, Du SM, Gao P, Zheng J, Verteporfin suppresses the proliferation, epithelial-mesenchymal transition and stemness of head and neck squamous carcinoma cells via inhibiting YAP1: J Cancer, 2019; 10; 4196-207
21. Meloche CA, Falany CN, Expression and characterization of the human 3β-hydroxysteroid sulfotransferases (SULT2B1a and SULT2B1b): J Steroid Biochem, 2001; 77; 261-69
22. Falany CN, He DN, Dumas NA, Human cytosolic sulfotransferase 2B1: Isoform expression, tissue specificity and subcellular localization: J Steroid Biochem, 2006; 102; 214-21
23. He DN, Meloche CA, Dumas NA, Different subcellular localization of sulphotransferase 2B1b in human placenta and prostate: Biochem J, 2004; 379; 533-40
24. Yang X, Xu Y, Guo F: PLoS One, 2013; 8; e60853
25. Hu L, Yang GZ, Zhang Y, Overexpression of SULT2B1b is an independent prognostic indicator and promotes cell growth and invasion in colorectal carcinoma: Lab Invest, 2015; 95; 1005-18
26. Chen W, Zhou H, Ye L, Zhan B, Overexpression of SULT2B1b promotes angiogenesis in human gastric cancer: Cell Physiol Biochem, 2016; 38; 1040-54
27. Zhang F, Zhang Y-Y, Sun Y-S: J Agric Food Chem, 2020; 68; 213-24
28. Sun Y-S, Thakur K, Hu F: Biomed Pharmacother, 2020; 125; 110013
29. Parodi M, La Spina C, Berchicci L, Photosensitizers and photodynamic therapy: Verteporfin: Dev Ophthalmol, 2015; 55; 330-36
30. Gibault F, Corvaisier M, Bailly F, Non-photoinduced biological properties of verteporfin: Curr Med Chem, 2016; 23; 1171-84
31. Chen R, Zhu S, Fan XG, High mobility group protein B1 controls liver cancer initiation through yes-associated protein-dependent aerobic glycolysis: Hepatology, 2018; 67; 1823-41
32. Jiang Y, Liu Y, Zhang ZVerteporfin inhibits proliferation, invasion and migration of MDA-MB-231 human breast cancer cells by down-regulating the expression of Yes-associated protein: Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi, 2017; 33(9); 1223-27
33. Ma YW, Liu YZ, Pan JX, Verteporfin induces apoptosis and eliminates cancer stem-like cells in uveal melanoma in the absence of light activation: Am J Cancer Res, 2016; 6; 2816-30
34. Xiao H, Wu L, Zheng H, Expression of yes-associated protein in cervical squamous epithelium lesions: Int J Gynecol Cancer, 2014; 24; 1575-82
35. Liu T, Liu Y, Gao H, Clinical significance of yes-associated protein overexpression in cervical carcinoma the differential effects based on histotypes: Int J Gynecol Cancer, 2013; 23; 735-43
36. Hui H, Lei W, Yan H, Effect of YAP1 expression down-regulated by siRNA interference on cell proliferation and invasion of human cervical cancer HeLa cells: J Mod Oncol, 2016; 24; 177-80
37. Rendic S, Guengerich F, Contributions of human enzymes in carcinogen metabolism: Chem Res Toxicol, 2012; 25; 1316-83
38. Hevir N, Ribič-Pucelj M, Rižner TL, Disturbed balance between phase I and II metabolizing enzymes in ovarian endometriosis: A source of excessive hydroxy-estrogens and ROS?: Mol Cell Endocrinol, 2013; 367; 74-84
39. Hevir N, Šinkovec J, Rižner TL, Disturbed expression of phase I and phase II estrogen-metabolizing enzymes in endometrial cancer: Lower levels of CYP1B1 and increased expression of S-COMT: Mol Cell Endocrinol, 2011; 331; 158-67
40. Fu J, Weise A, Falany J, Expression of estrogenicity genes in a lineage cell culture model of human breast cancer progression: Breast Cancer Res Treat, 2009; 120; 35-45
41. Bièche I, Girault I, Urbain E, Relationship between intratumoral expression of genes coding for xenobiotic-metabolizing enzymes and benefit from adjuvant tamoxifen in estrogen receptor alpha-positive postmenopausal breast carcinoma: Breast Cancer Res, 2004; 6; R252-63
42. Tozlu S, Girault I, Vacher S, Identification of novel genes that co-cluster with estrogen receptor alpha in breast tumor biopsy specimens, using a large-scale real-time reverse transcription-PCR approach: Endocr Relat Cancer, 2006; 13; 1109-20
Figures
 Figure 1. Effect of verteporfin (VP) on the proliferation of cervical cancer cells. (A) The viability of HeLa cells treated with different concentrations (0.5, 1, 2, and 5 μM) of VP was detected by CCK-8 assay. *** P<0.001 vs. Control group. @ P<0.05 and @@@ P<0.001 vs. 1 μM group. $$$ P<0.001 vs. 2 μM group. (B) The viability of H8 cells treated with different concentrations (0.5, 1, 2, and 5 μM) of VP was detected by CCK-8 assay. (C) The clone formation ability of HeLa cells treated with 5 μM VP was analyzed by clone formation assay.
Figure 1. Effect of verteporfin (VP) on the proliferation of cervical cancer cells. (A) The viability of HeLa cells treated with different concentrations (0.5, 1, 2, and 5 μM) of VP was detected by CCK-8 assay. *** P<0.001 vs. Control group. @ P<0.05 and @@@ P<0.001 vs. 1 μM group. $$$ P<0.001 vs. 2 μM group. (B) The viability of H8 cells treated with different concentrations (0.5, 1, 2, and 5 μM) of VP was detected by CCK-8 assay. (C) The clone formation ability of HeLa cells treated with 5 μM VP was analyzed by clone formation assay. Figure 2. Verteporfin (VP) inhibited the migration and invasion of cervical cancer cells. (A, C) The migration of HeLa cells treated with 5 μM VP was analyzed by wound healing assay. ** P<0.01 vs. Control group. (B, D) The migration of HeLa cells treated with 5 μM VP was analyzed by transwell assay. *** P<0.001 vs. Control group. (E) The expression of MMP2 and MMP9 in HeLa cells treated with 5 μM VP was determined by western blot analysis. *** P<0.001 vs. Control group.
Figure 2. Verteporfin (VP) inhibited the migration and invasion of cervical cancer cells. (A, C) The migration of HeLa cells treated with 5 μM VP was analyzed by wound healing assay. ** P<0.01 vs. Control group. (B, D) The migration of HeLa cells treated with 5 μM VP was analyzed by transwell assay. *** P<0.001 vs. Control group. (E) The expression of MMP2 and MMP9 in HeLa cells treated with 5 μM VP was determined by western blot analysis. *** P<0.001 vs. Control group. Figure 3. Verteporfin (VP) promoted the apoptosis of cervical cancer cells. (A) The apoptosis of HeLa cells treated with 5 μM VP was detected by TUNEL assay. (B, C) The expression of Bcl-2, Bax, caspase-3, pro-caspase-3, caspase-9, and pro-caspase-9 in HeLa cells treated with 5 μM VP was determined by western blot analysis. * P<0.05 and *** P<0.001 vs. Control group.
Figure 3. Verteporfin (VP) promoted the apoptosis of cervical cancer cells. (A) The apoptosis of HeLa cells treated with 5 μM VP was detected by TUNEL assay. (B, C) The expression of Bcl-2, Bax, caspase-3, pro-caspase-3, caspase-9, and pro-caspase-9 in HeLa cells treated with 5 μM VP was determined by western blot analysis. * P<0.05 and *** P<0.001 vs. Control group. Figure 4. Verteporfin (VP) suppressed the SULT2B1 expression in cervical cancer cells. (A) The mRNA expression of SULT2B1 in normal cervical cells and cervical cancer cells was detected by RT-qPCR analysis. * P<0.05 and *** P<0.001 vs. H8 group. (B) The protein expression of SULT2B1 in normal cervical cells and cervical cancer cells was detected by western blot analysis. ** P<0.01 and *** P<0.001 vs. H8 group. (C) The mRNA expression of SULT2B1 in HeLa cells treated with 5 μM VP was detected by RT-qPCR analysis. *** P<0.001 vs. Control group. (D) The protein expression of SULT2B1 in HeLa cells treated with 5 μM VP was detected by western blot analysis. ** P<0.01 vs. Control group.
Figure 4. Verteporfin (VP) suppressed the SULT2B1 expression in cervical cancer cells. (A) The mRNA expression of SULT2B1 in normal cervical cells and cervical cancer cells was detected by RT-qPCR analysis. * P<0.05 and *** P<0.001 vs. H8 group. (B) The protein expression of SULT2B1 in normal cervical cells and cervical cancer cells was detected by western blot analysis. ** P<0.01 and *** P<0.001 vs. H8 group. (C) The mRNA expression of SULT2B1 in HeLa cells treated with 5 μM VP was detected by RT-qPCR analysis. *** P<0.001 vs. Control group. (D) The protein expression of SULT2B1 in HeLa cells treated with 5 μM VP was detected by western blot analysis. ** P<0.01 vs. Control group. Figure 5. SULT2B1 overexpression weakened the inhibiting effect of verteporfin (VP) on the proliferation of cervical cancer cells. (A) The mRNA expression of SULT2B1 in HeLa cells after transfection was detected by RT-qPCR analysis. *** P<0.001 vs. Control group. ### P<0.001 vs. OE-NC group. (B) The protein expression of SULT2B1 in HeLa cells after transfection was detected by western blot analysis. * P<0.05 vs. Control group. # P<0.05 vs. OE-NC group. (C) The cell viability of HeLa cells after transfection and VP treatment was analyzed by CCK-8 assay. * P<0.05 and *** P<0.001 vs. Control group. ### P<0.001 vs. VP group. @@@ P<0.001 vs. VP+OE-NC group. (D) The clone formation ability of HeLa cells after transfection and VP treatment was determined by clone formation assay.
Figure 5. SULT2B1 overexpression weakened the inhibiting effect of verteporfin (VP) on the proliferation of cervical cancer cells. (A) The mRNA expression of SULT2B1 in HeLa cells after transfection was detected by RT-qPCR analysis. *** P<0.001 vs. Control group. ### P<0.001 vs. OE-NC group. (B) The protein expression of SULT2B1 in HeLa cells after transfection was detected by western blot analysis. * P<0.05 vs. Control group. # P<0.05 vs. OE-NC group. (C) The cell viability of HeLa cells after transfection and VP treatment was analyzed by CCK-8 assay. * P<0.05 and *** P<0.001 vs. Control group. ### P<0.001 vs. VP group. @@@ P<0.001 vs. VP+OE-NC group. (D) The clone formation ability of HeLa cells after transfection and VP treatment was determined by clone formation assay. Figure 6. SULT2B1 overexpression weakened the inhibiting effect of verteporfin (VP) on the migration and invasion of cervical cancer cells. (A, B) The migration of HeLa cells after transfection and VP treatment was analyzed by wound healing assay. ** P<0.01 and *** P<0.001 vs. Control group. # P<0.05 vs. VP group. @ P<0.05 vs. OE-NC group. (C, D) The migration of HeLa cells after transfection and VP treatment was analyzed by transwell assay. * P<0.05 and *** P<0.001 vs. Control group. ### P<0.001 vs. VP group. @@@ P<0.001 vs. OE-NC group. (E) The expression of MMP2 and MMP9 in HeLa cells after transfection and VP treatment was determined by western blot analysis. * P<0.05, ** P<0.01 and *** P<0.001 vs. Control group. ## P<0.01 and ### P<0.001 vs. VP group. @@ P<0.01 and @@@ P<0.001 vs. OE-NC group.
Figure 6. SULT2B1 overexpression weakened the inhibiting effect of verteporfin (VP) on the migration and invasion of cervical cancer cells. (A, B) The migration of HeLa cells after transfection and VP treatment was analyzed by wound healing assay. ** P<0.01 and *** P<0.001 vs. Control group. # P<0.05 vs. VP group. @ P<0.05 vs. OE-NC group. (C, D) The migration of HeLa cells after transfection and VP treatment was analyzed by transwell assay. * P<0.05 and *** P<0.001 vs. Control group. ### P<0.001 vs. VP group. @@@ P<0.001 vs. OE-NC group. (E) The expression of MMP2 and MMP9 in HeLa cells after transfection and VP treatment was determined by western blot analysis. * P<0.05, ** P<0.01 and *** P<0.001 vs. Control group. ## P<0.01 and ### P<0.001 vs. VP group. @@ P<0.01 and @@@ P<0.001 vs. OE-NC group. Figure 7. SULT2B1 overexpression weakened the promotion effect of verteporfin (VP) on the apoptosis of cervical cancer cells. (A) The apoptosis of HeLa cells after transfection and VP treatment was detected by TUNEL assay. (B) The expression of Bcl-2, Bax, caspase-3, pro-caspase-3, caspase-9 and pro-caspase-9 in HeLa cells after transfection and VP treatment was determined by western blot analysis. * P<0.05, ** P<0.01 and *** P<0.001 vs. Control group. # P<0.05 and ### P<0.001 vs. VP group. @ P<0.05 and @@@ P<0.001 vs. OE-NC group.
Figure 7. SULT2B1 overexpression weakened the promotion effect of verteporfin (VP) on the apoptosis of cervical cancer cells. (A) The apoptosis of HeLa cells after transfection and VP treatment was detected by TUNEL assay. (B) The expression of Bcl-2, Bax, caspase-3, pro-caspase-3, caspase-9 and pro-caspase-9 in HeLa cells after transfection and VP treatment was determined by western blot analysis. * P<0.05, ** P<0.01 and *** P<0.001 vs. Control group. # P<0.05 and ### P<0.001 vs. VP group. @ P<0.05 and @@@ P<0.001 vs. OE-NC group. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








