17 February 2021: Database Analysis
Prognostic Factors for Survival in Patients with Malignant Giant Cell Tumor of Bone: A Risk Nomogram Analysis Based on the Population
Xiaolong Zhu1BCDEF, Runzhi Huang12ABCDEF, Peng Hu1E, Penghui Yan1ABCDEF, Suna Zhai3D, Jie Zhang2AE, Junwei Zhuang1F, Huabin Yin4E, Tong Meng4AF*, Daoke Yang3F, Zongqiang Huang1AGDOI: 10.12659/MSM.929154
Med Sci Monit 2021; 27:e929154
Abstract
BACKGROUND: Malignant giant cell tumor of bone (MGCTB) is a rare histological type of malignant tumor that has a high tendency for local relapse and distant metastasis and ultimately leads to a poor prognosis. The purpose of this study was to describe the epidemiological features, identify the prognostic factors, and construct nomograms for patients with MGCTB.
MATERIAL AND METHODS: Patients with MGCTB that was histologically diagnosed between 1973 and 2014 were selected from the Surveillance, Epidemiology, and End Results (SEER) database as a training set. Survival analysis, Lasso regression, and random forests were used to identify the prognostic variables and establish the nomograms for patients with MGCTB, while an external cohort of 37 patients from our own institution and an external cohort of 163 patients from the SEER database in 2016 were used to validate the generalization performance of the nomograms.
RESULTS: In total, univariate and multivariable analysis indicated that age, International Classification of Diseases for Oncology, historical stage, primary site, surgery information, radiotherapy, and chemotherapy were independent prognostic variables for overall survival or cause-specific survival. Nomograms based on the multivariable models were built to predict survival, and we achieved a higher C-index in subsequent multidimensional validation.
CONCLUSIONS: Age, historical stage, and chemotherapy were independent prognostic variables for overall survival and cause-specific survival of MGCTB patients, and radiotherapy and primary site were independent prognostic variables for overall survival. Nomograms based on significant clinicopathological features and clinical experience can be effective in predicting the probability of survival for MGCTB patients.
Keywords: Giant Cell Tumor of Bone, nomograms, Biomarkers, Tumor, Bone Neoplasms, Cohort Studies, Databases, Genetic, Risk Assessment, Risk Factors, SEER Program
Background
A giant cell tumor of bone (GCTB) is an aggressive noncancerous skeletal tumor that consists of osteoclast-like multinucleated giant cells, spindle-like stromal cells, and monocytic round cells. Malignant GCTB (MGCTB) is the malignant form of GCTB and accounts for 2–9% of all cases [1–4]. Bone destruction is the prevailing clinical feature and results in local pain and pathological fracture. Previous studies have reported that local recurrence and distant metastasis are common, with relatively poor prognosis [5–9].
Due to the relatively infrequent incidence of MGCTB, there is little information available about treatment, and controversies concerning recommendations remain. Treatment protocols typically involve surgery alone or combination therapy of surgery, radiotherapy, and chemotherapy. Although en bloc tumor resection has been regarded as an effective therapeutic method for MGCTB that provides reduced recurrence rates, it is difficult to perform, especially in the axial skeleton. Furthermore, the therapeutic effects of chemotherapy and radiotherapy remain controversial and result in treatment dilemmas [9–11].
In order to improve the prognosis of MGCTB patients, there is a pressing need to identify the significant prognostic factors. Previous studies have reported that factors such as age, primary site, International Classification of Diseases for Oncology, presence and location of metastases, surgical strategy, and histological response to chemotherapy are relevant to the prognosis of patients [1,2,8,12]. However, the small sample sizes and single-center format limited the accuracy of these studies.
To accurately predict the prognosis of MGCTB patients, we selected patients from the Surveillance, Epidemiology, and End Results (SEER) database. Machine learning (random forest) and classic regression methods (Kaplan-Meier curve, Cox proportional hazards regression model, and Lasso regression) were used to identify independent prognostic variables, and nomograms were constructed to estimate overall survival (OS) and cause-specific survival (CSS). Moreover, a high-quality external validation from our own institution and the SEER database in 2016 and commonly used guidelines (Tumor Node Metastasis [TNM] and American Joint Committee on Cancer [AJCC] staging systems) were employed to evaluate the accuracy rate and applicability of the nomograms in clinical work.
Material and Methods
PATIENT SELECTION:
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the Ethics Committee of our institution (No. KEYAN-2018-LW-021), and informed consent was provided by all patients. Patients in the training cohort were selected from the SEER database, which contained cancer data covering around 34.6% of the US population. The SEER database includes information about patient demographics, tumor character information (primary tumor site, tumor morphology, and stage at diagnosis), treatment information, and vital status [13]. Only patients with MGCTB that was histologically diagnosed from 1973 to 2014 were included in our study. Patients were excluded if the MGCTB was not diagnosed by biopsy or if it was not their first tumor. Furthermore, patients whose race, marital status, surgery information, radiotherapy information, historical stage, and primary site were unknown were also excluded. The same criteria were applied in selecting patients from our own institution and the SEER database in 2016 to construct 2 testing cohorts.
DATA EXTRACTION:
In this study, variables from the training cohort were acquired from the SEER database on July 26, 2018, and included age at diagnosis, sex, race, primary site, radiotherapy, chemotherapy, surgery information, family income, marital status, education background, and employment status. We also extracted OS and CSS as the study endpoints.
STATISTICAL ANALYSIS:
Dichotomous variables are expressed as number (percentage), and continuous variables are presented as mean±standard deviation (SD) and median (range). Three statistical methods were applied to evaluate prognostic factors. First, the categorical variables were analyzed using the chi-square test. Second, the Kaplan-Meier method was used as an initial analysis to explore potential variables associated with OS and CSS. In order to select “different” cutoff points, we used X-tile plots to set the interception points (Supplementary Figure 1). Continuous variables were uncoupled to the new classification (age: <39 years, 39–68 years, and >68 years). In this study, random forest (ntree=500) was applied to all variables for further analysis. Mean Decrease Gini (MDG) was used to quantify the classification accuracy of each variable, with a higher MDG indicating that the grade of impurity from a category could be decreased the most by 1 variable, which suggests a significantly associated index [14].
After these programs, the statistically significant variables were selected to build the Cox proportional hazards model. The log-rank test was applied for model diagnosis. Additionally, Lasso regression was performed to ensure that the multifactor models were not overfitting. Finally, a model consisting of optimum variables was established. Based on multivariable analysis, nomograms were constructed to predict the probability of CSS and OS. The calibration, discrimination, and generalization of the nomograms were evaluated using calibration curves. The accuracy of the nomograms was then tested and compared with the TNM and AJCC staging systems.
A 2-sided
Results
PATIENT CHARACTERISTICS:
Among the 454 patients with MGCTB diagnosed from 1973 to 2014, 152 were excluded because they did not meet the inclusion criteria. In total, 302 patients were included in our training cohort. The process of data selection is shown in Figure 1. Based on the same process, the testing cohort consisted of 37 patients for external validation from our own institution, 163 patients for external validation from the SEER database in 2016, 201 patients labeled with TNM staging system, 53 patients labeled with sixth edition AJCC staging system, and 67 patients labeled with seventh edition AJCC staging system.
The patient characteristics are presented in Supplementary Table 1. The study population included 149 males and 153 females, predominantly white (77.2%), with a median age of 38.0 years (range: 1.0–91.0). The MGCTB was primarily localized or regional (83.8%). Among all MGCTB patients, the limbs accounted for the primary site in 72.5% of cases, followed by the trunk (21.9%) and the head-face-neck (5.6%). At the long-term follow-up stage, the median survival time was 75.5 months (range: 0–502.0). At the endpoint, 47 (15.6%) patients succumbed to MGCTB and 77 (25.5%) patients to all causes. The marital status, education levels, and family incomes exhibited relative hypodispersion among all patients.
UNIVARIATE ANALYSIS AND RANDOM FOREST:
The results of parametric or nonparametric tests, Kaplan-Meier survival analysis, and random forest for OS and CSS are described in Table 1. Six variables (age, primary site, radiotherapy, surgery information, chemotherapy information, and historical stage) showed statistical significance in parametric or nonparametric tests and Kaplan-Meier survival analysis (Figure 2). Moreover, these variables also ranked in the top 30% in MDG of random forest. Potential prognostic factors of these 6 variables were submitted to Cox proportional hazards analysis.
COX PROPORTIONAL HAZARDS MODEL AND LASSO REGRESSION:
The Cox proportional hazards regression model was constructed to confirm the effects of variables on the OS and CSS of patients (Table 2). The results of Lasso regression suggested that all variables incorporated into the final multivariate models were essential to modeling (Figure 3A–3D). Compared with patients younger than 39 years old, older patients had a poorer prognosis in OS (39–68 years: hazard ratio [HR], 4.933; 95% confidence interval [CI], 2.613 to 9.313; P<0.001; >68 years: HR, 20.043; 9.403 to 42.722; P<0.001), and CSS (39–68 years: HR, 3.772; 1.741 to 8.172; P<0.001; >68 years: HR, 8.733; 3.639 to 20.958; P<0.001).
In the historic stage, localized malignance (OS: HR, 0.281; 0.151 to 0.519;
Surprisingly, surgery information, which showed significant results in the survival curve (Figure 2), was not a significant prognostic indicator for either OS or CSS in the Cox proportional hazards model. Also, chemotherapy was associated with a worse OS (HR, 2.199; 1.293 to 3.739; P=0.004) and CSS (HR, 2.608; 1.373 to 4.954; P=0.003). Radiotherapy was found to be a favorable independent factor for OS (HR, 0.504; 0.276 to 0.920; P=0.026), but not for CSS. The receiver operating characteristic (ROC) curves suggested that the multivariate models had high accuracy (OS: area under the curve [AUC] of 3-year survival: 0.753; AUC of 5-year survival: 0.755; CSS: AUC of 3-year survival: 0.782; AUC of 5-year survival: 0.780) (Figure 3E, 3F).
NOMOGRAM AND VALIDATION:
Based on the Cox proportional hazards regression model, the nomograms were constructed with the training cohort (Figure 4A, 4B). The calibration plots of the cumulative incidence function are shown in detail in Figure 4C and 4D. The points further from the 45° line indicated a few inconsistencies between predictions and observations. Supplementary Table 2 shows the point assignment and prognostic score for each variable in the nomograms.
In order to verify the discrimination and practicability of the results in the nomograms, a multidimensional validation was performed. The external validation cohort contained 37 patients from our own institution, comprising 14 males and 23 females, with a median age of 31.0 years (range: 14.0–69.0) and a median survival time of 25.0 months. All patients had a histopathological diagnosis of MGCTB (Figure 5).
Data from the SEER database for patients who received a histological diagnosis of MGCTB in 2016 were used to further validate the model, which included 81 males and 82 females with a median age of 34.0 years (range: 9.0–87.0) and a median survival time of 102.0 months (range: 4.0–501.0). The 201 patients identified from the SEER database using the TNM staging system included 93 males and 108 females, with a median age of 41.0 years (range: 4.0–91.0) and a median survival time of 47.0 months (range: 0–129.0). The 53 patients identified from SEER using the sixth edition of the AJCC staging system included 25 males and 28 females, with a median age of 48.0 years (range: 16.0–91.0) and a median survival time of 26.0 months (range: 0–126.0). The 67 patients identified from SEER using the seventh edition of the AJCC staging system consisted of 31 males and 36 females, with a median age of 44.0 years (range: 17.0–91.0) and median survival time of 17.0 months (range: 0–59.0).
Compared with the current TNM staging system (0.772 [OS] and 0.837 [CSS]), and the AJCC sixth (0.624 [OS] and 0.641 [CSS]) and seventh (0.747 [OS] and 0.771 [CSS]) editions staging systems (Supplementary Figure 2), the nomograms achieved a higher C-index (internal validation: 0.836 [OS] and 0.827 [CSS]; external validation of our institution: 1.000 (OS) and 0.810 (CSS); external validation of the SEER in 2016: 0.877 (OS) and 0.894 (CSS).
Discussion
MGCTB is a rare histological type of malignant tumor with a high tendency for local relapse and distant metastasis. Considering the potentially deleterious effects of MGCTB on patients [10,11,15], evaluating the prognostic factors is useful in improving prognosis and assisting clinicians in making accurate survival evaluations and therapeutic decisions. In this study, we constructed a prognostic nomogram for patients with MGCTB based on the SEER database and validated it externally, the first of its kind. Machine learning models (chi-square test and random forest) and classic survival analysis methods (Kaplan-Meier analysis and Cox proportional hazards model) were used to explore the significant prognostic variables in patients with MGCTB. The results suggested that age, historical stage, and chemotherapy were independent prognostic variables for OS and CSS of MGCTB patients and that ICD-O-3 histology, radiotherapy, and primary site were independent prognostic variables for OS.
Similar to previous studies [1,6,16], the mean age in our study cohort was 40.7 years (median 38.0, range: 1.0 to 91.0) with an equal sex distribution. Sex, race, and marital status were not independent prognostic variables for OS and CSS. Similar results were also obtained for high school education, 9th grade education, bachelor’s degree (at least), median family income, families below the poverty line, unemployment, and white-collar employment.
Age was divided into 3 groups, with the cutoff value of young age (<39 years) (50.3%), middle age (39–68 years) (38.1%), and old age (>68 years) (11.6%). The results revealed that age was a significant prognostic factor for both CSS and OS, which was similar to previous reports [1,6,16]. Compared with the middle age group, patients in the young age group had a more favorable prognosis, and patients in the old age group had a poorer one. A realistic explanation is that older patients were more easily affected by complex therapeutic complications, which can lead to a poorer prognosis [8].
In this study, the primary site was divided into 3 categories: head-face-neck (5.6%), trunk (21.9%), and limbs (72.5%). Tumors occurring in the limbs were found to be favorable for prognosis, especially considering that a total resection and even amputation can be performed if necessary. In contrast, tumors in the trunk and head-face-neck typically involve essential nerves and vessels, which makes performing a clean resection technically challenging. Additionally, the historical stage was a statistically significant variable for OS and CSS. Compared with regional tumors (31.8%), patients with distant metastatic tumors (16.2%) had a poorer prognosis. Similarly, numerous studies have also found that distant metastasis was also related to poor prognosis [8,17,18]. It not only increased the tumor burden and damaged organ function, but also limited the application of the en bloc tumor resection [1].
Surgery is generally regarded to be the fundamental treatment option for patients with MGCTB [7,19]. In our study, surgery was found to be a significant factor in patients with MGCTB in the Kaplan-Meier survival analysis (OS,
Radiotherapy and chemotherapy are controversial treatment regimens for MGCTB [2,7,12,31], and both were demonstrated to be significant prognostic factors for patients with MGCTB in our study. Radiotherapy was found to be a favorable factor, while chemotherapy was a negative predictor. Although MGCTB was initially thought to be radiotherapy resistant and lead to radiotherapy-related malignant transformation [11,32], the efficacy and safety of radiotherapy have significantly improved in recent years [33–36]. Chemotherapy was not commonly used in MGCTB and was reserved only for advanced MGCTB that could not be cured by either surgery or radiotherapy. Thus, the prognosis of chemotherapy-applied patients with MGCTB was worse. Our subgroup analysis also demonstrated that patients who did not undergo surgical therapy preferred to receive chemotherapy (no surgery: OR, 5.944; 4.234 to 8.597;
Comprehensive nomograms were found to be useful and convenient tools to evaluate the prognosis of patients, and the nomograms developed in the current study were the first for MGCTB based on the SEER database. The nomograms were verified by internal (C-index: OS, 0.836; CSS, 0.827) and external validation (C-index: OS, 1.000; CSS, 0.810) using the database from our own institution, as well as external validation (C-index: OS, 0.877; CSS, 0.894) with additional data of patients with MGCTB from the SEER database in 2016. Moreover, the nomograms were also compared with the TNM staging system (C-index: OS, 0.772; CSS, 0.837), and the sixth (C-index: OS, 0.624; CSS, 0.641) and seventh editions of the AJCC staging system (C-index: OS, 0.747; CSS, 0.771) to verify their reliability. The C-index of our verification cohort was relatively high, although our sample size limited it. Therefore, we obtained additional data from patients with MGCTB from the SEER database in 2016 to further validate our model to reduce bias in external validation. Moreover, compared with the TNM and the AJCC (sixth and seventh editions) staging systems, the nomograms were found to exhibit higher sensitivity and specificity based on the C-index.
There were some additional limitations in our study that need to be addressed. First, although the SEER database contained a large sample size and multiple variables, it still presented some deficiencies. For example, because the SEER database contains information from multiple centers, its intergroup heterogeneity is not processed, even though we have strict inclusion and exclusion criteria to minimize this heterogeneity. Secondly, the median follow-up time of the external validation set from our own institution was not as long as the SEER database. In the future, stricter and more accurate nomograms for prediction need to be combined with genetic factors. Subsequent research should focus on the correlation between deep molecular mechanisms (eg, the newly discovered long noncoding RNA related to the prognosis of bone tumors) and the independent prognostic variables found in this study, with the assistance of weighted gene co-expression network analysis and deep learning [41].
Conclusions
Age, historical stage, and chemotherapy were independent prognostic variables for OS and CSS of MGCTB patients, and radiotherapy and primary site were independent prognostic variables for OS. Our nomograms were verified internally and externally based on significant clinicopathologic features and clinical experience and may assist clinicians in making more accurate survival evaluations in conjunction with the TNM and AJCC staging systems.
Supplementary Data
SUPPLEMENTARY MATERIALS A:
Supplementary Table 1 shows the baseline characteristics of patients with malignant giant cell tumor of bone.
Supplementary Table 2 shows the point assignment and prognostic score for each variable in nomograms for overall survival (OS) and cause-specific survival (CSS).
Supplementary Tables 3 and 4 show the analysis results of the cohort without variable “surgery information.”
Supplementary Tables 5–11 show the subgroup univariate analysis results.
SUPPLEMENTARY MATERIALS B:
The raw dataset of the training set consisted of 302 patients with malignant giant cell tumor of bone from the Surveillance, Epidemiology, and End Results database.
SUPPLEMENTARY MATERIALS C:
The raw dataset of the validated set consisted of 37 patients with malignant giant cell tumor of bone.
SUPPLEMENTARY MATERIALS D:
The raw dataset of the validated set consisted of 163 patients with malignant giant cell tumor of bone.
Figures
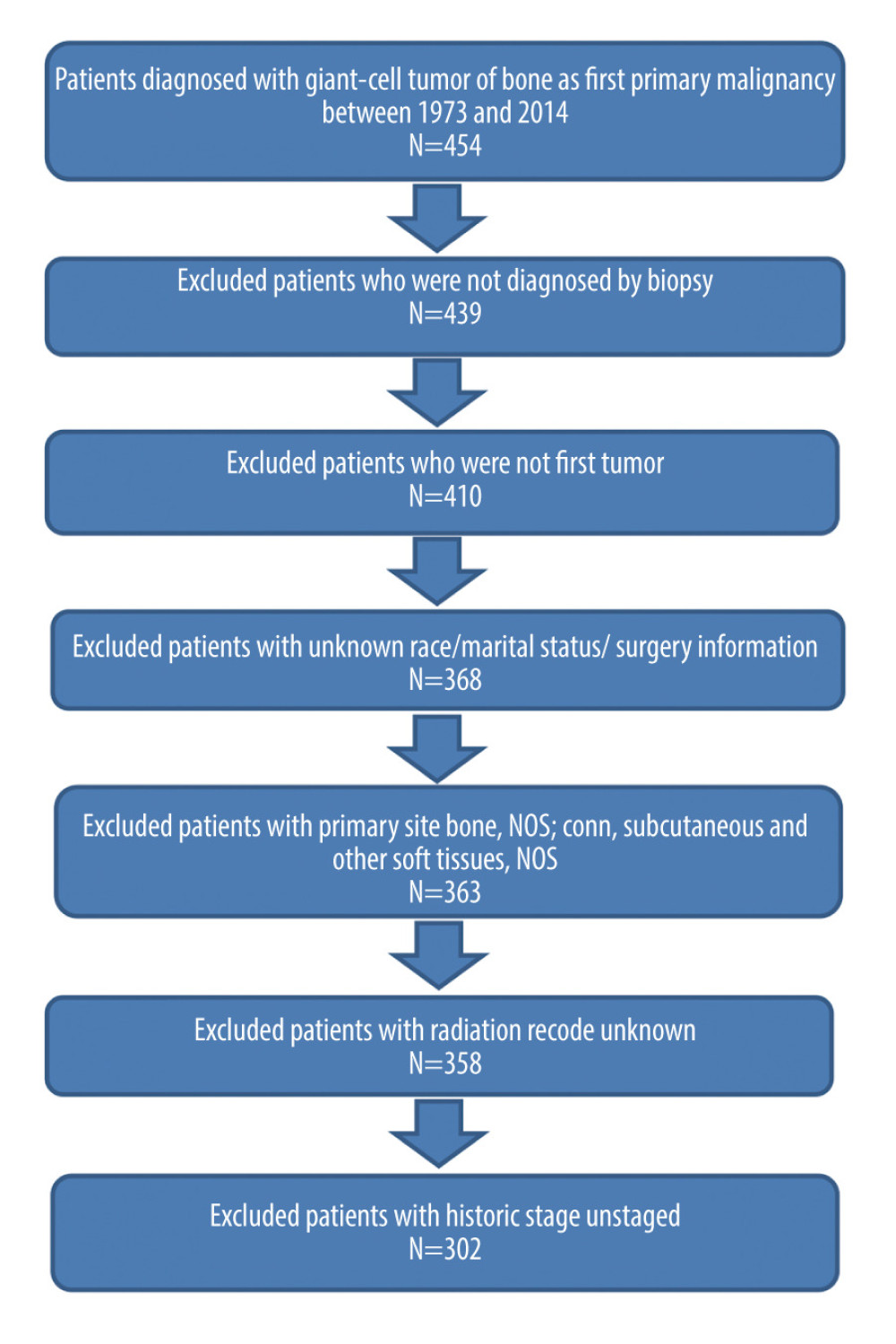 Figure 1. Flow chart showing the patient selection process from the Surveillance, Epidemiology, and End Results database.
Figure 1. Flow chart showing the patient selection process from the Surveillance, Epidemiology, and End Results database. 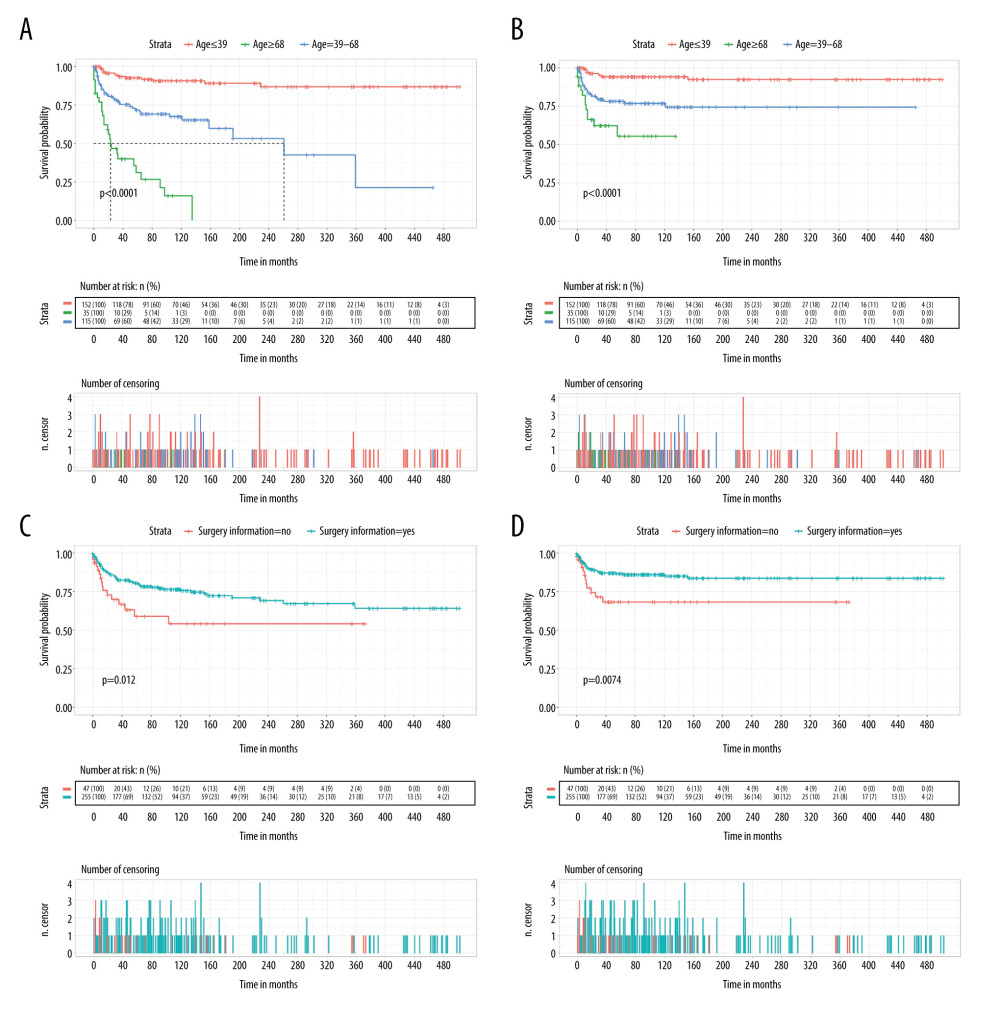 Figure 2. Survival curves of age (A, B) and surgery information (C, D) for overall survival (OS) and cause-specific survival (CSS).
Figure 2. Survival curves of age (A, B) and surgery information (C, D) for overall survival (OS) and cause-specific survival (CSS). 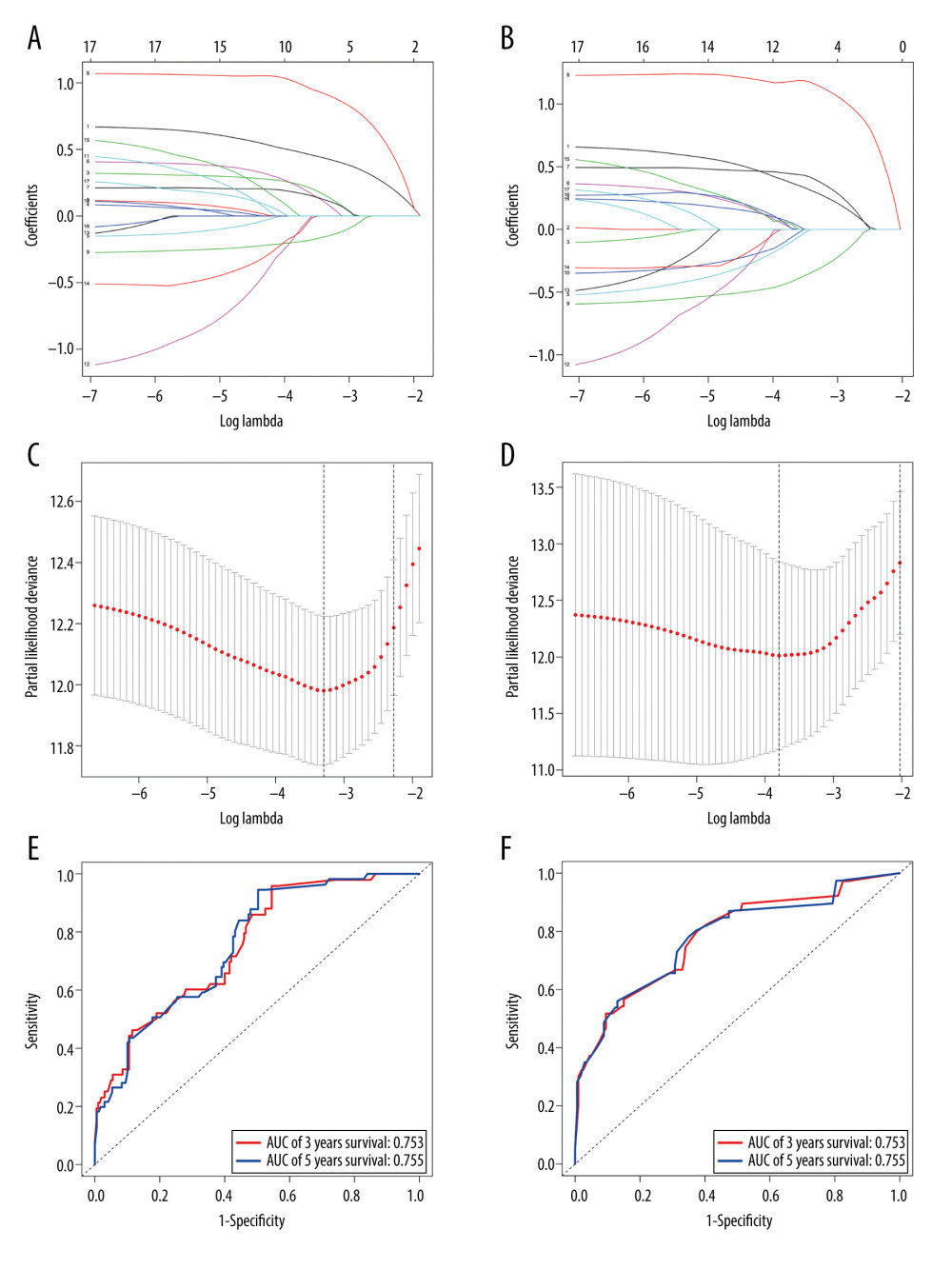 Figure 3. The results of the Lasso regression (A–D) and the receiver operating characteristic (ROC) curves (E, F). Lasso regression results suggested including 6 variables when overall survival (OS) was the endpoint (A, B), and 10 variables when cause-specific survival (CSS) (C, D) was the endpoint.
Figure 3. The results of the Lasso regression (A–D) and the receiver operating characteristic (ROC) curves (E, F). Lasso regression results suggested including 6 variables when overall survival (OS) was the endpoint (A, B), and 10 variables when cause-specific survival (CSS) (C, D) was the endpoint. 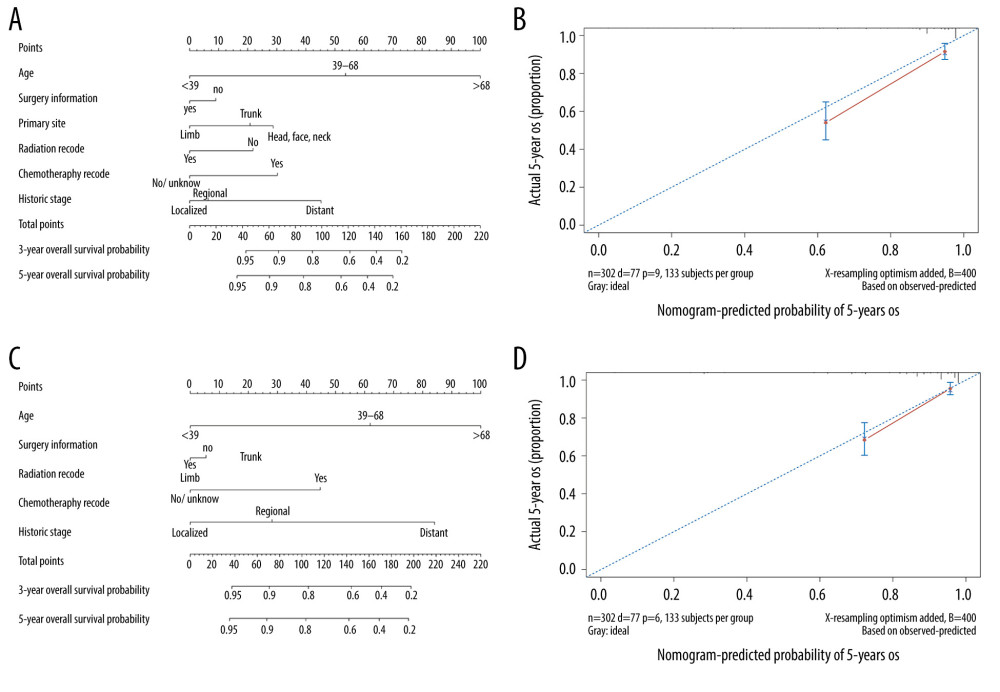 Figure 4. Nomograms (A, C) and calibration curves (B, D) of overall survival (OS) and cause-specific survival (CSS).
Figure 4. Nomograms (A, C) and calibration curves (B, D) of overall survival (OS) and cause-specific survival (CSS). 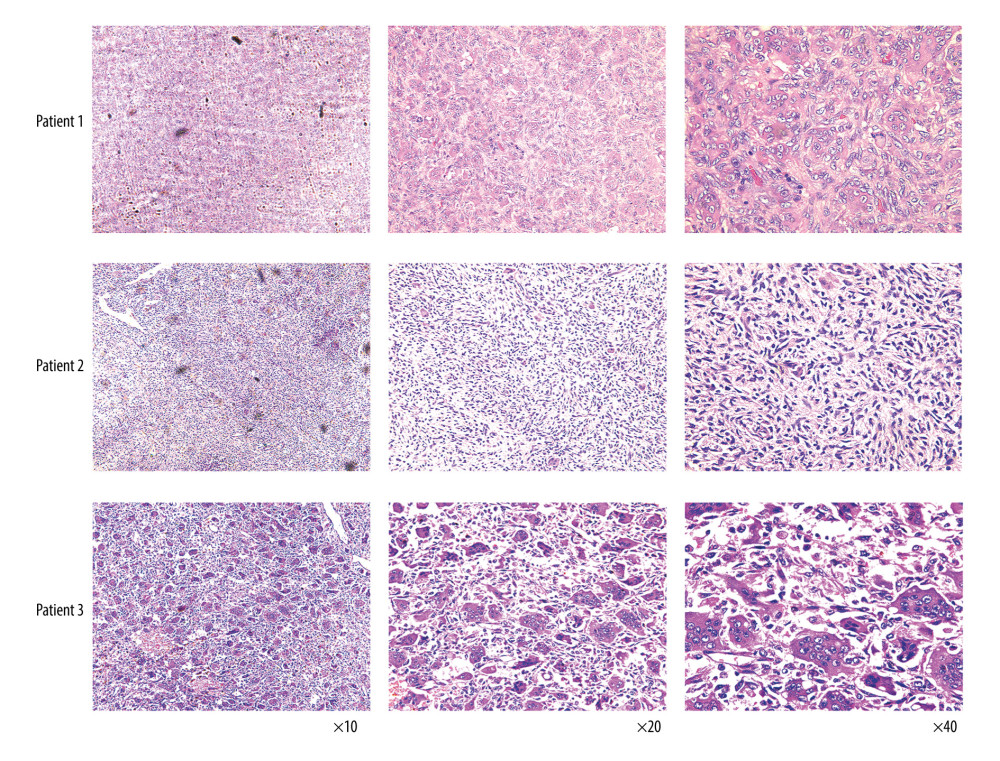 Figure 5. Hematoxylin and eosin (HE) slides and gross specimens of patients in the validation dataset with a histopathological diagnosis of malignant giant cell tumor of bone.
Figure 5. Hematoxylin and eosin (HE) slides and gross specimens of patients in the validation dataset with a histopathological diagnosis of malignant giant cell tumor of bone. References
1. Beebe-Dimmer JL, Cetin K, Fryzek JP, An analysis of data from the Surveillance, Epidemiology and End Results Program (1975–2004): Rare Tumors, 2009; 1; e52
2. Domovitov SV, Healey JH, Primary malignant giant-cell tumor of bone has high survival rate: Ann Surg Oncol, 2010; 17; 694-701
3. López-Pousa A, Martín Broto J, Garrido T, Vázquez J, Giant cell tumour of bone: New treatments in development: Clin Transl Oncol, 2015; 17; 419-30
4. Niu X, Xu H, Inwards CY, Epidemiologic comparison of 9200 patients treated at Beijing Ji Shui Tan Hospital, Beijing, China, with 10 165 patients at Mayo Clinic, Rochester, Minnesota: Arch Pathol Lab Med, 2015; 139; 1149-55
5. Raskin KA, Schwab JH, Mankin HJ, Giant cell tumor of bone: J Am Acad Orthop Surg, 2013; 21; 118-26
6. Amelio JM, Rockberg J, Hernandez RK, Population-based study of giant cell tumor of bone in Sweden (1983–2011): Cancer Epidemiol, 2016; 42; 82-89
7. Yin H, Cheng M, Li B, Treatment and outcome of malignant giant cell tumor in the spine: J Neurooncol, 2015; 124; 275-81
8. Gong L, Liu W, Sun X, Histological and clinical characteristics of malignant giant cell tumor of bone: Virchows Arch, 2012; 460; 327-34
9. Karamanakos PN, Jaaskelainen JE, Alafuzoff I, Malignant giant cell tumor in the posterior fossa of a neonate: J Neurosurg Pediatr, 2010; 5; 277-82
10. Sasagawa Y, Tachibana O, Shiraga S, Case report: Clin Neurol Neurosurg, 2012; 114; 786-88
11. Palmerini E, Picci P, Reichardt P, Downey G, Malignancy in giant cell tumor of bone: A review of the literature: Technol Cancer Res Treat, 2019; 18 1533033819840000
12. Bertoni F, Bacchini P, Staals EL, Malignancy in giant cell tumor: Skeletal Radiol, 2003; 32; 143-46
13. Howlader N, Noone AM, Krapcho M: SEER Cancer Statistics Review, 1975–2014, 2017, Bethesda, MD, National Cancer Institute Based on November 2016 SEER data submission, posted to the SEER website, April 2017. https://seer.cancer.gov/csr/1975_2014/
14. Huang ZJ, Book review. An introduction to statistical learning: With applications in R by Gareth James, Trevor Hastie, Robert Tibshirani, Daniela Witten: J Agric Biol Environ Stat, 2014; 19; 556-57
15. Leonard J, Gökden M, Kyriakos M, Case report and review of the literature: Neurosurgery, 2001; 48; 424-29
16. Rockberg J, Bach BA, Amelio J, Incidence trends in the diagnosis of giant cell tumor of bone in Sweden since 1958: J Bone Joint Surg Am, 2015; 97; 1756-66
17. Murshed KA, Elsayed AM, Szabados L, Locally aggressive giant cell tumor of bone with pulmonary distant metastasis and extrapulmonary seeding in pregnancy: J Am Acad Orthop Surg Glob Res Rev, 2020; 4(1); e19.00161
18. Kito M, Matusmoto S, Ae K, Clinical outcome prior to the introduction of molecular target therapy: Jpn J Clin Oncol, 2017; 47; 529-34
19. Montgomery C, Couch C, Emory CL, Nicholas R, Giant cell tumor of bone: Review of current literature, evaluation, and treatment options: J Knee Surg, 2019; 32; 331-36
20. Balke M, Streitbuerger A, Budny T, Treatment and outcome of giant cell tumors of the pelvis: Acta Orthop, 2009; 80; 590-96
21. Zhao Y, Tang X, Yan T, Risk factors for the local recurrence of giant cell tumours of the sacrum treated with nerve-sparing surgery: Bone Joint J, 2020; 102-B(10); 1392-98
22. Li D, Zhang J, Li Y, Surgery methods and soft tissue extension are the potential risk factors of local recurrence in giant cell tumor of bone: World J Surg Oncol, 2016; 14; 114
23. Luo W, Huang L, Liu H, Application of 3-dimensional printing technology in surgical design: Med Sci Monit, 2017; 23; 1691-700
24. Melican MC, Zimmerman MC, Dhillon MS, Three-dimensional printing and porous metallic surfaces: A new orthopedic application: J Biomed Mater Res, 2001; 55; 194-202
25. Ji T, Yang Y, Wang Y, Combining of serial embolization and denosumab for large sacropelvic giant cell tumor: Case report of 3 cases: Medicine (Baltimore), 2017; 96; e7799
26. Thomas D, Henshaw R, Skubitz K, An open-label, phase 2 study: Lancet Oncol, 2010; 11; 275-80
27. Freeman JL, Oushy S, Schowinsky J, A systematic review, meta-analysis, and case illustration: World Neurosurg, 2016; 96; 47-57
28. Griffin AM, Ferguson PC, Catton CN, Long-term outcome of the treatment of high-risk tenosynovial giant cell tumor/pigmented villonodular synovitis with radiotherapy and surgery: Cancer, 2012; 118; 4901-9
29. Yin H, Zhou W, Meng J, A retrospective analysis of 98 consecutive patients in a single center: Ann Surg Oncol, 2014; 21; 3572-78
30. Rao G, Suki D, Chakrabarti I, Surgical management of primary and metastatic sarcoma of the mobile spine: J Neurosurg Spine, 2008; 9; 120-28
31. Amanatullah DF, Clark TR, Lopez MJ, Giant cell tumor of bone: Orthopedics, 2014; 37; 112-20
32. Mahadevan A, Miksad R, Goldstein M, Induction gemcitabine and stereotactic body radiotherapy for locally advanced nonmetastatic pancreas cancer: Int J Radiat Oncol Biol Phys, 2011; 81; e615-22
33. Kishima H, Miyao Y, Shimizu K, Radiosensitive giant cell tumour of the sphenoid bone: Br J Neurosurg, 2001; 15; 171-74
34. Miszczyk L, Wydmański J, Spindel J, Efficacy of radiotherapy for giant cell tumor of bone: Given either postoperatively or as sole treatment: Int J Radiat Oncol Biol Phys, 2001; 49; 1239-42
35. Feigenberg SJ, Marcus RB, Zlotecki RA, Radiation therapy for giant cell tumors of bone: Clin Orthop Relat Res, 2003(411); 207-16
36. Skubitz KM, Giant cell tumor of bone: Current treatment options: Curr Treat Options Oncol, 2014; 15; 507-18
37. Lipplaa A, Dijkstra S, Gelderblom H, Challenges of denosumab in giant cell tumor of bone, and other giant cell-rich tumors of bone: Curr Opin Oncol, 2019; 31; 329-35
38. van der Heijden L, Dijkstra PDS, Blay JY, Gelderblom H, Giant cell tumour of bone in the denosumab era: Eur J Cancer, 2017; 77; 75-83
39. Lin P, Lin N, Teng W, A report of 10 cases: Orthop Surg, 2018; 10; 107-14
40. Ma Y, Li J, Pan J, Treatment options and prognosis for repeatedly recurrent giant cell tumor of the spine: Eur Spine J, 2016; 25; 4033-42
41. Zhang T, Wan CY, Mei XL, Long non-coding RNA HULC promotes progression of bone neoplasms: Med Sci Monit, 2018; 24; 5754-60
Figures
 Figure 1. Flow chart showing the patient selection process from the Surveillance, Epidemiology, and End Results database.
Figure 1. Flow chart showing the patient selection process from the Surveillance, Epidemiology, and End Results database. Figure 2. Survival curves of age (A, B) and surgery information (C, D) for overall survival (OS) and cause-specific survival (CSS).
Figure 2. Survival curves of age (A, B) and surgery information (C, D) for overall survival (OS) and cause-specific survival (CSS). Figure 3. The results of the Lasso regression (A–D) and the receiver operating characteristic (ROC) curves (E, F). Lasso regression results suggested including 6 variables when overall survival (OS) was the endpoint (A, B), and 10 variables when cause-specific survival (CSS) (C, D) was the endpoint.
Figure 3. The results of the Lasso regression (A–D) and the receiver operating characteristic (ROC) curves (E, F). Lasso regression results suggested including 6 variables when overall survival (OS) was the endpoint (A, B), and 10 variables when cause-specific survival (CSS) (C, D) was the endpoint. Figure 4. Nomograms (A, C) and calibration curves (B, D) of overall survival (OS) and cause-specific survival (CSS).
Figure 4. Nomograms (A, C) and calibration curves (B, D) of overall survival (OS) and cause-specific survival (CSS). Figure 5. Hematoxylin and eosin (HE) slides and gross specimens of patients in the validation dataset with a histopathological diagnosis of malignant giant cell tumor of bone.
Figure 5. Hematoxylin and eosin (HE) slides and gross specimens of patients in the validation dataset with a histopathological diagnosis of malignant giant cell tumor of bone. Tables
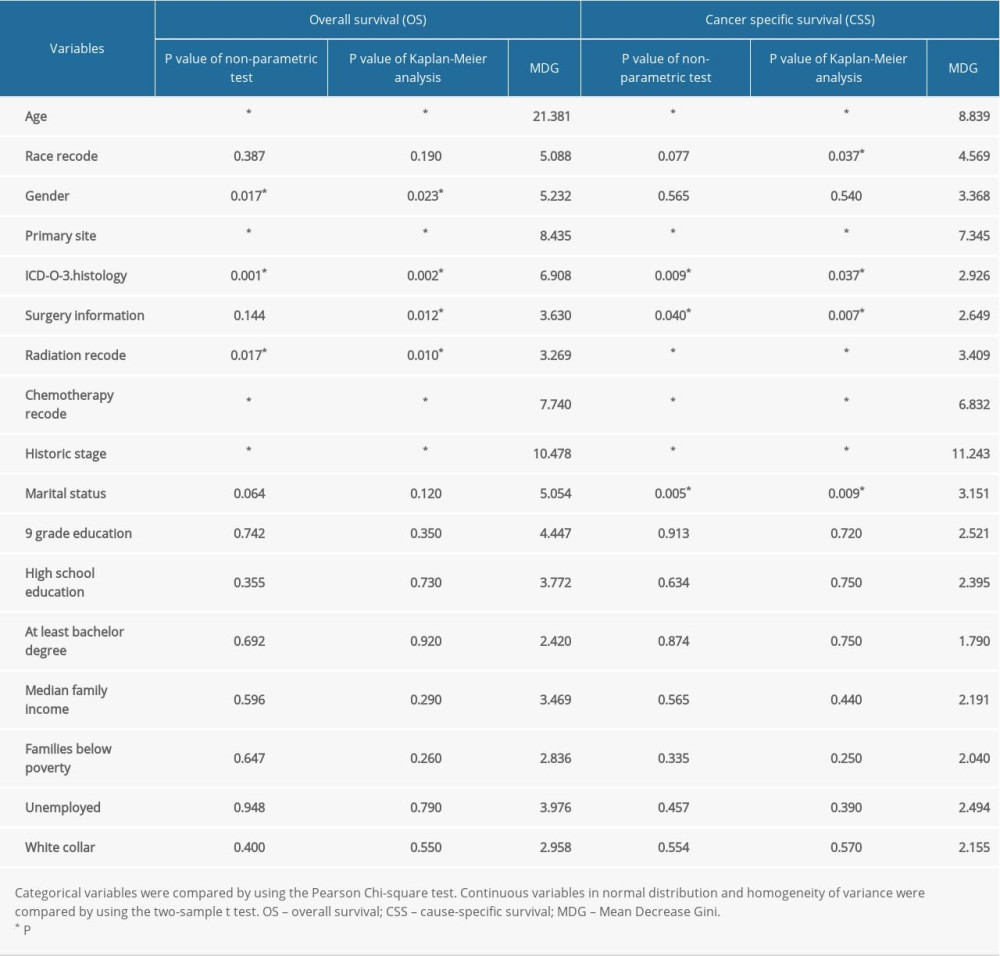 Table 1. Results of single factor analysis and random forest.
Table 1. Results of single factor analysis and random forest.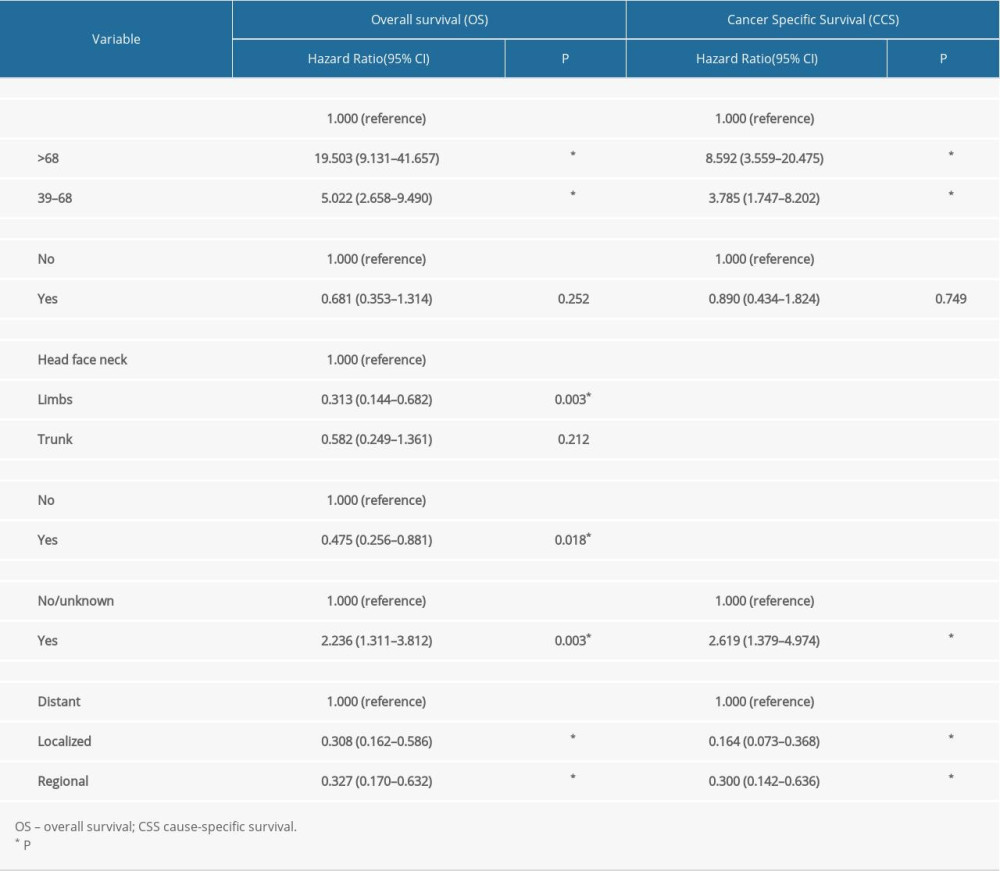 Table 2. Cox proportional hazards regression model for cancer–specific survival and overall survival in patients with malignant giant cell tumor of bone.
Table 2. Cox proportional hazards regression model for cancer–specific survival and overall survival in patients with malignant giant cell tumor of bone. Table 1. Results of single factor analysis and random forest.
Table 1. Results of single factor analysis and random forest. Table 2. Cox proportional hazards regression model for cancer–specific survival and overall survival in patients with malignant giant cell tumor of bone.
Table 2. Cox proportional hazards regression model for cancer–specific survival and overall survival in patients with malignant giant cell tumor of bone. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








