20 August 2021: Clinical Research
Association of Plasma Pro-Brain-Derived Neurotrophic Factor (proBDNF)/Mature Brain-Derived Neurotrophic Factor (mBDNF) Levels with Gene Val66Met Polymorphism in Alcohol Dependence
Min Mo1BCDEG, Xi-yue Fu2BCDF, Xu-lan Zhang1BD, Shao-chuan Zhang1BD, Hai-qing Zhang1CD, Li Wu1AF, Jia-lei Li1AF, Li Zhou1ACEFG*DOI: 10.12659/MSM.930421
Med Sci Monit 2021; 27:e930421
Abstract
BACKGROUND: In a previous study, we reported that pro-brain-derived neurotrophic factor (proBDNF) was involved in the pathology of alcohol dependence, and the single-nucleotide polymorphism (SNP) Val66Met was located at the prodomain of the brain-derived neurotrophic factor gene (BDNF). This polymorphism has been reported to affect intracellular trafficking and activity-dependent secretion of BDNF. Our present research investigated the relationships between the BDNF Val66Met polymorphism and the plasma levels of proBDNF and mature brain-derived neurotrophic factor (mBDNF) in patients with alcohol dependence.
MATERIAL AND METHODS: The BDNF gene Val66Met polymorphism was genotyped in 59 alcohol-dependent patients and 37 age- and sex-matched controls, and the plasma levels of proBDNF and mBDNF were assessed by enzyme-linked immunosorbent assay in all participants.
RESULTS: No association was found between the BDNF gene Val66Met polymorphism and alcohol dependence (P>0.05). In comparison with the control group, the level of plasma proBDNF in the alcohol-dependence group was notably increased (Z=-2.228, P=0.026), while the level of mBDNF was remarkedly decreased (Z=-2.014, P=0.044). In the alcohol-dependence group, significant associations were not found between the Val66Met polymorphisms and proBDNF and mBDNF plasma levels (P>0.05). The plasma level of proBDNF was positively correlated with the average daily alcohol consumption in the last month (r=0.344, P=0.008) and drinking history (r=0.317, P=0.014), while the plasma level of mBDNF had negative effects (r=-0.361, P=0.005, with the average daily alcohol consumption; r=-0.427, P=0.001, with drinking history).
CONCLUSIONS: The BDNF gene Val66Met polymorphism does not appear to affect the secretion of proBDNF and mBDNF in Chinese patients with alcohol dependence. Furthermore, this study reconfirmed that the plasma levels of proBDNF and mBDNF were correlated with the average daily alcohol consumption in the last month and with drinking history.
Keywords: Alcohol Abstinence, Brain-Derived Neurotrophic Factor, Polymorphism, Single Nucleotide, Protein Precursors, Alcoholism, Alleles, Amino Acid Substitution, Case-Control Studies, Disease Susceptibility, Enzyme-Linked Immunosorbent Assay, Genotype, Linkage Disequilibrium, young adult
Background
Alcohol is widely perceived as a neurotoxic agent that can harm physical and mental health and lead to serious damage to social function, professional function, and social adaptability. The effect of ethanol on the central nervous system is ebriety. The degree of ebriety caused by long-term alcohol consumption gradually declines, which is called tolerance. Individuals who continue to drink are considered to have alcohol dependence. Chronic and heavy alcohol exposure causes neurodegeneration and reduces the regeneration of neurons.
Brain-derived neurotrophic factor (BDNF) has an important role in the survival, growth, differentiation, and development of neurons, and it also participates in the development of substance addiction [1]. The single-nucleotide polymorphism (SNP) rs6265 is the substitution of valine for methionine (Val66Met) at the 66th codon of the
Material and Methods
STUDY PARTICIPANTS:
Study approval was granted by the Ethics Committee of Yunnan Mental Hospital Institution. Fifty-nine alcohol-dependent patients who were hospitalized in Yunnan Mental Hospital between September and November 2019 were recruited. Three senior psychiatrists used the Chinese version of the Mini International Neuropsychiatric Interview and the scale of alcohol use disorders identification test (AUDIT) to confirm that the patients met the diagnosis of alcohol dependence according to the International Statistical Classification of Diseases and Related Health Problems 10th Revision (ICD-10) criteria. Individuals with comorbidity with other non-nicotine substance abuse or dependence or with psychiatric disorders were excluded.
The control group consisted of 37 healthy subjects who underwent routine physical examinations at the Yunnan Psychiatric Hospital during the same period. Individuals were excluded from the control group if they met the diagnostic criteria for alcohol abuse or dependence in the past or if their average daily alcohol consumption was more than 100 g during the previous 3 months. Individuals were also excluded if they met the diagnostic criteria for schizophrenia or schizoaffective disorder or for a mood disorder according to ICD-10.
All participants were from the same city (Kunming City, Yunnan Province), and demographic information was matched between the alcohol-dependence group and the control group.
BDNF VAL66MET POLYMORPHISM GENOTYPING:
The next morning after hospitalization, 2–5 mL of peripheral blood was collected into anticoagulant tubes, and 200 μL of blood was used for genomic DNA extraction by using a Blood Genomic DNA Extraction Kit (Tiangen Biochemical Technology, Beijing, cat. no. DP348). The following primers were used:
The PCR conditions were 95°C for 5 min; 34 cycles of 95°C for 30 s, 60°C for 30 s, and 72°C for 30 s; and 72°C for 10 min. ABI7500 was conducted for genotyping. FinchTV (
DETERMINATION OF PROBDNF AND MBDNF CONCENTRATIONS WITH ELISA:
The blood samples were centrifuged at 1500 g for 10 min, and the plasma was frozen at −80°C for later analysis. The ELISA process was as follows [7,8]. The capture antibody (2 μg/mL, protein G-purified mouse anti-mBDNF monoclonal antibody, B34D10) was diluted in the coating buffer (50 mM carbonate), and 100 μL of capture antibody was added immediately to each well of the microplate. The microplate was incubated at 37°C for 1 h. The plate was then washed with phosphate-buffered saline 3 times. Next, 150 μL of blocking buffer (phosphate-buffered saline and bovine serum albumin) was added to each well, and the plate was again incubated for 1 h at 37°C. The standard mBDNF was diluted in a range from 2 to 0.125 ng/mL. One hundred microliters of diluted plasma was put into each well, and the plate was incubated at 37°C for 1 h. Then, the plate was washed with wash buffer 4 times. The next steps were to dilute the detecting antibody to 2.5 μg/mL in sample diluent and to add 100 μL to each well. The plate was incubated at 37°C for 1 h once more. The plate was washed 4 times with wash buffer, and the previous step was then repeated. Next, 100 μL of freshly prepared 3,3′,5,5′-tetramethylbenzidine substrate was added to each well, and color was allowed to develop for 10 to 15 min. Finally, 1 N sulfuric acid was used to stop the color reaction. The absorbance at 450 nm was measured with a microplate reader (Model Sunrise, TECAN, Germany) to determine the BDNF concentration according to the standard curve. To determine the specificity of the kit, mBDNF, proBDNF, nerve growth factor (NGF), neurotrophin 3 (NT-3), and neurotrophin 4 (NT-4) at 1 ng/mL or 0 to 20 ng/mL were added in the incubation step. The plasma level of proBDNF was determined by DuoSet human ELISA Development System (R&D, cat. no. DY3175) according to the manufacturer’s instructions.
STATISTICAL ANALYSIS:
SPSS version 19.0 (IBM, USA) was used for data analysis. The Hardy-Weinberg equilibrium was evaluated by chi-square test. The AUDIT scores and drinking life were expressed as mean±standard error (SE). The data for age, average ethanol consumption, and mBDNF and proBDNF ELISA results were expressed as median (M) plus the interquartile range (IQR [P25, P75]), and the Mann-Whitney
Results
DEMOGRAPHIC AND CLINICAL INFORMATION:
A total of 59 patients with alcohol dependence were included in the study. The patients’ median age was 44 (37, 49) years and ranged from 23 to 60 years old; the AUDIT score was 29.64±4.11 points (mean±SE); average alcohol consumption last month was converted to a median pure alcohol concentration of 500 (350, 600) g; duration of drinking alcohol was 20.695±1.043 years (mean±SE). The 37 cases in the control group had a median age of 38 (30, 49.5) years, ranging from 23 to 59 years. There was no statistically significant difference in age distribution between the 2 groups (
GENOTYPING AND LINKAGE DISEQUILIBRIUM EVALUATION:
There were no differences in the genotype distribution or allele frequencies of rs6265 between the patients and controls (
ASSOCIATION BETWEEN BDNF GENE RS6265 AND SUSCEPTIBILITY TO ALCOHOL DEPENDENCE:
Table 1 provides the allele and genotype frequencies of BDNF SNP (rs6265) in the alcohol-dependent patients and controls. There were no statistically significant differences between the 2 groups in genotype distributions of rs6265 (P>0.05).
PLASMA LEVELS OF PROBDNF AND MBDNF IN PATIENTS COMPARED WITH CONTROLS:
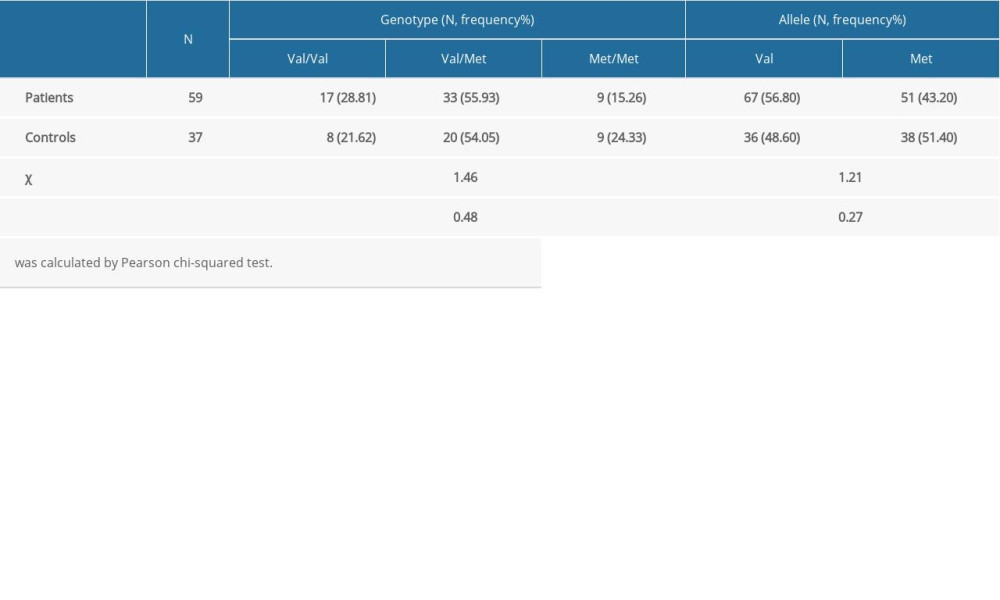
The median plasma level of proBDNF in the alcohol-dependent patients was 508.57 (378.97, 687.87) pg/mL, and the median plasma level of proBDNF in the controls was 404.06 (319.87, 597.54) pg/mL. The plasma level of proBDNF in the alcohol-dependent patients was remarkably higher than in the control participants (Z=−2.228, P=0.026, Mann-Whitney U test, Figure 1A). The median plasma level of mBDNF in the alcohol-dependent patients was 241.93 (155.67, 416.54) ng/mL, and the median plasma level of mBDNF in the controls was 295.67 (216.50, 530.31) ng/mL. The plasma level of mBDNF in the alcohol-dependent patients was significantly lower than in the control participants (Z=−2.014, P=0.044, Mann-Whitney U test, Figure 1B).
GENOTYPES OF BDNF GENE RS6265 AND PLASMA PROBDNF AND MBDNF LEVELS IN ALCOHOL-DEPENDENT PATIENTS:
Figure 2A and 2B illustrate the comparisons among different genotypes of BDNF gene rs6265 and plasma proBDNF and mBDNF levels in alcohol-dependent patients. There were 17 carriers of the GG genotype, 33 carriers of the AG genotype, and 9 carriers of the AA genotype. No significant differences were found among different genotypes of BDNF gene rs6265 and the proBDNF level (F2,56=1.056, P=0.3548, Figure 2A) and mBDNF level (F2,56=0.229, P=0.796, Figure 2B) in alcohol-dependent patients (1-way ANOVA test).
PLASMA PROBDNF AND MBDNF LEVELS AND THE AVERAGE DAILY ALCOHOL CONSUMPTION IN THE LAST MONTH IN ALCOHOL-DEPENDENT PATIENTS:
The plasma proBDNF level was directly correlated to the average daily alcohol consumption in the past month in the alcohol-dependent patients, as shown in Figure 3A (r=0.344, P=0.008, Spearman’s correlation test). The plasma mBDNF level was negatively correlated with the average daily alcohol consumption in the past month (r=−0.361, P=0.005, Spearman’s correlation test; Figure 3B).
PLASMA PROBDNF AND MBDNF LEVELS AND DRINKING HISTORY IN THE ALCOHOL-DEPENDENT PATIENTS:
The plasma proBDNF level in the alcohol-dependent patients was positively correlated with drinking history (r=0.317, P=0.014, Spearman’s correlation test; Figure 4A), and the plasma mBDNF level was negatively correlated with the drinking history (r=−0.427, P=0.001, Spearman’s correlation test; Figure 4B).
Discussion
LIMITATIONS:
The sample size of this study was small, and only 1 polymorphism of the
Conclusions
In summary, the correlation between the rs6265 polymorphism of the
Figures
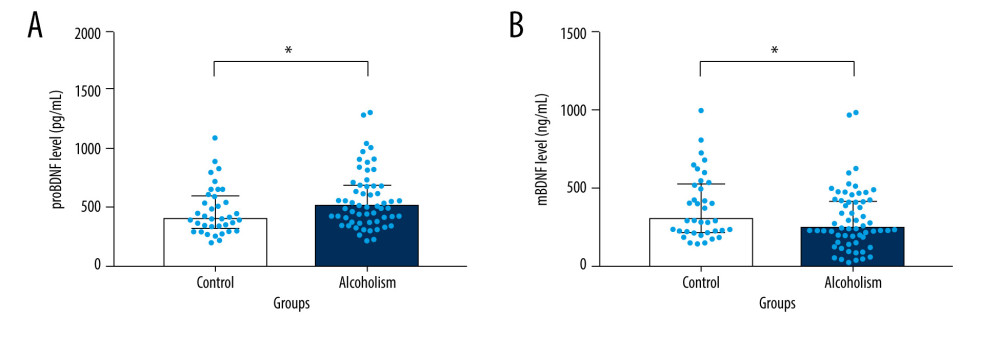 Figure 1. The plasma level of proBDNF was increased and mBDNF was decreased in the patients with alcohol dependence compared with controls. (A) The plasma level of proBDNF in the patients was significantly higher than in the controls (Z=−2.228, P=0.026, Mann-Whitney U test). (B) The plasma level of mBDNF in the patients was significantly reduced compared with the controls (Z=−2.014, P=0.044, Mann-Whitney U test). mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor.
Figure 1. The plasma level of proBDNF was increased and mBDNF was decreased in the patients with alcohol dependence compared with controls. (A) The plasma level of proBDNF in the patients was significantly higher than in the controls (Z=−2.228, P=0.026, Mann-Whitney U test). (B) The plasma level of mBDNF in the patients was significantly reduced compared with the controls (Z=−2.014, P=0.044, Mann-Whitney U test). mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor. 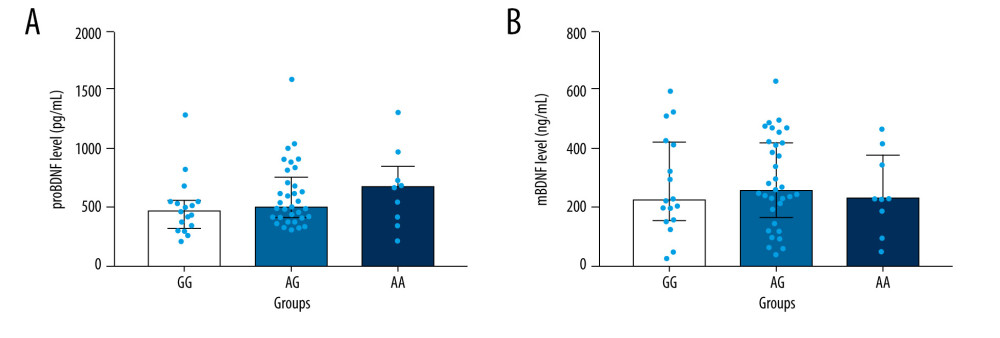 Figure 2. Comparisons between BDNF gene rs6265 and plasma proBDNF and mBDNF levels in alcohol-dependent patients. No significant differences were found between BDNF gene rs6265 and proBDNF level (A) or mBDNF level (B) in alcohol-dependent patients (One-way ANOVA, P>0.05). mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor.
Figure 2. Comparisons between BDNF gene rs6265 and plasma proBDNF and mBDNF levels in alcohol-dependent patients. No significant differences were found between BDNF gene rs6265 and proBDNF level (A) or mBDNF level (B) in alcohol-dependent patients (One-way ANOVA, P>0.05). mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor. 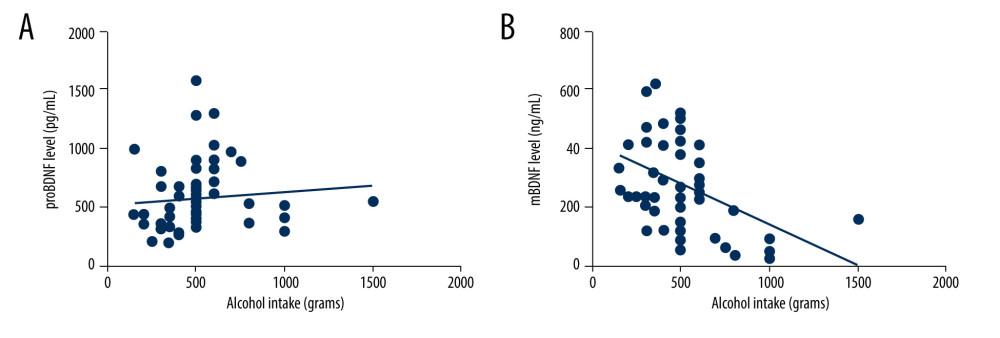 Figure 3. Correlations of plasma proBDNF and mBDNF levels with average daily ethanol consumption in the patients with alcohol dependence. Statistical analysis was performed with Spearman’s correlation test. (A) proBDNF (r=0.344, P=0.008) had a significant positive correlation with average daily ethanol consumption. (B) mBDNF (r=−0.361, P=0.005) had a significant negative correlation with total alcohol consumption. mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor.
Figure 3. Correlations of plasma proBDNF and mBDNF levels with average daily ethanol consumption in the patients with alcohol dependence. Statistical analysis was performed with Spearman’s correlation test. (A) proBDNF (r=0.344, P=0.008) had a significant positive correlation with average daily ethanol consumption. (B) mBDNF (r=−0.361, P=0.005) had a significant negative correlation with total alcohol consumption. mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor. 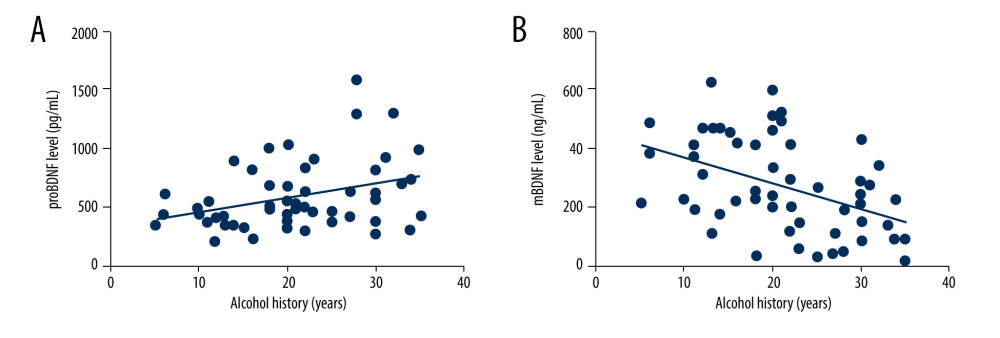 Figure 4. Correlations of plasma proBDNF and mBDNF levels with alcohol drinking history. Statistical analysis was performed with Spearman’s correlation test. (A) proBDNF (r=0.317, P=0.014) had a significant positive correlation with average daily ethanol consumption. (B) mBDNF (r=−0.427, P=0.001) had a significant negative correlation with total alcohol consumption. mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor.
Figure 4. Correlations of plasma proBDNF and mBDNF levels with alcohol drinking history. Statistical analysis was performed with Spearman’s correlation test. (A) proBDNF (r=0.317, P=0.014) had a significant positive correlation with average daily ethanol consumption. (B) mBDNF (r=−0.427, P=0.001) had a significant negative correlation with total alcohol consumption. mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor. References
1. Cheng M, Liu Q, Wang Y, MMP-9-BDNF pathway is implicated in cognitive impairment of male individuals with methamphetamine addiction during early withdrawal: Behav Brain Res, 2019; 366; 29-35
2. Chen ZY, Patel PD, Sant G, Variant brain-derived neurotrophic factor (BDNF) (Met66) alters the intracellular trafficking and activity-dependent secretion of wild-type BDNF in neurosecretory cells and cortical neurons: J Neurosci, 2004; 24(18); 4401-11
3. Bird CW, Baculis BC, Mayfield JJ, The BDNF val68met polymorphism modulates how developmental ethanol exposure impacts the hippocampus: Genes Brain Behav, 2018; 18(3); e12484
4. Warnault V, Darcq E, Morisot N, The BDNF Val68 to Met polymorphism increases compulsive alcohol drinking in mice that is reversed by Tropomyosin Receptor Kinase B activation: Biol Psychiatry, 2016; 79(6); 463-73
5. Zhou L, Xiong J, Ruan CS, ProBDNF/p75NTR/sortilin pathway is activated in peripheral blood of patients with alcohol dependence: Transl Psychiatry, 2018; 7(11); 2
6. Lu B, Pang PT, Woo NH, The yin and yang of neurotrophin action: Nat Rev Neurosci, 2005; 6(8); 603-14
7. Zhou L, Xiong J, Lim Y, Upregulation of blood proBDNF and its receptors in major depression: J Affect Disord, 2013; 150(3); 776-84
8. Lim Y, Zhong JH, Zhou XF, Development of mature BDNF-specific sandwich ELISA: J Neurochem, 2015; 134(1); 75-85
9. Beckley JT, Laguesse S, Phamluong K, The first alcohol drink triggers mTORC1-dependent synaptic plasticity in nucleus accumbens dopamine D1 receptor neurons: J Neurosci, 2016; 36(3); 701-13
10. Koob GF, Volkow ND, Neurocircuitry of addiction: Neuropsychopharmacology, 2010; 35(1); 217-38
11. Reichardt LF, Neurotrophin-regulated signalling pathways: Philos Trans R Soc Lond B Biol Sci, 2006; 361(1473); 1545-64
12. Baydyuk M, Xu B, BDNF signaling and survival of striatal neurons: Front Cell Neurosci, 2014; 8; 254
13. Gonzalez A, Moya-Alvarado G, Gonzalez-Billaut C, Bronfman FC, Cellular and molecular mechanisms regulating neuronal growth by brain-derived neurotrophic factor: Cytoskeleton (Hoboken), 2016; 73(10); 612-28
14. Egan MF, Kojima M, Callicott JH, The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function: Cell, 2003; 112(2); 257-69
15. Notaras M, Hill R, van de Buuse M, The BDNF gene Val66Met polymorphism as a modifier of psychiatric disorder susceptibility: Progress and controversy: Mol Psychiatry, 2015; 20(8); 916
16. Hensler JG, Ladenheim EE, Lyons WE, Ethanol consumption and serotonin-1A (5-HT1A) receptor function in heterozygous BDNF (+/−) mice: J Neurochem, 2003; 85; 1139-47
17. Tsai SJ, Liao DL, Yu YW, A study of the association of (Val66Met) polymorphism in the brain-derived neurotrophic factor gene with alcohol dependence and extreme violence in Chinese males: Neurosci Lett, 2005; 381(3); 340-43
18. Cheah SY, Lawford BR, Young RM, BDNF SNPs are implicated in comorbid alcohol dependence in schizophrenia but not in alcohol-dependent patients without schizophrenia: Alcohol Alcohol, 2014; 49(5); 491-97
19. Benzerouk F, Gierski F, Gorwood P, Brain-derived neurotrophic factor (BDNF) Val66Met polymorphism and its implication in executive functions in adult offspring of alcohol-dependent probands: Alcohol, 2013; 47(4); 271-74
20. Shin S, Stewart R, Ferri CP, An investigation of associations between alcohol use disorder and polymorphisms on ALDH2, BDNF, 5-HTTLPR, and MTHFR genes in older Korean men: Int J Geriatr Psychiatry, 2010; 25(5); 441-48
21. Klimkiewicz A, Mach A, Jakubczyk A, COMT and BDNF gene variants help to predict alcohol consumption in alcohol-dependent patients: J Addict Med, 2017; 11(2); 114-18
22. Lopez-Leon S, González-Giraldo Y, Wegman-Ostrosky T, Forero DA, Molecular genetics of substance use disorders: An umbrella review: Neurosci Biobehav Rev, 2021; 4(124); 358-69
23. Haerian BS, BDNF rs6265 polymorphism and drug addiction: A systematic review and meta-analysis: Pharmacogenomics, 2013; 14(16); 2055-65
24. Gerenu G, Martisova E, Ferrero H, Modulation of BDNF cleavage by plasminogen-activator inhibitor-1 contributes to Alzheimer’s neuropathology and cognitive deficits: Biochim Biophys Acta Mol Basis Dis, 2017; 1863(4); 991-1001
25. Ozsoy S, Durak AC, Esel E, Hippocampal volumes and cognitive functions in adult alcoholic patients with adolescent-onset: Alcohol, 2013; 47; 9-14
26. Trantham-Davidson H, Burnett EJ, Gass JT, Chronic alcohol disrupts dopamine receptor activity and the cognitive function of the medial prefrontal cortex: J Neurosci, 2014; 34(10); 3706
Figures
 Figure 1. The plasma level of proBDNF was increased and mBDNF was decreased in the patients with alcohol dependence compared with controls. (A) The plasma level of proBDNF in the patients was significantly higher than in the controls (Z=−2.228, P=0.026, Mann-Whitney U test). (B) The plasma level of mBDNF in the patients was significantly reduced compared with the controls (Z=−2.014, P=0.044, Mann-Whitney U test). mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor.
Figure 1. The plasma level of proBDNF was increased and mBDNF was decreased in the patients with alcohol dependence compared with controls. (A) The plasma level of proBDNF in the patients was significantly higher than in the controls (Z=−2.228, P=0.026, Mann-Whitney U test). (B) The plasma level of mBDNF in the patients was significantly reduced compared with the controls (Z=−2.014, P=0.044, Mann-Whitney U test). mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor. Figure 2. Comparisons between BDNF gene rs6265 and plasma proBDNF and mBDNF levels in alcohol-dependent patients. No significant differences were found between BDNF gene rs6265 and proBDNF level (A) or mBDNF level (B) in alcohol-dependent patients (One-way ANOVA, P>0.05). mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor.
Figure 2. Comparisons between BDNF gene rs6265 and plasma proBDNF and mBDNF levels in alcohol-dependent patients. No significant differences were found between BDNF gene rs6265 and proBDNF level (A) or mBDNF level (B) in alcohol-dependent patients (One-way ANOVA, P>0.05). mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor. Figure 3. Correlations of plasma proBDNF and mBDNF levels with average daily ethanol consumption in the patients with alcohol dependence. Statistical analysis was performed with Spearman’s correlation test. (A) proBDNF (r=0.344, P=0.008) had a significant positive correlation with average daily ethanol consumption. (B) mBDNF (r=−0.361, P=0.005) had a significant negative correlation with total alcohol consumption. mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor.
Figure 3. Correlations of plasma proBDNF and mBDNF levels with average daily ethanol consumption in the patients with alcohol dependence. Statistical analysis was performed with Spearman’s correlation test. (A) proBDNF (r=0.344, P=0.008) had a significant positive correlation with average daily ethanol consumption. (B) mBDNF (r=−0.361, P=0.005) had a significant negative correlation with total alcohol consumption. mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor. Figure 4. Correlations of plasma proBDNF and mBDNF levels with alcohol drinking history. Statistical analysis was performed with Spearman’s correlation test. (A) proBDNF (r=0.317, P=0.014) had a significant positive correlation with average daily ethanol consumption. (B) mBDNF (r=−0.427, P=0.001) had a significant negative correlation with total alcohol consumption. mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor.
Figure 4. Correlations of plasma proBDNF and mBDNF levels with alcohol drinking history. Statistical analysis was performed with Spearman’s correlation test. (A) proBDNF (r=0.317, P=0.014) had a significant positive correlation with average daily ethanol consumption. (B) mBDNF (r=−0.427, P=0.001) had a significant negative correlation with total alcohol consumption. mBDNF – mature brain-derived neurotrophic factor; proBDNF – pro-brain-derived neurotrophic factor. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387