16 August 2021: Meta-Analysis
Prognostic Significance of Microvascular Invasion in Pancreatic Ductal Adenocarcinoma: A Systematic Review and Meta-Analysis
Huangbao Li1ACEF, Weiwei Pan2AEF, Liu Xu1BE, Dong Yin3CDE, Shuqun Cheng34AEFG*, Fengqing Zhao5AEFDOI: 10.12659/MSM.930545
Med Sci Monit 2021; 27:e930545
Abstract
BACKGROUND: The incidence, pathogenesis, and prognostic effect of microvascular invasion on pancreatic ductal adenocarcinoma (PDAC) remain controversial. This study aimed to summarize the incidence, pathogenesis, role in clinical management, recurrence, and prognostic significance of microvascular invasion in PDAC.
MATERIAL AND METHODS: A literature review and meta-analysis were performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement. Systematic literature searches were conducted using PubMed and Google Scholar up to February 2021.
RESULTS: Seventeen studies were included in the meta-analysis. The incidence of microvascular invasion was 49.0% (95% confidence interval [CI], 43.8-54.5%) among PDAC patients who underwent surgery. The weighted multivariate Cox proportional hazards model hazard ratio for disease-free survival of 8 studies was 1.78 (95% CI 1.53-2.08, P<0.001), and there was no statistically significant difference between the subgroups (P=0.477). The hazard ratio for overall survival of 14 studies was 1.49 (95% CI 1.27-1.74, P<0.001), and there was no statistically significant difference between the subgroups (P=0.676).
CONCLUSIONS: Microvascular invasion occurred in nearly half of PDAC patients after surgery and was closely related to disease-free and overall survival. Understanding the role of microvascular invasion in PDAC will help provide more personalized and effective preoperative or postoperative strategies to achieve better survival outcomes.
Keywords: Incidence, Microvessels, Pancreatic Neoplasms, Carcinoma, Pancreatic Ductal, Disease-Free Survival, Proportional Hazards Models
Background
Pancreatic cancer (PC) remains a leading cause of cancer-related deaths, and 80–85% of patients develop unresectable locally advanced or distant metastases at the time of diagnosis [1]. Furthermore, even in ≥80% of patients who can undergo surgery, the long-term survival outcomes are unsatisfactory because of the high incidences of local tumor recurrence and distant metastasis [2], which often occur within 1 year [3,4]. Pancreatic ductal adenocarcinoma (PDAC) is the major histological subtype of pancreatic cancer, accounting for more than 85% of cases [5]. For PDAC patients undergoing surgery, microvascular invasion (MVI) is an observable pathological parameter; despite the controversy that exists [6–8], many studies still suggest that it may be a predictor of early recurrence and metastasis of PDAC [9–15].
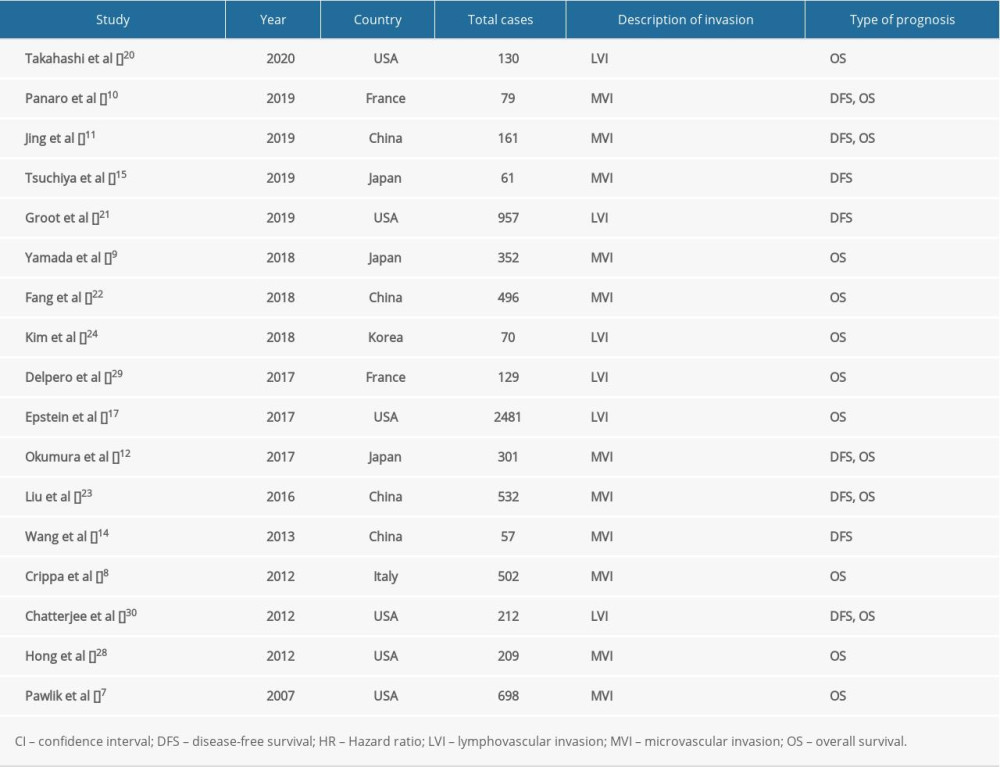
MVI is a microtubule invasion state that can only be observed around the tumor under a microscope [9,16]. It is described in the literature as “microvascular invasion” or “microscopic lymphovascular invasion” (LVI) [10,17]. MVI mostly describes microvessel invasion [18], while LVI is used to indicate micro-lymph-tubule invasion [19]. However, in the literature reviewed, only a few studies distinguished the 2 terms [9,15]; in most cases, they refer to the same pathological characteristics [7,11,12,14,20–23], which leads to confusion. In addition, the incidence of MVI in PDAC varies considerably [7–12,14,15,20–24] and the literature rarely discusses the potential mechanism of MVI in the progression of PDAC, so the mechanism and clinical significance of MVI in poor PDAC outcomes remains incompletely understood. We therefore conducted a systematic review and meta-analysis of all observational studies to examine the incidence, definition, clinical significance, and potential mechanism of MVI in PDAC, to help guide more personalized and effective preoperative or postoperative strategies to achieve better survival outcomes in patients with PC.
Material and Methods
SEARCH STRATEGY AND INCLUSION CRITERIA:
This systematic review and meta-analysis was reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [25]. We searched for all eligible studies in PubMed and Google Scholar in February 2021. In both databases, we used the uniform search strings: “pancreatic cancer, microvascular invasion” or “pancreatic cancer, lymphovascular invasion”. The literature search and reference screening were conducted by Li HB and Pan WW. If the title was too ambiguous to allow exclusion, we reviewed the abstract and full texts and decided whether or not to include the study.
We included all observational studies that (a) reported the type of disease as “pancreatic cancer” or “pancreatic ductal adenocarcinoma,” (b) reported the incidence of MVI or LVI, and (c) provided a relevant risk estimate in terms of the hazard ratio (HR) of disease-free survival (DFS) or overall survival (OS). We subsequently excluded papers that did not present full texts in English or Chinese and papers that reported the disease as “intraductal papillary carcinoma” or “pancreatic neuroendocrine tumors.” Conference abstracts were ineligible for inclusion.
DATA EXTRACTION:
Data extraction was performed independently by 2 authors (Xu L and Yin D), and disagreements were settled by consensus. For each study, we extracted descriptive information (author, year, country, study design, MVI or LVI cases, sample size, description of invasion, cancer type), and HR with 95% confidence interval (CI) of multivariate Cox regression analysis for DFS or OS.
ASSESSMENT OF STUDY QUALITY AND RISK OF BIAS:
The Strengthening of the Reporting of Observational Studies in Epidemiology (STROBE) guidelines were used to assess the quality of each study [26]. The risk of publication bias was assessed using funnel plots and Egger’s test.
STATISTICAL ANALYSES:
Fixed- or random-effects models were used to pool the study-specific risk estimates and calculated an overall effect estimate with associated 95% CI [27]. The I2 and Q tests were used to assess study heterogeneity. A multivariate Cox proportional hazards model was used to identify the factors which were independently associated with DFS and OS. P<0.05 was considered statistically significant. Statistical analyses were performed using R (version 4.0.2, www.r-project.org).
Results
LITERATURE SEARCH:
An electronic database search and survey of other sources identified 277 articles. By removing duplicate studies and studies with unmatched inclusion criteria, 32 full-text papers reported the incidence of MVI or LVI. After excluding the studies that did not report the relationship between MVI or LVI and prognosis, 17 articles were finally included in the systematic review and meta-analysis.
CHARACTERISTICS OF INCLUDED STUDIES AND QUALITY ASSESSMENT:
The characteristics of the 17 studies with a total of 7427 patients are summarized in Table 1 [7–12,14,15,17,20–24,28–30]. All articles were retrospective and observational studies, 4 of which were from China, 6 were from the USA, 3 were from Japan, and the others were from Italy, South Korea, and France. Six studies reported MVI as LVI. The risk of publication bias was assessed using funnel plots (Figure 1), and reports on the incidence of MVI were biased (Figure 1A).
INCIDENCES OF MVI IN PC:
We identified 17 reports of retrospective studies that described the incidence of MVI in PDAC cases (Figure 2) [7–12,14,15,17,20–24,28–30]. Thesestudies evaluated 7427 cases of PDAC, including 4029 cases with MVI. The I2 value was 96.2%, and a random-effects model was selected. The pooled incidence of MVI was 49.0% (95% CI, 43.8–54.5%) among PDAC patients who underwent surgery. Although the group defined as “LVI” seemed to have a higher incidence than the MVI group, there was no statistically significant difference between them (P=0.201).
RELATIONSHIP BETWEEN MVI AND PDAC PROGNOSIS:
Eight studies reported on DFS [10–12,14,15,21,23,30], with a weighted multivariate Cox proportional hazards model HR for DFS of 1.78 (95% CI, 1.53–2.08; P<0.001) and no statistically significant difference between the subgroups (P=0.477). The I2 value was 26% and a fixed-effects model was selected (Figure 3A). Fourteen studies reported on OS [7–12,17,20,22–24,28–30], with an HR for OS of 1.49 (95% CI, 1.27–1.74; P<0.001) and no statistically significant difference between the subgroups (P=0.676). The I2 value was 68.3% and a random-effects model was selected (Figure 3B). Since the OS reported by Epstein et al only included HR [17], its 95% CI was calculated according to the algorithm provided by Altman et al [31].
SENSITIVITY ANALYSIS:
Sensitivity analyses were further performed to determine the robustness of the results described above. For the incidences of MVI in PC and the relationship between MVI and PDAC prognosis, the results were not altered by deletion of any single study (Supplementary Figure 1).
Discussion
DEFINITION OF MVI:
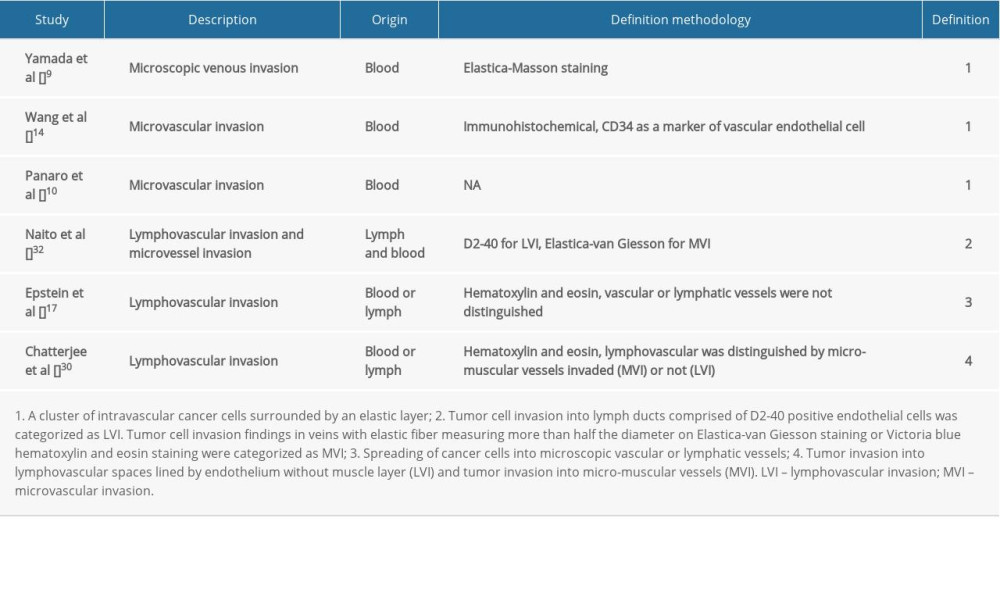
The definition of MVI in PDAC was not uniform (Table 2). Most studies described microvascular invasion of PDAC as MVI (11/17) [7–12,14,15,22,23,28], and the rest as LVI (6/17) [17,20,21,24,29,30]. In the studies that clearly explained the definition of microvascular or lymphatic invasion, 4 defined MVI as originating from blood vessels [9,10,14,32], 1 defined LVI as originating from lymphatic vessels [32], and 1 did not distinguish between them [17]. Although other studies specifically used the terms MVI or LVI, all of these studies’ research objectives were regarding MVI according to the articles’ descriptions [7,8,11,12,15,20–24,28,29]. In addition, from the data in Figure 3, we found that although different studies have different definitions of MVI, there was no significant difference between the 2 groups (MVI vs LVI) in either DFS or OS. Distinguishing between LVI or MVI requires immunohistochemical analyses with a combination of lymphatic markers (D2-40 and anti-podoplanin) and endothelial markers (ERG, CD31, CD34, and factor VIII) for accurate differentiation, which could be difficult to perform in a large sample or in routine clinical work [6,9,17,32]. It is difficult to distinguish between lymphatic and blood vessels in clinical work. This study proposes defining MVI as a cluster of tumor cells surrounded by microvessels lined by epithelium in the peritumoral domain or tumor microenvironment, and there is no need to distinguish its histological origin (ie, blood vessels or lymphatic vessels), as this does not affect prognostic evaluation.
INCIDENCES OF MVI:
The reported incidence of MVI varied widely (Figure 2). The primary reason is that different research centers have different definitions of MVI, as mentioned earlier. Given the critical role of MVI in prognostic evaluation, it is necessary for the pathologist to carefully check whether there is MVI when examining a sample, based on the above definition.
THE POTENTIAL MECHANISM FOR MVI IN PDAC:
Cancer metastasis involves a sequence of physical translocation steps from the primary tumor to distant organs [33]. During this process, MVI and the subsequent peripheral blood circulation of cancer cells and/or lymphatic metastasis are necessary and important routes of metastasis [9]. MVI even appears earlier than peripheral circulating tumor cells [33]; therefore, MVI is an essential early event in cancer metastasis.
Epithelial-to-mesenchymal transition (EMT) is a critical biological process by which cancer cells lose their polarized organization, degrade the surrounding extracellular matrix, and acquire migratory and invasive capabilities [34]. Multiple studies have directly confirmed that EMT is the biological basis of MVI in liver cancer [35–37]. Even though EMT is an important step in PDAC metastasis [38–41], the direct relationship between EMT and MVI in PDAC has rarely been reported in the literature. Stress-inducible phosphoprotein-1 (STIP1) is positively correlated with MVI in PDAC [11] and induces EMT in gastric and lung cancers [42,43], and it may be the potential key link between EMT and MVI in PDAC.
In addition to EMT (Figure 4A(a)), alterations in the endothelial permeability of the microvascular endothelial monolayer may also be a potential mechanism of MVI (Figure 4A(b)). Increased permeability of the microvascular endothelium, which acts as a metastatic barrier, facilitates invasion of tumor cells to the microvasculature, leading to MVI [44,45]. This phenomenon was confirmed in liver cancer [46]. The circular RNA IARS (circ-IARS), located within exosomes, promotes MVI in PDAC by increasing microvascular endothelial monolayer permeability via the downregulation of miR-122 [47].
THE POTENTIAL RELATIONSHIP BETWEEN MVI AND PDAC METASTASIS:
The precise mechanism underlying MVI in PDAC is controversial and complicated. Based on the existing literature, local recurrence may be attributed to residual MVI [15,18], while distant metastasis may be based on the following 3 routes (Figure 4B) [48–53]. First, cancer cells may directly enter the portal vein through the microvasculature originating from blood vessels and subsequently develop liver metastases (Figure 4B(1)) [52,53]. Second, perilesional lymphangiogenesis and luminal invasion may provide lymphatic structural support for MVI [54]. Based on the structure, cancer cells enter the microlymphatic vessels around the lesion and drain into the adjacent lymph nodes (Figure 4B(2)), which is supported by evidence that the rate of lymph node metastasis and the incidence of MVI are positively correlated [17]. Third, cancer cells enter the lymphatic ducts through the microlymphatic vessels and subsequently enter the venous system, which leads to liver, lung, and bone metastasis [55] (Figure 4B(3)). However, these 3 routes are interrelated [55]. Lymphatic and hematogenous metastases are positively related in PDAC [9], and patients with lymph node metastasis have higher rates of liver and lung metastasis than those without [56]. Lymphatic metastasis may occur before hematogenous metastasis due to the lack of the tight interendothelial junctions typically seen in blood vessels but not in lymphatic capillaries [48,51].
CLINICAL SIGNIFICANCE OF MVI:
MVI often indicates a higher rate of positive margins [9,30,57]. For patients at high risk of MVI in the preoperative evaluation, the distance from the tumor to the resection margin should be appropriately increased. For pathologically confirmed MVI cases after surgery, surgical specimen margins should be examined more carefully.
The histopathological presence of MVI in PDAC specimens can predict the risk of early recurrence [15,21], and patients with MVI had higher rates of locoregional recurrence, liver metastasis, and lung metastasis [9]. As such, MVI is an independent risk factor for DFS and OS (Figure 3). Neoadjuvant chemotherapy may be useful for reducing the MVI rate in cases of both resectable [28] and borderline resectable PDAC [29,32]. As PC patients cannot benefit from expanded surgery [58], patients with MVI should receive more aggressive postoperative adjuvant chemotherapy [59].
Preoperatively predicting the risk of MVI in liver cancer helps guide surgical decision-making and postoperative management based on radiographic and clinical parameters [60–64]. However, there are only a few similar studies involving PDAC cases. Yamada et al reported that MVI was significantly predicted by serum carbohydrate antigen 19-9, maximum standard uptake value from fluorodeoxyglucose positron emission tomography, and tumor size from preoperative computed tomography [9]. Andreasi et al also reported that plasma vasostatin-1 concentrations could be used to preoperatively predict the presence of MVI [65]. Other studies have revealed that MVI is positively correlated with STIP-1 and CD34 levels in PDAC [11,14]. Further research may help develop a model to predict the risk of MVI based on non-invasive parameters, similar to models for liver cancer, which will require studies to assess the relationships between MVI, radiographic parameters, and clinical parameters in PDAC cases.
In the era of precision medicine, accurate prognostication helps guide the selection of appropriate and effective treatment strategies [66]. Thus, many studies have aimed to develop prognostic models to predict outcomes in PDAC [67–69]. However, it is unfortunate that these studies have failed to consider the prognostic relevance of MVI. As MVI is an independent risk factor affecting postoperative recurrence and overall survival, it will be of clinical significance to develop a postoperative recurrence prediction model or prognosis model incorporating MVI to more precisely assess the prognosis.
Conclusions
MVI occurred in nearly half of PDAC patients after surgery and is closely related to DFS and OS. Understanding the role of MVI in PDAC helps provide more personalized and effective preoperative or postoperative strategies to achieve better survival outcomes. There are many important items that need to be resolved in the future. First, although previous literature analysis indicates that it may not be necessary to distinguish between microlymphatic or microvascular invasion, this inference still needs to be verified by strictly designed experiments. Second, robust prediction models for preoperative MVI status and postoperative prognosis are urgently needed. Finally, the details of molecules, pathways, and biological processes involved in MVI, along with the regulation of MVI and its role in PDAC metastases, remain to be completely elucidated.
Figures
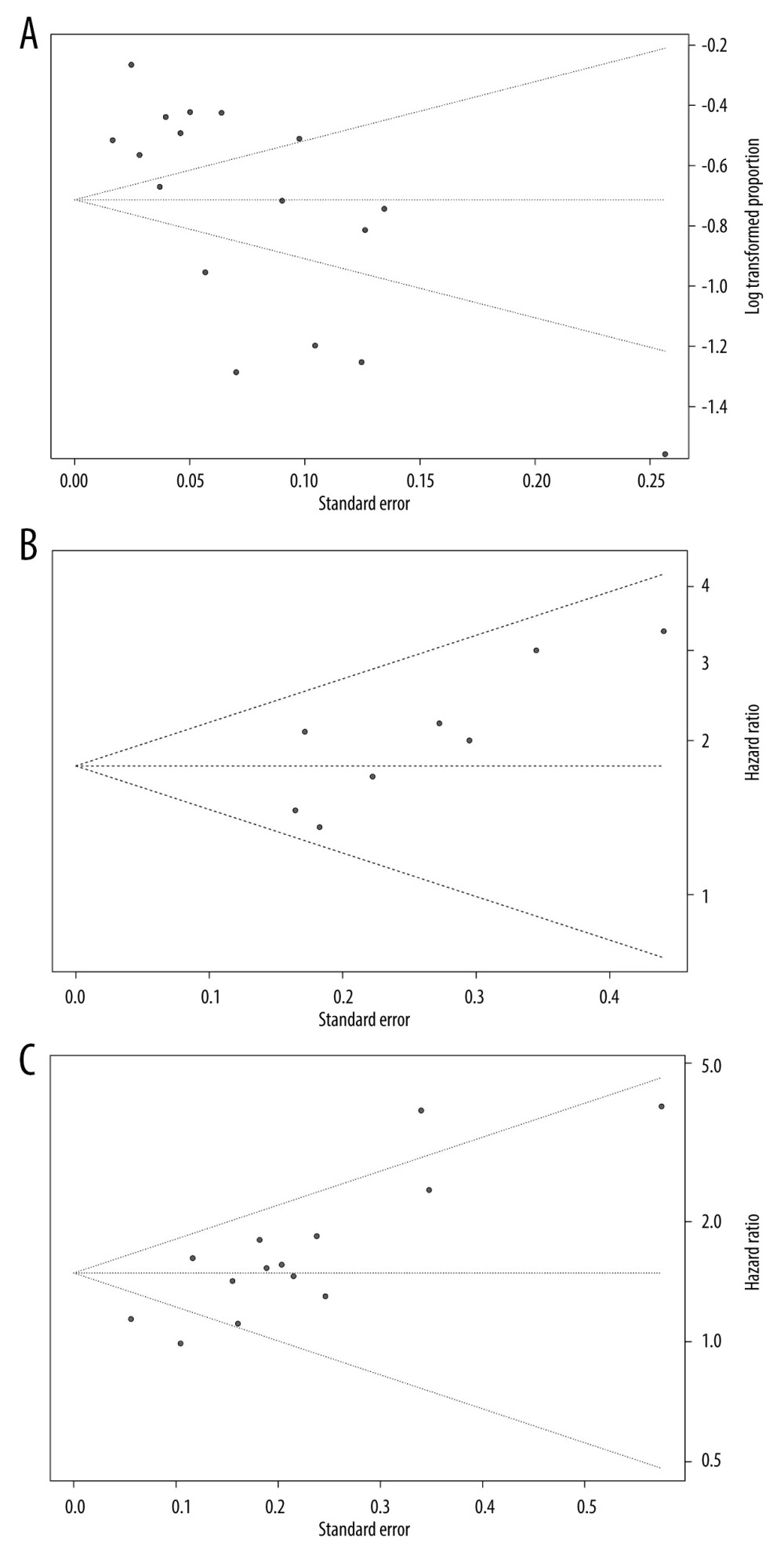 Figure 1. Funnel plots of microvascular invasion incidence (A), disease-free survival (B), and overall survival (C).
Figure 1. Funnel plots of microvascular invasion incidence (A), disease-free survival (B), and overall survival (C). 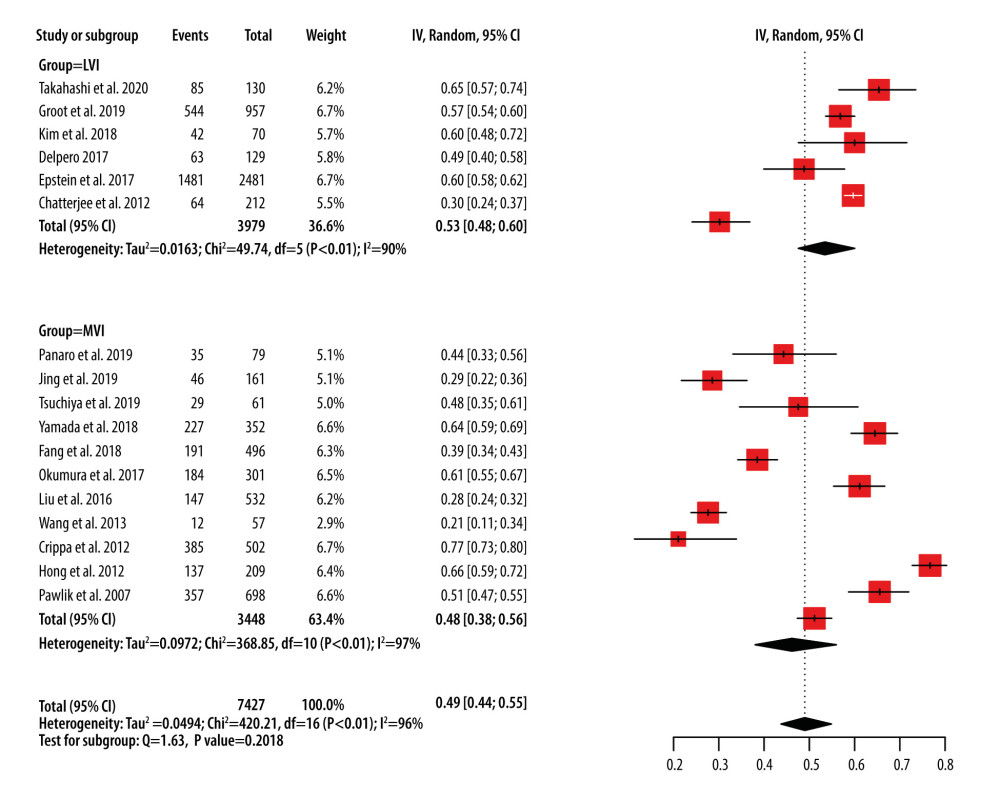 Figure 2. Forest plots of microvascular invasion incidence. CI – confidence interval; LVI – lymphovascular invasion; MVI – microvascular invasion.
Figure 2. Forest plots of microvascular invasion incidence. CI – confidence interval; LVI – lymphovascular invasion; MVI – microvascular invasion. 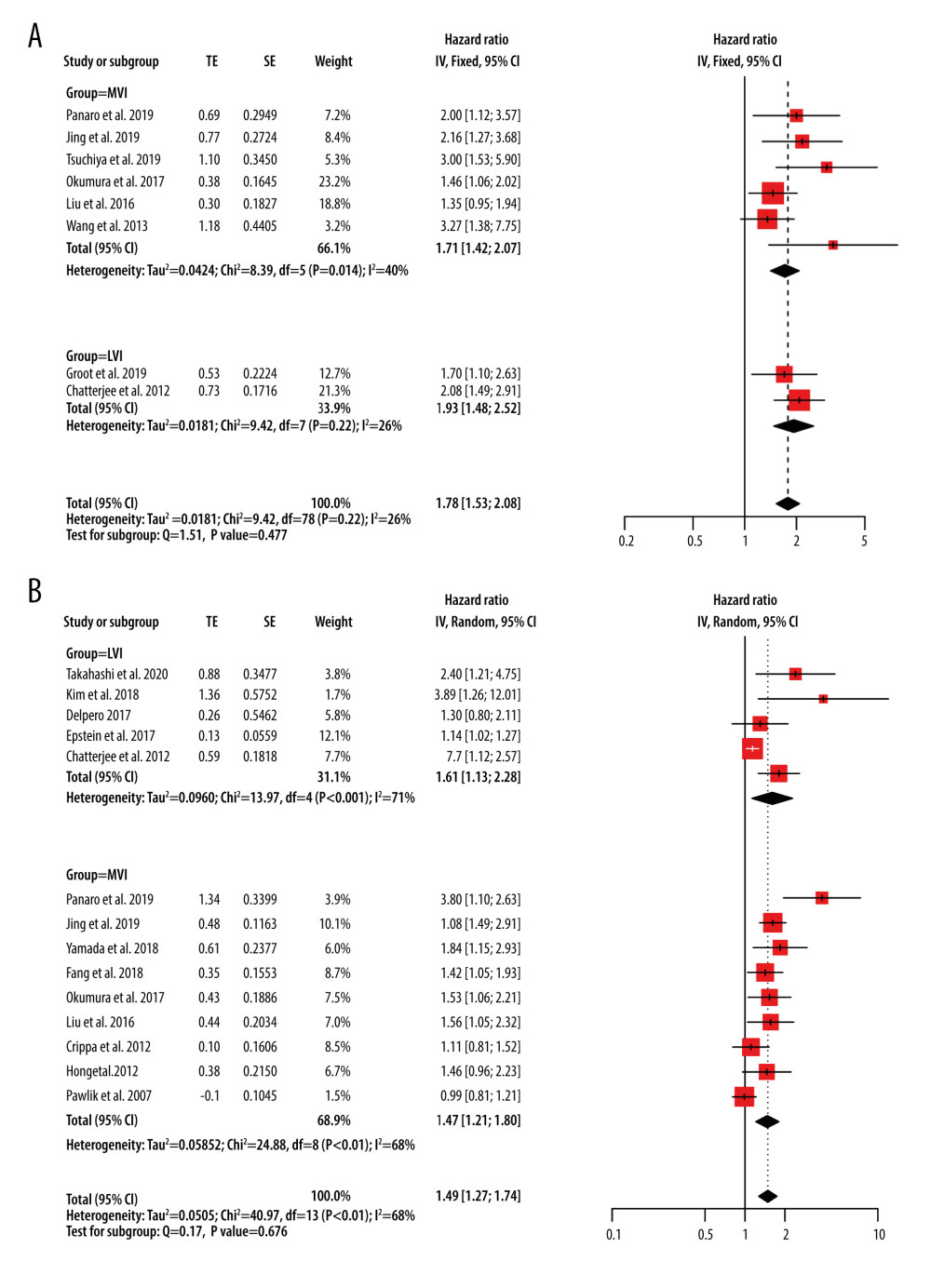 Figure 3. Forest plots of disease-free survival (A) and overall survival (B) for MVI. CI – confidence interval; LVI – lymphovascular invasion; MVI – microvascular invasion; SE – standard error of treatment estimate; TE – estimate of treatment effect, log hazard ratio.
Figure 3. Forest plots of disease-free survival (A) and overall survival (B) for MVI. CI – confidence interval; LVI – lymphovascular invasion; MVI – microvascular invasion; SE – standard error of treatment estimate; TE – estimate of treatment effect, log hazard ratio. 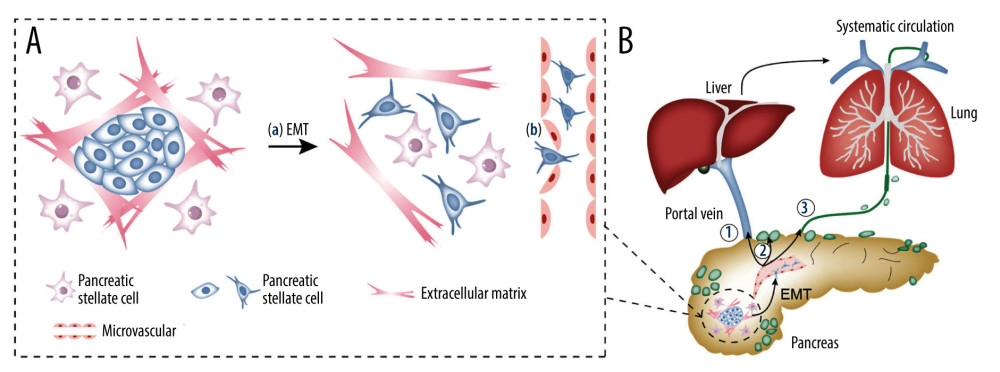 Figure 4. The mechanism of MVI in pancreatic cancer and the relationship between MVI and metastasis. (A) The mechanism of MVI in pancreatic cancer. (a) Tumor cells gain mobility and invasion capabilities through EMT. (b) Increased permeability of the microvascular endothelium facilitates tumor cells to invade into the microvasculature. (B) The relationship between MVI and metastasis. (1) Angiogenesis, leading to blood transfer and/or liver transfer through the portal vein. (2) Lymphangiogenesis, leading to metastasis to the draining lymph nodes. (3) Lymphangiogenesis, leading to metastasis to the lymphatic duct. EMT – epithelial-to-mesenchymal transition; MVI – microvascular invasion.
Figure 4. The mechanism of MVI in pancreatic cancer and the relationship between MVI and metastasis. (A) The mechanism of MVI in pancreatic cancer. (a) Tumor cells gain mobility and invasion capabilities through EMT. (b) Increased permeability of the microvascular endothelium facilitates tumor cells to invade into the microvasculature. (B) The relationship between MVI and metastasis. (1) Angiogenesis, leading to blood transfer and/or liver transfer through the portal vein. (2) Lymphangiogenesis, leading to metastasis to the draining lymph nodes. (3) Lymphangiogenesis, leading to metastasis to the lymphatic duct. EMT – epithelial-to-mesenchymal transition; MVI – microvascular invasion. References
1. McGuigan A, Kelly P, Turkington RC, Pancreatic cancer: A review of clinical diagnosis, epidemiology, treatment and outcomes: World J Gastroenterol, 2018; 24(43); 4846-61
2. Wu H, Guo JC, Yang SH, Postoperative imaging and tumor marker surveillance in resected pancreatic cancer: J Clin Med, 2019; 8(8); 1115
3. Groot VP, Rezaee N, Wu W, Patterns, timing, and predictors of recurrence following pancreatectomy for pancreatic ductal adenocarcinoma: Ann Surg, 2018; 267(5); 936-45
4. Daamen LA, Groot VP, Intven MPW, Postoperative surveillance of pancreatic cancer patients: Eur J Surg Oncol, 2019; 45(10); 1770-77
5. Luo G, Fan Z, Gong Y, Characteristics and outcomes of pancreatic cancer by histological subtypes: Pancreas, 2019; 48(6); 817-22
6. Cai SW, Yang SZ, Gao J, Prognostic significance of mast cell count following curative resection for pancreatic ductal adenocarcinoma: Surgery, 2011; 149(4); 576-84
7. Pawlik TM, Gleisner AL, Cameron JL, Prognostic relevance of lymph node ratio following pancreaticoduodenectomy for pancreatic cancer: Surgery, 2007; 141(5); 610-18
8. Crippa S, Partelli S, Zamboni G, Poorly differentiated resectable pancreatic cancer: Is upfront resection worthwhile?: Surgery, 2012; 152(3 Suppl 1); S112-19
9. Yamada M, Sugiura T, Okamura Y, Microscopic venous invasion in pancreatic cancer: Ann Surg Oncol, 2018; 25(4); 1043-51
10. Panaro F, Kellil T, Vendrell J, Microvascular invasion is a major prognostic factor after pancreatico-duodenectomy for adenocarcinoma: J Surg Oncol, 2019; 120(3); 483-93
11. Jing Y, Liang W, Liu J, Stress-induced phosphoprotein 1 promotes pancreatic cancer progression through activation of the FAK/AKT/MMP signaling axis: Pathol Res Pract, 2019; 215(11); 152564
12. Okumura S, Kaido T, Hamaguchi Y, Visceral adiposity and sarcopenic visceral obesity are associated with poor prognosis after resection of pancreatic cancer: Ann Surg Oncol, 2017; 24(12); 3732-40
13. Masui T, Sato A, Nakano K, Comparison of recurrence between pancreatic and duodenal neuroendocrine neoplasms after curative resection: A single-institution analysis: Ann Surg Oncol, 2018; 25(2); 528-34
14. Wang WQ, Liu L, Xu HX, Intratumoral alpha-SMA enhances the prognostic potency of CD34 associated with maintenance of microvessel integrity in hepatocellular carcinoma and pancreatic cancer: PLoS One, 2013; 8(8); e71189
15. Tsuchiya N, Matsuyama R, Murakami T, Risk factors associated with early recurrence of borderline resectable pancreatic ductal adenocarcinoma after neoadjuvant chemoradiation therapy and curative resection: Anticancer Res, 2019; 39(8); 4431-40
16. Roayaie S, Blume IN, Thung SN, A system of classifying microvascular invasion to predict outcome after resection in patients with hepatocellular carcinoma: Gastroenterology, 2009; 137(3); 850-55
17. Epstein JD, Kozak G, Fong ZV, Microscopic lymphovascular invasion is an independent predictor of survival in resected pancreatic ductal adenocarcinoma: J Surg Oncol, 2017; 116(6); 658-64
18. Erstad DJ, Tanabe KK, Prognostic and therapeutic implications of microvascular invasion in hepatocellular carcinoma: Ann Surg Oncol, 2019; 26(5); 1474-93
19. Aleskandarany MA, Sonbul SN, Mukherjee A, Molecular mechanisms underlying lymphovascular invasion in invasive breast cancer: Pathobiology, 2015; 82(3–4); 113-23
20. Takahashi H, Katsuta E, Yan L, Transcriptomic profile of lymphovascular invasion, a known risk factor of pancreatic ductal adenocarcinoma metastasis: Cancers (Basel), 2020; 12(8); 2033
21. Groot VP, Gemenetzis G, Blair AB, Defining and predicting early recurrence in 957 patients with resected pancreatic ductal adenocarcinoma: Ann Surg, 2019; 269(6); 1154-62
22. Fang LP, Xu XY, Ji YFactors influencing survival of patients with pancreatic adenocarcinoma after surgical resection: Zhonghua Yi Xue Za Zhi, 2018; 98(8); 606-11 [in Chinese]
23. Liu L, Xu HX, Wang WQ, Serum CA125 is a novel predictive marker for pancreatic cancer metastasis and correlates with the metastasis-associated burden: Oncotarget, 2016; 7(5); 5943-56
24. Kim HR, Seo M, Nah YW, Clinical impact of fluorine-18-fluorodeoxyglucose positron emission tomography/computed tomography in patients with resectable pancreatic cancer: Diagnosing lymph node metastasis and predicting survival: Nucl Med Commun, 2018; 39(7); 691-98
25. Liberati A, Altman DG, Tetzlaff J, The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration: PLoS Med, 2009; 6(7); e1000100
26. Vandenbroucke JP, von Elm E, Altman DG, Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration: PLoS Med, 2007; 4(10); e297
27. Kirkegard J, Mortensen FV, Cronin-Fenton D, Chronic pancreatitis and pancreatic cancer risk: A systematic review and meta-analysis: Am J Gastroenterol, 2017; 112(9); 1366-72
28. Hong SM, Goggins M, Wolfgang CL, Vascular invasion in infiltrating ductal adenocarcinoma of the pancreas can mimic pancreatic intraepithelial neoplasia: A histopathologic study of 209 cases: Am J Surg Pathol, 2012; 36(2); 235-41
29. Delpero JR, Jeune F, Bachellier P, Prognostic value of resection margin involvement after pancreaticoduodenectomy for ductal adenocarcinoma: Updates from a French prospective multicenter study: Ann Surg, 2017; 266(5); 787-96
30. Chatterjee D, Rashid A, Wang H, Tumor invasion of muscular vessels predicts poor prognosis in patients with pancreatic ductal adenocarcinoma who have received neoadjuvant therapy and pancreaticoduodenectomy: Am J Surg Pathol, 2012; 36(4); 552-59
31. Altman DG, Bland JM, How to obtain the confidence interval from a P value: BMJ, 2011; 343; d2090
32. Naito Y, Ishikawa H, Sadashima E, Significance of neoadjuvant chemoradiotherapy for borderline resectable pancreatic head cancer: Pathological local invasion and microvessel invasion analysis: Mol Clin Oncol, 2019; 11(3); 225-33
33. Chaffer CL, Weinberg RA, A perspective on cancer cell metastasis: Science, 2011; 331(6024); 1559-64
34. Dongre A, Weinberg RA, New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer: Nat Rev Mol Cell Biol, 2019; 20(2); 69-84
35. Zhang T, Guo J, Gu J, KIAA0101 is a novel transcriptional target of FoxM1 and is involved in the regulation of hepatocellular carcinoma microvascular invasion by regulating epithelial-mesenchymal transition: J Cancer, 2019; 10(15); 3501-16
36. Xu ZY, Ding SM, Zhou L, FOXC1 contributes to microvascular invasion in primary hepatocellular carcinoma via regulating epithelial-mesenchymal transition: Int J Biol Sci, 2012; 8(8); 1130-41
37. Lin J, He Y, Chen L, MYLK promotes hepatocellular carcinoma progression through regulating cytoskeleton to enhance epithelial-mesenchymal transition: Clin Exp Med, 2018; 18(4); 523-33
38. Chen Y, LeBleu VS, Carstens JL, Dual reporter genetic mouse models of pancreatic cancer identify an epithelial-to-mesenchymal transition-independent metastasis program: EMBO Mol Med, 2018; 10(10); e9085
39. Li H, Wang X, Wen C, Long noncoding RNA NORAD, a novel competing endogenous RNA, enhances the hypoxia-induced epithelial-mesenchymal transition to promote metastasis in pancreatic cancer: Mol Cancer, 2017; 16(1); 169
40. Beuran M, Negoi I, Paun S, The epithelial to mesenchymal transition in pancreatic cancer: A systematic review: Pancreatology, 2015; 15(3); 217-25
41. Houg DS, Bijlsma MF, The hepatic pre-metastatic niche in pancreatic ductal adenocarcinoma: Mol Cancer, 2018; 17(1); 95
42. Huang L, Zhai E, Cai S, Stress-inducible Protein-1 promotes metastasis of gastric cancer via Wnt/beta-catenin signaling pathway: J Exp Clin Cancer Res, 2018; 37(1); 6
43. Guo X, Yan Z, Zhang G, STIP1 regulates proliferation and migration of lung adenocarcinoma through JAK2/STAT3 signaling pathway: Cancer Manag Res, 2019; 11; 10061-72
44. Yoshioka K, Yoshida K, Cui H, Endothelial PI3K-C2alpha, a class II PI3K, has an essential role in angiogenesis and vascular barrier function: Nat Med, 2012; 18(10); 1560-69
45. Reymond N, d’Agua BB, Ridley AJ, Crossing the endothelial barrier during metastasis: Nat Rev Cancer, 2013; 13(12); 858-70
46. Fang JH, Zhang ZJ, Shang LR, Hepatoma cell-secreted exosomal microRNA-103 increases vascular permeability and promotes metastasis by targeting junction proteins: Hepatology, 2018; 68(4); 1459-75
47. Li J, Li Z, Jiang P, Circular RNA IARS (circ-IARS) secreted by pancreatic cancer cells and located within exosomes regulates endothelial monolayer permeability to promote tumor metastasis: J Exp Clin Cancer Res, 2018; 37(1); 177
48. Wong SY, Hynes RO, Lymphatic or hematogenous dissemination: How does a metastatic tumor cell decide?: Cell Cycle, 2006; 5(8); 812-17
49. Ren B, Cui M, Yang G, Tumor microenvironment participates in metastasis of pancreatic cancer: Mol Cancer, 2018; 17(1); 108
50. Ayres Pereira M, Chio IIC, Metastasis in pancreatic ductal adenocarcinoma: Current standing and methodologies: Genes (Basel), 2019; 11(1); 6
51. Hosch SB, Knoefel WT, Metz S, Early lymphatic tumor cell dissemination in pancreatic cancer: Frequency and prognostic significance: Pancreas, 1997; 15(2); 154-59
52. Tao L, Su L, Yuan C, Postoperative metastasis prediction based on portal vein circulating tumor cells detected by flow cytometry in periampullary or pancreatic cancer: Cancer Manag Res, 2019; 11; 7405-25
53. Hruban RH, Gaida MM, Thompson E, Why is pancreatic cancer so deadly? The pathologist’s view: J Pathol, 2019; 248(2); 131-41
54. Shen CN, Goh KS, Huang CR, Lymphatic vessel remodeling and invasion in pancreatic cancer progression: EBioMedicine, 2019; 47; 98-113
55. Fink DM, Steele MM, Hollingsworth MA, The lymphatic system and pancreatic cancer: Cancer Lett, 2016; 381(1); 217-36
56. Sahin IH, Elias H, Chou JF, Pancreatic adenocarcinoma: Insights into patterns of recurrence and disease behavior: BMC Cancer, 2018; 18(1); 769
57. Kimbrough CW, St Hill CR, Martin RC, Tumor-positive resection margins reflect an aggressive tumor biology in pancreatic cancer: J Surg Oncol, 2013; 107(6); 602-7
58. Jang JY, Kang MJ, Heo JS, A prospective randomized controlled study comparing outcomes of standard resection and extended resection, including dissection of the nerve plexus and various lymph nodes, in patients with pancreatic head cancer: Ann Surg, 2014; 259(4); 656-64
59. Strobel O, Neoptolemos J, Jager D, Optimizing the outcomes of pancreatic cancer surgery: Nat Rev Clin Oncol, 2019; 16(1); 11-26
60. Zhang X, Li J, Shen F, Significance of presence of microvascular invasion in specimens obtained after surgical treatment of hepatocellular carcinoma: J Gastroenterol Hepatol, 2018; 33(2); 347-54
61. Feng ST, Jia Y, Liao B, Preoperative prediction of microvascular invasion in hepatocellular cancer: A radiomics model using Gd-EOB-DTPA-enhanced MRI: Eur Radiol, 2019; 29(9); 4648-59
62. Nitta H, Allard MA, Sebagh M, Predictive model for microvascular invasion of hepatocellular carcinoma among candidates for either hepatic resection or liver transplantation: Surgery, 2019; 165(6); 1168-75
63. Lei Z, Li J, Wu D, Nomogram for preoperative estimation of microvascular invasion risk in hepatitis B virus-related hepatocellular carcinoma within the Milan Criteria: JAMA Surg, 2016; 151(4); 356-63
64. Zeng F, Chen B, Zeng J, Preoperative neutrophil-lymphocyte ratio predicts the risk of microvascular invasion in hepatocellular carcinoma: A meta-analysis: Int J Biol Markers, 2019; 34(3); 213-20
65. Andreasi V, Partelli S, Manzoni M, Association between preoperative Vasostatin-1 and pathological features of aggressiveness in localized nonfunctioning pancreatic neuroendocrine tumors (NF-PanNET): Pancreatology, 2019; 19(1); 57-63
66. Jin KT, Chen XY, Lan HR, Current progress in the clinical use of circulating tumor cells as prognostic biomarkers: Cancer Cytopathol, 2019; 127(12); 739-49
67. Li HB, Zhou J, Zhao FQ, A prognostic nomogram for disease-specific survival in patients with pancreatic ductal adenocarcinoma of the head of the pancreas following pancreaticoduodenectomy: Med Sci Monit, 2018; 24; 6313-21
68. Attiyeh MA, Chakraborty J, Doussot A, Survival prediction in pancreatic ductal adenocarcinoma by quantitative computed tomography image analysis: Ann Surg Oncol, 2018; 25(4); 1034-42
69. Okabayashi T, Shima Y, Sumiyoshi T, A novel physiobiological parameter-based grading system for resectable pancreatic cancer: Ann Surg Oncol, 2018; 25(7); 1889-95
Figures
 Figure 1. Funnel plots of microvascular invasion incidence (A), disease-free survival (B), and overall survival (C).
Figure 1. Funnel plots of microvascular invasion incidence (A), disease-free survival (B), and overall survival (C). Figure 2. Forest plots of microvascular invasion incidence. CI – confidence interval; LVI – lymphovascular invasion; MVI – microvascular invasion.
Figure 2. Forest plots of microvascular invasion incidence. CI – confidence interval; LVI – lymphovascular invasion; MVI – microvascular invasion. Figure 3. Forest plots of disease-free survival (A) and overall survival (B) for MVI. CI – confidence interval; LVI – lymphovascular invasion; MVI – microvascular invasion; SE – standard error of treatment estimate; TE – estimate of treatment effect, log hazard ratio.
Figure 3. Forest plots of disease-free survival (A) and overall survival (B) for MVI. CI – confidence interval; LVI – lymphovascular invasion; MVI – microvascular invasion; SE – standard error of treatment estimate; TE – estimate of treatment effect, log hazard ratio. Figure 4. The mechanism of MVI in pancreatic cancer and the relationship between MVI and metastasis. (A) The mechanism of MVI in pancreatic cancer. (a) Tumor cells gain mobility and invasion capabilities through EMT. (b) Increased permeability of the microvascular endothelium facilitates tumor cells to invade into the microvasculature. (B) The relationship between MVI and metastasis. (1) Angiogenesis, leading to blood transfer and/or liver transfer through the portal vein. (2) Lymphangiogenesis, leading to metastasis to the draining lymph nodes. (3) Lymphangiogenesis, leading to metastasis to the lymphatic duct. EMT – epithelial-to-mesenchymal transition; MVI – microvascular invasion.
Figure 4. The mechanism of MVI in pancreatic cancer and the relationship between MVI and metastasis. (A) The mechanism of MVI in pancreatic cancer. (a) Tumor cells gain mobility and invasion capabilities through EMT. (b) Increased permeability of the microvascular endothelium facilitates tumor cells to invade into the microvasculature. (B) The relationship between MVI and metastasis. (1) Angiogenesis, leading to blood transfer and/or liver transfer through the portal vein. (2) Lymphangiogenesis, leading to metastasis to the draining lymph nodes. (3) Lymphangiogenesis, leading to metastasis to the lymphatic duct. EMT – epithelial-to-mesenchymal transition; MVI – microvascular invasion. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387