17 December 2021: Animal Study
Galangin Enhances Anticancer Efficacy of 5-Fluorouracil in Esophageal Cancer Cells and Xenografts Through NLR Family Pyrin Domain Containing 3 (NLRP3) Downregulation
Cong Zhang1ABF, Cui-Lian Luo1BCD, Guan-Sheng Shang1DE, De-Xiong Jiang1BF, Qi Song2AG*DOI: 10.12659/MSM.931630
Med Sci Monit 2021; 27:e931630
Abstract
BACKGROUND: Galangin is believed to exert antioxidant effects by inhibition of the NLR family pyrin domain containing 3 (NLRP3) inflammasome, which has been linked to chemotherapy sensitivity in cancers. In this study, we explored the synergistic effect of galangin in combination with the chemotherapy agent 5-fluorouracil (5-FU) in esophageal cancer cells and xenografts.
MATERIAL AND METHODS: The esophageal squamous epithelium cell line Het-1A and 2 human esophageal cancer cell lines (Eca109, OE19) were used to investigate the effect of galangin with or without 5-FU in vitro through proliferation and invasion analyses, while apoptosis was analyzed in cancer cells. Furthermore, a subcutaneous xenograft tumor model in mice was used to study cancer development in vivo.
RESULTS: Compared with 5-FU monotherapy, combined galangin and 5-FU treatment reduced human esophageal cancer cell growth activities and invasion abilities. The results suggested that galangin had a chemotherapy-sensitized synergistic antitumor effect induced by 5-FU. The susceptibility of cancer cells to apoptosis, which is linked with chemotherapy sensitivity, was induced by 5-FU and further enhanced by galangin. NLRP3 was identified as being significantly activated by 5-FU, but galangin treatment reversed the effect and inhibited NLRP3 expression, which was accompanied by downregulated interleukin-1b levels. Further investigation showed that the induced apoptotic cascade can be mostly reversed by incubation with an NLRP3 activator, irrespective of AKT signaling. Using xenograft mouse models, we found that galangin exposure further restrained cancer development after 5-FU treatment and increased sensitivity to chemotherapy by suppressing the NLRP3 inflammasome pathway.
CONCLUSIONS: Our results indicated that galangin played a synergistic anticancer role through NLRP3 inflammasome inhibition when paired with FU-5.
Keywords: Esophageal Neoplasms, NLRP3 Protein, Human, Galangin, Animals, Antimetabolites, Antineoplastic, Antineoplastic Combined Chemotherapy Protocols, Blotting, Western, Cell Line, Tumor, Down-Regulation, Drug Synergism, Fluorouracil, Humans, Male, Mice, Mice, Nude, NLR Family, Pyrin Domain-Containing 3 Protein, Neoplasm Transplantation
Background
Esophageal cancer is a common malignant tumor with an increasing incidence worldwide during the past 3 decades. It is an aggressive malignancy and represents the seventh most common cause of cancer-related mortality. Adenocarcinoma is the most common form of esophageal cancer, especially in lower portion of the esophagus, while the major subtype in the top or middle of the esophagus is squamous cell carcinoma [1]. The cause of esophageal cancer remains unknown. Consuming alcohol, smoking, and being overweight are believed to be related to malignancy occurrence. Further, demographic characteristics, such as being male and African American, as well as environmental exposure to polycyclic aromatic hydrocarbons (from opium and indoor air pollution) and nutrient-deficient diets have been identified as risk factors of esophageal cancer [2,3]. Surgery or endoscopic resection is the main therapy for patients with early-stage esophageal cancer, with chemotherapy, radiation therapy, immune checkpoint inhibitor, or combined therapies (eg, pembrolizumab plus chemotherapy as recently approved) being recommended as standard therapies for metastatic diseases [4–6]. However, detecting esophageal cancer at an early stage is difficult and patients with late-stage disease or drug-resistant types continue to have a poor prognosis.
As one of the widely used anticancer drugs for gastric solid tumors, 5-fluorouracil (5-FU) interferes with nucleoside metabolism and can be incorporated into RNA and DNA, leading to cytotoxicity and cell death [7]. Resistance to 5-FU constitutes one of the causes of treatment failure. Recently, adaption to increased oxidative stress, overactivated inflammation, and impaired amino acid metabolism in cancer cells were reported to be involved in drug resistance to 5-FU [8,9]. For example, previous findings implied that high NLR family pyrin domain containing 3 (NLRP3) inflammasome expression and activation, which are responsible for the maturation and secretion of proinflammatory cytokines from immune responses, can confer a more malignant phenotype and promote chemoresistance to 5-FU-based therapy [10]. However, activated NLRP3-induced intracellular reactive oxygen species (ROS) levels in pancreatic cancer and oral squamous cell carcinoma were found to be related to increased interleukin (IL)-1β production and 5-FU chemoresistance [11]. Therefore, exploration of novel therapies that may increase sensitivity to 5-FU are urgent and could yield considerable benefits for cancer treatment.
Galangin (4
To better understand the synergistic antitumor effect of galangin in combination with 5-FU and its related molecular mechanism in esophageal cancer, we sought to determine whether galangin can further inactivate the NLRP3 pathway to suppress inflammation and inhibit tumorigenicity in addition to standard therapy. In the future, the combined therapies of galangin plus 5-FU targeting NLRP3 pathways might represent a promising therapeutic regimen to improve the activity of existing anticancer drugs in treating esophageal cancer.
Material and Methods
REAGENTS AND CELL LINES:
Galangin was purchased from Sigma (USA). A stock solution (5 mg/mL, 18.5 mM) consisting of 25 mg galangin and 5 mL of dimethyl sulfoxide (DMSO) was prepared. It was diluted to the required concentrations with the following cell culture media. The ready-to-use media were prepared with DMSO, which did not exceed 0.02% concentration in the final solution. Nigericin, an NLRP3 activator, was from Sigma Chemical Co. The normal esophageal squamous epithelium cell line Het-1A was purchased from American Type Culture Collection (ATCC) and cultured according to the product instructions using a BEGM kit (Lonza). Two human esophageal cancer cell lines (squamous cell carcinoma, Eca109; adenocarcinoma, OE19) were kindly provided by the Department of Pathology and grown in RPMI 1640 (Gibco) media supplemented with 10% fetal bovine serum (FBS, Sigma) in a humidified atmosphere of 95% air and 5% CO2 in a cell incubator at 37°C.
CELL PROLIFERATION ASSAY:
Using Cell Counting Kit-8 (CCK-8; Dojindo Laboratories), the cell proliferative capacities were determined. About 1×103 cells were seeded in 96-well plates and then treated with 5-FU, galangin, or both at different concentrations or with DMSO as the control. Following 2 days of culture, 10 μL of CCK-8 reagent was added to each well and the cells were cultured for an additional 1 h. A microplate reader (Bio-Rad Laboratories, USA) was then used to calculate the absorbance value around 450 nm of each well. A computer program CompuSyn (ComboSyn, Inc., USA) was used for quantitation of synergism and antagonism in drug combinations.
CELL INVASION ASSAY:
A Transwell cell invasion assay was performed using 8.0-μm pore polycarbonate filter inserts (Corning Costar, USA) coated with Matrigel (BD Biosciences, Franklin Lakes, NJ, USA). Briefly, esophageal cancer cells (1×104 cells/well) in conditioned or vehicle medium supplemented with less than 1% FBS were placed in the upper chamber. In the bottom chamber, complete medium with 10% FBS served as a chemoattractant to induce invasion. After incubation at 37°C for 24 h, the culture inserts were removed and the noninvasive cells on the upper surface of the culture inserts were removed using a cotton swab. The cells that invaded through the Matrigel were fixed with 4% paraformaldehyde and stained with 1% crystal violet at room temperature. Images were captured and the migrated cells were counted.
CELL APOPTOSIS ANALYSIS:
The esophageal cancer cells were harvested by trypsinization and washed twice with cold phosphate-buffered saline (PBS). The cells were resuspended in binding buffer at a density of 1.0×105 cells/mL. Next, 100 μL of the sample solution was transferred to a 5-mL culture tube and incubated with 5 μL of fluorescein isothiocyanate (FITC)-conjugated annexin V and 5 μL of propidium iodide (PI) for 15 min at room temperature in the dark according to the instruction of APOAF annexin V apoptosis kit (Sigma). Subsequently, 400 μL of binding buffer was applied to each sample and the samples were evaluated and analyzed by Canto II flow cytometer (BD Biosciences) and FlowJo 7.6 software (TreeStar, USA).
WESTERN BLOT ANALYSIS:
The total proteins of cells or tissues were isolated by lysis in RIPA buffer (Beyotime, China). A Pierce BCA protein assay kit (Thermo Fisher Scientific, USA) was used to rapidly calculate the protein concentration from the standard curve according to the manufacturer’s protocol. Protein was adjusted to the same amount (15 μg) among different samples, which were then loaded onto a 10% sodium dodecyl sulfate polyacrylamide gel for electrophoresis. After separation, the samples were transferred to a polyvinylidene difluoride (PVDF) membrane (Bio-Rad Laboratories, Germany). The PVDF membrane was blocked with 5% skimmed milk in Tris-buffered saline and Tween 20 (TBST) buffer on a shaker for 1 h at room temperature. The membrane was then incubated overnight with the following primary antibodies: anti-phospho-pAKT (Ser471) (9271), anti-AKT (9272), anti-IL-1β (12703), and anti-cleaved PARP (Asp214) (5625) from Cell Signaling Technology (USA); anti-NLRP3 (ABF-23) from Sigma; and anti-β-actin (sc-130656) from Santa Cruz Biotechnology (USA). After the PVDF membrane was washed 3 times on the shaker, it was incubated with adequate secondary antibody (7074, Cell Signaling Technology, USA) for 1 h at room temperature in the dark. The PVDF membrane was then incubated with chemiluminescence reagent in a foil-wrapped box for 5 min to generate signals (GE Healthcare Life Sciences, UK) and assessed by chemiluminescent detection.
TUMOR XENOGRAFT ASSAY:
All in vivo experiments were approved by the hospital Institutional Review Board/Institutional Animal Care and Use Committee. Esophageal cells were injected into the backs of male Balb/c nude mice (about 8 weeks old) at 1×107 cells in 200 μL of PBS per mouse (3 mice per group), and the mice were monitored for tumor growth. Four weeks after tumor implantation, when the tumor size reached ~100 mm3, the mice were randomly separated into the following treatment groups and received intraperitoneal injections as follows: (1) control group, which was treated with PBS; (2) the 5-FU group, which was treated with 5-FU, 20 mg/kg twice per week; and (3) the combined group, which was treated with 20 mg/kg 5-FU plus 10 mg/kg galangin twice per week. The tumor dimensions were carefully measured with a sliding caliper twice a week throughout the animal studies, and the estimated tumor volume was calculated using the following formula: volume (cm3)=(tumor length×[tumor width]2)/2. When the largest volume was more than 800 cm3, the experiment was stopped and the mice were killed by cervical dislocation. The mice were then dissected, and the tumor xenografts were harvested, weighed, and drop-fixed in a 10% formalin solution and subsequently prepared for immunohistochemistry assays.
STATISTICAL ANALYSIS:
The data are presented as the mean±standard error of the mean (SEM) unless otherwise noted. Analyses of all experiments were done using the
Results
GALANGIN TREATMENT INHIBITED THE GROWTH OF ESOPHAGEAL CANCER CELLS WITH 5-FU:
To investigate whether the combined treatment with galangin plus 5-FU showed antiviability effects, normal Het-1A and neoplastic Eca109 and OE19 cells were respectively treated with increasing concentrations of 5-FU (0–160 μmol/L), galangin (0–80 μmol/L), and combined therapy (2: 1 ratio) for 48–72 h. The viability of the cells was then examined by CCK-8 assays. As shown in Figure 1A–1C, the viability of the human esophageal cancer cell lines was diminished in a dose-dependent manner when incubated with 5-FU and galangin, respectively.
COMBINATION EFFECT OF GALANGIN AND 5-FU IN ESOPHAGEAL CANCER CELLS:
To further evaluate whether there was synergism of galangin and 5-FU, the combination index (CI) for cell growth inhibition was calculated in Het-1A, Eca109, and OE19 cells, respectively. As shown in Figure 2 and calculated from the CompuSyn program, the combination of galangin and 5-FU from 48 to 72 h presented a synergetic inhibitory effect on Eca109 cells with a fraction affected (Fa) value of more than 0.5 (CI <1). Therefore, the combination of 5-FU and galangin (2: 1 ratio) for 48 h achieved a synergistic effect when the concentration of 5-FU was greater than 10.03 μmol/L and galangin was greater than 5.02 μmol/L. Consistently, with 72-h drug incubation, the combination of 5-FU and galangin (2: 1 ratio) still exhibited a synergistic effect, with an Fa value of more than 0.6 (CI <1), when the concentration of 5-FU was higher than 8.55 μmol/L and galangin was higher than 4.28 μmol/L. The combination of galangin and 5-FU also showed a synergistic effect in OE19 cells for 48 and 72 h of combined exposure, with the Fa values being more than 0.25 (CI <1) and 0.35 (CI <1) when the concentration of 5-FU and galangin were greater than 10.05 and 5.03 μmol/L and 8.90 and 4.45 μmol/L, respectively.
COMBINED USE OF 5-FU AND GALANGIN INHIBITS INVASION OF ESOPHAGEAL CANCER CELLS:
The effect of galangin plus 5-FU on the invasion properties of esophageal cancer cells was then investigated, and the results showed that the invasive capabilities of Eca109 and OE19 cells were significantly inhibited by add-on treatment with galangin compared with 5-FU alone (Figure 3A–3C).
DETECTION OF INCREASED APOPTOSIS WITH GALANGIN TREATMENT IN HUMAN ESOPHAGEAL CANCER CELL LINES:
To determine whether the induction of cell death by galangin at a low concentration was due to induced apoptosis in Eca109 and OE19 cells, we used flow cytometry analysis to concurrently detect viable, necrotic, early apoptotic, and late apoptotic cells based on distinct double-staining patterns with a combination of FITC-conjugated annexin V and PI. As shown in Figure 4A–4C, the combined treatment of galangin (10 μmol/L) and 5-FU (20 μmol/L) for 36 h increased the ratio of apoptotic tumor cells from quantitative analysis.
DOWNREGULATION OF THE INFLAMMASOME PATHWAY WITHOUT IMPAIRMENT OF AKT SIGNALING BY GALANGIN IN HUMAN ESOPHAGEAL CANCER CELL LINES:
The pharmacological action of many flavonoids has been examined and established to lead to inflammasome formation accompanied by NLRP3 expression. Consistent with some other flavonoids, our results demonstrated that galangin regulated proliferation and death through inhibition of NLRP3 in both Eca109 and OE19 cells. The esophageal cancer cells were treated with vehicle or 20 μmol/L 5-FU or 20 μmol/L 5-FU plus 10 μmol/L galangin for 48 h, after which alterations in the expression of NLRP3 and its downstream cytokine IL-1β were examined. As demonstrated in Figure 5, after treatment with 5-FU monotherapy, the expression level of NLRP3 and IL-1β in the Eca109 and OE19 cells increased, followed by significant reductions when galangin was added. In addition, the apoptotic effect represented by cleaved PARP fragment (PARP1) was dramatically increased by the combined treatment. To further investigate the effect of galangin on the NLRP3-related inflammasome pathway, we added 20 μM nigericin, a NLRP3 pathway activator, for 4 h, and all signaling, including downregulated IL-1β levels and pro-apoptotic signals, were partially reversed in esophageal cancer cells exposed to the combined treatment.
PI3K/AKT signaling is known to be critical for cancer cell survival. To determine whether AKT-related changes are involved in the synergistic effects of galangin and 5-FU, the levels of p-AKT and AKT in Eca109 and OE19 cells were examined by western blot analysis. As shown in Figure 5, phosphorylated AKT expression was significantly decreased in esophageal cancer cells once 5-FU was used compared with the control group. However, when combined treatment using galangin with 5-FU was used, we found that the inactivation of AKT signaling was still stable. A similar pattern of AKT inactivation was identified in the group in which additional treatment of nigericin was introduced after 5-FU and galangin therapies, despite the NLRP3 signaling and PARP cleavage being greatly restored.
ANTITUMOR ACTIVITY OF COMBINED 5-FU AND GALANGIN AGAINST HUMAN ESOPHAGEAL TUMOR XENOGRAFT IN VIVO:
The anticancer effect and toxicity of 5-FU and galangin on esophageal tumor cells were investigated in a mouse xenograft model. As shown in Figure 6A, 6B, galangin plus 5-FU injection demonstrated synergistic growth inhibition of OE19 cells in vivo compared with 5-FU monotherapy and the control. Finally, it is consistent with the western blot analyses in vitro that, in comparison with the control group, the activated NLRP3 regulation in the 5-FU monotherapy group and reversed regulation in the combined group were confirmed in xenograft tissues by immunohistology assays (Figure 6C).
Discussion
Esophageal cancer is one of the least understood and most lethal cancers worldwide among human solid tumors, primarily owing to its highly aggressive nature and low survival rate [22]. A better understanding of the impaired biological process, signaling pathway, and molecular mechanisms could lead to an improved outcome in patients, presented by less aggressive modalities with novel treatment methods. In our study, we explored the enhanced inhibitory effect of a combined regimen composed of the classical chemotherapy agent 5-FU and the flavonoid galangin. Our results showed that the combined treatment using galangin and 5-FU significantly reduced human esophageal cancer cell growth activities and invasive abilities. The CI was calculated and it suggested that galangin had a synergistic antitumor effect based on 5-FU through a chemo-sensitized mechanism. Increased chemotherapy sensitivity, which was reflected by apoptosis activation, was further identified when NLRP3 was suppressed by galangin after an initial 5-FU treatment. Moreover, western blot assays showed that the chemo-sensitized effect due to galangin could be mostly reversed by incubation with an NLRP3 activator, irrespective of AKT signaling. In in vivo experiments, tumor volume was significantly reduced in mice treated with galangin plus 5-FU compared with those treated with a single agent alone. Therefore, the regimen combining 5-FU chemotherapy with galangin appears to be a promising therapeutic option for patients with esophageal cancer.
Natural reagents are promising products in pharmacology considering their multiple components and the related advantages of multi-targets actions. They also appear to offer a broad platform for anticancer drug screening. Vincristine, irinotecan, paclitaxel, and etoposide are typical examples of compounds that are extracted from plants, and many medicines have been produced and used in the clinic. Interest has increased in investigating flavonoids for treating malignancies. Flavonoids have been identified as the active ingredients in traditional herbal medicines as well as the standard of cares for the prevention or treatment of cancer in recent years. However, the results of several large-scale, long-term epidemiological studies have suggested that total flavonoids do not have strong protective effects on esophageal cancer in men in the United States [23] or in patients from European countries [24]. A meta-analysis provided weak evidence that total flavonoids were marginally associated with decreased esophageal cancer risk (OR=0.78, 95% confidence interval: 0.59–1.04) [25]. In conclusion, these studies suggested that dietary intake of total flavonoids might slightly reduce the risk of esophageal cancer in a population. However, results from noninterventional studies indicated that the associations remained significant before adjusting for dietary fiber, which was strongly correlated with flavonoid consumption. Furthermore, there were insufficient analyses of risks by esophageal cancer subgroup, such as esophageal adenocarcinoma and esophageal squamous cell carcinoma; flavonoid subclasses, such as flavonols, flavones, flavan-3-ols, flavanones, anthocyanins, and isoflavones; and individuals’ characteristics such as never smoker, former smoker, and current smoker [26,27].
For the standard treatment of esophageal cancer, 5-FU remains the primary chemotherapy agent. However, little is known about gastrointestinal tract flavonoid absorption, flavonoid metabolism varies by individual, and the degree to which flavonoids can have direct effects on epithelial surfaces when they cross and act on the esophagus remains uncertain [28]. Even though dietary flavonoid intake has not been proven to be significantly related to overall esophageal cancer incidence, the common effects of antioxidant, anti-inflammatory, and apoptosis induction may contribute to the decreased mortality from population-based findings [29]. Wang et al [30] reported that myricetin intake exhibited a cooperative inhibitory effect with 5-FU by arresting the G0/G1 phase of esophageal cancer cells. Moreover, Hong et al [31] discovered that myricetin can directly activate autophagy in a dose-dependent manner to markedly suppress esophageal cancer cell migration and invasion abilities. Therefore, our results are consistent with previous ones, and galangin, as one of flavonoids, obtained a similar chemosensitization potential in vitro and in vivo, further indicating that it can function as a powerful chemosensitizer for esophageal cancer when an adequate dose of 5-FU is administered.
Standard regimens, especially chemotherapies, such as 5-FU plus platinum and paclitaxel plus carboplatin, produce anticancer effects that may be accompanied by nephrotoxicity, hepatotoxicity, cardiotoxicity, and other adverse effects. Moreover, chemoresistance eventually develops in almost all patients with solid cancers. To obtain a sustainably significant effect, increasing the dosage of the drugs is necessary. However, this often leads to increased toxicity. Therefore, our finding of a synergistic antitumor effect of galangin with 5-FU has important clinical significance and most patients may benefit from prolonged progression-free survival time with the addition of galangin to current standard regimens. Furthermore, galangin is a natural compound and has a proven a safety profile in normal cells from the study.
Dysregulation of NLRP3 inflammasome signaling in cancer cells contributes to tumor pathogenesis, although its role in cancer development and progression remains controversial due to inconsistent findings [14]. Until now, little has been done to determine its exact effect in antitumor treatment. Feng et al [11] found that activation of the NLRP3 inflammasome was clearly increased in oral squamous cell carcinoma tissues of patients who received 5-FU-based chemotherapy. This finding implied that the NLRP3 inflammasome promoted 5-FU resistance, and targeting the ROS/NLRP3 inflammasome signaling pathway may boost 5-FU-based adjuvant chemotherapy in malignancies. Huang et al [32] reported that upregulated NLRP3 could help cancer stem cell self-renewal activation and may be involved in the residual disease recurrence after antitumor definitive treatment. Therefore, it is urgent to investigate the synergistic effect of simultaneous suppression of NLRP3 in addition to chemotherapy treatment to minimize the tumor burden in the body as much as possible.
Both apoptosis and pyroptosis involve caspase-dependent programmed cell death. In general, PARP1 cleavage by caspase-3/7 is a hallmark of apoptosis, which is mainly regulated by the PI3K/AKT/mTOR pathway, while NLRP3 inflammasome activation briefly triggers pyroptosis owing to increased oxidative stress. In addition, there are several crossover channels; for example, Malireddi et al [33] reported that caspase-7-mediated PARP1 cleavage is related to proinflammatory gene expression. It was recently discovered that NLRP3 inflammasomes activate both apoptotic and pyroptotic death pathways, and it can be concluded that PARP1 processing is a general strategy used by cells undergoing programmed cell death to preserve the cellular energy stores to allow proper execution of the cell death program [33]. 5-FU induces apoptosis in tumor cells, but the development of chemoresistance represents a substantial challenge in patients with esophageal cancer. The underlying mechanisms of this resistance have multiple aspects, and impaired NLRP3 inflammasome expression by increased intracellular ROS may be a major component. In our results, we did not detect significant AKT signaling changes after additional treatment with galangin. This outcome suggested that the proapoptotic effects induced by galangin are more likely related to NLRP3 regulation, as demonstrated in the nigericin rescue experiments. Similar to previous reports [34], our results suggest that attenuating NLRP3 inflammasome activation by a natural product (ie, galangin) could mediate increased chemotherapy sensitivities of 5-FU through rapid, increased cell apoptosis.
There were also some limitations in this study. For example, no clinical specimens were investigated. Further, previous studies have confirmed G0/G1 arrest in the cell cycle due to downregulation of cyclins D, E, and A and apoptosis as the main mechanisms underlying the antitumor effect of galangin in other cancers; however, similar proapoptotic changes have not been observed in esophageal cancers or after monotherapy with 5-FU [35].
Therefore, the exact effect of galangin on esophageal cancer is far from known, especially when combined with 5-FU. The results of this study underline the significance of galangin as a promising synergistic antitumor agent in addition to chemotherapy in esophageal cancers by targeting NLRP3-related inflammasome signaling but not the PI3K/AKT pathway. Further characterization research focused on this compound in the future will provide more solid evidence on the potential of galangin as a therapeutic option for combined treatment of esophageal cancer.
Conclusions
Galangin combined with 5-FU exhibited synergistic anticancer effects in esophageal cancer cells and xenografts through NLRP3 inhibition. This finding suggests that galangin is a promising antineoplastic molecule and can be used to develop innovative drugs in the future.
Figures
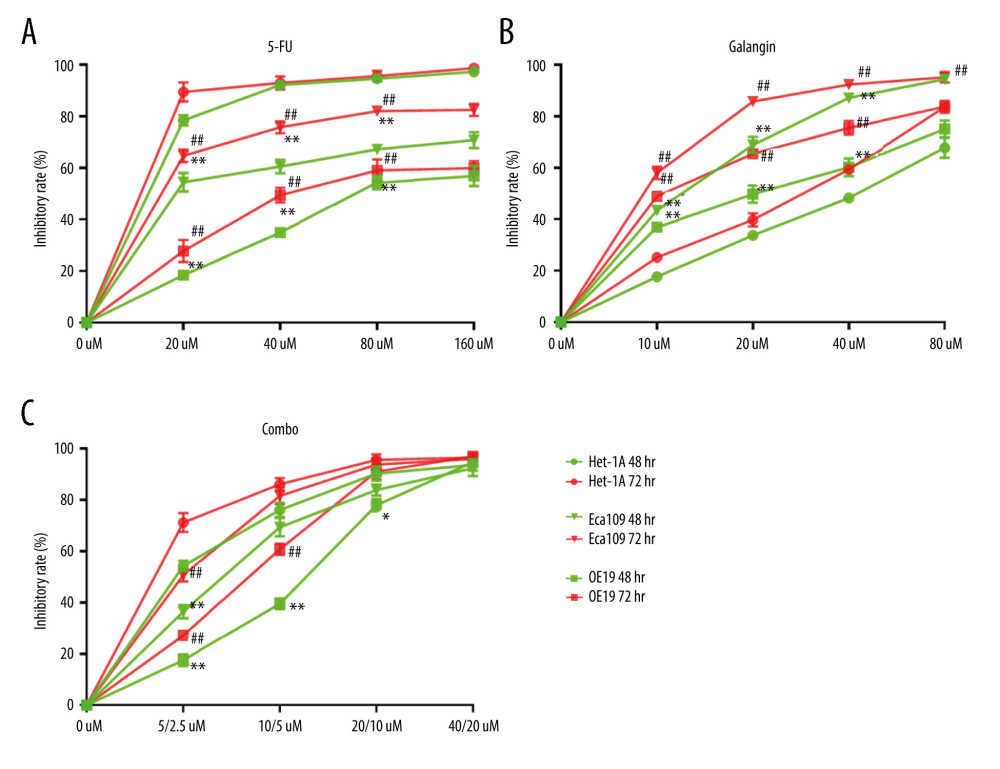 Figure 1. Effects of 5-fluorouracil (5-FU) and galangin on the proliferation of normal human esophageal cells and malignant cells. Het-1A, Eca109, and OE19 cells were incubated with different concentrations of 5-FU (A), galangin (B), and combined therapy (C) for 48–72 h, and the absorbance by CCK-8 assay was calculated compared with the control group independently. Data are shown as the mean±SEM of 3 independent experiments. * P<0.05 vs control group, ** P<0.01 vs control group for 48 h; # P<0.05 vs control group, ## P<0.01 vs control group for 72 h.
Figure 1. Effects of 5-fluorouracil (5-FU) and galangin on the proliferation of normal human esophageal cells and malignant cells. Het-1A, Eca109, and OE19 cells were incubated with different concentrations of 5-FU (A), galangin (B), and combined therapy (C) for 48–72 h, and the absorbance by CCK-8 assay was calculated compared with the control group independently. Data are shown as the mean±SEM of 3 independent experiments. * P<0.05 vs control group, ** P<0.01 vs control group for 48 h; # P<0.05 vs control group, ## P<0.01 vs control group for 72 h. 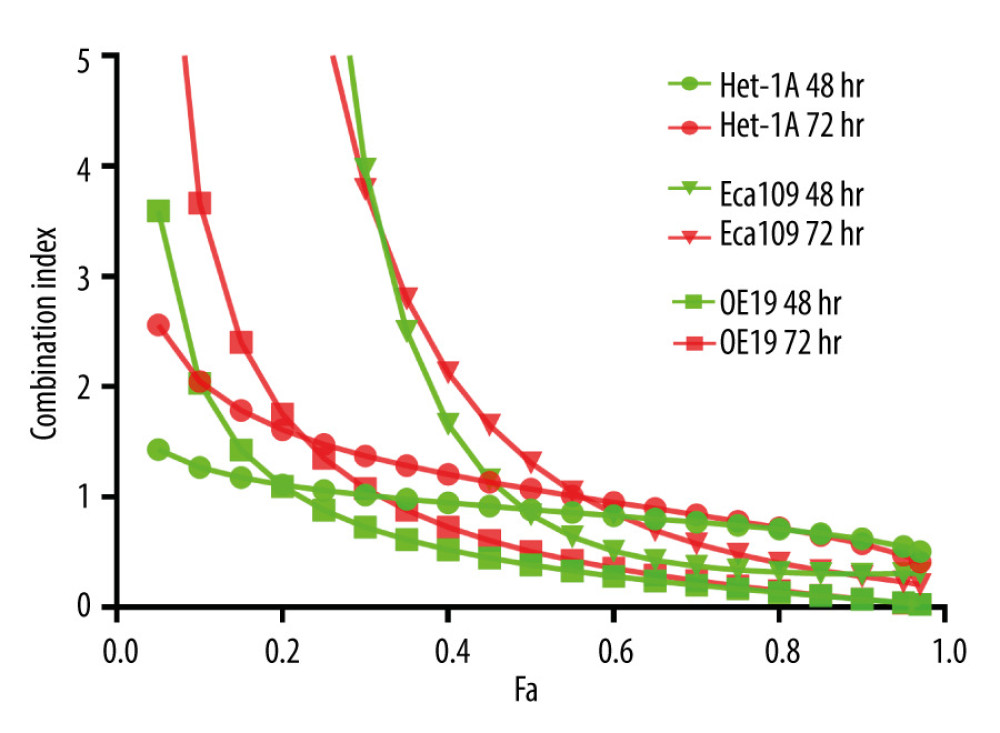 Figure 2. Exponential effect curves of the combination of fluorouracil (5-FU) and galangin in Het-1A, Eca109, and OE19 cells for 48- or 72-h treatment. Each point is related to a combination index (CI) value of different combined treatment effects.
Figure 2. Exponential effect curves of the combination of fluorouracil (5-FU) and galangin in Het-1A, Eca109, and OE19 cells for 48- or 72-h treatment. Each point is related to a combination index (CI) value of different combined treatment effects. 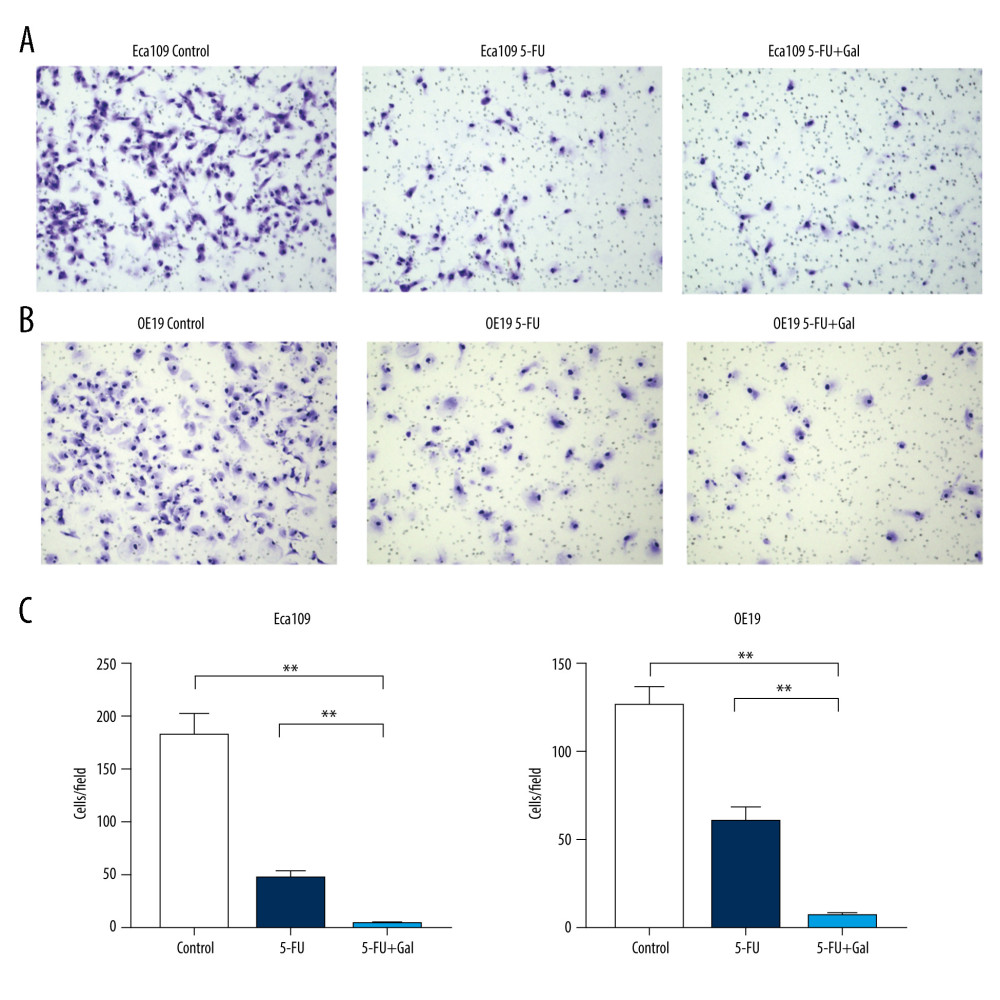 Figure 3. Effects of fluorouracil (5-FU) and galangin on the invasion of human esophageal cancer cells. (A) Eca109 and (B) OE19 cells were treated with 5-FU, galangin, and combined therapy for 24 h and then analyzed with a Transwell invasion assay. (C) Quantitative analyses were conducted, and the data are shown as the mean±SEM of 3 independent experiments. * P<0.05 vs control group; ** P<0.01 vs control group.
Figure 3. Effects of fluorouracil (5-FU) and galangin on the invasion of human esophageal cancer cells. (A) Eca109 and (B) OE19 cells were treated with 5-FU, galangin, and combined therapy for 24 h and then analyzed with a Transwell invasion assay. (C) Quantitative analyses were conducted, and the data are shown as the mean±SEM of 3 independent experiments. * P<0.05 vs control group; ** P<0.01 vs control group. 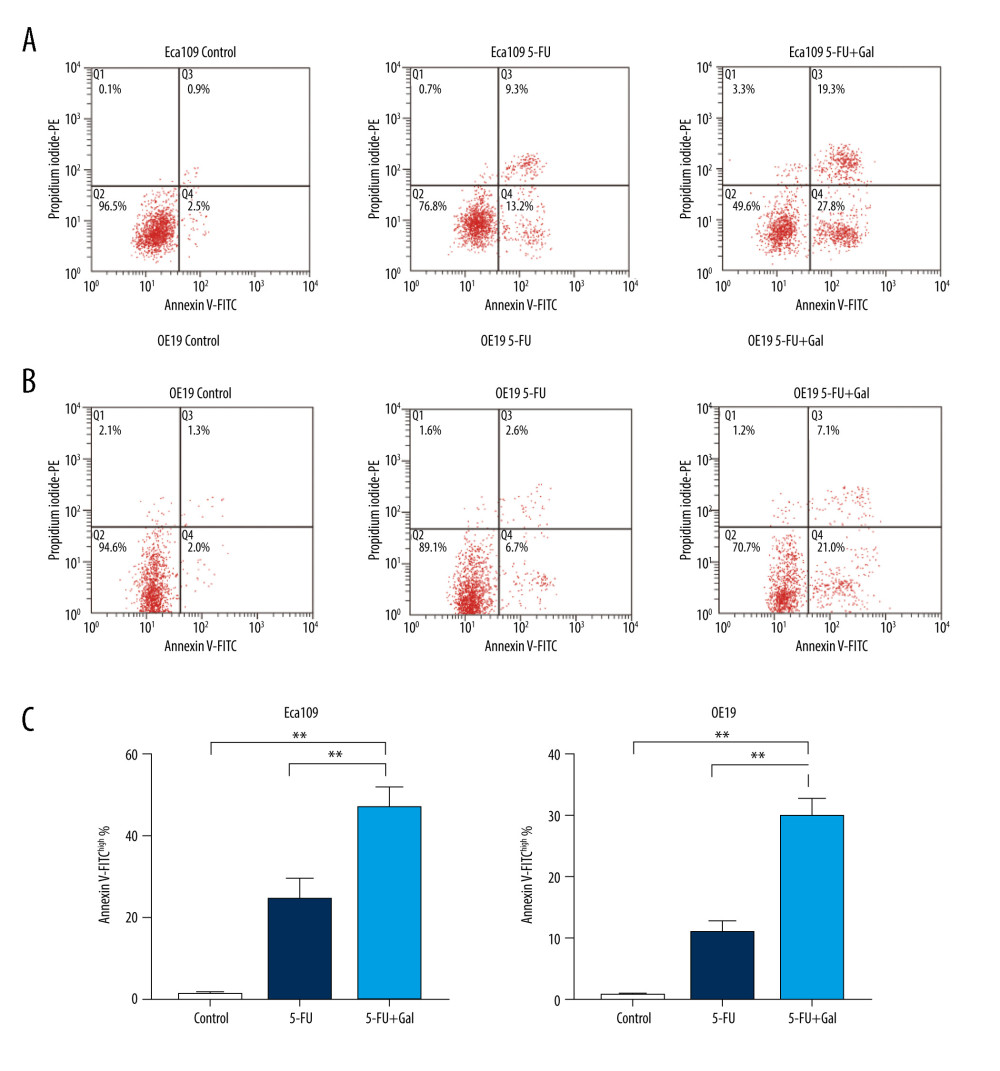 Figure 4. Effects of fluorouracil (5-FU) and galangin on the apoptosis of human esophageal cancer cells. (A) Eca109 and (B) OE19 cells were treated with 5-FU, galangin, and combined therapy for 36 h and then analyzed with annexin V/propidium iodide double stain. (C) Quantitative analyses were conducted, and representative diagrams of flow cytometry analyses are displayed. Data are presented as the mean±SEM of 3 independent experiments. * P<0.05 vs control group; ** P<0.01 vs control group.
Figure 4. Effects of fluorouracil (5-FU) and galangin on the apoptosis of human esophageal cancer cells. (A) Eca109 and (B) OE19 cells were treated with 5-FU, galangin, and combined therapy for 36 h and then analyzed with annexin V/propidium iodide double stain. (C) Quantitative analyses were conducted, and representative diagrams of flow cytometry analyses are displayed. Data are presented as the mean±SEM of 3 independent experiments. * P<0.05 vs control group; ** P<0.01 vs control group. 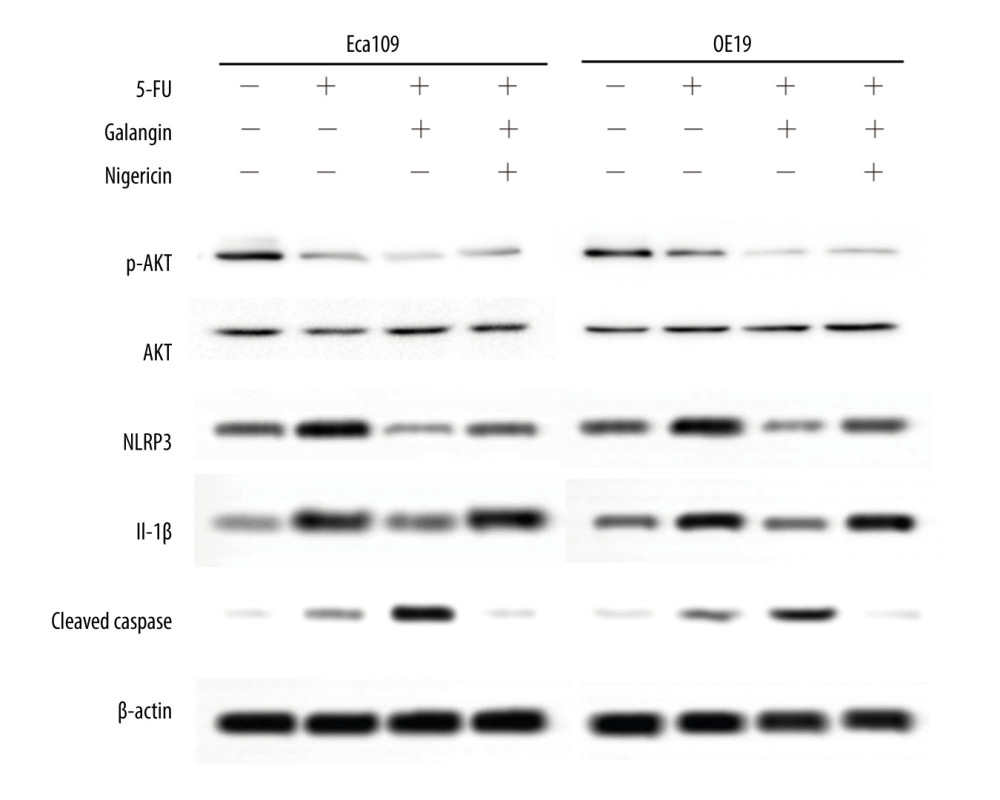 Figure 5. Galangin modulated cell death through the NLRP3 inflammasome but not the AKT pathway in human esophageal cancer cells after fluorouracil (5-FU) treatment. Total cell extracts were obtained and subjected to western blot analysis, which included anti-AKT, phospho-AKT, NLRP3, IL-1β, cleaved PARP (PARP1), and β-actin. The Eca109 and OE19 cells were also treated with DMSO (control), 5-FU, 5-FU+galangin, and 5-FU+galangin+nigericin solution. AKT – protein kinase B; DMSO – dimethyl sulfoxide; IL-1β – interleukin 1β; NLRP3 – NLR family pyrin domain containing 3; PARP – poly (ADP-ribose) polymerase.
Figure 5. Galangin modulated cell death through the NLRP3 inflammasome but not the AKT pathway in human esophageal cancer cells after fluorouracil (5-FU) treatment. Total cell extracts were obtained and subjected to western blot analysis, which included anti-AKT, phospho-AKT, NLRP3, IL-1β, cleaved PARP (PARP1), and β-actin. The Eca109 and OE19 cells were also treated with DMSO (control), 5-FU, 5-FU+galangin, and 5-FU+galangin+nigericin solution. AKT – protein kinase B; DMSO – dimethyl sulfoxide; IL-1β – interleukin 1β; NLRP3 – NLR family pyrin domain containing 3; PARP – poly (ADP-ribose) polymerase. 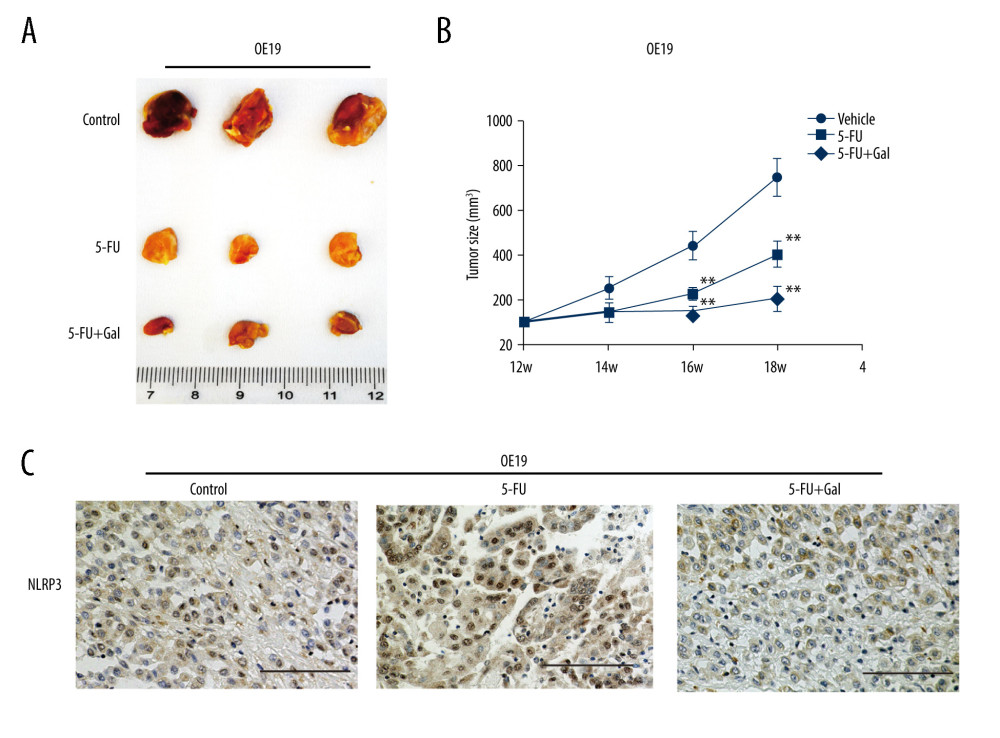 Figure 6. Galangin attenuated the malignant growth of esophageal cancer cells in mice. (A, B) Tumor volumes and representative esophageal tissues after resection in mouse xenograft models. (C) Galangin plus fluorouracil (5-FU) inhibited tumor NLR family pyrin domain containing 3 (NLRP3) expression by immunohistology stain assays. Scale bar, 100 μm.
Figure 6. Galangin attenuated the malignant growth of esophageal cancer cells in mice. (A, B) Tumor volumes and representative esophageal tissues after resection in mouse xenograft models. (C) Galangin plus fluorouracil (5-FU) inhibited tumor NLR family pyrin domain containing 3 (NLRP3) expression by immunohistology stain assays. Scale bar, 100 μm. References
1. Abbas G, Krasna M, Overview of esophageal cancer: Ann Cardiothorac Surg, 2017; 6(2); 131-36
2. Huang FL, Yu SJ, Esophageal cancer: Risk factors, genetic association, and treatment: Asian J Surg, 2018; 41(3); 210-15
3. Sheikh M, Poustchi H, Pourshams A, Individual and combined effects of environmental risk factors for esophageal cancer based on results from the Golestan cohort study: Gastroenterology, 2019; 156(5); 1416-27
4. Watanabe M, Otake R, Kozuki R, Recent progress in multidisciplinary treatment for patients with esophageal cancer: Surg Today, 2020; 50(1); 12-20
5. Triantafyllou T, Wijnhoven BPL, Current status of esophageal cancer treatment: Chin J Cancer Res, 2020; 32(3); 271-86
6. Kato K, Sun JM, Shah MA, Pembrolizumab plus chemotherapy versus chemotherapy as first-line therapy in patients with advanced esophageal cancer: The phase 3 KEYNOTE-590 study: Ann Oncol, 2020; 31(S4); S1192-93
7. Thomas DM, Zalcberg JR, 5-Fluorouracil: A pharmacological paradigm in the use of cytotoxics: Clin Exp Pharmacol Physiol, 1998; 25(11); 887-95
8. Hwang IT, Chung YM, Kim JJ, Drug resistance to 5-FU linked to reactive oxygen species modulator 1: Biochem Biophys Res Commun, 2007; 359(2); 304-10
9. De Angelis PM, Svendsrud DH, Kravik KL, Stokke T, Cellular response to 5-fluorouracil (5-FU) in 5-FU-resistant colon cancer cell lines during treatment and recovery: Mol Cancer, 2006; 5; 20
10. Ghiringhelli F, Bruchard M, Apetoh L, Immune effects of 5-fluorouracil: Ambivalence matters: Oncoimmunology, 2013; 2(3); e23139
11. Feng X, Luo Q, Zhang H, The role of NLRP3 inflammasome in 5-fluorouracil resistance of oral squamous cell carcinoma: J Exp Clin Cancer Res, 2017; 36(1); 81
12. Mukhtar H, Ahmad N, Tea polyphenols: Prevention of cancer and optimizing health: Am J Clin Nutr, 2000; 71(6 Suppl); 1698S-702S discussion 1703S–4S
13. Lim H, Min DS, Park H, Kim HP, Flavonoids interfere with NLRP3 inflammasome activation: Toxicol Appl Pharmacol, 2018; 355; 93-102
14. Moossavi M, Parsamanesh N, Bahrami A, Role of the NLRP3 inflammasome in cancer: Mol Cancer, 2018; 17(1); 158
15. Liu D, You P, Luo Y, Galangin induces apoptosis in MCF-7 human breast cancer cells through mitochondrial pathway and phosphatidylinositol 3-kinase/Akt inhibition: Pharmacology, 2018; 102(1–2); 58-66
16. Zhao X, Zhang JInhibitory effect of galangin on DNA topoisomerases in lung cancer cells: Zhong Nan Da Xue Xue Bao Yi Xue Ban, 2015; 40(5); 479-85 [in Chinese]
17. Zhang HT, Luo H, Wu J, Galangin induces apoptosis of hepatocellular carcinoma cells via the mitochondrial pathway: World J Gastroenterol, 2010; 16(27); 3377-84
18. Wang HX, Tang C, Galangin suppresses human laryngeal carcinoma via modulation of caspase-3 and AKT signaling pathways: Oncol Rep, 2017; 38(2); 703-14
19. Kong Y, Feng Z, Chen A, The natural flavonoid galangin elicits apoptosis, pyroptosis, and autophagy in glioblastoma: Front Oncol, 2019; 9; 942
20. Yu S, Gong LS, Li NF, Galangin (GG) combined with cisplatin (DDP) to suppress human lung cancer by inhibition of STAT3-regulated NF-kappaB and Bcl-2/Bax signaling pathways: Biomed Pharmacother, 2018; 97; 213-24
21. Tomar A, Vasisth S, Khan SI, Galangin ameliorates cisplatin induced nephrotoxicity in vivo by modulation of oxidative stress, apoptosis and inflammation through interplay of MAPK signaling cascade: Phytomedicine, 2017; 34; 154-61
22. Domper Arnal MJ, Ferrandez Arenas A, Lanas Arbeloa A, Esophageal cancer: Risk factors, screening and endoscopic treatment in Western and Eastern countries: World J Gastroenterol, 2015; 21(26); 7933-43
23. Bobe G, Peterson JJ, Gridley G, Flavonoid consumption and esophageal cancer among black and white men in the United States: Int J Cancer, 2009; 125(5); 1147-54
24. Vermeulen E, Zamora-Ros R, Duell EJ, Dietary flavonoid intake and esophageal cancer risk in the European prospective investigation into cancer and nutrition cohort: Am J Epidemiol, 2013; 178(4); 570-81
25. Cui L, Liu X, Tian Y, Flavonoids, flavonoid subclasses, and esophageal cancer risk: A meta-analysis of epidemiologic studies: Nutrients, 2016; 8(6); 350
26. Sardana RK, Chhikara N, Tanwar B, Panghal A, Dietary impact on esophageal cancer in humans: a review: Food Funct, 2018; 9(4); 1967-77
27. Wang QL, Xie SH, Li WT, Lagergren J, Smoking cessation and risk of esophageal cancer by histological type: Systematic review and meta-analysis: J Natl Cancer Inst, 2017; 109(12); djx115
28. Nobili S, Lippi D, Witort E, Natural compounds for cancer treatment and prevention: Pharmacol Res, 2009; 59(6); 365-78
29. Petrick JL, Steck SE, Bradshaw PT, Dietary intake of flavonoids and oesophageal and gastric cancer: Incidence and survival in the United States of America (USA): Br J Cancer, 2015; 112(7); 1291-300
30. Wang L, Feng J, Chen X, Myricetin enhance chemosensitivity of 5-fluorouracil on esophageal carcinoma in vitro and in vivo: Cancer Cell Int, 2014; 14; 71
31. Hong P, Liu QW, Xie Y, Echinatin suppresses esophageal cancer tumor growth and invasion through inducing AKT/mTOR-dependent autophagy and apoptosis: Cell Death Dis, 2020; 11(7); 524
32. Huang CF, Chen L, Li YC, NLRP3 inflammasome activation promotes inflammation-induced carcinogenesis in head and neck squamous cell carcinoma: J Exp Clin Cancer Res, 2017; 36(1); 116
33. Malireddi RK, Ippagunta S, Lamkanfi M, Kanneganti TD, Cutting edge: proteolytic inactivation of poly(ADP-ribose) polymerase 1 by the Nlrp3 and Nlrc4 inflammasomes: J Immunol, 2010; 185(6); 3127-30
34. Abotaleb M, Samuel SM, Varghese E, Flavonoids in cancer and apoptosis: Cancers (Basel), 2018; 11(1); 28
35. Ha TK, Kim ME, Yoon JH, Galangin induces human colon cancer cell death via the mitochondrial dysfunction and caspase-dependent pathway: Exp Biol Med (Maywood), 2013; 238(9); 1047-54
Figures
 Figure 1. Effects of 5-fluorouracil (5-FU) and galangin on the proliferation of normal human esophageal cells and malignant cells. Het-1A, Eca109, and OE19 cells were incubated with different concentrations of 5-FU (A), galangin (B), and combined therapy (C) for 48–72 h, and the absorbance by CCK-8 assay was calculated compared with the control group independently. Data are shown as the mean±SEM of 3 independent experiments. * P<0.05 vs control group, ** P<0.01 vs control group for 48 h; # P<0.05 vs control group, ## P<0.01 vs control group for 72 h.
Figure 1. Effects of 5-fluorouracil (5-FU) and galangin on the proliferation of normal human esophageal cells and malignant cells. Het-1A, Eca109, and OE19 cells were incubated with different concentrations of 5-FU (A), galangin (B), and combined therapy (C) for 48–72 h, and the absorbance by CCK-8 assay was calculated compared with the control group independently. Data are shown as the mean±SEM of 3 independent experiments. * P<0.05 vs control group, ** P<0.01 vs control group for 48 h; # P<0.05 vs control group, ## P<0.01 vs control group for 72 h. Figure 2. Exponential effect curves of the combination of fluorouracil (5-FU) and galangin in Het-1A, Eca109, and OE19 cells for 48- or 72-h treatment. Each point is related to a combination index (CI) value of different combined treatment effects.
Figure 2. Exponential effect curves of the combination of fluorouracil (5-FU) and galangin in Het-1A, Eca109, and OE19 cells for 48- or 72-h treatment. Each point is related to a combination index (CI) value of different combined treatment effects. Figure 3. Effects of fluorouracil (5-FU) and galangin on the invasion of human esophageal cancer cells. (A) Eca109 and (B) OE19 cells were treated with 5-FU, galangin, and combined therapy for 24 h and then analyzed with a Transwell invasion assay. (C) Quantitative analyses were conducted, and the data are shown as the mean±SEM of 3 independent experiments. * P<0.05 vs control group; ** P<0.01 vs control group.
Figure 3. Effects of fluorouracil (5-FU) and galangin on the invasion of human esophageal cancer cells. (A) Eca109 and (B) OE19 cells were treated with 5-FU, galangin, and combined therapy for 24 h and then analyzed with a Transwell invasion assay. (C) Quantitative analyses were conducted, and the data are shown as the mean±SEM of 3 independent experiments. * P<0.05 vs control group; ** P<0.01 vs control group. Figure 4. Effects of fluorouracil (5-FU) and galangin on the apoptosis of human esophageal cancer cells. (A) Eca109 and (B) OE19 cells were treated with 5-FU, galangin, and combined therapy for 36 h and then analyzed with annexin V/propidium iodide double stain. (C) Quantitative analyses were conducted, and representative diagrams of flow cytometry analyses are displayed. Data are presented as the mean±SEM of 3 independent experiments. * P<0.05 vs control group; ** P<0.01 vs control group.
Figure 4. Effects of fluorouracil (5-FU) and galangin on the apoptosis of human esophageal cancer cells. (A) Eca109 and (B) OE19 cells were treated with 5-FU, galangin, and combined therapy for 36 h and then analyzed with annexin V/propidium iodide double stain. (C) Quantitative analyses were conducted, and representative diagrams of flow cytometry analyses are displayed. Data are presented as the mean±SEM of 3 independent experiments. * P<0.05 vs control group; ** P<0.01 vs control group. Figure 5. Galangin modulated cell death through the NLRP3 inflammasome but not the AKT pathway in human esophageal cancer cells after fluorouracil (5-FU) treatment. Total cell extracts were obtained and subjected to western blot analysis, which included anti-AKT, phospho-AKT, NLRP3, IL-1β, cleaved PARP (PARP1), and β-actin. The Eca109 and OE19 cells were also treated with DMSO (control), 5-FU, 5-FU+galangin, and 5-FU+galangin+nigericin solution. AKT – protein kinase B; DMSO – dimethyl sulfoxide; IL-1β – interleukin 1β; NLRP3 – NLR family pyrin domain containing 3; PARP – poly (ADP-ribose) polymerase.
Figure 5. Galangin modulated cell death through the NLRP3 inflammasome but not the AKT pathway in human esophageal cancer cells after fluorouracil (5-FU) treatment. Total cell extracts were obtained and subjected to western blot analysis, which included anti-AKT, phospho-AKT, NLRP3, IL-1β, cleaved PARP (PARP1), and β-actin. The Eca109 and OE19 cells were also treated with DMSO (control), 5-FU, 5-FU+galangin, and 5-FU+galangin+nigericin solution. AKT – protein kinase B; DMSO – dimethyl sulfoxide; IL-1β – interleukin 1β; NLRP3 – NLR family pyrin domain containing 3; PARP – poly (ADP-ribose) polymerase. Figure 6. Galangin attenuated the malignant growth of esophageal cancer cells in mice. (A, B) Tumor volumes and representative esophageal tissues after resection in mouse xenograft models. (C) Galangin plus fluorouracil (5-FU) inhibited tumor NLR family pyrin domain containing 3 (NLRP3) expression by immunohistology stain assays. Scale bar, 100 μm.
Figure 6. Galangin attenuated the malignant growth of esophageal cancer cells in mice. (A, B) Tumor volumes and representative esophageal tissues after resection in mouse xenograft models. (C) Galangin plus fluorouracil (5-FU) inhibited tumor NLR family pyrin domain containing 3 (NLRP3) expression by immunohistology stain assays. Scale bar, 100 μm. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








