24 November 2021: Clinical Research
Discoidin Domain Receptor Tyrosine Kinase 1 (DDR1): A Novel Predictor for Recurrence of Hepatocellular Carcinoma After Curative Resection
Meng Xu1CDEFG, Chenghao Cui1AB*DOI: 10.12659/MSM.933109
Med Sci Monit 2021; 27:e933109
Abstract
BACKGROUND: Previous studies showed that the discoidin domain receptor tyrosine kinase 1 (DDR1) is significantly elevated in a variety of cancers, and it is closely related to the occurrence and development of tumors. However, its clinical significance in hepatocellular carcinoma (HCC) is not fully elucidated. So, in this study, we aimed to systemically evaluate the prognostic value of DDR1 in HCC.
MATERIAL AND METHODS: A total of 200 individuals were enrolled in this study (including 120 HCC patients, 40 chronic hepatitis patients, and 40 health individuals). The contents of DDR1 in serum was measured by enzyme-linked immunosorbent assay (ELISA), while the expression level of DDR1 in para-tumor and tumor tissue was detected by immunohistochemistry staining. Kaplan-Meier, Cox regression analyses, and log-rank test were used to assess the prognostic value.
RESULTS: The contents of DDR1 in serum of HCC patients was significantly higher compared with chronic hepatitis patients (P<0.01) and health individuals (P<0.001). The expression level of DDR1 in tumors was higher than that in normal liver tissue, and it had relatively strong correlation with DDR1 in serum. We next demonstrated that high DDR1 has utility as a prognostic risk factor for tumor recurrence and metastasis, and it still retains its discrimination ability in low-risk groups (BCLC 0+A). Moreover, DDR1 is as an independent predictor of prognosis in HCC patients with microvascular invasion (MVI), and is strongly associated with epithelial-mesenchymal transition (EMT)-related protein.
CONCLUSIONS: DDR1 is a novel predictor for HCC recurrence. Integration of serum and tumor DDR1 detection into clinical management would provide convenience and enhanced accuracy in clinical practice.
Keywords: DDR1 Protein, Human, HCC-1(9-74), MVI-12, Biomarkers, Tumor, Carcinoma, Hepatocellular, Discoidin Domain Receptor 1, Humans, Liver Neoplasms, Male
Background
As one of the leading causes of cancer-related death worldwide, the incidence and mortality rates of hepatocellular carcinoma (HCC) are quite high in China [1]. Currently, although great improvements have been made in treating this malignancy, the prognosis of HCC patients still remains poor. Approximately 30–50% of HCC patients have recurrence and metastasis within 5 years after curative resection [2,3]. Therefore, a reliable biomarker is urgently needed during clinical diagnosis to provide advanced treatment for patients.
Discoidin domain receptor tyrosine kinase 1 (DDR1) is a member of the receptor tyrosine kinase (RTK) family [4], and its main function is to combine with ECM components to perform further biological functions [5,6]. It is highly expressed in epithelial cells and tissues, and acts as a cellular sensor, which can also trigger several transmembrane receptors, such as TGF-β and Notch and other adhesive receptors [7]. Consistently, accumulating evidence shows that DDR1 participates in cell physiological and pathophysiological process that regulate tumor cells progression, including polarity, adhesion, proliferation, and migration [8,9]. Recent studies revealed that activated DDR1 can stimulate matrix metalloproteinase (MMP) expression and activation [10,11], suggesting that DDR1 plays a vital role in tumor metastasis. Interestingly, after activation, DDR1 is secreted into peripheral blood, which strongly suggests DDR1 is a promising biomarker for HCC. However, the clinical significance of DDR1 in HCC is not fully clarified.
Hence, we conducted a retrospective study to evaluated the prognostic value of DDR1 in pretreatment serum and tumors, and found a correlation between DDR1 and pathological parameters.
Material and Methods
ENROLLMENT AND SPECIMEN COLLECTION:
We recruited 120 patients with HCC who received curative resections at the Huashan Hospital, Fudan University from January 2015 to September 2016, as well as 40 patients with chronic hepatitis and 40 health individuals without history of cancer or cardiovascular disease. In the clinic, the diagnosis of HCC was based on biochemistry tests and magnetic resonance imaging (MRI). Simultaneously, 2 senior pathologists performed further histopathology to confirm the diagnosis according to the guidelines of the American Association for the Study of Liver Diseases [12]; staging was defined by Barcelona Clinic Liver Cancer (BCLC) guidelines, and BCLC 0+A was considered to be early-stage disease. Tumor differentiation was defined based on the Edmondson grading system. The Child-Pugh scoring system was used to access liver function. This study was approved by the Research Ethics Committee of Huashan Hospital. The approval for the use of human subjects was obtained from the Research Ethics Committee of Huashan Hospital (approval number 2014-6-5), and written informed consent was received from every patient and healthy individual in this study.
SAMPLE MANAGEMENT:
Peripheral blood samples of HCC patients were obtained in the morning before their surgery, and the samples from chronic hepatitis patients and healthy individuals were collected in the morning as well. The blood sample was centrifuged at 3000 rpm for 15 min immediately after being accepted from the ward, and about 2 ml of serum was obtained from each participant. The serum samples were stored at −80°C before analysis. DDR1 concentration levels were measured using an enzyme-linked immunosorbent assay (ELISA) kit (Sino Biological, China).
TISSUE MICROARRAY (TMA):
Tumor tissues and paired para-tumor normal tissues were collected for immunohistochemical staining (IHC) by tissue microarray. The dilution of antibody including DDR1, E-cadherin, N-cadherin, and vimentin (Cell Signaling Technology, USA), was 1: 1000. TMA was cut into 4-μm-thick sections and paraffin-embedded. All slides were incubated with the primary antibodies for 12 h at 4°C, followed by incubation with secondary antibody (rabbit anti-mouse) for 1 h on the next day. Phosphate-buffered saline was used instead of the primary antibody for negative controls. Two senior pathologists were responsible for the results of IHC. The IHC scores were determined based on staining proportion and intensity. The staining proportion was scored as follows: <10% scored 0 (negative), 10–40% scored 1 (weakly positive), 40–70% scored 2 (moderately positive), and >70% scored 3 (strongly positive). The staining intensity was scored as follows: 0 for negative, 1 for weak, 2 for intermediate, and 3 for strong. The final IHS scores were the sum of the intensity and proportion scores, which was divided into 7 categories (0–2 was considered as low level, while 3–6 was considered high level).
PATIENT FOLLOW-UP:
The patients were monitored by serum testing (including AFP, ALT, AST, DDR1) every month and abdomen ultrasonography every 2 months after the surgery. Time to recurrence (TTR) of HCC was the interval between curative resection and recurrence or metastasis that was confirmed based on imaging and serologic testing.
STATISTICAL ANALYSIS:
The statistical analysis was performed using SPSS 20.0 software (IBM, Chicago, USA). The Figure was created using GraphPad Prism 8 (GraphPad Software, CA, USA). The experimental results are expressed in the form of mean±SD. Differences between the 2 groups were analyzed using Fisher’s exact probability test, the chi-square test, and the
Results
CLINICAL CHARACTERISTICS OF INDIVIDUALS ENROLLED IN THE STUDY:
A total of 200 individuals were enrolled in the research, including 120 HCC patients who receiving curative resection, 40 chronic hepatitis patients, and 40 healthy donors. The mean age of the HCC patients was 52 years (range, 45–60), 53 (range, 42–64) for chronic hepatitis patients, and 53 (range, 45–57) for healthy individuals. A total of 68 HCC patients had recurrence and distant metastasis. The median follow-up period was 26.1 months (range, 7.8–42.0 months). The clinical characteristics of patients and health individuals are summarized in Table 1.
DDR1 WAS SIGNIFICANTLY ELEVATED IN HCC PATIENTS:
To evaluate the clinical significance of DDR1 in HCC patients, we first used an ELISA kit to measure the contents of DDR1 in serum. The result showed DDR1 was dramatically higher in HCC patients (523.23±76.33 pg/ml) compared with chronic hepatitis patients (331.45±65.21 pg/ml) and health individuals (177.87±61.19 pg/ml) (P<0.001) (Figure 1A), suggesting that serum DDR1 can distinguish HCC patients from hepatitis patients and health individuals. Meanwhile, DDR1 expression levels in serum from HCC patients were continuously monitored after surgery. The results showed DDR1 decreased significantly with tumor burden removed and alleviation from the second month after patients received curative resection (Figure 1A).
IHC was used to evaluate the expression of DDR1 in tumor tissue and para-tumor tissues in HCC patients. Interestingly, DDR1was upregulated in most tumor tissues; 59.17% of HCC patients were classified as DDR1 high expression, and 40.83% were considered as low DDR1 expression. Meanwhile, 65.83% para-tumors were considered as DDR1 low expression and 34.17% were high expression, which was a significant difference (Figure 1B). Further investigation through logistic regression analysis showed DDR1 expression in serum had a strong correlation with that in tumor tissue (P<0.001), suggesting that HCC patients who had high DDR1 expression in tumors were very likely to have a high expression level in serum (Figure 1C). These results reveal that DDR1 in serum was greatly elevated in HCC patients and it might originate from tumor tissue.
INCREASED EXPRESSION OF DDR1 IN SERUM AND TUMOR PREDICTED POOR PROGNOSIS:
The clinical significance of DDR1 in HCC patients’ prognosis was further investigated. As planned, the HCC patients were stratified into 2 groups – the high-DDR1 group and the low-DDR1 group – based on the cutoff value of DDR1 (514.77 pg/ml) in serum results from X-tile analysis, and we found 61.98% of patients had recurrence or distant metastasis in the high-DDR1 group, while only 49.98% of patients had tumor recurrence in the low-DDR1 group. Consistently, the same trend was also observed in the high- and low-DDR1 tumor expression groups (Figure 2A). Consistently, ROC curve analysis demonstrated that DDR1 in serum and in tumors had a large AUC-ROC (DDR1 in serum AUC=0.654, 0.513–0.795; DDR1 in tumor AUC=0.685, 0.553–0.817) for predicting recurrence among all variates investigated (Figure 2B).
Kaplan-Meier analysis showed HCC patients with high level of DDR1 in serum had shorter TTR, as well as those patients with high DDR1 expression in tumors. Next, the prognostic value of DDR1 in HCC low-risk groups (the AFP ≤400 ng/ml group, single-tumor group, and tumor size ≤5 cm group) was further analyzed, and we found that DDR1 in serum and in tumors retained the ability to distinguished the HCC patients who were more likely to have recurrence (Figure 3A, 3B).
THE CORRELATION BETWEEN DDR1 AND CLINICOPATHOLOGICAL PARAMETERS:
We next investigated the association between DDR1 and other clinicopathological parameters. The results are shown in Table 2. HCC patients with high DDR1 in serum or in tumors tended to have higher possibility of microvascular invasion (MVI) and more circulating tumor cells (CTCs), while there was no difference in other clinicopathological parameters, including age, sex, AFP, cirrhosis, ALT, tumor number, tumor size, satellite lesion, Edmondson stage, and BCLC stage. This result suggested that patients with high DDR1 expression tended to have a tumor phenotype that presented more aggressively. Thus, we further evaluated whether epithelial-mesenchymal transition (EMT)-related protein had a relationship with DDR1. The results showed that DDR1 expression had a positive correlation with N-cadherin (P=0.015) and vimentin (P=0.022) and a negative correlation with E-cadherin (P=0.023) (Figure 4A, 4B).
DDR1 IS A PROMISING MARKER OF PROGNOSIS FOR HCC PATIENTS:
Univariate and multivariate Cox regression analyses were applied for analyzing independent risk factors for recurrence in the HCC patients. Univariate analyses showed that AFP (HR: 1.774, 95%, 1.100–2.862, P=0.019), tumor size (HR: 3.426, 95%, 2.090–5.615, P=0.000), tumor number (HR: 1.783, 95%, 1.092–2.912, P=0.021), DDR1 in serum (HR: 1.902, 95%, 1.150–3.146, P=0.012) and DDR1 in tumors (HR: 2.199, 95%, 1.275–3.790, P=0.005) were correlated with HCC recurrence. Multivariate analyses showed tumor number (HR: 2.855, 95%, 1.686–4.636, P=0.000), DDR1 in serum (HR: 1.520, 95%, 1.078–2.640, P=0.045), and DDR1 in tumors (HR: 1.811, 95%, 1.117–3.241, P=0.032) were independent predictors for TTR (Table 3).
Discussion
DDR1 is a type of tyrosine kinase receptor, which contains a discoidin homology (DS) domain in its extracellular region, and it can be effectively activated by collagen [13,14]. Unlike typical RTKs, after binding collagens, DDR1 starts autophosphorylation with very slow and sustained kinetics [15], then breaking away from the cell surface and slowly entering the peripheral circulation. This might explain why DDR1 in serum had a close correlation with that in tumors. We also found that after patients received curative resection, DDR1 in peripheral circulation decreased dramatically, further suggesting that a large amount of DDR1 in peripheral circulation of HCC patients comes from tumor tissues. To the best of our knowledge, we were the first team to find an association between DDR1 in circulation and tumor tissue. This discovery suggests DDR1 is an effective biomarker for HCC recurrence.
However, previous studies have demonstrated that DDR1 participates in progression of multiple cancers, including breast, head, and neck, lung, brain, esophagus, and prostate cancers [16,17]. Dysregulation and overexpression of DDR1 was reported to be correlated with unfavorable outcomes [18]. Here, we showed that DDR1 in serum was significantly higher in HCC patients than in people with chronic hepatitis and health individuals, and the same trend was also observed in cancerous and normal liver tissue, suggesting the potential role of DDR1 in HCC prognosis. Further research indicated that DDR1 in serum and in tumor could be independent risk factors for HCC recurrence, and DDR1 in tumor tissues showed a better discrimination ability.
Clinically, tumor progression is usually monitored by serological testing, among which AFP is one of the most widely used biomarker for evaluating the status of HCC patients. However, it was challenging to monitor patients with low AFP levels, as well as those in the single-tumor group, small tumor size group, and BCLC 0-A groups; over 50% of these patients would still have recurrence and distant metastasis after surgery [19]. We found DDR1 retains its prognostic ability in those low-risk patients. In summary, our data show that DDR1 is a powerful and effective biomarker for HCC recurrence, and it might play a vital role in tumor management when combined with other monitoring methods such as AFP testing, CT or MRI.
Next, we analyzed the unfavorable correlation between DDR1 and clinicopathological parameters. Intriguingly, DDR1 is associated with MVI and CTC. Accumulating clinical evidence confirms that MVI and CTC are related with the poor outcome of HCC patients [20–24]. Some investigators even suggested referral for liver transplantation of all patients who were found to have MVI after hepatic resection, due to the significant risk of recurrence [25]. However, MVI is a pathological characteristic, only visible in resected HCC tissue or based on biopsy, which poses a serious problem in clinical practice. Since we found that DDR1 in serum had a close correlation with MVI, measurement of DDR1 in serum would make it possible to predict the MVI without performing complex surgery, which would help to improve the clinical management of HCC patients. Meanwhile, CTC was reported originating from solid tumors with epithelial–mesenchymal transition. When shedding from a tumor and entering the circulation, CTC is transferred to distant sites, causing local or distant metastasis. In this process, EMT plays an important role in generating CTCs. Coincidently, DDR1 has been reported to upregulate matrix metalloproteinase (MMP) expression after binding collagen, and further affects cell migration ability [26]. Zhao et al showed that overexpression of DDR1 in prostate cancer cells activated the ERK signaling pathway, thus increasing tumor invasion and metastasis [27]. While, Wang et al and Lin et al found that DDR1 can activate the STAT3 pathway, which drives EMT and increases the expressions of MMPs in tumor progression [28,29].
Therefore, we investigated the correlation between EMT-related protein and DDR1 in HCC, and we found DDR1 was positively correlated with N-cadherin and vimentin, and it was negatively correlated with E-cadherin. These results indicate that DDR1 might influence EMT-related protein expression to promote tumor recurrence and metastasis. However, the underlying mechanism needs to be further investigated in HCC.
Conclusions
In summary, our research showed that DDR1 had a great prognostic value and close correlation with tumor progression, as a new independent risk factor for HCC recurrence and metastasis. It also has potential to predict MVI before surgery, which provides convenience and enhanced accuracy in clinical practice. However, this study still has certain limitations. First of all, the number of patients and healthy people recruited in the study was limited, and this was only a single-center retrospective study. Although we found DDR1 can promote HCC progression through the mechanism of EMT, we were still not able to clarify the details. Since we found satisfactory performance of DDR1, our team will conduct more researches on it.
Figures
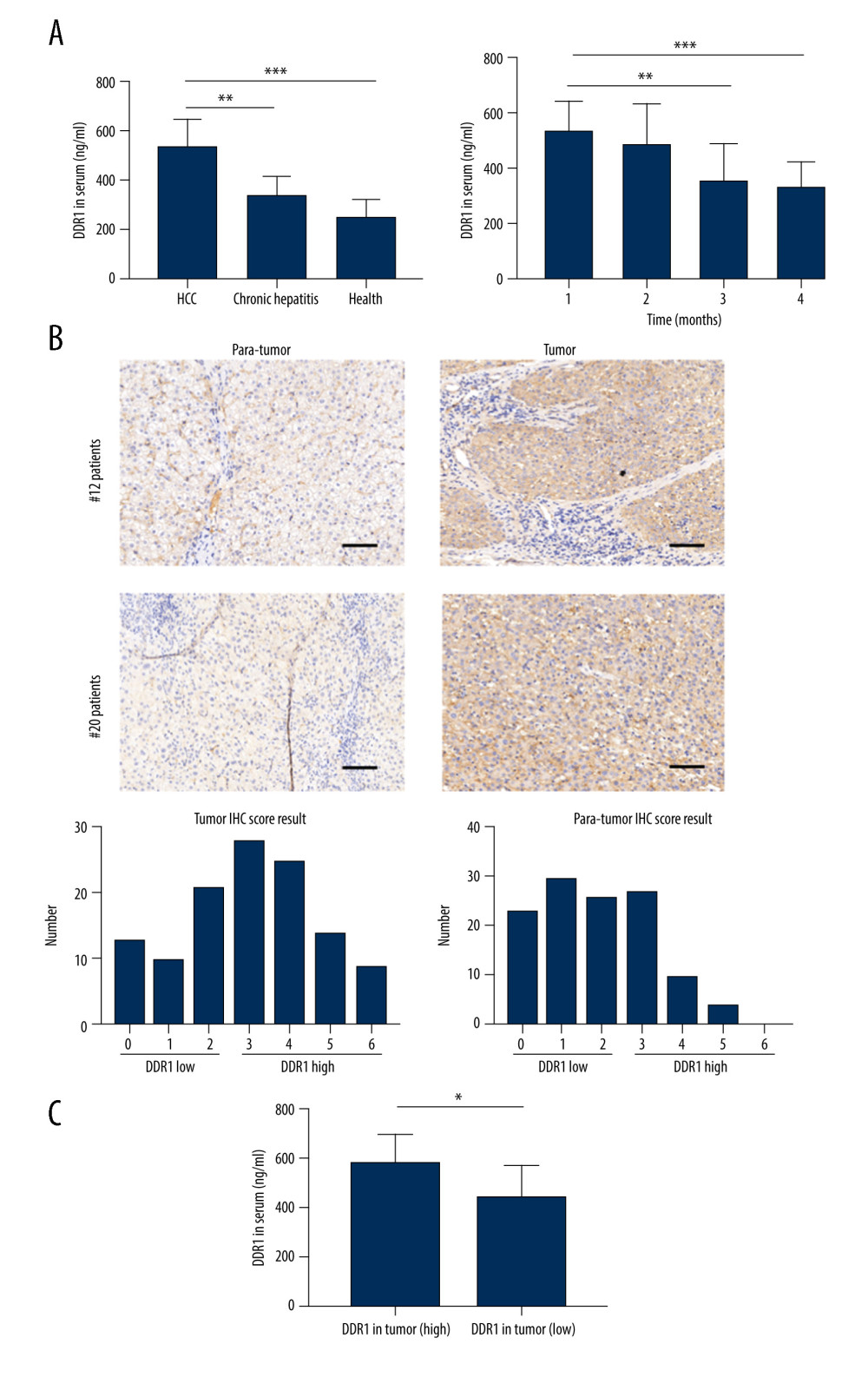 Figure 1. The expression of DDR1 was increased in serum and tumor in HCC patients. (A) Distributions of serum DDR1 in HCC patients (n=120), CHB patients (n=40), and healthy donors (n=40) and its expression level after HCC patients receiving curative resection. (B) The expression of DDR1 in tumor and para-tumor tissues in HCC patients. (C) Strong correlation between DDR1 in serum and in tumors. Scar bar: 100 nm.
Figure 1. The expression of DDR1 was increased in serum and tumor in HCC patients. (A) Distributions of serum DDR1 in HCC patients (n=120), CHB patients (n=40), and healthy donors (n=40) and its expression level after HCC patients receiving curative resection. (B) The expression of DDR1 in tumor and para-tumor tissues in HCC patients. (C) Strong correlation between DDR1 in serum and in tumors. Scar bar: 100 nm. 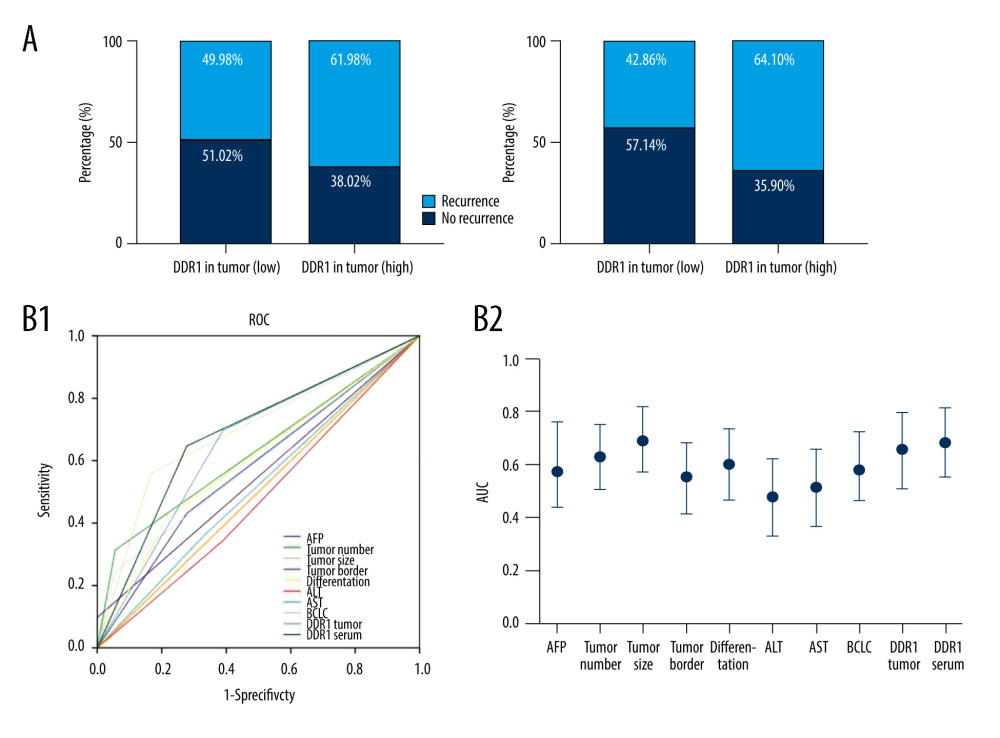 Figure 2. Increased DDR1 in serum and in tumors was associated with poor outcome of HCC patients. (A) HCC patients with high expression of DDR1 trended to have high possibility of recurrence and metastasis. (B) ROC analysis of different variables for predicting recurrence (B1) and AUC (B2).
Figure 2. Increased DDR1 in serum and in tumors was associated with poor outcome of HCC patients. (A) HCC patients with high expression of DDR1 trended to have high possibility of recurrence and metastasis. (B) ROC analysis of different variables for predicting recurrence (B1) and AUC (B2). 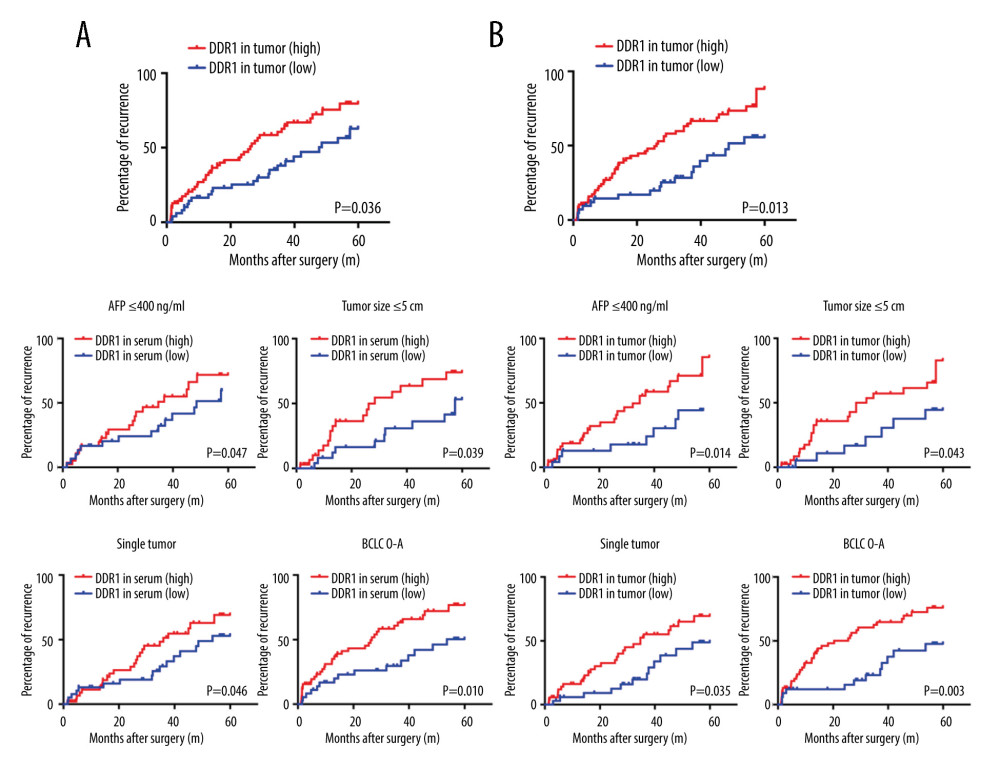 Figure 3. DDR1 was a promising marker of prognosis for HCC patients in the low-risk HCC group. (A) Kaplan-Meier analysis of TTR evaluated with DDR1 expression in serum and tumors. (B) Kaplan-Meier analyses of TTR according to DDR1 in the low AFP (≤400 ng/ml) group, the single-tumor group, the tumor size ≤5 cm group, and the BCLC 0-A group.
Figure 3. DDR1 was a promising marker of prognosis for HCC patients in the low-risk HCC group. (A) Kaplan-Meier analysis of TTR evaluated with DDR1 expression in serum and tumors. (B) Kaplan-Meier analyses of TTR according to DDR1 in the low AFP (≤400 ng/ml) group, the single-tumor group, the tumor size ≤5 cm group, and the BCLC 0-A group. 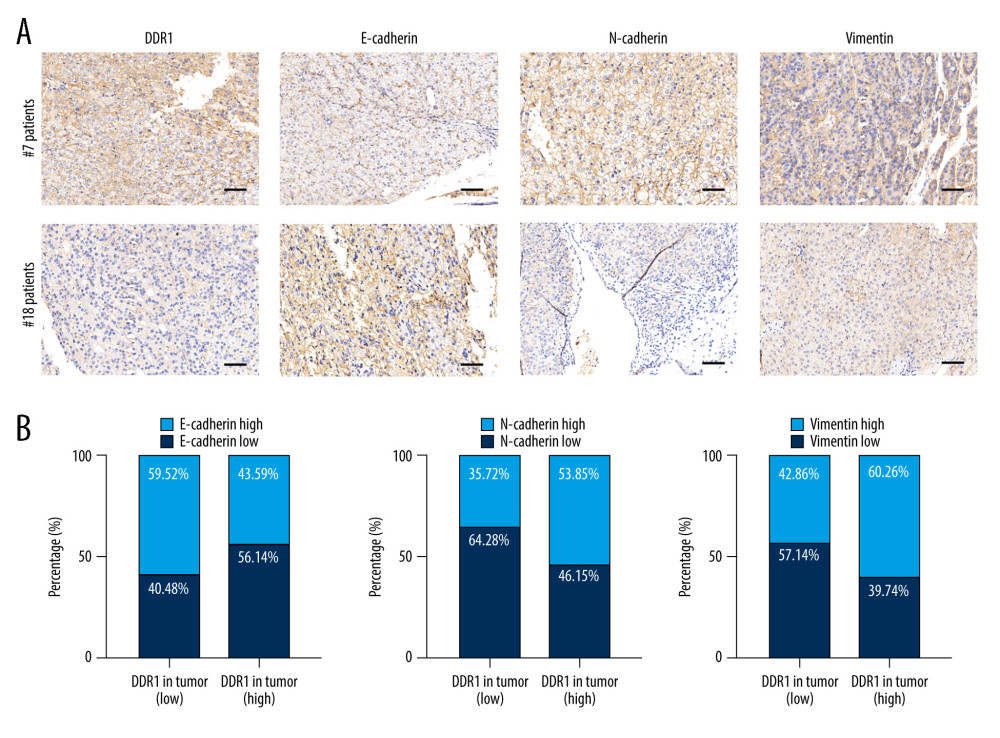 Figure 4. The correlation between DDR1 and EMT-related protein. (A) Representative images of E-cadherin, N-cadherin, vimentin, and DDR1 immunohistochemistry in tissue microarray slides are shown. (B) Patients with high DDR1 tended to have higher N-cadherin and vimentin and lower E-cadherin. Scar bar: 100 nm.
Figure 4. The correlation between DDR1 and EMT-related protein. (A) Representative images of E-cadherin, N-cadherin, vimentin, and DDR1 immunohistochemistry in tissue microarray slides are shown. (B) Patients with high DDR1 tended to have higher N-cadherin and vimentin and lower E-cadherin. Scar bar: 100 nm. References
1. Lu C, Rong D, Zhang B, Current perspectives on the immunosuppressive tumor microenvironment in hepatocellular carcinoma: Challenges and opportunities: Mol Cancer, 2019; 18; 130
2. Bruix J, Reig M, Sherman M, Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma: Gastroenterology, 2016; 150; 835-53
3. Galle PR, Tovoli F, Foerster F, The treatment of intermediate stage tumours beyond TACE: from surgery to systemic therapy: J Hepatol, 2017; 67; 173-83
4. Valiathan RR, Marco M, Leitinger B, Discoidin domain receptor tyrosine kinases: New players in cancer progression: Cancer Metastasis Rev, 2012; 31; 295-321
5. Shrivastava A, Radziejewski C, Campbell E, An orphan receptor tyrosine kinase family whose members serve as nonintegrin collagen receptors: Mol Cell, 1997; 1; 25-34
6. Vogel W, Gish GD, Alves F, Pawson T, The discoidin domain receptor tyrosine kinases are activated by collagen: Mol Cell, 1997; 1; 13-23
7. Gao H, Chakraborty G, Zhang Z, Multi-organ site metastatic reactivation mediated by non-canonical discoidin domain receptor 1 signaling: Cell, 2016; 166; 47-62
8. Vogel WF, Aszódi A, Alves F, Pawson T, Discoidin domain receptor 1 tyrosine kinase has an essential role in mammary gland development: Mol Cell Biol, 2001; 21; 2906-17
9. Labrador JP, Azcoitia V, Tuckermann J, The collagen receptor DDR2 regulates proliferation and its elimination leads to dwarfism: EMBO Rep, 2001; 2; 446-52
10. Hou G, Vogel WF, Bendeck MP, Tyrosine kinase activity of discoidin domain receptor 1 is necessary for smooth muscle cell migration and matrix metalloproteinase expression: Circ Res, 2002; 90; 1147-49
11. Ferri N, Carragher NO, Raines EW, Role of discoidin domain receptors 1 and 2 in human smooth muscle cell-mediated collagen remodeling: potential implications in atherosclerosis and lymphangioleiomyomatosis: Am J Pathol, 2004; 164; 1575-85
12. Bruix J, Sherman M, Management of hepatocellular carcinoma: Hepatology, 2005; 42; 1208-36
13. Leitinger B, Discoidin domain receptor functions in physiological and pathological conditions: Int Rev Cell Mol Biol, 2014; 310; 39-87
14. Kadler KE, Baldock C, Bella J, Boot-Handford RP, Collagens at a glance: J Cell Sci, 2007; 120; 1955-58
15. Shrivastava A, Radziejewski C, Campbell E, An orphan receptor tyrosine kinase family whose members serve as nonintegrin collagen receptors: Mol Cell, 1997; 1; 25-34
16. Valiathan RR, Marco M, Leitinger B, Discoidin domain receptor tyrosine kinases: New players in cancer progression: Cancer Metastasis Rev, 2012; 31; 295-321
17. Jian ZX, Sun J, Chen W, Involvement of discoidin domain 1 receptor in recurrence of hepatocellular carcinoma by genome-wide analysis: Med Oncol, 2012; 29; 3077-82
18. Cader FZ, Vockerodt M, Bose S, The EBV oncogene LMP1 protects lymphoma cells from cell death through the collagen-mediated activation of DDR1: Blood, 2013; 122; 4237-45
19. Yamao T, Yamashita YI, Imai K, Clinical significance of preoperative hepatocellular carcinoma with high lens culinaris agglutinin-reactive fraction of alpha-fetoprotein, but low alpha-fetoprotein: Anticancer Res, 2019; 39; 883-89
20. Finn RS, Zhu AX, Farah W, Therapies for advanced stage hepatocellular carcinoma with macrovascular invasion or metastatic disease: A systematic review and meta-analysis: Hepatology, 2018; 67; 422-35
21. Banerjee S, Wang DS, Kim HJ, A computed tomography radiogenomic biomarker predicts microvascular invasion and clinical outcomes in hepatocellular carcinoma: Hepatology, 2015; 62; 792-800
22. Lee S, Kim SH, Lee JE, Preoperative gadoxetic acid-enhanced MRI for predicting microvascular invasion in patients with single hepatocellular carcinoma: J Hepatol, 2017; 67; 526-34
23. Ahn JC, Teng PC, Chen PJ, Detection of circulating tumor cells and their implications as a novel biomarker for diagnosis, prognostication, and therapeutic monitoring in hepatocellular carcinoma: Hepatology, 2021; 73; 422-36
24. Qi LN, Xiang BD, Wu FX, Circulating tumor cells undergoing EMT provide a metric for diagnosis and prognosis of patients with hepatocellular carcinoma: Cancer Res, 2018; 78; 4731-44
25. Sala M, Fuster J, Llovet JM, High pathological risk of recurrence after surgical resection for hepatocellular carcinoma: An indication for salvage liver transplantation: Liver Transpl, 2004; 10; 1294-300
26. Page-McCaw A, Ewald AJ, Werb Z, Matrix metalloproteinases and the regulation of tissue remodelling: Nat Rev Mol Cell Biol, 2007; 8; 221-33
27. Zhao Z, Zhao S, Luo L, miR-199b-5p-DDR1-ERK signalling axis suppresses prostate cancer metastasis via inhibiting epithelial-mesenchymal transition: Br J Cancer, 2021; 124(5); 982-94
28. Wang Z, Sun X, Bao Y, E2F1 silencing inhibits migration and invasion of osteosarcoma cells via regulating DDR1 expression: Int J Oncol, 2017; 51(6); 1639-50
29. Lin Y, Jin H, Wu X, The cross-talk between DDR1 and STAT3 promotes the development of hepatocellular carcinoma: Aging (Albany NY), 2020; 12(14); 14391-405
Figures
 Figure 1. The expression of DDR1 was increased in serum and tumor in HCC patients. (A) Distributions of serum DDR1 in HCC patients (n=120), CHB patients (n=40), and healthy donors (n=40) and its expression level after HCC patients receiving curative resection. (B) The expression of DDR1 in tumor and para-tumor tissues in HCC patients. (C) Strong correlation between DDR1 in serum and in tumors. Scar bar: 100 nm.
Figure 1. The expression of DDR1 was increased in serum and tumor in HCC patients. (A) Distributions of serum DDR1 in HCC patients (n=120), CHB patients (n=40), and healthy donors (n=40) and its expression level after HCC patients receiving curative resection. (B) The expression of DDR1 in tumor and para-tumor tissues in HCC patients. (C) Strong correlation between DDR1 in serum and in tumors. Scar bar: 100 nm. Figure 2. Increased DDR1 in serum and in tumors was associated with poor outcome of HCC patients. (A) HCC patients with high expression of DDR1 trended to have high possibility of recurrence and metastasis. (B) ROC analysis of different variables for predicting recurrence (B1) and AUC (B2).
Figure 2. Increased DDR1 in serum and in tumors was associated with poor outcome of HCC patients. (A) HCC patients with high expression of DDR1 trended to have high possibility of recurrence and metastasis. (B) ROC analysis of different variables for predicting recurrence (B1) and AUC (B2). Figure 3. DDR1 was a promising marker of prognosis for HCC patients in the low-risk HCC group. (A) Kaplan-Meier analysis of TTR evaluated with DDR1 expression in serum and tumors. (B) Kaplan-Meier analyses of TTR according to DDR1 in the low AFP (≤400 ng/ml) group, the single-tumor group, the tumor size ≤5 cm group, and the BCLC 0-A group.
Figure 3. DDR1 was a promising marker of prognosis for HCC patients in the low-risk HCC group. (A) Kaplan-Meier analysis of TTR evaluated with DDR1 expression in serum and tumors. (B) Kaplan-Meier analyses of TTR according to DDR1 in the low AFP (≤400 ng/ml) group, the single-tumor group, the tumor size ≤5 cm group, and the BCLC 0-A group. Figure 4. The correlation between DDR1 and EMT-related protein. (A) Representative images of E-cadherin, N-cadherin, vimentin, and DDR1 immunohistochemistry in tissue microarray slides are shown. (B) Patients with high DDR1 tended to have higher N-cadherin and vimentin and lower E-cadherin. Scar bar: 100 nm.
Figure 4. The correlation between DDR1 and EMT-related protein. (A) Representative images of E-cadherin, N-cadherin, vimentin, and DDR1 immunohistochemistry in tissue microarray slides are shown. (B) Patients with high DDR1 tended to have higher N-cadherin and vimentin and lower E-cadherin. Scar bar: 100 nm. Tables
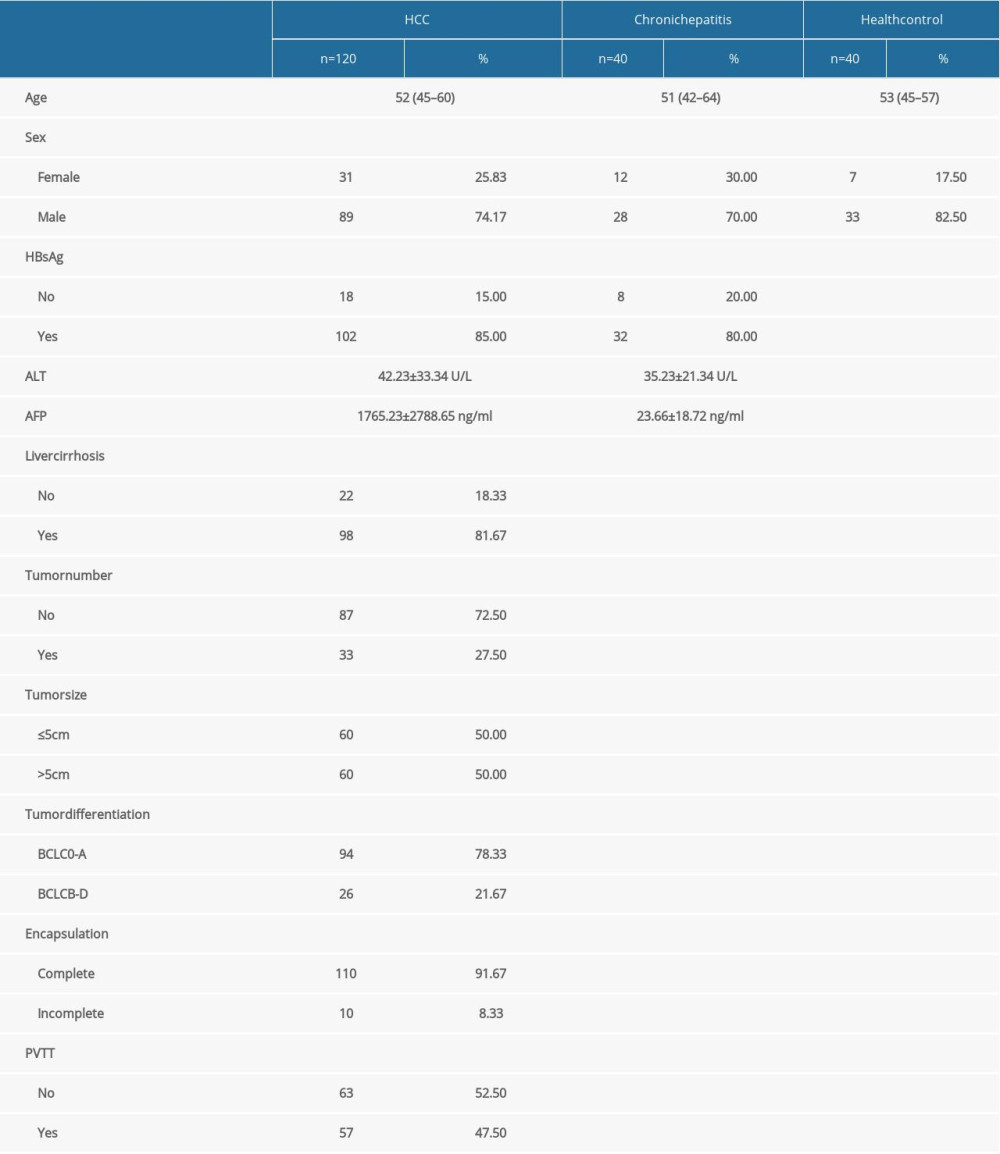 Table 1. Clinical characteristic of individuals enrolled in the study.
Table 1. Clinical characteristic of individuals enrolled in the study.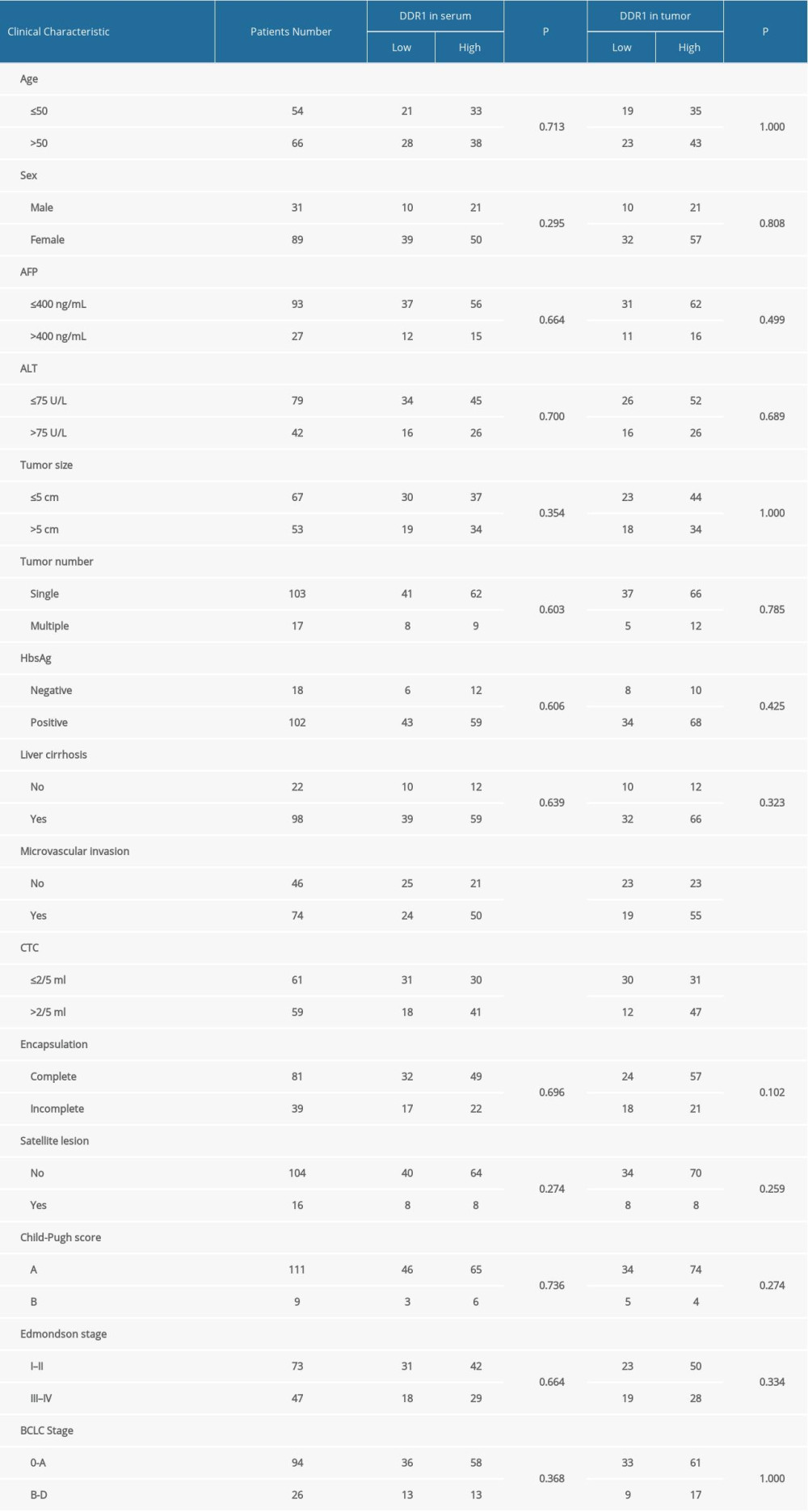 Table 2. Correlation between DDR1 and clinicopathologic characteristics.
Table 2. Correlation between DDR1 and clinicopathologic characteristics.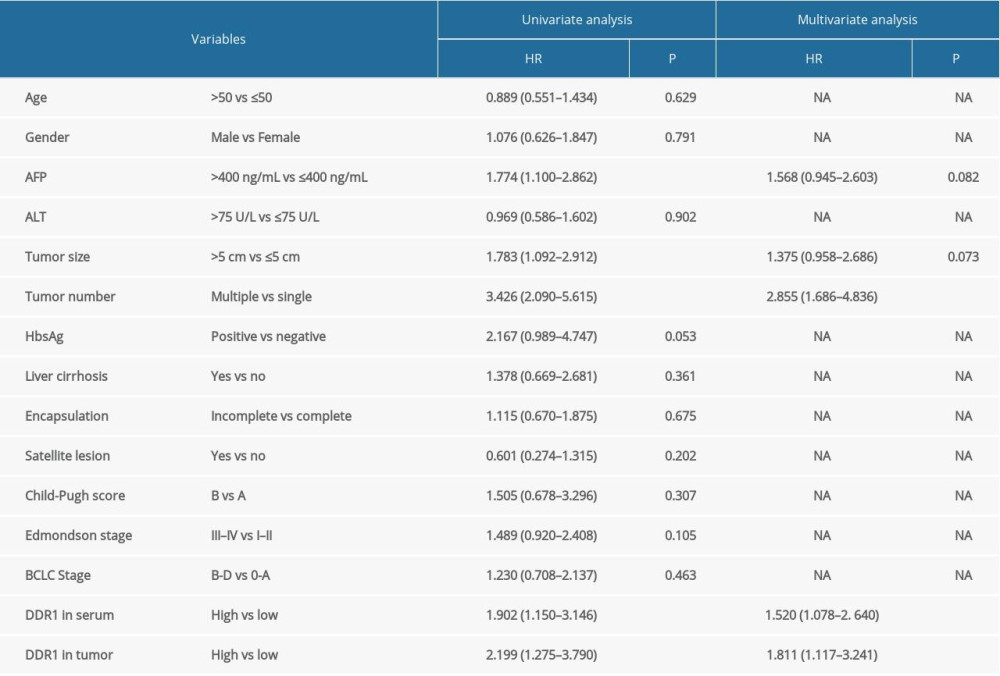 Table 3. Univariate and multivariate Cox regression analysis of factors associated with recurrence.
Table 3. Univariate and multivariate Cox regression analysis of factors associated with recurrence. Table 1. Clinical characteristic of individuals enrolled in the study.
Table 1. Clinical characteristic of individuals enrolled in the study. Table 2. Correlation between DDR1 and clinicopathologic characteristics.
Table 2. Correlation between DDR1 and clinicopathologic characteristics. Table 3. Univariate and multivariate Cox regression analysis of factors associated with recurrence.
Table 3. Univariate and multivariate Cox regression analysis of factors associated with recurrence. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








