31 January 2022: Meta-Analysis
Utility of Pretreatment Blood Platelet-To-Lymphocyte Ratio in Prediction of Clinical Outcomes and Chemosensitivity in Patients with Advanced Gastric Cancer: A Meta-Analysis
Xiulan Peng1AE*, Wei Zeng2BF, Bing Tang3CF, Anbing He1B, Min Zhang1B, Renfeng Luo4ABDOI: 10.12659/MSM.933449
Med Sci Monit 2022; 28:e933449
Abstract
BACKGROUND: The results of previous studies that evaluated the association between pretreatment blood platelet-to-lymphocyte ratio (PLR) and clinical outcomes and chemosensitivity in patients with advanced gastric cancer are inconsistent. Therefore, this study was designed to investigate the association between pretreatment blood PLR and clinical outcomes and chemosensitivity in advanced gastric cancer patients.
MATERIAL AND METHODS: We performed a systematic literature search in PubMed, Web of Science, EMBASE, and the Cochrane Library up to Mar 9, 2021. Hazard ratios (HRs) for overall survival (OS) and disease-free survival (DFS) were pooled for meta-analysis. The quality of the included studies was measured by the Newcastle-Ottawa Quality Assessment Scale.
RESULTS: We included 17 studies comprising 3499 patients with advanced GC in this meta-analysis. Pooled results demonstrated that high PLR was correlated with poor OS (HR=1.429, 95% CI=1.246-1.639, P<0.001) and DFS (HR=1.47, 95% CI=1.14-1.88, P=0.003) compared with low PLR in patients with advanced GC. Moreover, high PLR was associated with a lower response to chemotherapy in patients with advanced GC (OR=1.395, 95% CI=1.056-1.841, P=0.019). However, there was no significant correlation between PLR and clinicopathological features.
CONCLUSIONS: This meta-analysis suggests that high PLR is a risk factor for unfavorable OS, DFS, and chemosensitivity in patients with advanced GC.
Keywords: Stomach Neoplasms, Disease-Free Survival, Humans, Lymphocyte Count, Platelet Count
Background
Gastric cancer (GC), a global problem of human health, is the fifth most common malignancy and the third leading cause of cancer-related death worldwide [1]. Annually, there are more than 1 033 701 newly diagnosed patients with GC [2]. Tumor stage is the strongest prognostic factor for patients with GC; it is a potential curative disease at an early stage, but it is almost always diagnosed at the advanced stage of disease [3]. Although great progress has been made in the diagnosis and treatment of gastric cancer, the outcome of advanced gastric cancer is still extremely poor [4]. The role of surgical treatment still remains quite significant in conquering GC [5]. For patients with advanced GC, adequate surgical resection is not the only curative therapeutic regimen [6], and chemotherapy is regarded as an important part of or main treatment in advanced GC patients [7]. However, the response rate to first-line treatment is only 27–54%, and many patients who show no response to chemotherapy have fatal outcomes because of delayed effective therapy [8,9]. There are still no consistent and recognized biomarkers for chemotherapeutic drugs in advanced GC patients; therefore, a predictive biomarker for prognosis and prediction of chemosensitivity to improve therapeutic strategies in patients with advanced GC is required.
Studies have demonstrated that the prognosis of different types of cancer is influenced not only by its features, but also by the tumor microenvironment derived from the immunological state of the patient [10–12]. Recently, studies have assumed that the systemic inflammatory response has a critical role in the occurrence and progression of cancer, as well as in the response to therapy [13–16]. Pretreatment systemic inflammation response biomarkers, such as hypersensitive C-reactive protein (hs-CRP), PLT count, platelet-to-lymphocyte ratio (PLR), neutrophil-to-lymphocyte ratio (NLR), and lymphocyte-to-monocyte ratio (LMR) have been identified as prognostic indicators in a variety of malignant tumors, including GC [17–20]. Liu et al showed that PLR is associated with response to chemotherapy and OS in patients with advanced non-small cell lung cancer [21]. Among these prognostic biomarkers, the prognostic value of PLR in GC may be the most controversial [22,23]. The utility of PLR for prediction of prognosis and chemosensitivity in patients with advanced GC is unclear. Thus, we conducted a meta-analysis to evaluate the utility of PLR in prediction of prognosis and chemosensitivity, as well as the correlations between the pretreatment PLR and clinicopathological features, in patients with advanced GC.
Material and Methods
LITERATURE SEARCH:
This meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines [24] and it was conducted according to the mandatory 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and checklist. We conducted a comprehensive literature search in PubMed, Web of Science, EMBASE, and the Cochrane Library up to March 9, 2021. The search strategy was (“Neoplasm, Stomach” or “Stomach Neoplasm” or “Neoplasms, Stomach” or “Gastric Neoplasms” or “Gastric Neoplasm” or “Neoplasm, Gastric” or “Neoplasms, Gastric” or “Cancer of Stomach” or “Stomach Cancers” or “Gastric Cancer” or “Cancer, Gastric” or “Cancers, Gastric” or “Gastric Cancers” or “Stomach Cancer” or “Cancer, Stomach” or “Cancers, Stomach” or “Cancer of the Stomach” or “Gastric Cancer, Familial Diffuse”) AND (“PLR” or “platelet/lymphocyte ratio” or “platelet to lymphocyte ratio” or “platelet lymphocyte ratio” “platelet-to-lymphocyte ratio” or “platelet-lymphocyte ratio”). The references were manually examined to collect potentially relevant studies. Because this meta-analysis does not include individual patient information, ethics committee approval was not needed.
INCLUSION AND EXCLUSION CRITERIA:
The inclusion criteria were as follows: (1) stage III and IV gastric cancer diagnosis was histopathologically confirmed in patients; (2) hazard ratios (HRs) and corresponding 95% confidence intervals (CIs) of PLR with survival outcomes including overall survival (OS) and/or progression-free survival(PFS) were reported, or the association between PLR and chemosensitivity of advanced gastric cancer was reported, or the association between PLR and clinicopathological characteristics of gastric cancer was reported; (3) a definite cut-off value of pretreatment PLR was determined; and (4) full-text articles published in English.
The exclusion criteria were as follows: (1) duplicate publications; (2) reviews, editorials, conference abstracts, letters and case reports; (3) basic research or animal studies; (4) studies without sufficient data.
OUTCOME ASSESSMENT:
Our primary outcome was the correlation between PLR and OS, DFS, and chemosensitivity in advanced GC patients. Secondary outcomes included the association between PLR and clinicopathological features in advanced GC patients.
DATA COLLECTION AND QUALITY ASSESSMENT:
Studies assessing high PLR versus low PLR in predicting prognostic and chemosensitivity and survival of advanced gastric cancer patients were selected. Two investigators (WZ and BT) independently reviewed the articles retrieved in the literature search and extracted the relevant data. Any disagreements were resolved by discussion with a third reviewer (XP). The following data were independently collected from the retrieved articles by AH and RL: first author’s name, publication year, study country, study period, number of cases, age, sex, time of follow-up, treatment, cut-off value of PLR, prognostic endpoints (OS or DFS), HRs with corresponding 95% CIs, chemosensitivity, and clinicopathological characteristics.
Quality assessment was performed in accordance with the Newcastle-Ottawa scale (NOS) [25]. The NOS scores range from 0 to 9, and if the score was higher than 6, the study was considered to be high-quality. Two reviewers (BT and MZ) independently calculated the NOS scores, and any disagreements were resolved by discussion with a third reviewer (RL). The study was independently examined by 3 investigators (XP, WZ, and AH). Any disagreements were resolved by discussion with a third reviewer (RL). All authors reviewed the manuscript, any disagreements were resolved by discussion to approve the final manuscript.
STATISTICAL ANALYSIS:
The pooled HRs and 95% CIs were used to assess the prognostic role of PLR on the survival (OS/DFS) of patients with advanced GC. HR >1 with P<0.05 indicated a poor prognosis in patients with high PLR. Statistical heterogeneity among studies was determined by using the Cochran Q-test and I2 test [26,27]. I2 >50% or P<0.10 were considered to indicate significant heterogeneity. A random-effects model was used if substantial heterogeneity was detected among studies; otherwise, a fixed-effects model was applied.
The subgroup analysis was performed by ethnicity, cut-off value, cut-off selection, sample size, treatment, and NOS score. The odd ratios (ORs) and 95% CIs were calculated to assess the correlations between PLR and clinicopathological features and chemosensitivity of patients with advanced GC. Sensitivity analysis was conducted to evaluate the reliability of the results. Publication bias was examined by using the Begg’s test and Egger’s test.
Results
SEARCH RESULTS:
The detailed selection process of literature selection is shown in Figure 1. A total of 282 records were identified after the initial literature search, and 225 duplicate articles were removed. After examining the title and abstract, 34 articles were discarded. Subsequently, the full text of the remaining 17 articles were evaluated, and 6 were discarded for the following reasons: 5 studies lacked necessary data and 1 study did not include advanced-stage patients. Finally, 17 studies comprising 3499 patients with advanced GC were finally identified in the meta-analysis [8,28–43].
CHARACTERISTICS OF THE INCLUDED STUDIES:
The specific characteristics of 17 studies are summarized in Table 1. All selected studies were published between 2010 and 2021, and were conducted in 5 countries, including 1 in Italy [10], 1 in Korea [34], 2 in Turkey [31,32], 3 in Japan [29,39,42], and 10 in China [28,30,33,35–38,40,41,43]. There was only 1 prospective trail [40], and the sample sizes ranged from 33 to 537. The cut-off values for PLR in 17 studies ranged from 107.7 to 284. As for outcomes, 16 studies evaluated the association between PLR and OS [10,28–39,41–43], and 3 studies investigated the prognostic role of PLR in DFS [33,40,41]. Three studies investigated the association between the PLR and chemosensitivity [28–30], and 11 studies reported the correlation between the PLR and clinicopathological features [10,28,30,34,35,38–43]. In addition, the NOS scores of the quality of 17 included studies ranged from 6 to 9, indicating high quality.
OVERALL QUALITY OF THE INCLUDED STUDIES:
The Newcastle-Ottawa Scale was used to assess the quality of all the included studies. The NOS scores of the quality of 17 included studies ranged from 6 to 9, indicating high quality.
CORRELATION BETWEEN PLR AND OS IN ADVANCED GC PATIENTS:
Sixteen studies containing 3153 patients with advanced GC evaluated the prognostic value of PLR in OS [10,28–39,41–43]. As shown in Figure 2, moderate heterogeneity existed among the included studies, so a random-effects model was used (I2=54.1%, P=0.05, Figure 2 and Table 2). The median score was used as the cut-off value to categorize low or high PLR. The pooled HRs and 95% CIs demonstrated that patients with a high PLR were associated with poor OS in advanced GC (HR=1.429, 95% CI=1.246–1.639, P<0.001).
SUBGROUP ANALYSIS OF CORRELATION BETWEEN PLR AND OS:
Due to the moderate heterogeneity, we performed subgroup analyses to minimize the impact of differences among studies on the pooled results. Subgroup analysis was introduced for further investigation, stratified by ethnicity, cut-off value, cut-off selection, sample size, treatment, and NOS score. The data showed that there were significant correlations between high PLR and sample size, treatment, cut-off value, and NOS score (Table 2).
CORRELATION BETWEEN PLR AND DFS IN ADVANCED GC PATIENTS:
Three clinical studies investigated the association between PLR and DFS in resected GC [33,40,41]. Pooled results demonstrated that high PLR was correlated to poor DFS (HR=1.47, 95% CI=1.14–1.88, P=0.003) compared with low PLR in patients with advanced GC. As shown in Figure 3, a random-effects model was applied for analysis due to moderate heterogeneity (I2=59.8%, P=0.083).
SUBGROUP ANALYSIS OF CORRELATION BETWEEN PLR AND DFS:
Similar to the OS, subgroup analysis revealed that there was significant correlation between high PLR and ethnicity, sample size, and NOS score (Table 2).
ASSOCIATION BETWEEN PLR AND CHEMOSENSITIVITY:
The association between the PLR and chemosensitivity was analyzed from 3 studies [28–30]. As show in Figure 4, moderate heterogeneity existed among the included studies, so a random-effects model was used (I2=59.8%, P=0.083 Figure 4). The analysis revealed that low PLR was associated with a higher response to chemotherapy in patients with advanced GC (OR=1.395, 95% CI=1.056–1.841, P=0.019).
ASSOCIATION BETWEEN PLR AND CLINICOPATHOLOGICAL FEATURES:
To evaluate the associations between PLR and clinicopathological features of GC, we identified 6 clinical features. The features included sex (male versus female), age (years) (≥60 versus <60), tumor differentiation (poor versus moderate/well-differentiated), tumor size (≥5 cm versus <5 cm), CEA (≥5 versus <5), and ECOG (≥2 versus <2) (Table 3 and Figure 5). However, there was no significant correlation between PLR and clinicopathological features, sex (OR=0.940, 95% CI=0.794–1.112, P=0.468), age (OR=1.398, 95% CI=0.827–2.362, P=0.211), tumor differentiation (OR=0.985, 95% CI=0.902–1.075, P =0.734), tumor size (OR=1.130, 95% CI=0.567–2.253, P=0.729), CEA (OR=1.128, 95% CI=0.965–1.320, P=0.131), or ECOG (OR=1.355, 95% CI=0.492–3.731, P=0.557).
SENSITIVITY ANALYSIS:
Sensitivity analysis was conducted to evaluate the stability of pooled HRs for OS and DFS. As shown in Figure 6, the sensitivity analysis demonstrated the credibility of the results of this meta-analysis.
PUBLICATION BIAS:
The Begg’s funnel and Egger’s test were used to estimate the potential publication bias. The results showed that there was no significant bias for OS (Begg’s p=0.260, Egger’s p=0.346) or PFS (Begg’s P=1.000, Egger’s P=0.288) (Figure 7).
Discussion
Several studies have reported the association between PLR and the prognosis of gastric cancer, but they yielded conflicting results, and these studies including many early-stage GC patients [21,22,44,45]. Thus, the prognostic value of the PLR remains unclear in advanced GC, and there have no study in predictor of chemosensitivity value of PLR for advanced GC. Therefore, we conducted this meta-analysis to evaluate the ability of PLR to predict prognosis and chemosensitivity in patients with advanced GC. Based on the available data (N=3499), the results of the present meta-analysis have some clinical implications. Our meta-analysis demonstrated that higher PLR was associated with worse OS, PFS, and chemosensitivity in patients with advanced GC. Moreover, the association of an elevated PLR with OS remained substantial in studies with heterogeneous sample sizes, treatments, cut-off values, and NOS scores. However, we did not find a significant correlation between PLR and clinicopathological features in patients with advanced GC. As the PLR is a blood-derived parameter that is inexpensive and readily available, it could be routinely used to assess disease progression and chemosensitivity in patients with advanced GC. The results of our study may assist in predicting cancer prognosis, and, more importantly, help develop optimal treatment strategies.
PLR is based on peripheral platelet and lymphocyte counts. A high PLR corresponds to high platelet and/or low lymphocyte counts. The interplay between inflammatory responses and tumor invasion may suggest a possible explanation. Platelets are involved in the tumorigenesis and metastasis of cancer cells by secreting tumor growth factors [46]. Lymphocytes are an important component of the immune system and play a vital role in the suppression of tumor progression [47]. Based on the above evidence, it is reasonable to speculate that a normal range of PLR signifies the balance between the degree of tumor progression and the status of anti-tumor immunity. High PLR represents elevated platelets or decreased lymphocytes, implying the weakened anti-tumor immune response and rapid deterioration. Hence, it is reasonable that high PLR predicts unfavorable OS and DFS in advanced GC.
Systemic inflammation and tumor microenvironments play an important role in modulating chemotherapy resistance [48–50]. The association of high PLR with low chemotherapeutic efficacy could result from an elevated platelet count and/or a decreased lymphocyte count. Activated platelets create a procoagulant microenvironment that allows tumor cells to coat themselves with platelets and escaped the patients’ immune system [51]. It was shown that overexpression of TGF-β receptor 2 (TGFBR2) decreased 5-fluorouracil sensitivity of GC cells [52]. VEGF-C mediated cisplatin resistance in GC cells, while microRNA-101 that targeted VEGF-C could reverse the resistance [53]. It was reported that the expression of ERCC1 and GSTM1 positive genotype is predictive of chemotherapy treatment outcome in patients with advanced gastric cancer [54,55], but none of these factors is currently used clinically because they are invasive, required specific laboratory equipment and complex methodology. PLR has the advantages of simplicity, convenience, affordability, and reproducibility, and it may be helpful for guiding treatment and monitoring treatment efficacy.
Although we performed this meta-analysis according to the PRISMA guidelines, this meta-analysis still has several limitations. First, no randomized controlled trials were enrolled in this study and almost all included studies were retrospective, which may have caused some heterogeneity and higher I2 when integrated. Second, the cut-off value of PLR was not uniform among the included studies, which might be the main source of the substantial heterogeneity and higher I2. Third, 1 HR was extracted from univariate analyses, and may have overestimated effect sizes. In addition, only 3 studies assessed the ability of PLR to predict DFS and chemosensitivity, and this may have reduced the accuracy of the correlation between PLR and DFS and chemosensitivity. Prospective studies on this topic are needed.
Conclusions
Our study revealed that high pretreatment PLR was obviously associated with unfavorable OS, PFS, and chemosensitivity in patients with advanced GC. These results indicate that pretreatment PLR could be an easily accessible and cost-effective prognostic and clinical decision-making biomarker for advanced GC in clinical practice.
Figures
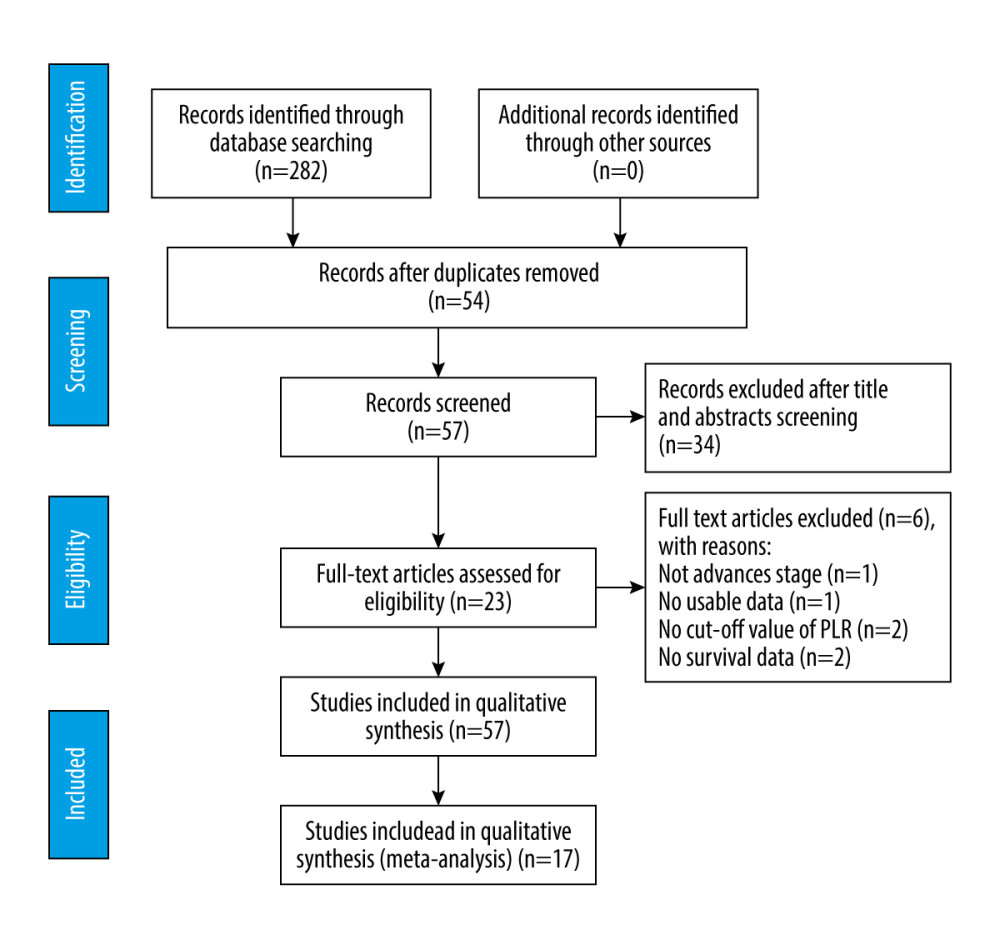 Figure 1. Flow chart of literature search and study selection.
Figure 1. Flow chart of literature search and study selection. 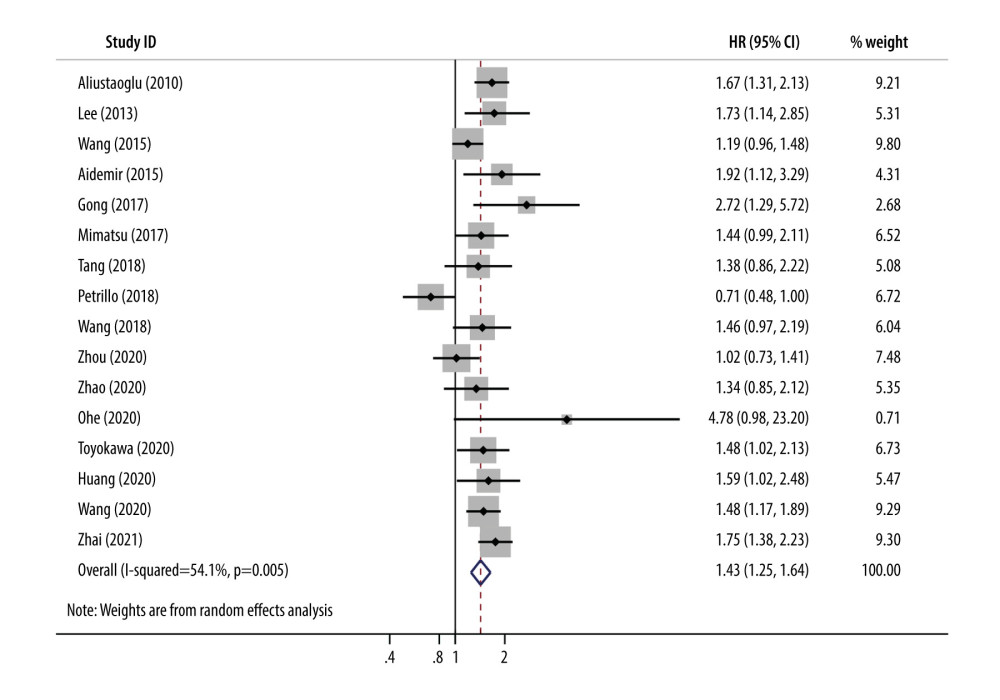 Figure 2. Forest plots for the association platelet to lymphocyte ratio and overall survival in patients with advanced gastric cancer (HR=1.429, 95% CI=1.246–1.639, P<0.001). CI – confidence interval; HR – hazard ratio.
Figure 2. Forest plots for the association platelet to lymphocyte ratio and overall survival in patients with advanced gastric cancer (HR=1.429, 95% CI=1.246–1.639, P<0.001). CI – confidence interval; HR – hazard ratio. 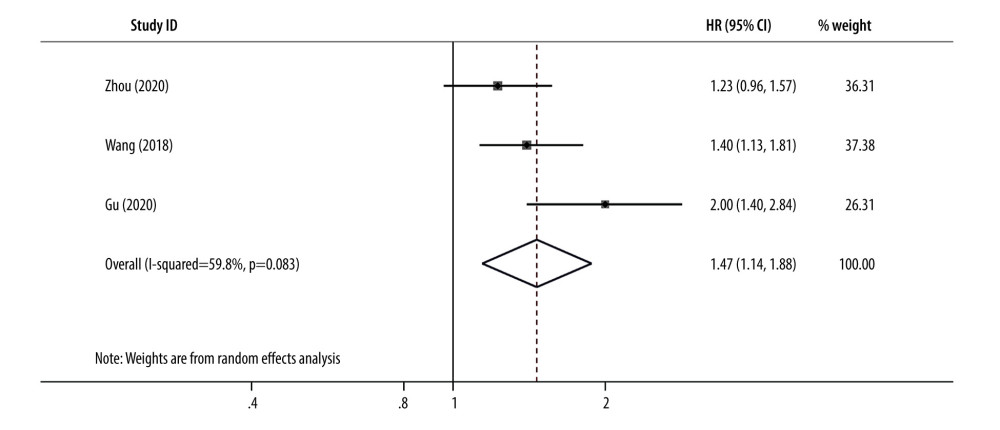 Figure 3. Forest plots for the association platelet to lymphocyte ratio and disease-free survival in patients with advanced gastric cancer (HR=1.47, 95% CI=1.14–1.88, P=0.003). CI – confidence interval; HR – hazard ratio.
Figure 3. Forest plots for the association platelet to lymphocyte ratio and disease-free survival in patients with advanced gastric cancer (HR=1.47, 95% CI=1.14–1.88, P=0.003). CI – confidence interval; HR – hazard ratio. 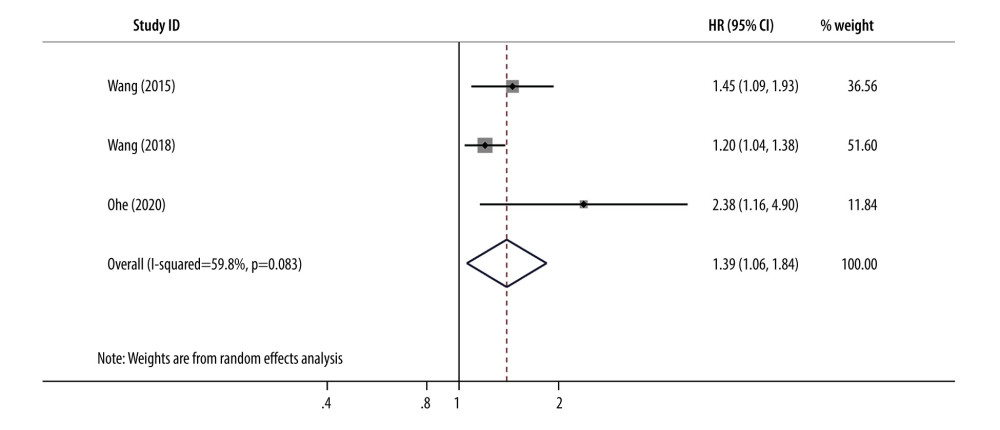 Figure 4. Forest plots of the correlation between platelet to lymphocyte ratio and chemosensitivity in advanced gastric cancer (OR=1.395, 95% CI=1.056–1.841, P=0.019).
Figure 4. Forest plots of the correlation between platelet to lymphocyte ratio and chemosensitivity in advanced gastric cancer (OR=1.395, 95% CI=1.056–1.841, P=0.019). 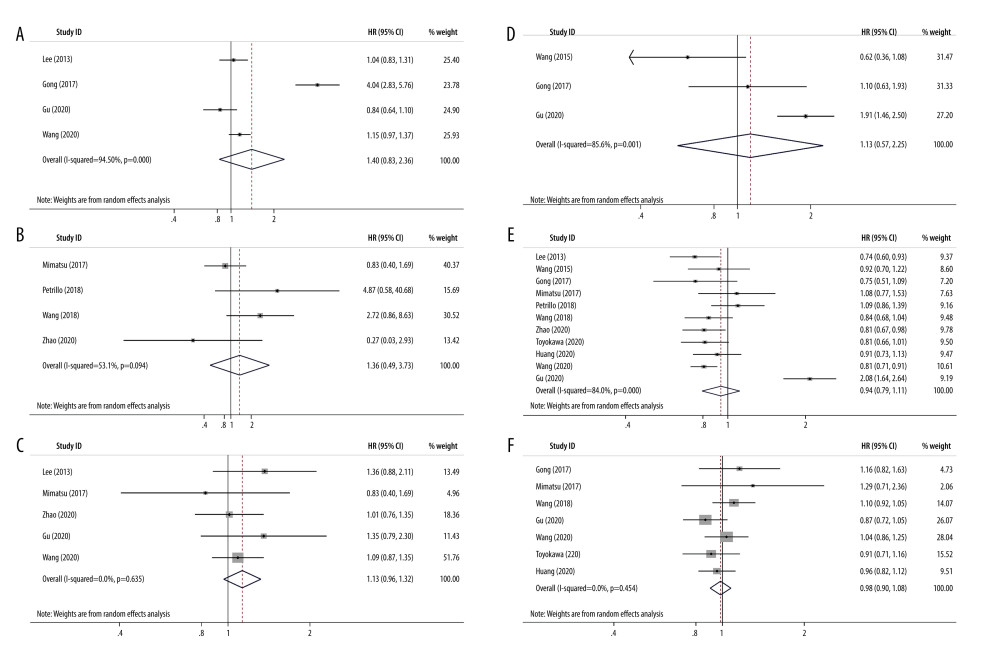 Figure 5. Forest plots of the correlation between platelet-to-lymphocyte ratio and clinical features in advanced gastric cancer. (A) Age (years) (≥60 versus <60, OR=1.398, 95% CI=0.827–2.362, P=0.211); (B) ECOG (≥2 versus <2, OR=1.355, 95% CI=0.492–3.731, P=0.557); (C) CEA (≥5 versus <5, OR=1.128, 95% CI=0.965–1.320, P=0.131); (D) Tumor size (≥5 cm versus <5 cm, OR=1.130, 95% CI=0.567–2.253, P=0.729); (E) Sex (male versus female, OR=0.940, 95% CI=0.794–1.112, P=0.468); (F) Tumor differentiation (poor versus moderate/well, OR=0.985, 95% CI=0.902–1.075, P=0.734).
Figure 5. Forest plots of the correlation between platelet-to-lymphocyte ratio and clinical features in advanced gastric cancer. (A) Age (years) (≥60 versus <60, OR=1.398, 95% CI=0.827–2.362, P=0.211); (B) ECOG (≥2 versus <2, OR=1.355, 95% CI=0.492–3.731, P=0.557); (C) CEA (≥5 versus <5, OR=1.128, 95% CI=0.965–1.320, P=0.131); (D) Tumor size (≥5 cm versus <5 cm, OR=1.130, 95% CI=0.567–2.253, P=0.729); (E) Sex (male versus female, OR=0.940, 95% CI=0.794–1.112, P=0.468); (F) Tumor differentiation (poor versus moderate/well, OR=0.985, 95% CI=0.902–1.075, P=0.734). 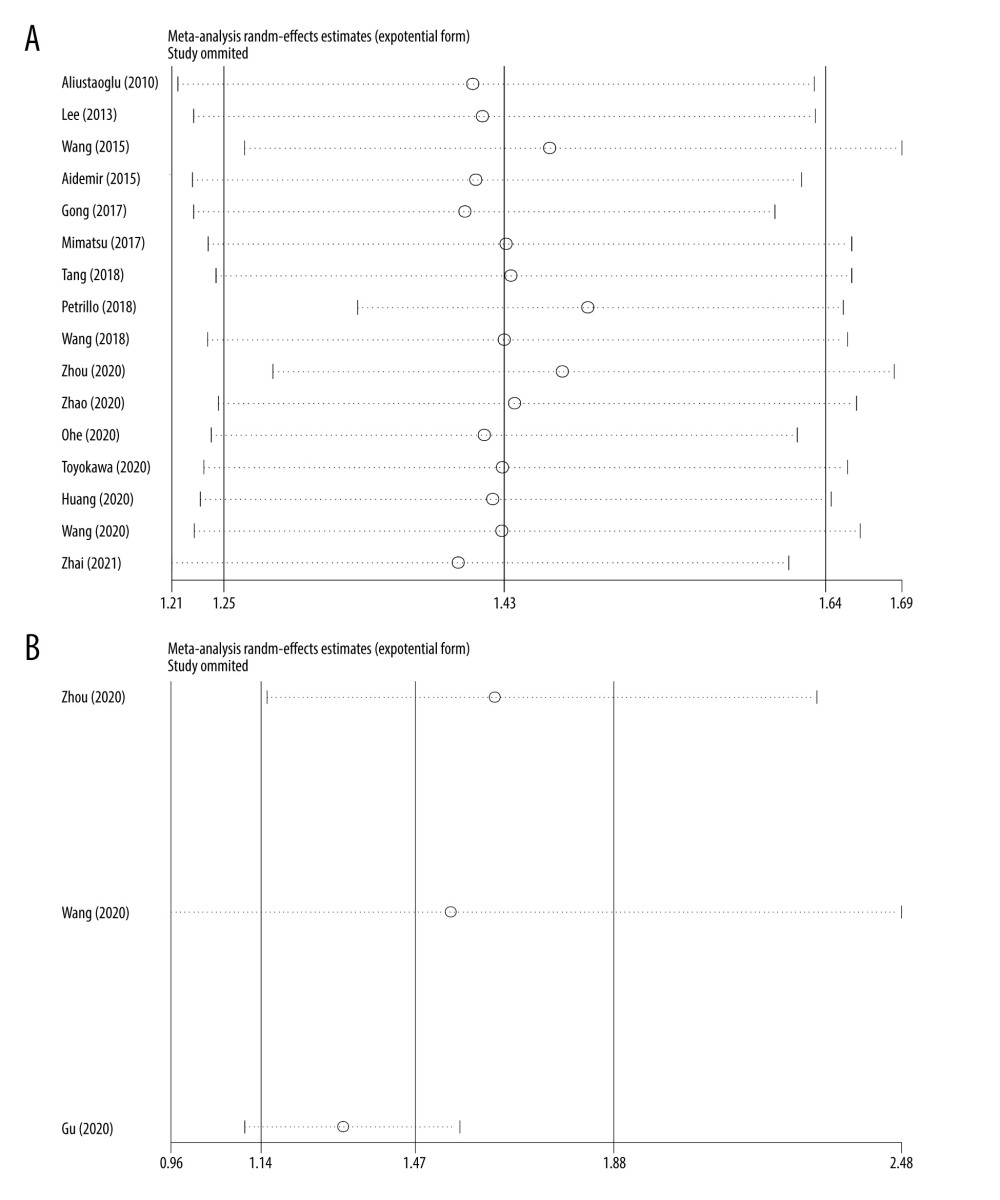 Figure 6. Sensitivity analysis for the association between platelet-to-lymphocyte ratio with survival in advanced gastric cancer. (A) Overall survival, (B) Disease-free survival.
Figure 6. Sensitivity analysis for the association between platelet-to-lymphocyte ratio with survival in advanced gastric cancer. (A) Overall survival, (B) Disease-free survival. 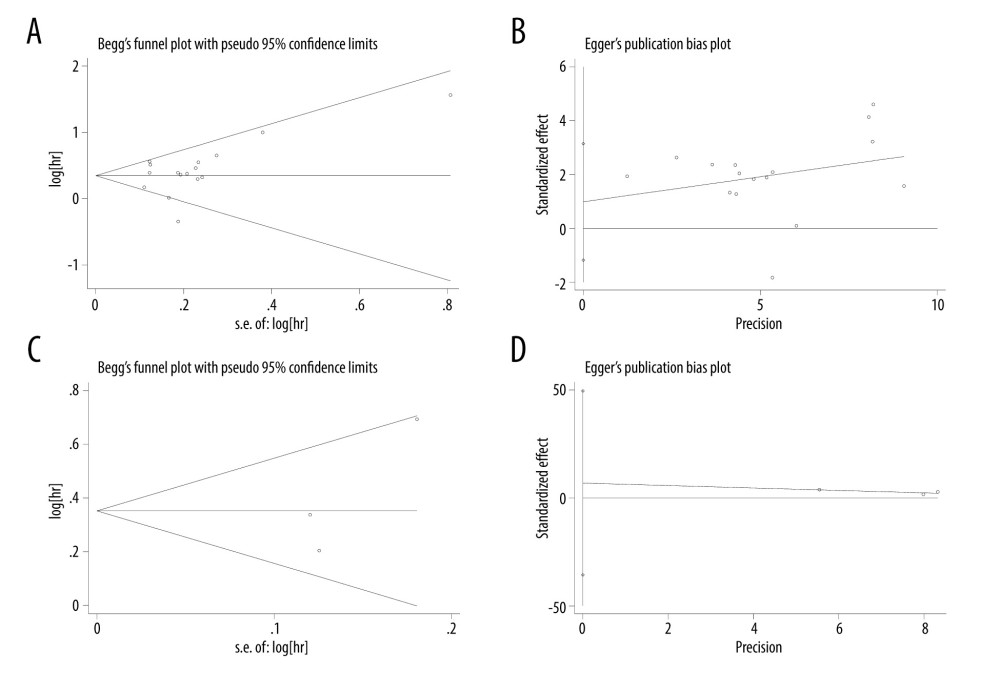 Figure 7. Publication bias assessed by Begg’s test and Egger’s test. (A) Begg’s test the publication bias for overall survival, P=0.260; (B) Egger’s test for overall survival, P=0.346; (C) Begg’s test the publication bias for ratio (disease-free survival, P=1.000; (D) Egger’s test for disease-free survival, P=0.288.
Figure 7. Publication bias assessed by Begg’s test and Egger’s test. (A) Begg’s test the publication bias for overall survival, P=0.260; (B) Egger’s test for overall survival, P=0.346; (C) Begg’s test the publication bias for ratio (disease-free survival, P=1.000; (D) Egger’s test for disease-free survival, P=0.288. References
1. Torre LA, Siegel RL, Ward EM, Global cancer incidence and mortality rates and trends: An update: Cancer Epidemiol Biomarkers Prev, 2016; 25(1); 16-27
2. Bray F, Ferlay J, Soerjomataram I, Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries: Cancer J Clin, 2018; 68; 394-424
3. Rawla P, Barsouk A, Epidemiology of gastric cancer: Global trends, risk factors and prevention: Prz Gastroenterol, 2019; 14(1); 26-38
4. Song Z, Wu Y, Yang J, Progress in the treatment of advanced gastric cancer: Tumour Biol, 2017; 39(7); 1010428317714626
5. Choi YY, Cho M, Kwon IG, ten thousand consecutive gastrectomies for gastric cancer: Perspectives of a master surgeon: Yonsei Med J, 2019; 60(3); 235-42
6. Lutz MP, Zalcberg JR, Ducreux M, Highlights of the EORTC St. Gallen International Expert Consensus on the primary therapy of gastric, gastroesophageal and oesophageal cancer-differential treatment strategies for subtypes of early gastroesophageal cancer: Eur J Cancer, 2012; 48(16); 2941-53
7. Smyth EC, Nilsson M, Grabsch HI, Gastric cancer: Lancet, 2020; 396(10251); 635-48
8. Koizumi W, Kim YH, Fujii M, Addition of docetaxel to S-1 without platinum prolongs survival of patients with advanced gastric cancer: A randomized study (START): J Cancer Res Clin Oncol, 2014; 140(2); 319-28
9. Bang YJ, Van Cutsem E, Feyereislova A, Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial: Lancet, 2010; 376(9742); 687-97
10. Petrillo A, Laterza MM, Tirino G, Systemic inflammation-based score can predict prognosis in metastatic gastric cancer patients before first-line chemotherapy: Future Oncol, 2018; 14(24); 2493-505
11. Okamura Y, Ashida R, Ito T, Preoperative neutrophil to lymphocyte ratio and prognostic nutritional index predict overall survival after hepatectomy for hepatocellular carcinoma: World J Surg, 2015; 39(6); 1501-9
12. Park JH, Watt DG, Roxburgh CS, Colorectal cancer, systemic inflammation, and outcome: Staging the tumor and staging the host: Ann Surg, 2016; 263(2); 326-36
13. Coffelt SB, Visser KED, Cancer: Inflammation lights the way to metastasis: Nature, 2014; 507(7490); 48-49
14. Mantovani A, Allavena P, Sica A, Cancer-related inflammation: Nature, 2008; 454(7203); 436-44
15. Shabnam S, Michael K, Immunity, inflammation, and cancer: An eternal fight between good and evil: J Clin Invest, 2015; 125(9); 3347-55
16. Wagner DD, New links between inflammation and thrombosis: Arterioscler Thromb Vasc Biol, 2005; 25(7); 1321-24
17. Yu JJ, Li JD, Yang T, Association between neutrophil-lymphocyte ratio and prognosis after potentially curative resection for gastric cancer: J Surg Oncol, 2018; 117(8); 1854
18. Choi JH, Suh YS, Choi Y, Comprehensive analysis of the neutrophil-to-lymphocyte ratio for preoperative prognostic prediction nomogram in gastric cancer: World J Surg, 2018; 42(8); 2530-41
19. Baert T, Van Camp J, Vanbrabant L, Influence of CA125, platelet count and neutrophil to lymphocyte ratio on the immune system of ovarian cancer patients: Gynecol Oncol, 2018; 150(1); 31-37
20. Jiang T, Qiao M, Zhao C, Pretreatment neutrophil-to-lymphocyte ratio is associated with outcome of advanced-stage cancer patients treated with immunotherapy: A meta-analysis: Cancer Immunol Immunother, 2018; 67(5); 713-27
21. Liu H, Wu Y, Wang Z, Pretreatment platelet-to-lymphocyte ratio (PLR) as a predictor of response to first-line platinum-based chemotherapy and prognosis for patients with non-small cell lung cancer: J Thorac Dis, 2013; 5(6); 783-89
22. Xu Z, Xu W, Cheng H, The prognostic role of the platelet-lymphocytes ratio in gastric cancer: A meta-analysis: PLoS One, 2016; 11(9); e0163719
23. Gu X, Gao XS, Cui M, Clinicopathological and prognostic significance of platelet to lymphocyte ratio in patients with gastric cancer: Oncotarget, 2016; 7(31); 49878-87
24. Vrabel M, Preferred reporting items for systematic reviews and meta-analyses: Oncol Nurs Forum, 2015; 42(5); 552-54
25. Stang A, Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses: Eur J Epidemiol, 2010; 25(9); 603-5
26. Cochran W, The combination of estimates from different experiments: Biometrics, 1954; 10; 101-29
27. Higgins JP, Thompson SG, Quantifying heterogeneity in a meta-analysis: Stat Med, 2002; 21; 1539-58
28. Wang F, Liu ZY, Xia YY, Changes in neutrophil/lymphocyte and platelet/lymphocyte ratios after chemotherapy correlate with chemotherapy response and prediction of prognosis in patients with unresectable gastric cancer: Oncol Lett, 2015; 10(6); 3411-18
29. Ohe Y, Fushida S, Yamaguchi T, Peripheral blood platelet-lymphocyte ratio is good predictor of chemosensitivity and prognosis in gastric cancer patients: Cancer Manag Res, 2020; 12; 1303-11
30. Wang J, Qu J, Li Z, Pretreatment platelet-to-lymphocyte ratio is associated with the response to first-line chemotherapy and survival in patients with metastatic gastric cancer: J Clin Lab Anal, 2018; 32(1); e22185
31. Aliustaoglu M, Bilici A, Ustaalioglu BB, The effect of peripheral blood values on prognosis of patients with locally advanced gastric cancer before treatment: Med Oncol, 2010; 27(4); 1060-65
32. Aldemir MN, Turkeli M, Simsek M, Prognostic value of baseline neutrophil-lymphocyte and platelet-lymphocyte ratios in local and advanced gastric cancer patients: Asian Pac J Cancer Prev, 2015; 16(14); 5933-37
33. Zhou D, Wu Y, Zhu Y, The prognostic value of neutrophil-to-lymphocyte ratio and monocyte-to-lymphocyte ratio in metastatic gastric cancer treated with systemic chemotherapy: J Cancer, 2020; 11(14); 4205-12
34. Lee S, Oh SY, Kim SH, Prognostic significance of neutrophil lymphocyte ratio and platelet lymphocyte ratio in advanced gastric cancer patients treated with FOLFOX chemotherapy: BMC Cancer, 2013; 13; 350
35. Zhao G, Liu N, Wang S, Prognostic significance of the neutrophil-to-lymphocyte and platelet-to-lymphocyte ratio in patients with metastatic gastric cancer: Medicine (Baltimore), 2020; 99(10); e19405
36. Zhai Z, Gao J, Zhu Z, The ratio of the hemoglobin to red cell distribution width combined with the ratio of platelets to lymphocytes can predict the survival of patients with gastric cancer liver metastasis: Biomed Res Int, 2021; 2021 8729869
37. Tang C, Cheng X, Yu S, Platelet-to-lymphocyte ratio and lymphocyte-to-white blood cell ratio predict the efficacy of neoadjuvant chemotherapy and the prognosis of locally advanced gastric cancer patients treated with the oxaliplatin and capecitabine regimen: Onco Targets Ther, 2018; 11; 7061-75
38. Huang C, Li Z, Zhang Z, Prognostic value and association of systemic inflammation for patients with stage IV gastric cancer: Acta Gastroenterol Belg, 2020; 83(2); 255-63
39. Toyokawa T, Muguruma K, Yoshii M, Clinical significance of prognostic inflammation-based and/or nutritional markers in patients with stage III gastric cancer: BMC Cancer, 2020; 20(1); 517
40. Gu L, Wang M, Cui X, Clinical significance of peripheral blood-derived inflammation markers in advanced gastric cancer after radical resection: BMC Surg, 2020; 20(1); 219
41. Wang H, Ding Y, Li N, Prognostic value of neutrophil-lymphocyte ratio, platelet-lymphocyte ratio, and combined neutrophil-lymphocyte ratio and platelet-lymphocyte ratio in stage IV advanced gastric cancer: Front Oncol, 2020; 10; 841
42. Mimatsu K, Fukino N, Ogasawara Y, Utility of inflammatory marker- and nutritional status-based prognostic factors for predicting the prognosis of stage IV gastric cancer patients undergoing non-curative surgery: Anticancer Res, 2017; 37(8); 4215-22
43. Gong W, Zhao L, Dong Z, After neoadjuvant chemotherapy platelet/lymphocyte ratios negatively correlate with prognosis in gastric cancer patients: J Clin Lab Anal, 2018; 32(5); e22364
44. Cao W, Yao X, Cen D, The prognostic role of platelet-to-lymphocyte ratio on overall survival in gastric cancer: A systematic review and meta-analysis: BMC Gastroenterol, 2020; 20(1); 16
45. Zhang X, Zhao W, Yu Y, Clinicopathological and prognostic significance of platelet-lymphocyte ratio (PLR) in gastric cancer: An updated meta-analysis: World J Surg Oncol, 2020; 18(1); 191
46. Qi C, Li B, Guo S, P-selectin-mediated adhesion between platelets and tumor cells promotes intestinal tumorigenesis in Apc(Min/+) Mice: Int J Biol Sci, 2015; 11(6); 679-87
47. Dunn GP, Old LJ, Schreiber RD, The immunobiology of cancer immunosurveillance and immunoediting: Immunity, 2004; 21(2); 137-48
48. Wu Y, Zhou BP, Inflammation: A driving force speeds cancer metastasis: Cell Cycle, 2009; 8(20); 3267-73
49. Hanahan D, Weinberg RA, Hallmarks of cancer: The next generation: Cell, 2011; 144(5); 646-74
50. de Visser KE, Jonkers J, Towards understanding the role of cancer-associated inflammation in chemoresistance: Current Pharm Des, 2009; 15(16); 1844-53
51. Sasako M, Sakuramoto S, Katai H, Five-year outcomes of a randomized Phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer: J Clin Oncol, 2011; 29(33); 4387-93
52. Nakayama N, Koizumi W, Sasaki T, A multicenter, Phase I dose-escalating study of docetaxel, cisplatin and S-1 for advanced gastric cancer (KDOG0601): Oncology, 2008; 75(1–2); 1-7
53. Grivennikov SI, Greten FR, Karin M, Immunity, inflammation and cancer: Cell, 2010; 140(6); 883-99
54. Kwon HC, Roh MS, Oh SY, Prognostic value of expression of ERCC1, thymidylate synthase, and glutathione S-transferase P1 for 5-fluorouracil/oxaliplatin chemotherapy in advanced gastric cancer: Ann Oncol, 2007; 18(3); 504-9
55. Seo BG, Kwon HC, Oh SY, Comprehensive analysis of excision repair complementation group 1, glutathione S-transferase, thymidylate synthase and uridine diphosphate glucuronosyl transferase 1A1 polymorphisms predictive for treatment outcome in patients with advanced gastric cancer treated with FOLFOX or FOLFIRI: Oncol Rep, 2009; 22(1); 127-36
Figures
 Figure 1. Flow chart of literature search and study selection.
Figure 1. Flow chart of literature search and study selection. Figure 2. Forest plots for the association platelet to lymphocyte ratio and overall survival in patients with advanced gastric cancer (HR=1.429, 95% CI=1.246–1.639, P<0.001). CI – confidence interval; HR – hazard ratio.
Figure 2. Forest plots for the association platelet to lymphocyte ratio and overall survival in patients with advanced gastric cancer (HR=1.429, 95% CI=1.246–1.639, P<0.001). CI – confidence interval; HR – hazard ratio. Figure 3. Forest plots for the association platelet to lymphocyte ratio and disease-free survival in patients with advanced gastric cancer (HR=1.47, 95% CI=1.14–1.88, P=0.003). CI – confidence interval; HR – hazard ratio.
Figure 3. Forest plots for the association platelet to lymphocyte ratio and disease-free survival in patients with advanced gastric cancer (HR=1.47, 95% CI=1.14–1.88, P=0.003). CI – confidence interval; HR – hazard ratio. Figure 4. Forest plots of the correlation between platelet to lymphocyte ratio and chemosensitivity in advanced gastric cancer (OR=1.395, 95% CI=1.056–1.841, P=0.019).
Figure 4. Forest plots of the correlation between platelet to lymphocyte ratio and chemosensitivity in advanced gastric cancer (OR=1.395, 95% CI=1.056–1.841, P=0.019). Figure 5. Forest plots of the correlation between platelet-to-lymphocyte ratio and clinical features in advanced gastric cancer. (A) Age (years) (≥60 versus <60, OR=1.398, 95% CI=0.827–2.362, P=0.211); (B) ECOG (≥2 versus <2, OR=1.355, 95% CI=0.492–3.731, P=0.557); (C) CEA (≥5 versus <5, OR=1.128, 95% CI=0.965–1.320, P=0.131); (D) Tumor size (≥5 cm versus <5 cm, OR=1.130, 95% CI=0.567–2.253, P=0.729); (E) Sex (male versus female, OR=0.940, 95% CI=0.794–1.112, P=0.468); (F) Tumor differentiation (poor versus moderate/well, OR=0.985, 95% CI=0.902–1.075, P=0.734).
Figure 5. Forest plots of the correlation between platelet-to-lymphocyte ratio and clinical features in advanced gastric cancer. (A) Age (years) (≥60 versus <60, OR=1.398, 95% CI=0.827–2.362, P=0.211); (B) ECOG (≥2 versus <2, OR=1.355, 95% CI=0.492–3.731, P=0.557); (C) CEA (≥5 versus <5, OR=1.128, 95% CI=0.965–1.320, P=0.131); (D) Tumor size (≥5 cm versus <5 cm, OR=1.130, 95% CI=0.567–2.253, P=0.729); (E) Sex (male versus female, OR=0.940, 95% CI=0.794–1.112, P=0.468); (F) Tumor differentiation (poor versus moderate/well, OR=0.985, 95% CI=0.902–1.075, P=0.734). Figure 6. Sensitivity analysis for the association between platelet-to-lymphocyte ratio with survival in advanced gastric cancer. (A) Overall survival, (B) Disease-free survival.
Figure 6. Sensitivity analysis for the association between platelet-to-lymphocyte ratio with survival in advanced gastric cancer. (A) Overall survival, (B) Disease-free survival. Figure 7. Publication bias assessed by Begg’s test and Egger’s test. (A) Begg’s test the publication bias for overall survival, P=0.260; (B) Egger’s test for overall survival, P=0.346; (C) Begg’s test the publication bias for ratio (disease-free survival, P=1.000; (D) Egger’s test for disease-free survival, P=0.288.
Figure 7. Publication bias assessed by Begg’s test and Egger’s test. (A) Begg’s test the publication bias for overall survival, P=0.260; (B) Egger’s test for overall survival, P=0.346; (C) Begg’s test the publication bias for ratio (disease-free survival, P=1.000; (D) Egger’s test for disease-free survival, P=0.288. Tables
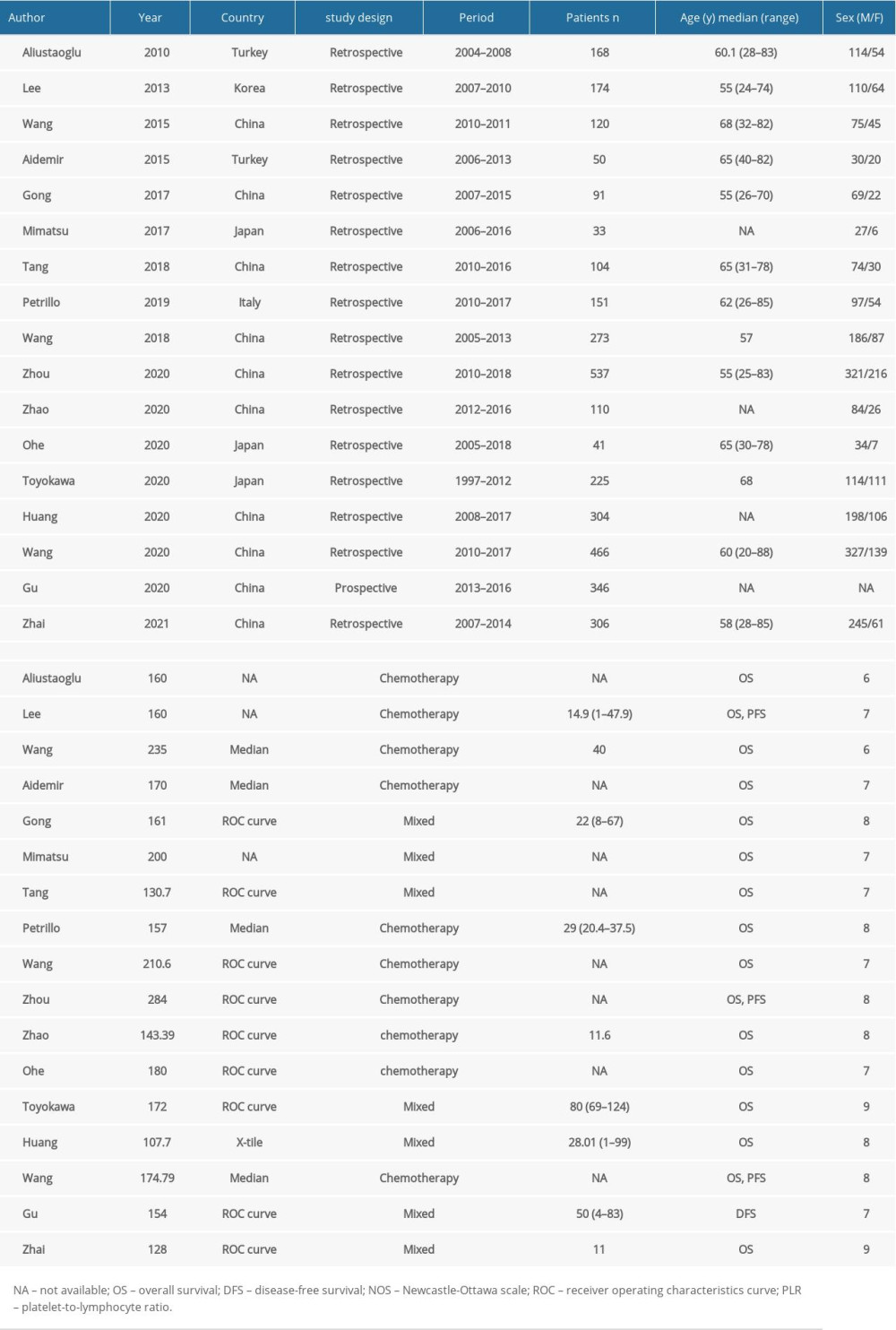 Table 1. Baseline characteristics of eligible studies for this meta-analysis.
Table 1. Baseline characteristics of eligible studies for this meta-analysis.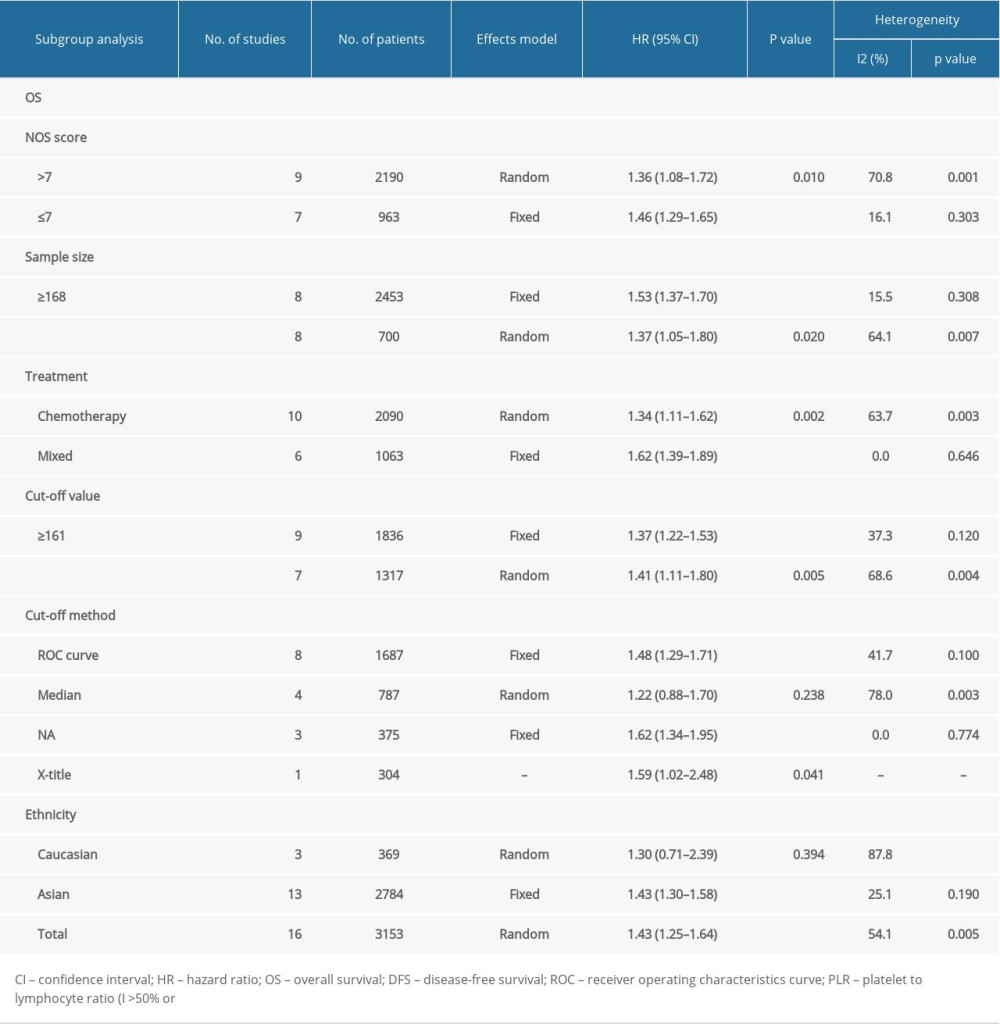 Table 2. Subgroup analyses of pooled HRs and 95% CIs for OS based on different factors.
Table 2. Subgroup analyses of pooled HRs and 95% CIs for OS based on different factors.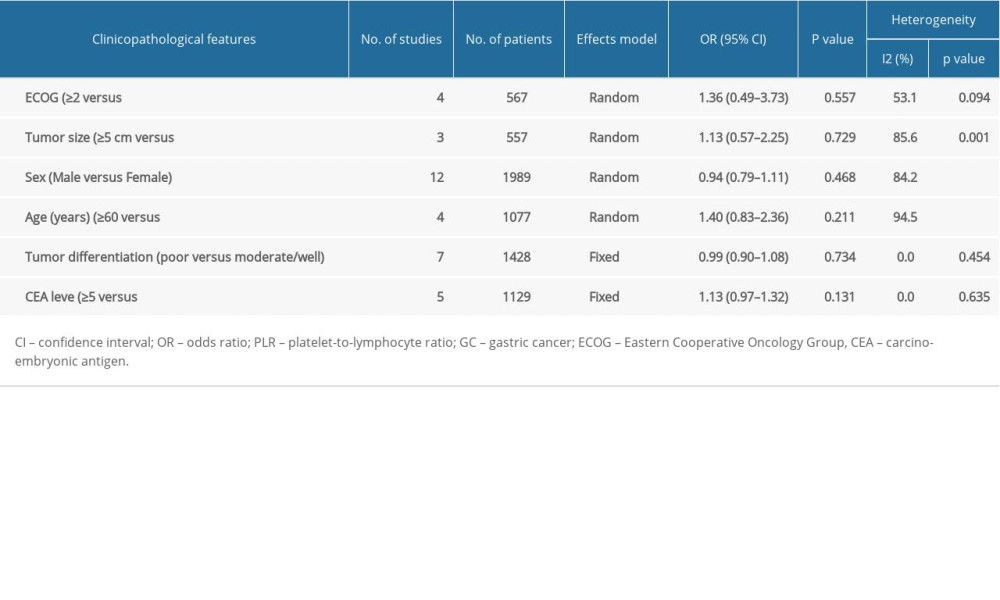 Table 3. Correlations of PLR and clinicopathological characteristics in patients with advanced GC.
Table 3. Correlations of PLR and clinicopathological characteristics in patients with advanced GC. Table 1. Baseline characteristics of eligible studies for this meta-analysis.
Table 1. Baseline characteristics of eligible studies for this meta-analysis. Table 2. Subgroup analyses of pooled HRs and 95% CIs for OS based on different factors.
Table 2. Subgroup analyses of pooled HRs and 95% CIs for OS based on different factors. Table 3. Correlations of PLR and clinicopathological characteristics in patients with advanced GC.
Table 3. Correlations of PLR and clinicopathological characteristics in patients with advanced GC. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








