27 February 2022: Clinical Research
Serum Uric Acid Levels in Relation to Atrial Fibrillation: A Case-Control Study
Xia Zhong1ABCDEF*, Huachen Jiao2ABCFG, Dongsheng Zhao1BD, Jing Teng1CFDOI: 10.12659/MSM.934007
Med Sci Monit 2022; 28:e934007
Abstract
BACKGROUND: Although serum uric acid (SUA) levels have been reported to be associated with atrial fibrillation (AF), the specific associations remain unclear. The purpose of this study was to investigate the potential relationship of serum uric acid levels to atrial fibrillation.
MATERIAL AND METHODS: We retrospectively analyzed clinical data of 970 consecutive hospitalized patients (M/F, 519/451; age, 64.78±13.49 years). The study included 478 patients with AF, and 492 age-matched patients with sinus rhythm and no history of arrhythmia as a control group. The t test, ANOVA, chi-squared test, or Fisher exact test were performed to analyze clinical baseline data.
RESULTS: Compared with the control group, patients with AF exhibited higher SUA levels (5.66±1.90 vs 5.35±1.55 mg/dL, P=0.006), especially women (P<0.001). Pearson correlation analysis showed SUA was influenced by A/G, PAB, and APOA1 in patients with AF. Logistic regression analysis showed SUA was associated with AF (total: OR=1.002, 95% CI: 1.000-1.003; women: OR=1.005, 95% CI: 1.003-1.007). After adjustment for clinical related factors for AF, SUA was still associated with AF (total: OR=1.004, 95% CI: 1.002-1.006; women: OR=1.005, 95% CI: 1.002-1.009). Also, elevated SUA was positively correlated with A/G and PAB and negatively correlated with APOA1. There were no significant differences in SUA levels in AF subtypes and complications.
CONCLUSIONS: Elevated SUA levels were associated with AF, but the independent association was significant only in women. Elevated SUA may promote other AF-related factors and participate in the pathological process of AF.
Keywords: Risk, Gender Identity, Atrial Fibrillation, Accident Prevention, Case-Control Studies, Female, Humans, Male, Risk Factors, Uric Acid
Background
Atrial fibrillation (AF) is an increasingly common arrhythmia, which has a considerable impact on patient health and socioeconomic factors. Currently, AF affects 33 million people worldwide [1], and this rate is predicted to increase [2], which also means there will be an increased risk of ischemic stroke, systemic embolism, and heart failure [3–5]. Although ablation approaches and antiarrhythmic drugs are widely used as effective therapies for AF, the recurrence and mortality rates still remain high. In addition, evidence suggests that the present long-term outcome of catheter ablation of AF is not ideal, particularly for patients with paroxysmal atrial fibrillation, with single-procedure success rates of only 66.6% [6] and a 5-year success rate of less than 30% after a single procedure [7]. Given the increasing risk of AF, its morbidity and mortality, and its significant health and socioeconomic impact, the prevention and treatment of AF cannot be ignored.
Although the pathological mechanism of AF can be an inconclusive measurement, inflammatory and oxidative stress have been shown to play an important role in the pathological progress of AF. Serum uric acid (SUA) is a byproduct of purine catabolism and is catalyzed by xanthine oxidoreductase [8]. Several studies have demonstrated that elevated SUA levels are associated with activated pro-inflammatory and oxidative stress pathways [9,10], and xanthine oxidoreductase is generally considered to be an important link in this process. Regardless of the predictor or causative factor, an increasing data have demonstrated the positive association between increased SUA levels and AF [11–13]. Nevertheless, in most instances, the pathological progression of AF and SUA levels can be influenced by multiple parameters, including drugs and diseases, and in particular, patient sex [14,15]. From the clinical standpoint, the associations between SUA levels and AF appear to be ambiguous and create ongoing controversy.
In the present study, we aimed to understand the pathological process of AF by investigating the potential relationship between SUA levels and AF.
Material and Methods
DATA SOURCE AND STUDY DESIGN:
This was a case-control study. Data were obtained from the electronic medical record database of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine from January 2019 to May 2021. We retrospectively analyzed the clinical data of 970 consecutive hospitalized patients (male/female, 519/451; age, 62.17±15.48 years). The sample included 478 patients with AF aged 31 to 85 years old, and 492 age- and sex-matched patients with sinus rhythm and no history of arrhythmia served as a control group. Inclusion criteria were as follows: patients with AF, regardless of paroxysmal or permanent, and complete clinical data. Exclusion criteria were as follows: valve diseases, structural heart disease, cardiac surgery, heart failure, hepatic or renal dysfunction, hyperthyroidism, malignant tumor, uric acid lowering drugs, diuretics, allopurinol, and pregnancy. We collected and analyzed baseline data, including AF categories, age, sex, complications, medication, and laboratory data, from a medical record review. Patients with AF were divided into 6 groups according to SUA levels and sex as follows: men (low level group: SUA <5.5 mg/dL; middle level group: 5.5≤ SUA ≤6.5 mg/dL; and high level group: SUA >6.5 mg/dL) and women (low level group: SUA <4.2 mg/dL; middle level group: 4.2≤ SUA ≤5.0 mg/dL; and high level group: SUA >5.0 mg/dL). This study was approved by the Ethics Committee on Medical Research of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine and was performed according to the principles of the Declaration of Helsinki. Informed consent was waived.
DEFINITION OF AF:
According to guidelines [16], AF was considered to be an arrhythmia lasting long enough for a 12-lead electrocardiogram to be recorded, or lasting for at least 30 s. In addition, paroxysmal AF was specifically defined as AF that terminated spontaneously or with intervention within 7 days of initiation. Permanent AF was defined as the presence of recognized AF and sinus rhythm that would not be further restored or maintained.
DEFINITION OF HYPERURICEMIA:
The measurements of SUA were reported in μmol/L and converted as 1 mg/dL=59.48 μmol/L. SUA levels were measured by the uricase method. Hyperuricemia was defined as an SUA level >7.0 mg/dL in men and an SUA level >5.7 mg/dL in women [17].
SCREENED INDICATORS:
We screened basic indicators, including age, sex, blood pressure, complications, and medication and biochemical indicators, including serum creatinine (SCr), fasting blood glucose (FBG), alanine aminotransferase (ALT), aspartate aminotransferase (AST), ALT/AST, serum apolipoprotein A1 (APOA1), APOB (serum apolipoprotein B), albumin (ALB), Ig, A/G, prealbumin (PAB), and lipoprotein (a) (Lp [a]) in all participants.
STATISTICAL ANALYSIS:
Analyses were performed using SPSS version 26.0 (IBM Corp, Armonk, NY, USA) and GraphPad Prism software (version 9.0.0). Continuous variables were expressed as mean±standard deviation or median (interquartile range [IQR]). Categorical variables were presented as percentages or ratios. The comparisons of parametric and nonparametric continuous variables were performed by ANOVA or the
Results
BASELINE CHARACTERISTICS OF THE STUDY POPULATION:
A total of 478 patients were included in the AF group: 253 (52.9%) were men and the mean age was 64.87±10.75 years; 492 participants without AF were included in the control group: 266 (54.1%) were men and the mean age was 64.69±15.72 years. The AF group had more patients with hyperuricemia than did the control group (n=142 vs 102, P=0.001; men: 70 vs 66, women: 72 vs 36, P=0.017, Table 1). Patients with AF had significantly higher SUA levels (5.66±1.90 vs 5.35±1.55 mg/dL, P=0.006, Table 1 and Figure 1; women: 5.33±1.88 vs 4.58±1.21 mg/dL, P<0.001, Table 1 and Figure 2), SCr (78.23±55.15 vs 66.95±34.43 μmoI/L, P<0.001, Table 1), and ALT/AST (1.28±0.55 vs 1.15±0.51, P<0.001, Table 1) than did controls. In addition, patients with AF had significantly lower ALB (37.75±4.59 vs 40.30±4.04 g/L, P<0.001, Table 1), APOA1 (1.08±0.25 vs 1.22±0.25 g/L, P<0.001, Table 1), APOB (0.81±0.48 vs 1.00±0.25 g/L, P<0.001, Table 1), A/G (1.35±0.24 vs 1.46±0.22, P<0.001, Table 1), and PAB (190.63±60.78 vs 227.69±52.68 mg/L, P<0.001, Table 1) than did controls. Obviously, the AF group was more prone to have a history of hypertension, diabetes, and coronary heart disease (all P<0.001, Table 1) and to be taking β-blockers, calcium channel blockers (CCBs), angiotensin-converting enzyme inhibitors (ACEI/ARBs), and statins (all P<0.001, Table 1). We also observed that SUA levels were positively correlated with A/G (r=0.318, P=0.046, Figure 3A), PAB (r=0.138, P=0.002, Figure 3B) and negatively correlated with APOA1 (r=−0.201, P<0.001, Figure 3C) in the AF group.
ASSOCIATION BETWEEN SUA LEVELS AND AF:
To check the association between SUA levels and AF using logistic regression analysis, we adjusted for sex, hypertension, diabetes, coronary heart disease (CHD), β-blockers, CCBs, ACEI/ARB, and statins (OR=1.002, 95% CI: 1.000–1.004, P=0.004). After adjusting for SCr, AST, ALT/AST, ALB, APOA1, APOB, A/G, and PAB, SUA levels remained a significant factor correlated with AF (OR=1.003, 95% CI: 1.002–1.005, P=0.001). After further adjustment for all these factors, SUA remained a significant indicator for AF (OR=1.004, 95% CI: 1.002–1.006, P=0.014). Furthermore, the independent association was significant in women (P<0.05) but not in men (P>0.05) (Table 2, Figure 4).
ASSOCIATION OF SUA LEVELS AND AF BY SUBTYPES AND COMPLICATIONS:
As shown in Table 3, the SUA levels in patients with paroxysmal AF and permanent AF were 5.54±1.95 vs 5.73±1.87 mg/dL, respectively (P=0.290). The SUA levels in patients with AF with hypertension, diabetes, and CHD were 5.71±1.91 vs 5.40±2.13 vs 5.61±1.86 mg/dL, respectively (P=0.292). There were no significant differences between subtypes and complications in patients with AF.
ASSOCIATION OF SUA LEVELS AND METABOLIC INDICATORS IN PATIENTS WITH AF BY SEX:
As shown in Table 4, regardless of sex, patients with higher SUA levels had higher SCr (75.20±34.22 vs 79.79±16.55 vs 89.49±31.89 μmoI/L, P=0.005) and ALB (36.44±4.90 vs 39.28±4.75 vs 37.77±4.47mg/L, P<0.001. In male patients with AF, those with higher SUA levels had higher PAB (169.78±60.93 vs 205.28±65.03 vs 209.18±64.06 mg/L, P<0.001).
Discussion
In the present study, we observed that elevated SUA levels were associated with AF and that the independent association was significant only in women (
SUA is the final product of purine of purine metabolism catalyzed by xanthine oxidase in humans and is produced in the liver and muscles and is mainly excreted by the kidneys [18,19]. Overproduction and insufficient excretion can promote elevated SUA levels. Because of the uricosuric effect of estrogen, although SUA levels increase after menopause, SUA levels in men are higher than those in women throughout life [20,21]. In general, normal serum SUA levels are 6.5 to 7 mg/dL for men and 6 to 6.5 mg/dL for women [22]. The recommended goal is a concentration below 5 mg/dL in patients with higher cardiovascular risk [23,24].
To date, although the pathogenesis of AF is still challenging, inflammation and oxidative stress have been demonstrated to play important roles in the occurrence and development of AF [25,26]. In this regard, hyperuricemia-induced AF, occurring through an inflammatory pathway, has been receiving attention [27]. Evidence has indicated that the formation of free radical superoxide anion and oxidative stress contribute to the electrical remodeling of the left atria, and SUA plays an important role in this process [28,29]. Moreover, the release of pro-inflammatory cytokines and local activation of the renin-angiotensin system are also associated with SUA levels [30,31]. Notably, inflammation also promotes the production of SUA through augmented cell destruction [21]. In addition, chronic inflammation promotes the activation of the Janus kinase/signal transducer and activator of transcription pathway, which also plays an important role in left atrial electrical remodeling [32,33]. Studies have identified that hyperuricemia is associated with paroxysmal or persistent AF as well as with the risk of AF in patients undergoing surgery [34].
Clinical evidence has shown that SUA level is associated with an increased risk for future AF in both sexes [21]. Indeed, to date, the effects of sex on the association between SUA and AF are still controversial. In a cohort study of 7155 patients, regardless of sex, SUA levels significantly increased the crude prevalence of AF (
Furthermore, in the present study, we observed a difference in the association of SUA with AF by subtypes and complications. However, few studies tested the association of SUA with AF by subtypes and complications. In a cross-sectional study of 45 patients with paroxysmal AF, 41 patients with permanent AF and 48 controls were tested, and the results indicated that a significant difference in SUA level was evident between patients with paroxysmal AF (5.7 mg/dL), patients with permanent AF (6.7 mg/dL), and control participants (5.1 mg/dL) (
Multiple parameters, including drugs and diseases, can influence AF. In the present study, we observed that SUA, sex, hypertension, diabetes, CHD, β-blockers, CCBs, ACEI/ARBs, statins, SCr, AST, ALT/AST, ALB, APOA1, APOB, A/G, and PAB were associated with AF. After further analysis, we found that SUA level was positively correlated with A/G and PAB and negatively correlated with APOA1. Meanwhile, SUA levels were independently associated with SCr and ALB in patients with AF and independently associated with PAB only in men. Inflammation and oxidative stress have been proposed as the important mechanisms contributing to the occurrence of AF [38,39]. Serum albumin exerts anti-inflammatory and anti-oxidative functions in physiological conditions [40,41]. A/G, the indicator of measuring nutritional status, has been confirmed to be involved in the systemic inflammatory process [42]. Generally, low A/G represents poor nutritional status and chronic inflammation. Many studies have suggested that low A/G is inversely associated with cardiovascular events [42,43]. A previous study demonstrated that serum albumin level is independently and inversely related to AF [44]. Furthermore, evidence supports that the alteration of A/G may contribute to sympathovagal imbalance, which is closely related to the onset of AF [45,46]. PAB is also an indicator that reflects the body’s nutritional and immune status. Generally, PAB suggests low levels in inflammatory states and impaired heart function [47], and low PAB is associated with cardiovascular risk [48]. ApoA1, the major apolipoprotein of high-density lipoprotein (HDL), mediates the antiatherogenic and cardioprotective functions of HDL [49]. A previous study, based on patients with AF treated with oral anticoagulation, reported that higher ApoA1 levels were independently related with a lower risk of ischemic cardiovascular outcomes. Therefore, it is speculated that ApoA1 may mediate the ischemic outcome of AF [50]. Additionally, APOA1 can exert anti-inflammatory and anti-oxidant effects through related enzymes [51,52]. A study demonstrated that compared with levels in a healthy population, ApoA1 levels were significantly lower in patients with AF [53]. Another small study also showed that compared with that in a healthy population, patients with AF had an approximately 30% lower expression of ApoA1. Further studies are needed to determine their causalities and effects on AF.
We conducted a retrospective case-control study to investigate the potential relation of SUA levels to AF and the effects of sex on the association between SUA and AF. Moreover, we evaluated the associations between SUA and other clinical parameters in patients with AF. These findings may contribute to the understanding of the pathological process for AF. However, this study had several potential limitations. First, the small sample size was the largest limitation. Second, patients with persistent AF were not observed, and several asymptomatic patients with AF may have been ignored. Accordingly, results cannot be generalized to all patients with AF. Third, this was a single-center retrospective study, and we only investigated associations and not causalities. Fourth, markers of inflammation and oxidative stress were not included. Lastly, due to the retrospective nature of this study, there may still be potential confounding factors. Further prospective studies should focus on confirming our results and elucidating the specific associations between SUA levels and AF.
Conclusions
In conclusion, elevated SUA levels were associated with AF in the present study; however, the independent association was significant only in women. We also observed that elevated SUA levels were positively correlated with A/G and PAB and negatively correlated with APOA1. Nevertheless, there were no significant differences found between subtypes and complications of AF. The findings may be useful in understanding the pathological progress of AF and may contribute to its prevention.
Figures
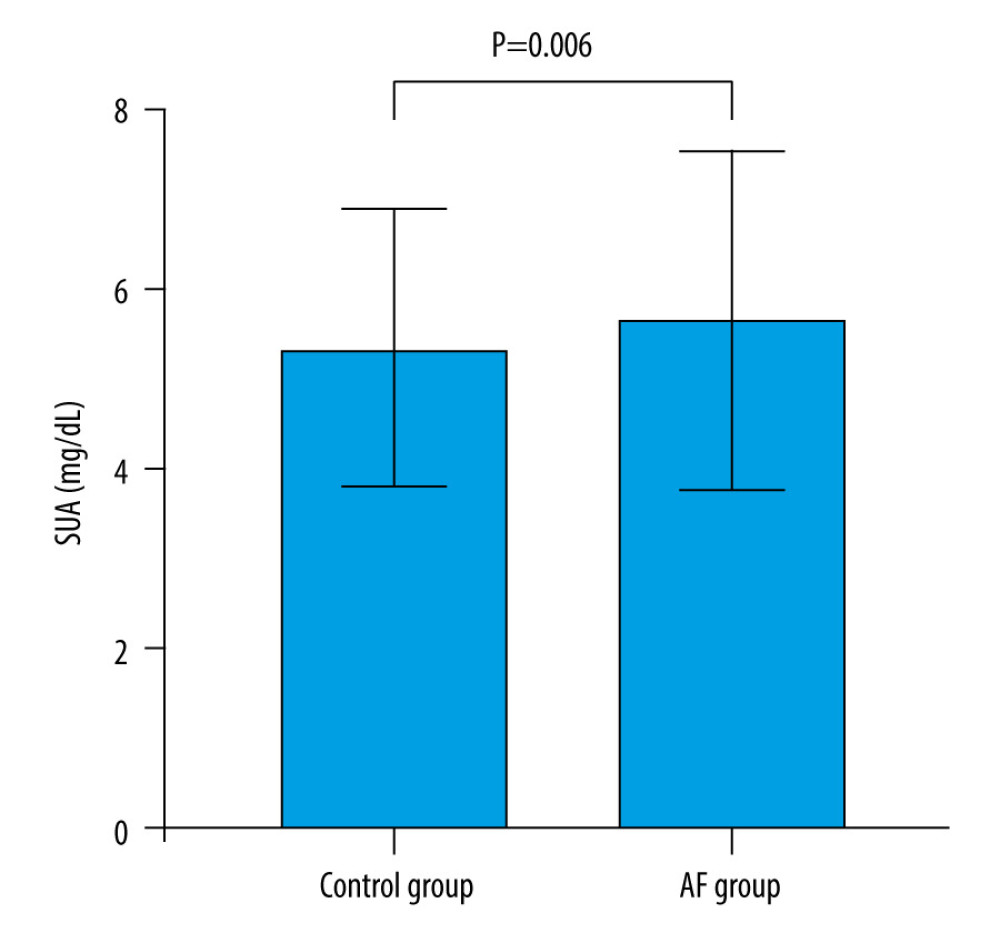 Figure 1. SUA levels in AF group and controls. Serum uric acid levels in AF group were significantly increased than controls (5.66±1.90 vs 5.35±1.55 mg/dL, P=0.006). Abbreviations as in Table 1. The figure was created by GraphPad Prism software (version 9.0.0).
Figure 1. SUA levels in AF group and controls. Serum uric acid levels in AF group were significantly increased than controls (5.66±1.90 vs 5.35±1.55 mg/dL, P=0.006). Abbreviations as in Table 1. The figure was created by GraphPad Prism software (version 9.0.0). 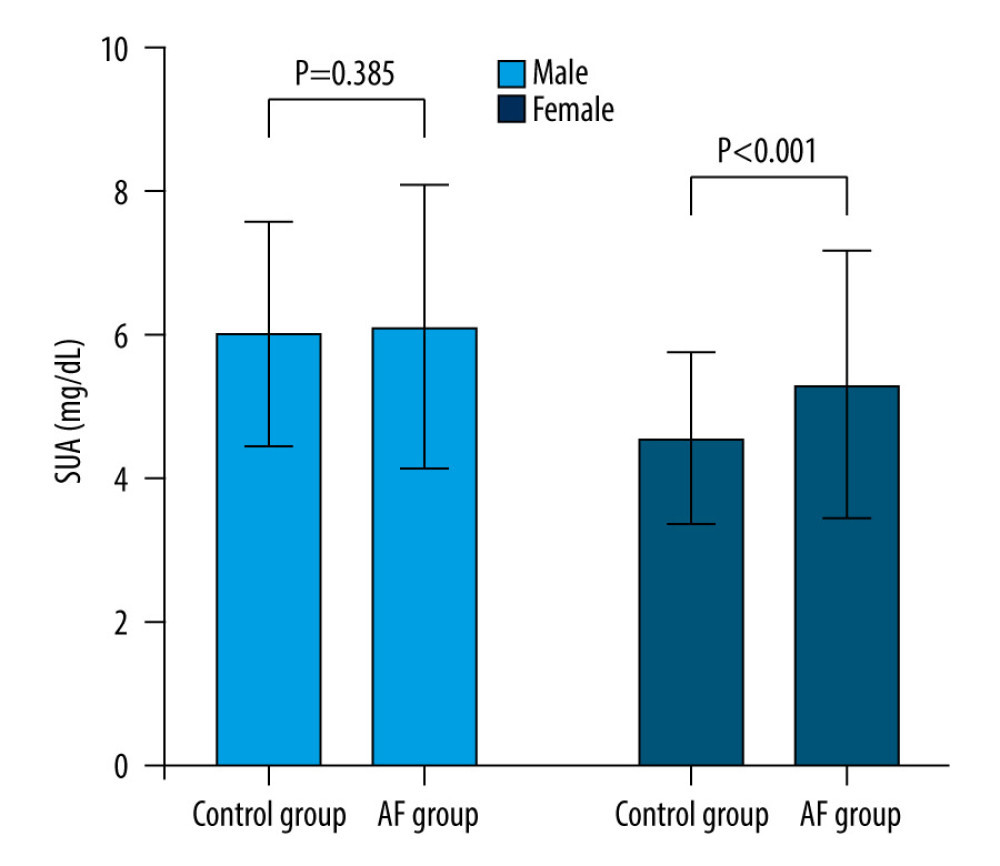 Figure 2. SUA levels in AF and control groups by gender. SUA levels were significantly higher AF patients than in the female controls(5.33±1.88 vs 4.58±1.21 mg/dL, P<0.001). The difference was not observed in male of two groups. Abbreviations as in Table 1. The figure was created by GraphPad Prism software (version 9.0.0).
Figure 2. SUA levels in AF and control groups by gender. SUA levels were significantly higher AF patients than in the female controls(5.33±1.88 vs 4.58±1.21 mg/dL, P<0.001). The difference was not observed in male of two groups. Abbreviations as in Table 1. The figure was created by GraphPad Prism software (version 9.0.0). 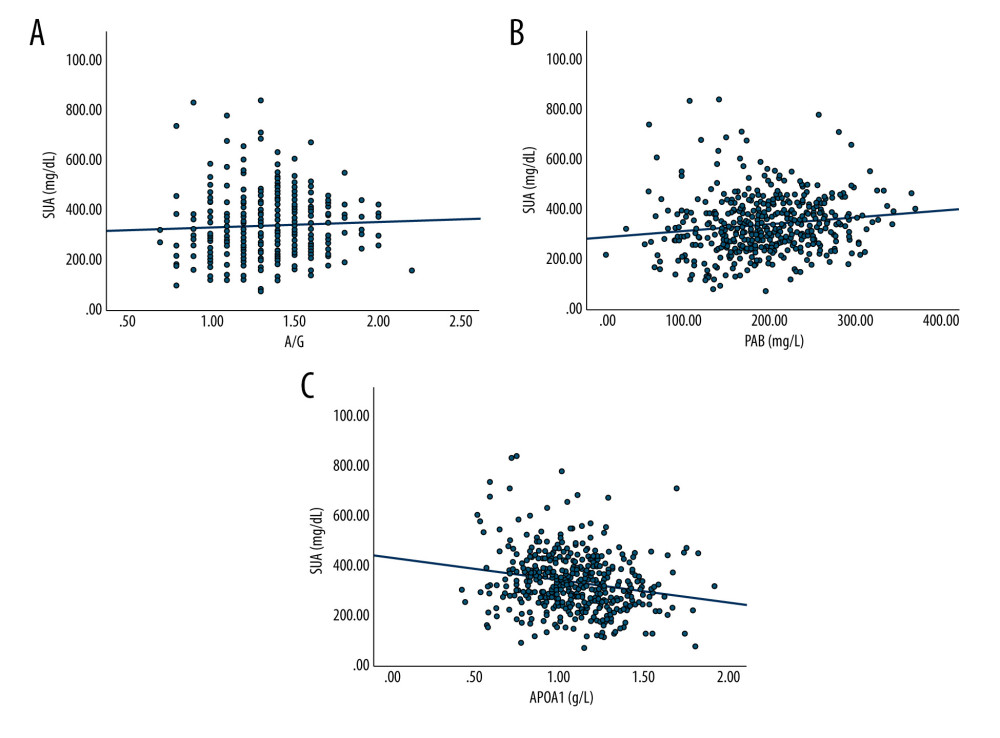 Figure 3. The influence factors of SUA levels in AF patients. (A) Correlation between SUA levels and A/G in AF patients (r=0.318, p=0.046). (B) Correlation between SUA levels and PAG in AF patients(r=0.138, p=0.002). (C) Correlation between SUA levels and A1 (APOA1) in AF patients (r=−0.201, p<0.001). Abbreviations as in Table 1. The figures were created by SPSS version 26.0 (SPSS Inc., Chicago, IL, USA).
Figure 3. The influence factors of SUA levels in AF patients. (A) Correlation between SUA levels and A/G in AF patients (r=0.318, p=0.046). (B) Correlation between SUA levels and PAG in AF patients(r=0.138, p=0.002). (C) Correlation between SUA levels and A1 (APOA1) in AF patients (r=−0.201, p<0.001). Abbreviations as in Table 1. The figures were created by SPSS version 26.0 (SPSS Inc., Chicago, IL, USA). 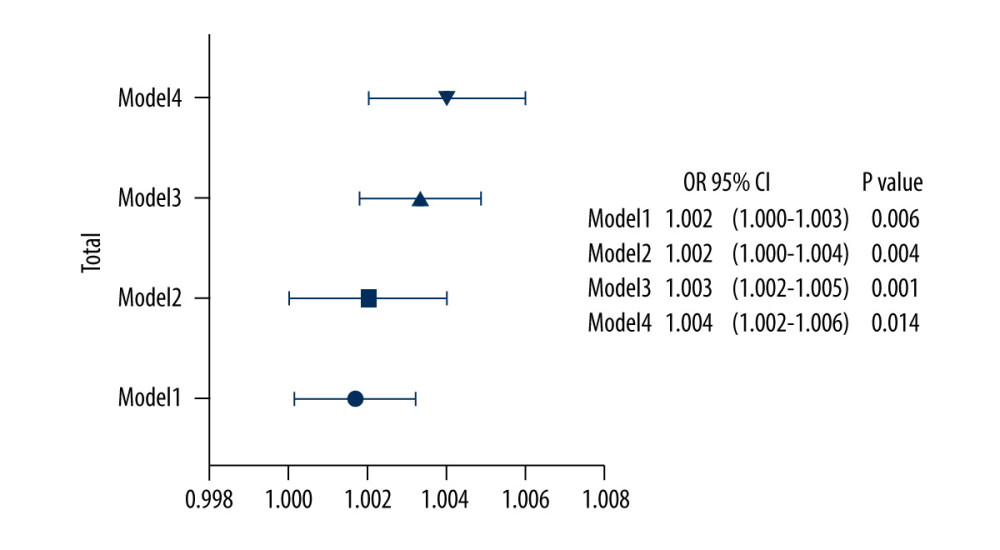 Figure 4. Multiple-adjusted odds ratios (ORs), with 95% confidence intervals (95% CIs) of the association between SUA levels and AF. The figure was created by GraphPad Prism software (version 9.0.0).
Figure 4. Multiple-adjusted odds ratios (ORs), with 95% confidence intervals (95% CIs) of the association between SUA levels and AF. The figure was created by GraphPad Prism software (version 9.0.0). Tables
Table 1. Demographic and clinical characteristics of control and atrial fibrillation group.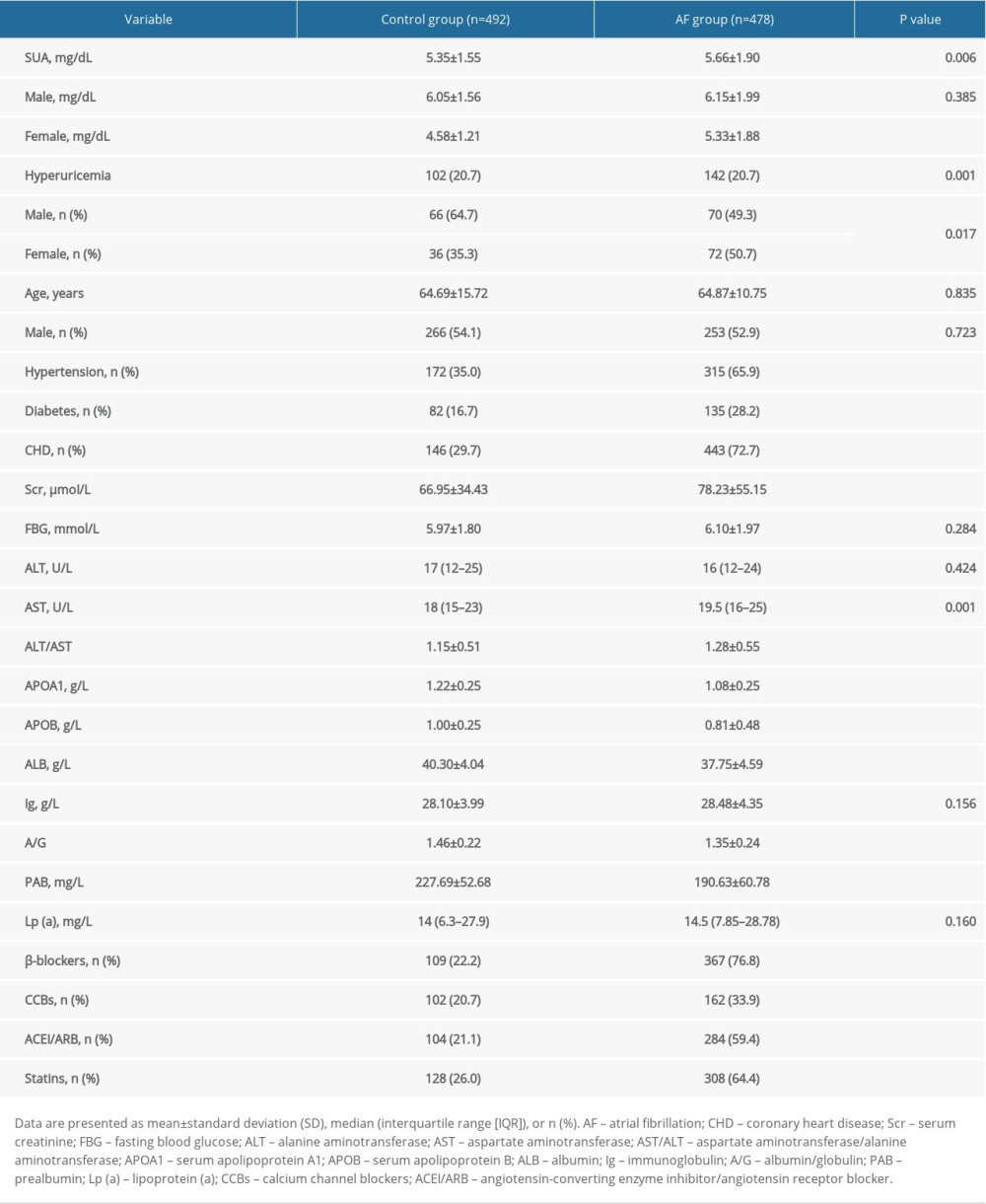 Table 2. Association between serum uric acid levels and atrial fibrillation.
Table 2. Association between serum uric acid levels and atrial fibrillation.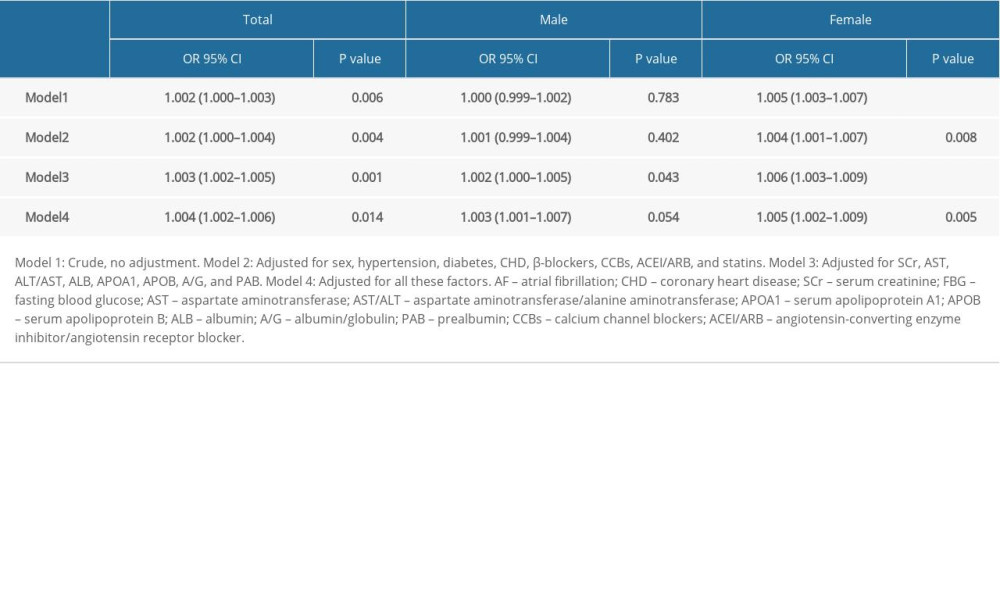 Table 3. Association of serum uric acid levels and atrial fibrillation by subtypes and complications.
Table 3. Association of serum uric acid levels and atrial fibrillation by subtypes and complications.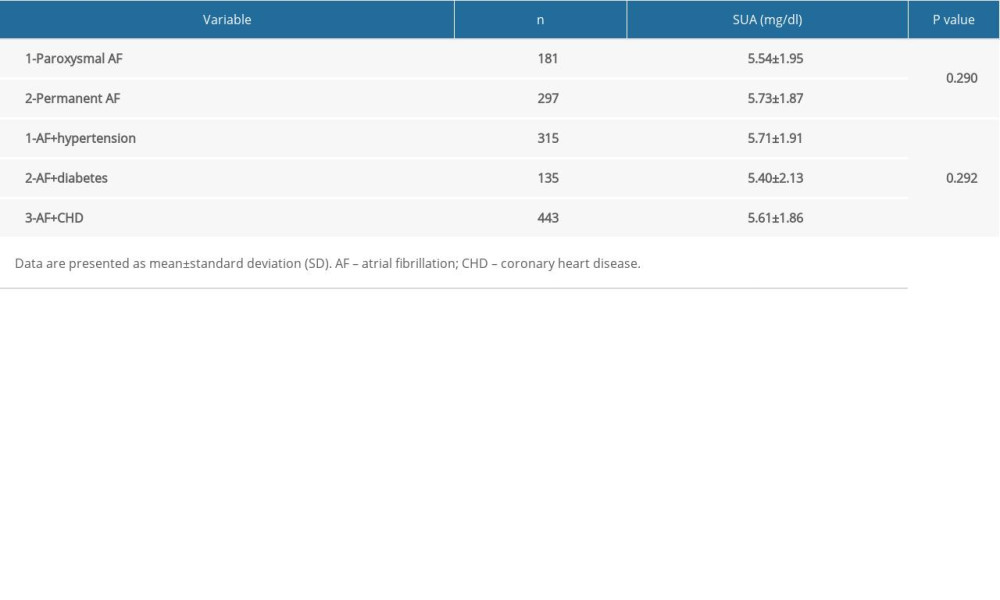 Table 4. Association of serum uric acid levels and metabolic indicators in patients with atrial fibrillation by sex.
Table 4. Association of serum uric acid levels and metabolic indicators in patients with atrial fibrillation by sex.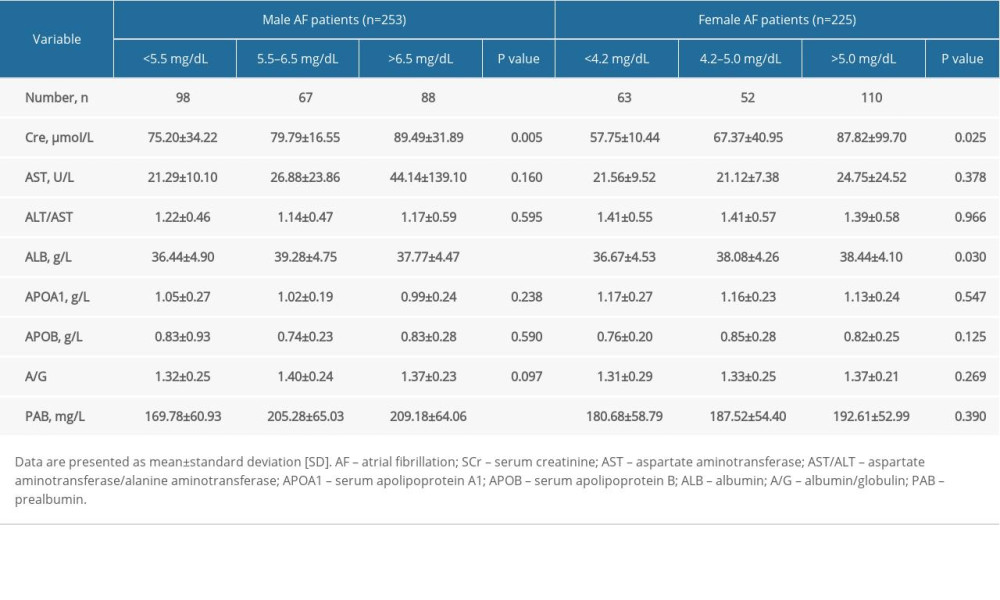
References
1. Chugh SS, Havmoeller R, Narayanan K, Worldwide epidemiology of atrial fibrillation: A Global Burden of Disease 2010 Study: Circulation, 2014; 129(8); 837-47
2. Ren J, Yang Y, Zhu J, The characteristics, long-term outcomes, risk factors, and antithrombotic therapy in Chinese patients with atrial fibrillation and bioprosthetic valves: Front Cardiovasc Med, 2021; 8; 665124
3. Jing Z, Xuewen L, Study on the correlation between inflammatory factors, oxidative stress and atrial fibrillation myocardial remodeling: J Integr Trad Chin West Med Cardio Cerebrovasc Dis, 2014; 12(05); 565-67
4. Seesing MFJ, Scheijmans JCG, Borggreve AS, The predictive value of new-onset atrial fibrillation on postoperative morbidity after esophagectomy: Dis Esophagus, 2018; 31(11); doy028
5. Bencivenga L, Komici K, Nocella P, Atrial fibrillation in the elderly: A risk factor beyond stroke: Ageing Res Rev, 2020; 61; 101092
6. Ganesan AN, Shipp NJ, Brooks AG, Long-term outcomes of catheter ablation of atrial fibrillation: A systematic review and meta-analysis: J Am Heart Assoc, 2013; 2(2); e004549
7. Weerasooriya R, Khairy P, Litalien J, Catheter ablation for atrial fibrillation: Are results maintained at 5 years of follow-up?: J Am Coll Cardiol, 2011; 57; 160-66
8. Tamariz L, Agarwal S, Soliman EZ, Association of serum uric acid with incident atrial fibrillation (from the Atherosclerosis Risk in Communities [ARIC] study): Am J Cardiol, 2011; 108(9); 1272-76
9. Glantzounis GK, Tsimoyiannis EC, Kappas AM, Galaris DA, Uric acid and oxidative stress: Curr Pharm Des, 2005; 11(32); 4145-51
10. Ruggiero C, Cherubini A, Ble A, Uric acid and inflammatory markers: Eur Heart J, 2006; 27; 11
11. Wannamethee SG, Papacosta O, Lennon L, Whincup PH, Serum uric acid as a potential marker for heart failure risk in men on antihypertensive treatment: The British Regional Heart Study: Int J Cardiol, 2018; 252; 187-92
12. Canpolat U, Aytemir K, Yorgun H, Usefulness of serum uric acid level to predict atrial fibrillation recurrence after cryoballoon-based catheter ablation: Europace, 2014; 16(12); 1731-37
13. He XN, Li SN, Zhan JL, Serum uric acid levels correlate with recurrence of paroxysmal atrial fibrillation after catheter ablation: Chin Med J (Engl), 2013; 126(5); 860-64
14. Tamariz L, Hernandez F, Bush A, Association between serum uric acid and atrial fibrillation: A systematic review and meta-analysis: Heart Rhythm, 2014; 11(7); 1102-8
15. Kuwabara M, Niwa K, Nishihara S, Hyperuricemia is an independent competing risk factor for atrial fibrillation: Int J Cardiol, 2017; 231; 137-42
16. Calkins H, Hindricks G, Cappato R, 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation: Europace, 2018; 20(1); e1-160
17. Zhu Y, Pandya BJ, Choi HK, Prevalence of gout and hyperuricemia in the US general population: The National Health and Nutrition Examination Survey 2007–2008: Arthritis Rheum, 2011; 63; 3136-41
18. Ono K, How is uric acid related to atrial fibrillation?: Circ J, 2019; 83(4); 705-6
19. Yang Y, Qin X, Li Y, Relationship between serum uric acid and mortality risk in hemodialysis patients: A multicenter prospective cohort study: Am J Nephrol, 2020; 51(10); 823-32
20. Watanabe E, Uric acid and atrial fibrillation-cause or other association?: Circulation, 2012; 76; 584-85
21. Nyrnes A, Toft I, Njølstad I, Uric acid is associated with future atrial fibrillation: An 11-year follow-up of 6308 men and women – the Tromso Study: Europace, 2014; 16(3); 320-26
22. Tian TT, Li H, Chen SJ, Serum uric acid as an independent risk factor for the presence and severity of early-onset coronary artery disease: A case-control study: Dis Markers, 2018; 2018; 1236837
23. Borghi C, Tykarski A, Widecka K, Expert consensus for the diagnosis and treatment of patient with hyperuricemia and high cardiovascular risk: Cardiol J, 2018; 25(5); 545-63
24. Borghi C, Domienik-Karłowicz J, Tykarski A, Expert consensus for the diagnosis and treatment of patient with hyperuricemia and high cardiovascular risk: 2021 update: Cardiol J, 2021; 28(1); 1-14
25. Kanellis J, Kang DH, Uric acid as a mediator of endothelial dysfunction, inflammation, and vascular disease: Semin Nephrol, 2005; 25; 39-42
26. Strazzullo P, Puig JG, Uric acid and oxidative stress: relative impact on cardiovascular risk?: Nutr Metab Cardiovasc Dis, 2007; 17; 409-14
27. Pak S, Yatsynovich Y, Valencia D, Chen T, serum uric acid and atrial fibrillation: Meta-analysis: Crit Pathw Cardiol, 2018; 17(3); 161-66
28. Korantzopoulos P, Kolettis TM, Galaris D, The role of oxidative stress in the pathogenesis and perpetuation of atrial fibrillation: Int J Cardiol, 2007; 115; 135-43
29. DudLey SC, Hoch NE, McCann LA, Atrial fibrillation increases production of superoxide by the left atrium and left atrial appendage: Role of the NADPH and xanthine oxidases: Circ J, 2005; 112; 1266-73
30. Kang DH, Han L, Ouyang X, Uric acid causes vascular smooth muscle cell proliferation by entering cells via a functional urate transporter: Am J Nephrol, 2005; 25; 425-33
31. Baldwin W, McRae S, Marek G, Hyperuricemia as a mediator of the proinflammatory endocrine imbalance in the adipose tissue in a murine model of the metabolic syndrome: Diabetes, 2011; 60; 1258-69
32. Tsai CT, Lai LP, Kuo KT, Angiotensin II activates signal transducer and activators of transcription 3 via Rac1 in atrial myocytes and fibroblasts: Implication for the therapeutic effect of statin in atrial structural remodeling: Circulation, 2008; 117; 344-55
33. Korantzopoulos P, Letsas KP, Liu T, Xanthine oxidase and uric acid in atrial fibrillation: Front Physiol, 2012; 3; 150
34. Maharani N, Kuwabara M, Hisatome I, Hyperuricemia and atrial fibrillation: Int Heart J, 2016; 57(4); 395-99
35. Suzuki S, Sagara K, Otsuka T, Gender-specific relationship between serum uric acid level and atrial fibrillation prevalence: Circ J, 2012; 76; 607-11
36. Sun GZ, Guo L, Wang J, Association between hyperuricemia and atrial fibrillation in rural China: A cross-sectional study: BMC Cardiovasc Disord, 2015; 15; 98
37. Letsas KP, Korantzopoulos P, Filippatos GS, Uric acid elevation in atrial fibrillation: Hellenic J Cardiol, 2010; 51; 209-13
38. Zacharia E, Papageorgiou N, Ioannou A, Inflammatory biomarkers in atrial fibrillation: Curr Med Chem, 2019; 26(5); 837-54
39. Youn JY, Zhang J, Zhang Y, Oxidative stress in atrial fibrillation: An emerging role of NADPH oxidase: J Mol Cell Cardiol, 2013; 62; 72-79
40. Arques S, Human serum albumin in cardiovascular diseases: Eur J Intern Med, 2018; 52; 8-12
41. Roche M, Rondeau P, Singh NR, The antioxidant properties of serum albumin: FEBS Lett, 2008; 582; 1783-87
42. Li K, Fu W, Bo Y, Zhu Y, Effect of albumin-globulin score and albumin to globulin ratio on survival in patients with heart failure: A retrospective cohort study in China: BMJ Open, 2018; 8(7); e022960
43. Azab B, Bibawy J, Harris K, Value of albumin-globulin ratio as a predictor of all-cause mortality after non-ST elevation myocardial infarction: Angiology, 2013; 64(2); 137-45
44. Liao LZ, Zhang SZ, Li WD, Serum albumin and atrial fibrillation: Insights from epidemiological and mendelian randomization studies: Eur J Epidemiol, 2020; 35(2); 113-22
45. Pal GK, Shyma P, Habeebullah S, Association of albumin-globulin ratio with sympathovagal imbalance in pregnancy-induced hypertension: Indian J Physiol Pharmacol, 2011; 55(2); 128-38
46. Wessel N, Berg K, Kraemer JF, Cardiac autonomic dysfunction and incidence of de novo atrial fibrillation: heart rate variability vs heart rate complexity: Front Physiol, 2020; 11; 596844
47. Kawano H, Arakawa S, Satoh O, Effect of pimobendan in addition to standard therapy for heart failure on prevention of readmission in elderly patients with severe chronic heart failure: Geriatr Gerontol Int, 2014; 14(1); 109-14
48. Franco J, Formiga F, Trullas JC, Impact of prealbumin on mortality and hospital readmission in patients with acute heart failure: Eur J Intern Med, 2017; 43; 36-41
49. Chistiakov DA, Orekhov AN, Bobryshev YV, ApoA1 and ApoA1-specific self-antibodies in cardiovascular disease: Lab Invest, 2016; 96(7); 708-18
50. Pol T, Held C, Westerbergh J, Dyslipidemia and risk of cardiovascular events in patients with atrial fibrillation treated with oral anticoagulation therapy: Insights from the ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation) trial: J Am Heart Assoc, 2018; 7(3); e007444
51. Du R, Winarsih I, Ho B, Ding JL, Lipid-free apolipoprotein A-I exerts an antioxidative role against cell-free hemoglobin: Am J Clin Exp Immunol, 2012; 1; 33-48
52. Barter PJ, Puranik R, Rye KA, New insights into the role of hdL as an anti-inflammatory agent in the prevention of cardiovascular disease: Curr Cardiol Rep, 2007; 9; 493-98
53. Çınar T, Tanık VO, Gürkan K, Comparison of apolipoprotein-A1 levels between paroxysmal atrial fibrillation patients and healthy subjects: J Cardiovasc Thorac Res, 2020; 12(2); 140-44
Errate
Figures
 Figure 1. SUA levels in AF group and controls. Serum uric acid levels in AF group were significantly increased than controls (5.66±1.90 vs 5.35±1.55 mg/dL, P=0.006). Abbreviations as in Table 1. The figure was created by GraphPad Prism software (version 9.0.0).
Figure 1. SUA levels in AF group and controls. Serum uric acid levels in AF group were significantly increased than controls (5.66±1.90 vs 5.35±1.55 mg/dL, P=0.006). Abbreviations as in Table 1. The figure was created by GraphPad Prism software (version 9.0.0). Figure 2. SUA levels in AF and control groups by gender. SUA levels were significantly higher AF patients than in the female controls(5.33±1.88 vs 4.58±1.21 mg/dL, P<0.001). The difference was not observed in male of two groups. Abbreviations as in Table 1. The figure was created by GraphPad Prism software (version 9.0.0).
Figure 2. SUA levels in AF and control groups by gender. SUA levels were significantly higher AF patients than in the female controls(5.33±1.88 vs 4.58±1.21 mg/dL, P<0.001). The difference was not observed in male of two groups. Abbreviations as in Table 1. The figure was created by GraphPad Prism software (version 9.0.0). Figure 3. The influence factors of SUA levels in AF patients. (A) Correlation between SUA levels and A/G in AF patients (r=0.318, p=0.046). (B) Correlation between SUA levels and PAG in AF patients(r=0.138, p=0.002). (C) Correlation between SUA levels and A1 (APOA1) in AF patients (r=−0.201, p<0.001). Abbreviations as in Table 1. The figures were created by SPSS version 26.0 (SPSS Inc., Chicago, IL, USA).
Figure 3. The influence factors of SUA levels in AF patients. (A) Correlation between SUA levels and A/G in AF patients (r=0.318, p=0.046). (B) Correlation between SUA levels and PAG in AF patients(r=0.138, p=0.002). (C) Correlation between SUA levels and A1 (APOA1) in AF patients (r=−0.201, p<0.001). Abbreviations as in Table 1. The figures were created by SPSS version 26.0 (SPSS Inc., Chicago, IL, USA). Figure 4. Multiple-adjusted odds ratios (ORs), with 95% confidence intervals (95% CIs) of the association between SUA levels and AF. The figure was created by GraphPad Prism software (version 9.0.0).
Figure 4. Multiple-adjusted odds ratios (ORs), with 95% confidence intervals (95% CIs) of the association between SUA levels and AF. The figure was created by GraphPad Prism software (version 9.0.0). Tables
 Table 1. Demographic and clinical characteristics of control and atrial fibrillation group.
Table 1. Demographic and clinical characteristics of control and atrial fibrillation group. Table 2. Association between serum uric acid levels and atrial fibrillation.
Table 2. Association between serum uric acid levels and atrial fibrillation. Table 3. Association of serum uric acid levels and atrial fibrillation by subtypes and complications.
Table 3. Association of serum uric acid levels and atrial fibrillation by subtypes and complications. Table 4. Association of serum uric acid levels and metabolic indicators in patients with atrial fibrillation by sex.
Table 4. Association of serum uric acid levels and metabolic indicators in patients with atrial fibrillation by sex. Table 1. Demographic and clinical characteristics of control and atrial fibrillation group.
Table 1. Demographic and clinical characteristics of control and atrial fibrillation group. Table 2. Association between serum uric acid levels and atrial fibrillation.
Table 2. Association between serum uric acid levels and atrial fibrillation. Table 3. Association of serum uric acid levels and atrial fibrillation by subtypes and complications.
Table 3. Association of serum uric acid levels and atrial fibrillation by subtypes and complications. Table 4. Association of serum uric acid levels and metabolic indicators in patients with atrial fibrillation by sex.
Table 4. Association of serum uric acid levels and metabolic indicators in patients with atrial fibrillation by sex. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








