10 May 2022: Lab/In Vitro Research
Impact of Factors Secreted by Tumor Cells on Response of Pleural Mesothelial Cells to Different Sclerosing Agents in an In Vitro Model
Michał MierzejewskiDOI: 10.12659/MSM.936065
Med Sci Monit 2022; 28:e936065
Abstract
BACKGROUND: Chemical pleurodesis is one of the major therapeutic options for patients with recurrent malignant pleural effusion. Mesothelial cells are considered to play a pivotal role in the response to different chemical compounds (sclerosants) used for pleurodesis. Malignant cells might have an impact on the mesothelial response to applied sclerosing agents and, in consequence, on the efficacy of pleurodesis. We aimed to evaluate the impact of cancer cell paracrine on mesothelial cell response to different sclerosing agents.
MATERIAL AND METHODS: The study used mesothelial cell (MeT-5A) cultures stimulated with sclerosing agents (talc, doxycycline, iodopovidone, and TGF-β for 24 h) in the presence or absence of supernatants from adenocarcinoma cultures (HCC827). The mesothelial mRNA expression and protein levels of IL-6, IL-8, and TGF-β was assessed. Further, lung fibroblasts were cultured with and without cell supernatants from previously established cell cultures for 24 h. Then, concentration of soluble collagen was evaluated in culture supernatants.
RESULTS: The exposure of mesothelial cells to sclerosants decreased the concentration of IL-6 and IL-8 protein. The addition of mediators secreted by adenocarcinoma altered the inflammatory response of the mesothelial cells to sclerosing agents. IL-8 concentration in cultures stimulated with talc and adenocarcinoma supernatant was higher compared to cultures stimulated with talc only. The exposure of lung fibroblasts to supernatant from mesothelial cell (with or without adenocarcinoma) did not affect collagen secretion.
CONCLUSIONS: An addition of soluble factors produced by adenocarcinoma altered the inflammatory response of the pleural mesothelial cells after stimulation with sclerosing agents. Our observations suggest that the tumor paracrine effect affects biological pathways of pleurodesis.
Keywords: Pleurodesis, Sclerosing Solutions, Talc, Adenocarcinoma, Collagen, Humans, Interleukin-6, Interleukin-8, Pleura, Pleural Effusion, Malignant
Background
The pleura is a thin membrane that covers the outer surface of the lungs as well as the mediastinum, diaphragm, and inner surface of the chest wall. Together, these surfaces delimit the pleural cavity, which resembles a collapsed sac that encases the lung. Physiologically, this cavity contains a minimal amount of lubricating fluid. A balance between pleural fluid formation and reabsorption can be disrupted in numerous diseases, leading to accumulation pleural effusion, which is clinically manifested primarily as exertional or resting dyspnea.
Pleural effusion is a common and debilitating complication of various malignancies. It has been estimated that approximately 200 000 and 50 000 new cases of malignant pleural effusion (MPE) are diagnosed each year in the United States and United Kingdom, respectively [1,2]. Lung and breast carcinoma have been reported to be responsible for 53–75.2% of all MPEs [3,4]. There are data showing that up to 30% of all lung cancer patients and 7–11% of breast cancer patients develop pleural effusion in the course of the disease [5]. Furthermore, it is estimated that MPE occurs in nearly 50% of all patients with metastatic cancers [6]. Since malignant pleural involvement is usually a manifestation of advanced (metastatic) disease, curative treatment is only occasionally possible, and pleurodesis is 1 of only 2 therapeutic options (the other is indwelling pleural catheter [IPC]) that can offer a long-term symptomatic effect. Chemical pleurodesis is the administration of a sclerotizing agent (eg, talc) into the pleural cavity, aimed at ablation of the pleural cavity. Its objective is to obtain an adhesion of the parietal and visceral pleura to form a permanent symphysis. It is performed to prevent accumulation of fluid from pleural cavity, which is common in neoplastic disease with pleural metastases. Accumulation of the fluid in the pleura often causes shortness of breath and decreased exercise tolerance. Procedures that can prevent pleural fluid recurrence are of great importance in palliative care. The latter method is less invasive and therefore is widely preferred to treat patients with recurrent MPE. The efficacy of chemical pleurodesis in patients with MPE is estimated at between 41.3% and almost 100% [7–9], depending on a range of factors, including the underlying cancer type and sclerosing agent used. A wide variety of sclerosants have been used in the past, but nowadays talc and doxycycline are mainly used in everyday practice worldwide. Povidone-iodine (PVP-I) is a less commonly used but promising sclerotic agent. Research on this chemical compound is ongoing. TGF-β has not been used in human studies, but it is an interesting subject of research due to its unique properties – it creates pleural adhesions without causing inflammation. Although there has been more than 100 years of experience with chemical pleurodesis, information on the mechanisms of action of various sclerosing agents is very incomplete [10].
Contrary to an early belief, the pleura is not merely a mechanical transmitter of pressure changes in the chest, but is an active organ involved in fluid exchange, cell-to-cell interactions and secretion of mediators. Histologically, the pleura is composed of a single layer of mesothelial cells on the layer of connective tissue. Due to the large number of these cells and their presence in the entire pleura, mesothelial cells play a pivotal role in the response to pleural injury. A growing amount of data emphasizes the role of the mesothelial cells as one of the most important structural elements that determine the efficacy of pleurodesis. The cascade of reactions leading to the formation of pleural adhesions is believed to be triggered by action of the sclerosing agent on normal mesothelial cells. They seem to be the main source of cytokines and initiate the early stages of inflammation by increasing the influx of other cells (including neutrophils and fibroblasts), eventually leading to pleural fibrosis. In response to chemical stimulants, mesothelial cells secrete interleukin 8 (IL-8), monocyte chemoattractant protein (MCP-1), vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), transforming growth factor β (TGF-β), basic fibroblast growth factor (bFGF), and a variety of other mediators [11], eventually leading to fibroblast proliferation and production of collagen and extracellular matrix components. Thus, in the pleurodesis process, this group of cells is primarily affected by the administered drug (sclerosant), and the effectiveness of the pleurodesis process depends on their correct response. The mechanisms of pleurodesis are still poorly understood. It seems possible that the magnitude of some mesothelial cell responses is related to the chemical used for pleurodesis [12]. The extent of malignant pleural involvement is thought to be another important factor adversely affecting the efficacy of pleurodesis. It is well-known that the procedure is less successful in patients with extensive malignant pleural involvement. In part, this may be explained by impaired lung expandability and the lack of proper apposition of visceral and parietal pleura, which can occur by reducing the number of healthy mesothelial cells by infiltrating the tumor. This phenomenon is also probably linked to a number of mechanisms, including physical obstacles in the close contact between 2 pleural layers, more rapid fluid reaccumulation, lower pH of the fluid, and lower number of intact mesothelial cells able to respond to the sclerosant [13–15]. It is possible that the direct action of the substances secreted from cancer cells on the interactions and their intensity between the normal mesothelium and other cells, including fibroblasts, is linked to the lower efficiency of pleurodesis in cancer patients. There are scant data on the direct influence of factors released from malignant cells on the response of mesothelial cells to sclerosing agents. Considering the large number of cytokines, growth factors, and other bioactive substances that are secreted by malignant cells [16], it can be hypothesized that they affect the mesothelial response to the applied sclerosing agents and, in consequence, on the efficacy of pleurodesis. This largely unknown interplay may affect our understanding of the mechanisms of pleurodesis and the ability to predict its outcome. However, an additional mechanism in which the paracrine action of cancer cells impairs the response of mesothelial cells to the applied sclerosants has been postulated. Considering the aforementioned gap in knowledge of the interaction among the sclerosing agent, mesothelial cells, and tumor cells, we undertook the present study to shed more light on this issue.
This in vitro study was designed to evaluate possible mechanisms affecting the response of mesothelial cells to different sclerosing agents. We assumed that this response, including an interplay between mesothelial cells and fibroblasts, can be affected by active mediators secreted by tumor cells. In the vast majority of cases, neoplastic lesions in the pleura are metastatic, with the most common being adenocarcinoma. Therefore, we used adenocarcinoma cells to assess the role of neoplastic cells in our experiment. Fibroblasts were cultured to simulate the final stage of pleurodesis—fibrosis. Therefore, 3 cell types were selected for in vitro experiments and cultures: mesothelial cells, fibroblasts, and adenocarcinoma cells. We used supernatant from adenocarcinoma as a model of the diverse set of factors (eg, proteins, lipids, exosomes, and other small molecular messengers) secreted and released from adenocarcinoma. The aim of this study was to evaluate the impact of biologically active mediators secreted by cancer cells on (1) mesothelial cell responses to different sclerosing agents and (2) the effect of mesothelial cell-derived mediators on collagen production by lung fibroblasts.
Material and Methods
GENERAL STUDY DESIGN:
The design of this study included MeT-5A cells, which are normal, non-cancerous pleura cells, and their culture was intended to represent a healthy pleura. The human adenocarcinoma culture was the source of supernatant containing all the biochemical factors secreted by this group of cells. The supernatant from adenocarcinoma was added to an experimental group of mesothelial cells to simulate a cancer paracrine effect. Fibroblasts were cultured with supernatants from mesothelial cells to evaluate the effect on collagen production.
This was an experimental study based on human mesothelial cell (MC) cultures: MeT-5A, human adenocarcinoma cell (ADC) cultures (HCC827), and human lung fibroblast cultures (CC-2512). The study was conducted in 2 phases. In the first, ADC and MC were cultured. This phase included the evaluation of MC responses to different sclerosing agents tested in the presence and absence of supernatants from ADC cultures. ADC cells were cultured until 100% confluence, then the supernatant from cells was collected. MeT-5A cultures were divided into 2 groups: cultured with standard medium or cultured with ADC supernatant addition. All MC cultures (with or without ADC) were incubated with or without sclerosing agent for 24 h (Figure 1). Briefly, the experiment included 4 MC cultures:
Importantly, there were 4 different subsets of sclerosant-treated cultures, each with a different type of chemical used: talc, PVP-I, doxycycline, and TGF-β (see the section
The second phase of the study was designed to assess the profibrotic activity of mediators secreted by the mesothelial cells stimulated with sclerosants incubated with or without ADC supernatant. For this purpose, lung fibroblasts (at 80% of confluency) were cultured with or without cell supernatants from 2 different sets of previously established cell cultures (150 μL per total 500 μL) for 24 h (Figure 1). Then, the concentration of soluble collagen was evaluated in culture supernatants.
Different controls were used in this study. For mesothelium: (1) unstimulated cells, (2) cells with 150 μL of pure RPMI per total 500 μL – this control stands for A+ cultures and controls if response of mesothelial cells (MeT-5A) with ADC supernatant is not influenced by medium used for cancer cell cultivation (RPMI), which is different than in MeT-5A cells. (3) For controlling the level of mediators produced by the cancer cell line, we used the control in which mesothelial cells (MeT-5A) were cultivated for 24 h with supernatant from ADC without any sclerosants (A+S−): unstimulated cells with 150 μL of ADC supernatant per total 500 μL – this control evaluates the profile of inflammatory load added with ADC supernatant to MeT-5A cells. For fibroblasts, the control with untreated fibroblasts was used to measure a background collagen production.
CELL CULTURES:
Human immortalized mesothelial cells isolated from pleural fluids obtained from non-cancerous individuals MeT-5A (ATCC CRL-9444) were purchased from ATCC (VA, USA). These cells were cultured in M199 medium (Sigma Aldrich, MO, USA) supplemented with 3.3 nM mouse epidermal growth factor (EGF) (Corning, NY, USA), 400 nM hydrocortisone (Sigma Aldrich, MO, USA), 870 nM zinc-free bovine insulin (Sigma Aldrich, MO, USA), 20 mM HEPES (Gibco, Thermo Fisher, MA, USA), and 0.001% Trace elements (Corning, NY, USA). Human lung fibroblasts (CC-2512) were purchased from Lonza (NY, USA) and cultured in Minimum Essential Medium (MEM) (Gibco, Thermo Fisher, MA, USA) supplemented with 1% MEM Non-Essential Amino Acids (Gibco, Thermo Fisher, MA, USA). Human lung adenocarcinoma cells HCC827 (ATCC CRL-2868) were provided by ATCC (VA, USA) and cultured in RPMI (Gibco, Thermo Fisher, MA, USA). All culture media were supplemented with 10% fetal bovine serum (Thermo Fisher, MA, USA) and 1% antibiotic cocktail (Gibco, Thermo Fisher, MA, USA). Cells were cultured at 37ºC in 5% CO2 at passage 3–5.
SCLEROSANTS:
The following agents were used as sclerosants: Talc (Novatech, France) at a dose of 20 μg/mL, PVP-I (ChemCruz, TX, USA) at a concentration of 0.001%, doxycycline (Sigma Aldrich, MO, USA) at a dose of 2 μg/mL, and TGF-β (R&D Systems, MI, USA) at a dose of 0.2 μg/mL.
PROTEIN CONCENTRATION MEASUREMENT:
The levels of the IL-6, IL-8, and TGF-β in cell culture supernatants were measured using commercially available streptavidin-biotin-based ELISA kits: human IL-6 (88–7066), human IL-8 (88–8086), and human/mouse TGF-β (88–8350) (Thermo Fisher, MA, USA). The sensitivity of kits was 2 pg/mL, 2 pg/mL, and 8 pg/mL for IL-6, IL-8, and TGF-β, respectively. Briefly, ELISAs were performed in microplates (Costar, Cambridge, MA, USA) sensitized with specific monoclonal antibodies for each cytokine, according to the manufacturer’s instructions. After washing and distribution of the samples, specific biotin-conjugated antibodies were added. After incubation with streptavidin-peroxidase, substrate was added for development of the reaction. The absorbance was measured on an Infnite M Nanono microplate reader (Tecan, Switzerland) at 450 nm with reference to 570 nm. All standards and samples were measured in duplicate.
COLLAGEN LEVEL MEASUREMENTS:
Collagen concentration in supernatants from fibroblast cultures was measured by colorimetric Soluble Collagen Assay Sircol (Biocolor, UK) according to the manufacturer’s procedure (test sensitivity 1.0 μg/100 μL collagen). Briefly, 1 mL of Sircol dye was added to 100 μL of each sample, incubated for 30 min, and then centrifuged at 13 000×g for 10 min. The pellets were washed with Acid-Salt Wash Reagent and drained. We added 250 μL of alkaline metal reagent to each tube before vortex mixing. We transferred 200 μL of the sample onto a 96-well plate, and absorbance was measured on an Infnite M Nanono microplate reader (Tecan, Switzerland) at 555 nm. Absolute values were obtained with a standard curve (collagen type I standard supplied with the kit) in the range 0.5 to 15 μg/100 μL.
RNA ISOLATION AND REVERSE TRANSCRIPTION:
Total RNA was isolated from the cells using Trizol (Sigma Aldrich, MO, USA). The concentration and purity of isolated RNA was measured on a DU650 spectrophotometer (Beckman Coulter, CA, USA). One microgram of total RNA was used for reverse transcription (Thermo Fisher, MA, USA).
REAL-TIME PCR:
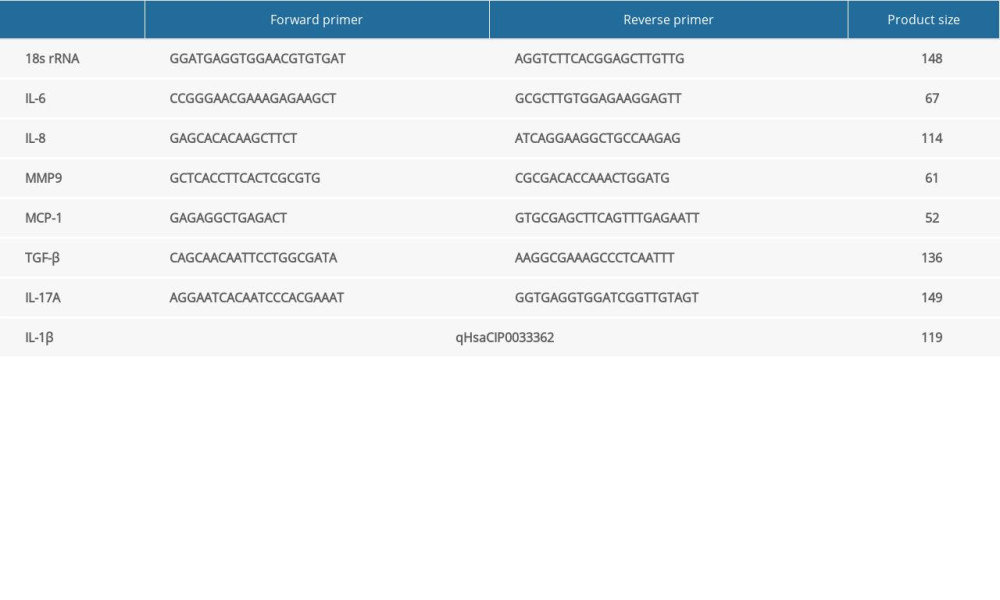
The real-time PCR was performed with an ABI-Prism 7500 Sequence Detector System (Applied Biosystems, Foster City, USA). For PCR reaction, 0.8 μL of cDNA was amplified in 16 μL PCR volume, containing a Power SYBR Green PCR Mastermix (Thermo Fisher, MA, USA) with 150 nM of specific primers (Thermo Fisher, MA, USA, BioRad, CA, USA) (Table 1) as standard procedure (95°C for 15 s and 60°C for 60 s for 40 cycles). We applied 18s rRNA for each sample as an endogenous control to normalize gene expression levels. The unstimulated cells from each experiment were used as a calibrator. Each sample was measured in duplicate. Relative quantification values were calculated by the 2−ΔΔCT method according to Livak et al [17]. Data are expressed as expression levels relative to the mean ΔCT of control unstimulated mesothelial cells. The results are expressed as relative quantification units (fold change).
STATISTICAL ANALYSIS:
Statistical analyses were performed using Statistica 12.0 software (StatSoft, Inc., Tulsa, USA) and GraphPad (version 9.3.1). Data are presented as median and interquartile range (IQR). Data distribution was tested using the Shapiro-Wilk test. Differences between continuous variables were tested using the nonparametric Mann-Whitney U test. Differences were considered statistically significant at
Results
MRNA EXPRESSION OF MEDIATORS IN MESOTHELIAL CELLS:
The impact of sclerosants on inflammatory mediators was weakly seen on mRNA level in 24 h (Figures 2A-2D, 3A–3C). Generally, sclerosants upgraded mRNA expression of evaluated markers (Figures 2A, 2B, 2D, 3B, 3C), with statistical significance found for IL-17 mRNA after talc treatment (14.92-fold change [0.30- to 17.52-fold change]) compared to controls (0.36-fold change [0.20- to 30.84-fold change]), P=0.04 (Figure 2D). MMP9 mRNA expression was elevated in A+S+ (TGF-β) culture (4.21-fold change [2.22- to 6.26-fold change]) compared to control with ADC (A+S−) (0.79-fold change [0.44- to 0.88-fold change]), P=0.03 (Figure 3B). Talc was the only sclerosant that caused increase of IL-6 mRNA expression in A+S+ culture (1.82-fold change [1.22- to 2.01-fold change]) compared to A-S+ culture (1.05-fold change [0.85- to 1.55-fold change]), P=0.04 (Figure 2B), with simultaneous lack of significant difference compared to or between untreated controls.
IL-6, IL-8, AND TGF-β PROTEIN LEVEL:
In the group of mesothelial cells without ADC application, all sclerosants except TGF-β were associated with decreased IL-6 production by MeT-5A cells (A-S+) after 24 h of incubation. A significant downregulation of IL-6 level was found for doxycycline (18.95 pg/mL [15.98 to 20.68 pg/mL], P=0.03), PVP-I (21.86 pg/mL [18.84 to 23.85 pg/mL], P=0.03 and talc (23.22 pg/mL [21.41 to 24.16 pg/mL], P=0.048 compared to unstimulated controls (A-S−) (28.06 pg/mL [27.90 to 37.07 pg/mL]) (Figure 4A). The same was observed for IL-8. Doxycycline (43.12 pg/mL [41.02 to 54.77 pg/mL]), PVP-I (55.54 pg/mL [51.46 to 61.04 pg/mL]) and talc (54.93 pg/mL [47.64 to 62.69 pg/mL]) decreased IL-8 production by mesothelial cells in A-S+ cultures compared to A-S− controls (69.79 pg/mL [63.60 to 89.85 pg/mL]) (Figure 4B).
The analysis of mesothelial cells with ADC supernatant showed an elevated concentration of IL-6 in A+S+ cultures regardless of the sclerosant used: for doxycycline, 55.47 pg/mL (46.22–58.61 pg/mL) (P=0.002); for PVP-I, 57.89 pg/mL (52.55–78.78 pg/mL) (P=0.003); for talc, 50.44 pg/mL (49.40–61.98 pg/mL) (P=0.002); and for TGF-β, 96.14 pg/mL (87.49–153.04 pg/mL) (P=0.003). However, due to an elevated level of IL-6 in A+S− control, results were 63.19 pg/mL (55.69 to 78.84 pg/mL) vs unstimulated control cells (A-S−) 28.06 pg/mL (27.89 to 37.07 pg/mL) (P=0.03) – this observation is related to inflammatory load of ADC supernatant rather than the effect of sclerosants on mesothelial inflammatory response per se (Figure 4A).
A similar pattern of changes was observed for IL-8 protein. The cultures with ADC supernatant were characterized by elevated level of IL-8 compared to mesothelial cells alone in both control and sclerosant-treated MeT-5A cells. An upgraded concentration of IL-8 was found in controls with ADC (A+S−) 97.61 pg/mL (93.42 to 99.8 pg/mL) compared to controls with RPMI (A-S−) 48.92 pg/mL (40.0 to 54.68 pg/mL) (P=0.004). IL-8 levels were increased in all A+S+ compared to A-S+ cultures (P=0.003, p=0.003, P=0.002, and P=0.002) for doxycycline, PVP-I, talc, and TGF-β respectively but not compared to A+S− controls, suggesting that the inflammatory profile of A+S+ is related to ADC load but is not caused by sclerosants. The only exception was found for A+S+ mesothelial cultures with talc – 89.75 pg/mL (80.23 to 92.12 pg/mL) – where the production of IL-8 was lower than in A+S− controls (P=0.02) (Figure 4B).
A significantly higher concentration of TGF-β protein was found in the cultures in which TGF-β was used as a sclerosant: A-S+ 59.13 pg/mL (53.49 to 62.05 pg/mL) and A+S+ 55.60 pg/mL (48.21–60.97 pg/mL) vs A-S− 2.68 pg/mL (2.52 to 3.00 pg/mL). However, it seems that due to the lack of increase in mRNA expression in these cultures, the increased protein concentration resulted from the presence of sclerosant and not from the increased protein production by cells (Figure 4C).
COLLAGEN SECRETION:
Cell supernatants from mesothelial/mesothelial+ADC cultures tended to stimulate (but not significantly) collagen production by lung fibroblasts. Fibroblasts stimulated with control (A-S−) mesothelial supernatants produced higher levels of collagen (1.00 μg [0.89 to 1.045 μg]) than fibroblasts stimulated with A-S+ (doxycycline (0.82 μg (0.72 to 0.83 μg) (P=0.01), and PVP-I (0.82 μg (0.78 to 0.84 μg) (P=0.02) – as well as with A+S+ cultures (doxycycline was 0.79 μg (0.74 to 0.84 μg) (P=0.01) and talc was 0.83 μg (0.63–0.89 μg) (P=0.048) (Figure 5).
Discussion
The present study showed various effects of various sclerosing agents on the inflammatory response of mesothelial cells. In general, the response of mesothelial cells to applied sclerosants was rather weak, with some exceptions, including a significant increase of IL-17A mRNA expression in cultures stimulated with PVP-I. The exposure of mesothelial cells to doxycycline, PVP-I, talc, and TGF-β was associated with decreased concentration of IL-6 and IL-8 protein in the culture supernatants. Although the reactions to sclerosants observed in our model were not particularly pronounced, the addition of supernatant from ADC cells altered the inflammatory response of the pleural mesothelial cells after sclerosant stimulation. Importantly, simultaneous treatment of mesothelial cells with sclerosing agents and ADC cell supernatant did not inhibit the expression of different inflammatory cytokines or the secretion of collagen by lung fibroblasts exposed to soluble factors produced by mesothelial cells. In contrast, some mesothelial cell responses to sclerosing agents (IL-6 and IL-8 concentration) were stronger compared to stimulation with the sclerosant alone.
The effect of selected sclerosants on mesothelial cells had been evaluated previously. In normal conditions, cytokines like IL-8 are produced by mesothelial cells [18] as attractants for neutrophils to the pleural cavity [19]. Talc was shown to increase production of pro-inflammatory cytokines, including IL-1, IL-6 [20], and IL-8 [21] in human pleural mesothelial cells in vitro and in vivo [22]. Furthermore, iodopovidone and doxycycline upgraded IL-8 secretion in a rabbit model of pleurodesis. This was in contrast to TGF-β, which was shown to stimulate collagen production without IL-8-mediated inflammation [23,24]. In a rabbit study of talc pleurodesis, Marchi et al found that IL-8 concentration in pleural fluid peaked at 6 h and its level remained high until 24 h after talc administration [22]. In an in vitro study with rabbit pleural mesothelial cells stimulated with talc, Acencio et al [25] showed that IL-8 concentration increased in a time-dependent manner. It has to be emphasized that animal studies demonstrated that nonsteroidal anti-inflammatory drugs (NSAIDs) and corticosteroids reduce the efficacy of pleurodesis mediated by tetracycline [26], talc [27], doxycycline [28], and iodopovidone [29]. The results of these studies show the important role of inflammation in pleurodesis.
Some our results differ from what we had expected based on the available literature. This is probably due to several reasons. The conditions used in similar studies were highly variable in terms of the time, doses of sclerosants, and type of cells used. For example, in vitro and in vivo animal studies by Lee et al [30] used primary pleural mesothelial cells obtained from C57BL/6 mice and TGF-β in doses of 0.01 to 100 ng/cm2 (0.2 μg/cm2 in our study), talc in doses of 1 to 100 μg/cm2 (10.5 μg/cm2 in our study), and doxycycline in doses of 0.1 to 1000 μg/cm2 (1.05 μg/cm2 in our study) for 4, 8, and 24 h. Antony et al [31] used human primary pleural mesothelial cells isolated from pleural fluid and stimulated with talc (4 μg/cm2) for 24 h. Ghio et al [21], who evaluated cell iron homeostasis after talc pleurodesis, used the same cell line as in our study (MeT-5A ATCC [VA, USA]), and applied 100 μg/mL talc (intended to be a “massive” dose) for 4 h. Our previous study [32], which used an in vitro model of primary mesothelial cells, found that among all evaluated sclerosing agents, talc presented the most efficient pro-inflammatory properties associated with modulation of IL-1β level. The results of this work differ from the previous one due to other mesothelial cells (immortalized normal mesothelial cell line) used in experiments. However, in both of our in vitro mesothelial studies, talc was found to cause the most relevant effect in inflammatory mesothelial response, confirming the hypothesis that this sclerosant has the highest potency in pleurodesis.
It was shown that high amounts of sclerosing factors along with long-term exposure to these agents decreased cell viability [33]. The experimental conditions applied in this study were based on our earlier work [32], which used primary cells isolated from the patient’s pleura specimen. It is possible that the in vitro conditions used in previous experiments were not suitable for the immortalized cell line. Discrepancies between our results and the results of other authors may be due to the different characteristics of rabbit and human cells, as well as different experimental conditions.
In the context of pleurodesis, TGF-β is a particularly interesting and important sclerosing agent. It is also a natural mediator associated with pleural mesothelial cells. It controls proliferation and differentiation and modulates the inflammatory process. In pleurodesis, TGF-β is a chemoattractant for fibroblasts and shows profibrotic properties. In a rabbit study by Marchi et al [22], the concentration of TGF-β after talc pleurodesis increased in a time-dependent manner, whereas Acencio et al [25], who used an in vitro cell culture, showed rapid elevation of TGF-β level, which remained stable after 48 h. In our study, the influence of the sclerosants themselves on the TGF-β concentration level was not visible, which may be due to methodological differences. As a sclerosant, TGF-β alone did not induce any significant effects on cytokine secretion. The only exception was a significantly higher concentration of TGF-β protein found in the cultures in which TGF-β was used as a sclerosant, but it seems that it results from the presence of sclerosant and not from the increased protein production by cells. Interestingly, we observed a significant increase in the concentrations of IL-6 and IL-8 after using TGF-β as a sclerosant with the soluble mediators secreted by ADC, pointing to an inflammatory response. According to the literature [34], TGF-β used as a sclerosant had not been associated with the intensification of the inflammatory process so far. Lee et al [24] showed that TGF-β induced pleurodesis without IL-8 release or an acute inflammatory response. This conclusion was also supported by the observation that high-dose parenteral corticosteroids do not inhibit TGF-β-mediated pleurodesis [35]. Possibly, TGF-β pleural symphysis occurs without an accompanying inflammatory reaction only in the non-cancerous pleura, which could explain the differences between the earlier findings of other authors and our results. However, this requires further research.
Besides of the assessment of the effect of various sclerosants on mesothelial cell inflammatory response, our study was also designed to evaluate the influence of the biologically active compounds produced by tumor cells alone and combined with different pleural sclerosants. The paracrine effect of the soluble factors secreted by tumor cells on other cells in the tumor microenvironment has been well described in neoplasms such as breast cancer [36]. Therefore, it can be assumed that it can also affect the process of pleurodesis. In our study, mesothelial culture cells stimulated with both sclerosing agents and supernatant from tumor cells demonstrated more pronounced inflammatory response (at least in some aspects, such as being expressed as an increased concentration of IL-6 and IL-8) than cells exposed to sclerosants alone. Moreover, the IL-8 concentrations were not significantly higher in the A+S− cultures compared to controls. Hence, elevated IL-8 levels in A+S+ compared to control seem to be related to the altered effect of the sclerosants. In contrast to IL-8, IL-6 concentrations were significantly higher in both the A+S− and A+S+ cultures compared to controls. Thus, it can be hypothesized that the effect was directly related to the mesothelial response to ADC supernatant and not to the sclerosants themselves. Either way, the above observations suggest that the tumor paracrine effect can influence the biological pathways of pleurodesis.
Among the tested sclerosants, the most pronounced inflammatory reaction was observed after stimulation with talc. This effect was visible in the increased expression of IL-17A in the A-S+ group and increased IL-6 and IL-8 concentration in the A+S+ in comparison to the A-S− and control groups. Such an effect is not surprising, as talc is regarded as the criterion standard for pleurodesis [37]. Talc induces pleuritis with histiocytic and granulomatous foreign body reaction [38]. However, inflammation is not the goal of pleurodesis per se, but is merely a means leading to pleural symphysis, as well as being the trigger of most adverse effects [10].
Previous studies provided evidence that an extensive malignant pleural involvement was associated with reduced probability of successful pleurodesis. In a retrospective study of 450 cases, Bielsa et al [39] showed a negative correlation between the extent of malignant pleural disease and the success rate of talc pleurodesis. This effect may be related not only to impaired lung re-expansion, but also to the disruption of mechanisms related to the biochemical pathways of pleurodesis [13,15]. This assumption seems to be in line with the observation by Martínez-Moragón et al [40] that low values of pleural glucose and pH (corresponding in part to the advancement of the neoplastic disease) are significantly associated with the probability of pleurodesis failure. Recently, the effect of pleural progression of lung carcinoma on the inflammatory process and fibrosis was investigated in an animal study by Sabbion et al [41]. They found that mice that received intrapleural injection of Lewis lung carcinoma cells and underwent pleurodesis had lower fibrosis and inflammation scores compared to controls that underwent pleurodesis alone. According to the authors, this may be due to the increased amount of fluid in the pleura in the neoplastic group, a reduced number of mesothelial cells, and a higher concentration of cytokines such as VEGF. It is possible that the inflammatory reaction of mesothelial cells on sclerosants and the effects of cancer are strictly dependent on their individual properties and local cytokine milieu. The present study was performed using immortalized mesothelial cells isolated from pleural fluids obtained from non-cancerous individuals and aimed to find basic inflammatory mechanisms of mesothelial cells in such conditions. It should be kept in mind that the response of pleural mesothelial cells to sclerosing agents and mediators produced by adenocarcinoma cell can vary and differ among individuals.
Although this study provides some new information about the inflammatory response of mesothelial cells, several limitations must be addressed before more definitive conclusions can be drawn. First, some significant changes could have been missed due to the relatively small sample size. Second, as already mentioned, this study used a commercially available cell line, in contrast to our previous study, in which primary cells isolated from pleura biopsies (with other experimental conditions the same) were used. Differences in cell biology may have caused bias. However, it should be stressed that the primary objective of the study was not the time- and dose-dependent analysis of mesothelial cell response to sclerosing agents, but the evaluation of the impact of cancer cells on sclerosant-related mesothelial cell reactions. Moreover, this study utilized monocultures in which the cell–cell interplay was impossible to analyze. Mesothelial cells/fibroblasts co-cultures might be a better model for evaluation of collagen production. Examination of the profibrotic effect, such as in co-cultures, remains to be considered in the future.
More research is needed using a wider range of stimulant doses and stimulation times. Future research should focus on identifying the mechanisms of pleurodesis related to the biology of neoplastic cells. It can be assumed that different types of cancer will have different effects on the pathways of pleurodesis. This might be consistent with the clinical observation that patients with MPE in the course of some malignancies (eg, breast cancer) have a better chance of achieving pleurodesis compared to others [39].
Conclusions
Our study showed that the addition of supernatant from adenocarcinoma cells altered the inflammatory response of the pleural mesothelial cells after sclerosant stimulation. This suggests that the tumor paracrine effect affects the biological pathways of pleurodesis. Talc resulted in the most pronounced inflammatory reaction of all sclerosants tested. Further evaluation of these phenomena in models with more complex co-cultures is warranted.
Figures
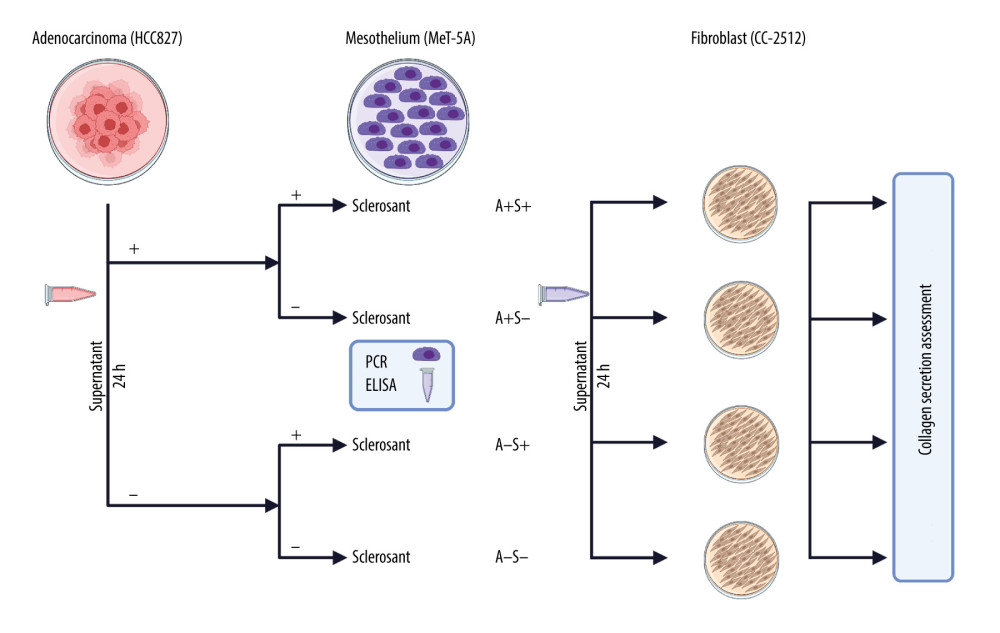 Figure 1. A flow chart presenting the general study design, including phase 1 and 2. Initially, adenocarcinoma (ADC) cells were cultured and supernatant was collected. After that, mesothelial cell cultures where incubated with or without supernatant from ADC and treated or not treated with a sclerosant. After 24 h, the levels of cytokines in cell culture supernatants and mRNA in cell lysates were measured (end of phase 1). Then, supernatants from mesothelial cell cultures were used to stimulate fibroblast culture (phase 2). The fibroblast culture was assessed for collagen secretion. The whole experiment was repeated for each of the following sclerosants: talc (20 μg/mL), PVP-I (0.001%), doxycycline (2 μg/mL), and TGF-β (0.2 μg/mL). Created with BioRender.com.
Figure 1. A flow chart presenting the general study design, including phase 1 and 2. Initially, adenocarcinoma (ADC) cells were cultured and supernatant was collected. After that, mesothelial cell cultures where incubated with or without supernatant from ADC and treated or not treated with a sclerosant. After 24 h, the levels of cytokines in cell culture supernatants and mRNA in cell lysates were measured (end of phase 1). Then, supernatants from mesothelial cell cultures were used to stimulate fibroblast culture (phase 2). The fibroblast culture was assessed for collagen secretion. The whole experiment was repeated for each of the following sclerosants: talc (20 μg/mL), PVP-I (0.001%), doxycycline (2 μg/mL), and TGF-β (0.2 μg/mL). Created with BioRender.com. 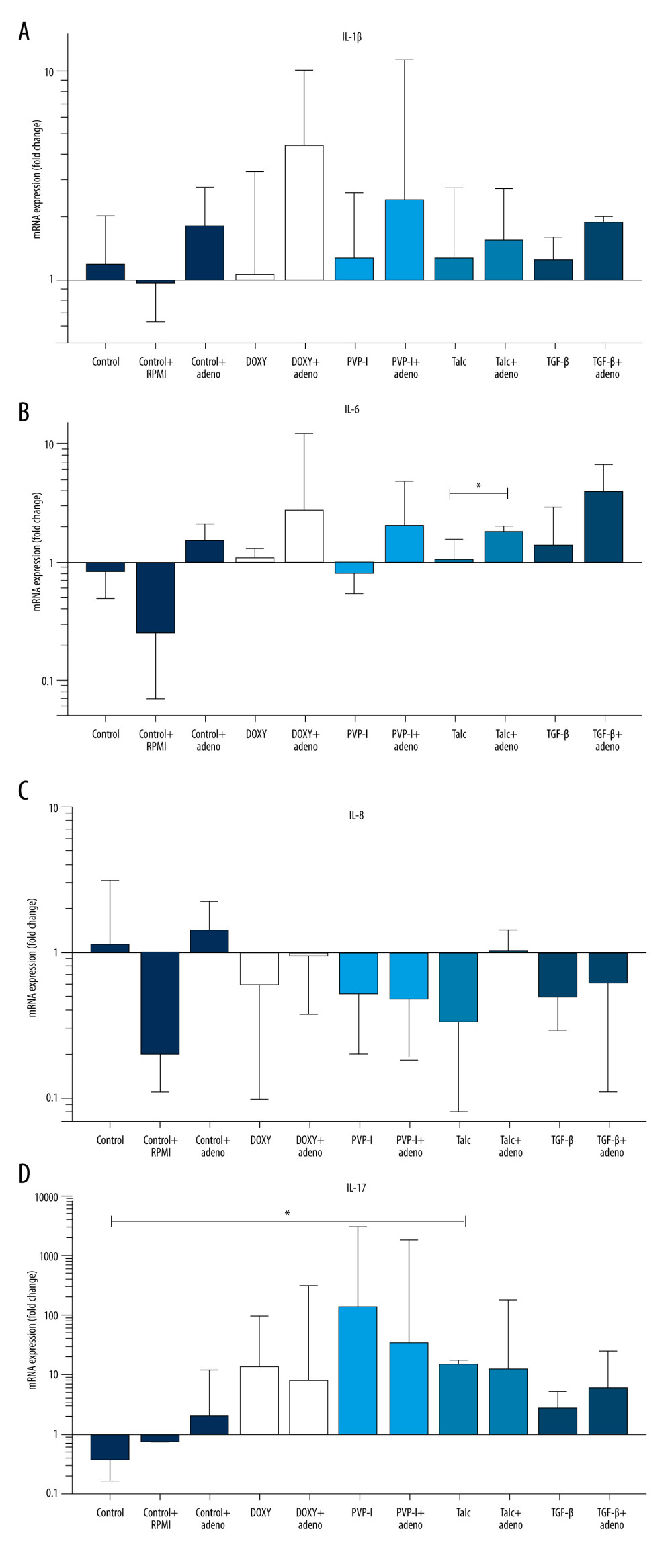 Figure 2. (A–D) IL-1β, IL-6, IL-8, IL-17A mRNA expression in mesothelial cells stimulated with sclerosants (doxycycline, PVP-I, talc and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline, PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA.
Figure 2. (A–D) IL-1β, IL-6, IL-8, IL-17A mRNA expression in mesothelial cells stimulated with sclerosants (doxycycline, PVP-I, talc and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline, PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA. 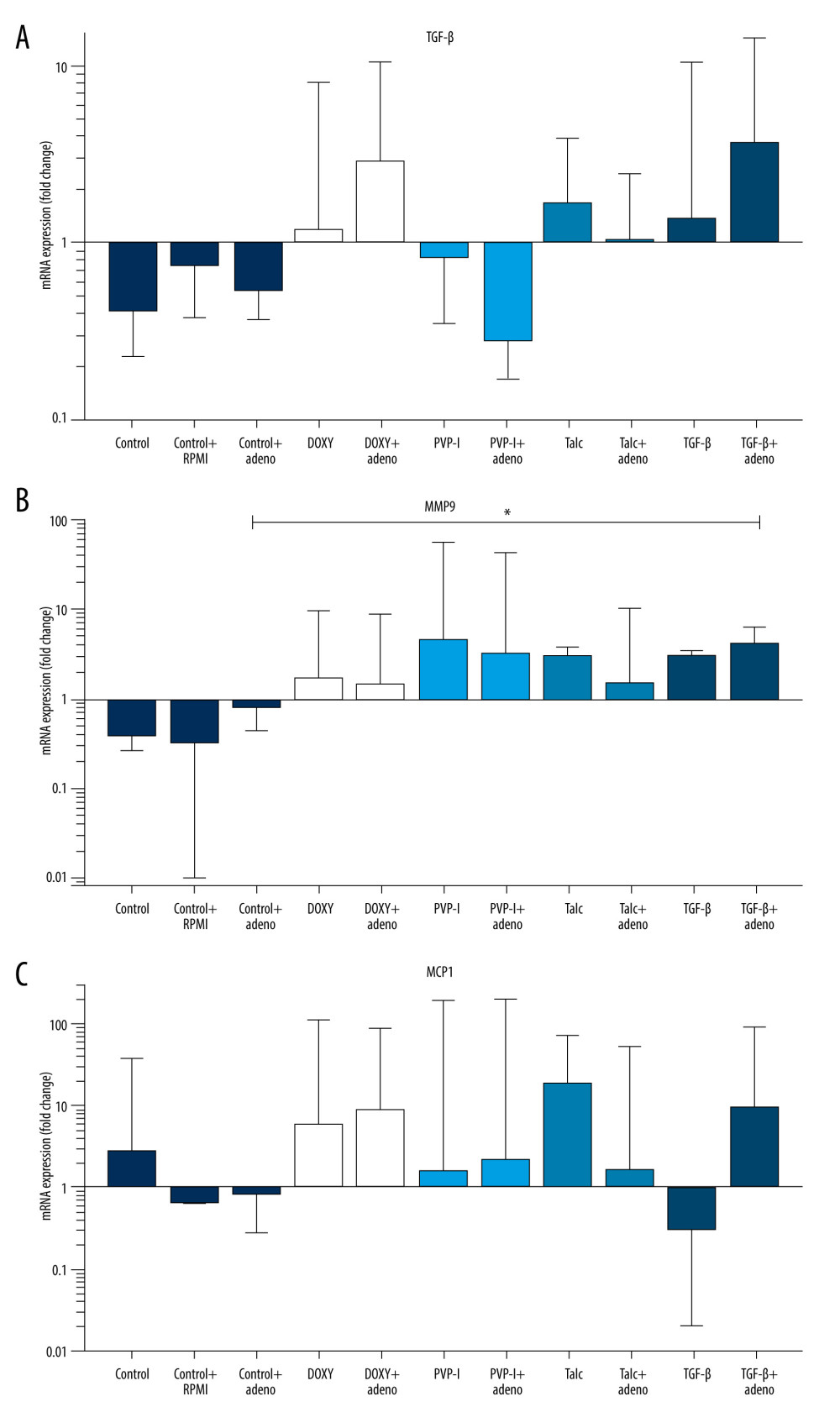 Figure 3. (A–C) TGF-β, MMP9, MCP-1 mRNA expression in mesothelial cells stimulated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA.
Figure 3. (A–C) TGF-β, MMP9, MCP-1 mRNA expression in mesothelial cells stimulated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA. 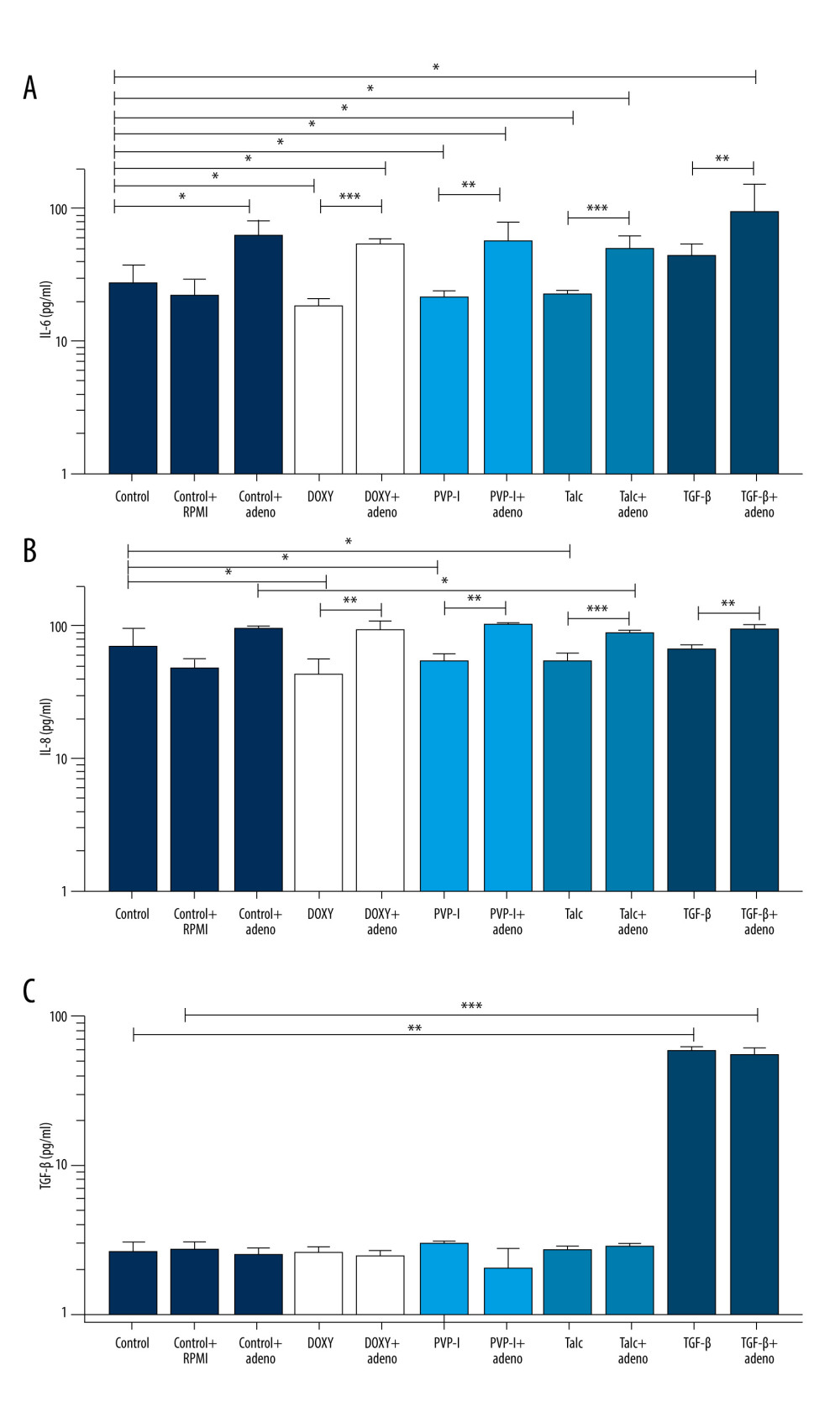 Figure 4. (A–C) IL-6, IL-8, and TGFβ protein levels in mesothelial cell culture supernatants stimulated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA.
Figure 4. (A–C) IL-6, IL-8, and TGFβ protein levels in mesothelial cell culture supernatants stimulated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA. 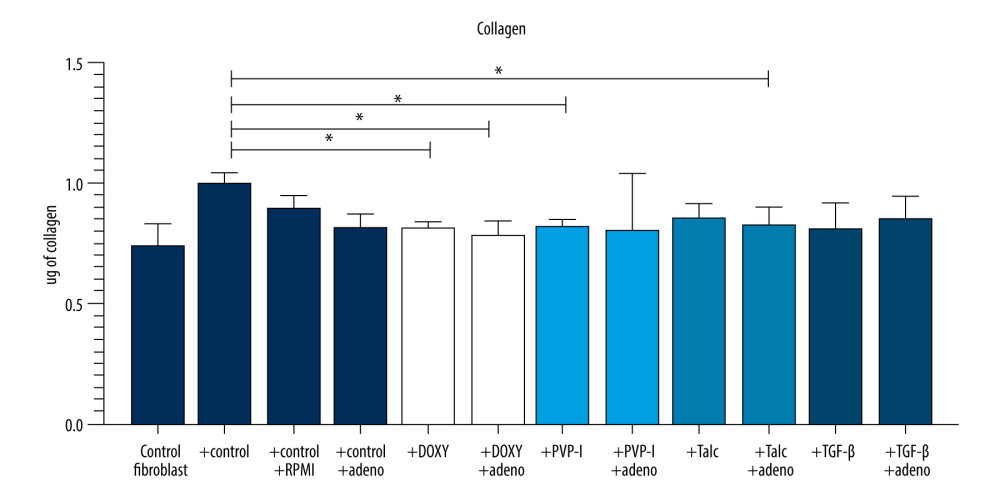 Figure 5. Collagen secretion by lung fibroblasts stimulated with supernatant from mesothelial cells treated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) with or without adenocarcinoma cell supernatant addition. Collagen concentrations measured in supernatants. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA.
Figure 5. Collagen secretion by lung fibroblasts stimulated with supernatant from mesothelial cells treated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) with or without adenocarcinoma cell supernatant addition. Collagen concentrations measured in supernatants. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA. References
1. Light RW, Pleural effusions: Med Clin North Am, 2011; 95; 1055-70
2. Rahman NM, Ali NJ, Brown G, Local anaesthetic thoracoscopy: British Thoracic Society Pleural Disease Guideline 2010: Thorax, 2010; 65(Suppl 2); ii54-60
3. Taghizadeh N, Fortin M, Tremblay A, US hospitalizations for malignant pleural effusions: Data from the 2012 National Inpatient Sample: Chest, 2017; 151; 845-54
4. Koegelenberg CFN, Bennji SM, Boer E, The current aetiology of malignant pleural effusion in the Western Cape Province, South Africa: South Afr Med J, 2018; 108; 275-77
5. Penz E, Watt KN, Hergott CA, Management of malignant pleural effusion: Challenges and solutions: Cancer Manag Res, 2017; 9; 229-41
6. Neragi-Miandoab S, Malignant pleural effusion, current and evolving approaches for its diagnosis and management: Lung Cancer Amst Neth, 2006; 54; 1-9
7. Zahid I, Routledge T, Billè A, Scarci M, What is the best treatment for malignant pleural effusions?: Interact Cardiovasc Thorac Surg, 2011; 12; 818-23
8. Kennedy L, Sahn SA, Talc pleurodesis for the treatment of pneumothorax and pleural effusion: Chest, 1994; 106; 1215-22
9. Huggins JT, Doelken P, Sahn SA, Intrapleural therapy: Respirol Carlton Vic, 2011; 16; 891-99
10. Mierzejewski M, Korczynski P, Krenke R, Janssen JP, Chemical pleurodesis – a review of mechanisms involved in pleural space obliteration: Respir Res, 2019; 20; 247
11. Schwarz Y, Star A, Role of talc modulation on cytokine activation in cancer patients undergoing pleurodesis: Pulm Med, 2012; 2012; 806183
12. Marchi E, Vargas FS, Teixeira LR, Intrapleural low-dose silver nitrate elicits more pleural inflammation and less systemic inflammation than low-dose talc: Chest, 2005; 128; 1798-804
13. Rodríguez-Panadero F, López Mejías J, Low glucose and pH levels in malignant pleural effusions. Diagnostic significance and prognostic value in respect to pleurodesis: Am Rev Respir Dis, 1989; 139; 663-67
14. Rodriguez-Panadero F, Montes-Worboys A, Mechanisms of pleurodesis: Respir Int Rev Thorac Dis, 2012; 83; 91-98
15. Aelony Y, Yao JF, King RR, Prognostic value of pleural fluid pH in malignant epithelial mesothelioma after talc poudrage: Respir Int Rev Thorac Dis, 2006; 73; 334-39
16. da Cunha BR, Domingos C, Stefanini ACB, Cellular interactions in the tumor microenvironment: The role of secretome: J Cancer, 2019; 10; 4574-87
17. Livak KJ, Schmittgen TD, Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method: Methods San Diego Calif, 2001; 25; 402-8
18. Light RW, Gary Lee Y: Textbook of pleural diseases [Internet], Routledge CRC Press [cited 2021 Feb 2]. Available from: https://www.routledge.com/Textbook-of-Pleural-Diseases/Light-Lee/p/book/9781482222500
19. Psathakis K, Calderón-Osuna E, Romero-Romero B, The neutrophilic and fibrinolytic response to talc can predict the outcome of pleurodesis: Eur Respir J, 2006; 27; 817-21
20. Acencio MMP, Silva BR, Teixeira LR, Evaluation of cellular alterations and inflammatory profile of mesothelial cells and/or neoplastic cells exposed to talc used for pleurodesis: Oncotarget, 2020; 11; 3730-36
21. Ghio AJ, Soukup JM, Dailey LA, Disruption of iron homeostasis in mesothelial cells after talc pleurodesis: Am J Respir Cell Mol Biol, 2012; 46; 80-86
22. Marchi E, Vargas FS, Acencio MM, Evidence that mesothelial cells regulate the acute inflammatory response in talc pleurodesis: Eur Respir J, 2006; 28; 929-32
23. Teixeira LR, Vargas FS, Puka J, Effectiveness and safety of iodopovidone in an experimental pleurodesis model: Clin Sao Paulo Braz, 2013; 68; 557-62
24. Lee YCG, Lane KB, Zoia O, Transforming growth factor-beta induces collagen synthesis without inducing IL-8 production in mesothelial cells: Eur Respir J, 2003; 22; 197-202
25. Acencio MMP, Vargas FS, Marchi E, Pleural mesothelial cells mediate inflammatory and profibrotic responses in talc-induced pleurodesis: Lung, 2007; 185; 343-48
26. Ors Kaya S, Bir F, Atalay H, Effect of diclofenac on experimental pleurodesis induced by tetracycline in rabbits: J Investig Med, 2005; 53; 267-70
27. Xie C, Teixeira LR, McGovern JP, Light RW, Systemic corticosteroids decrease the effectiveness of talc pleurodesis: Am J Respir Crit Care Med, 1998; 157; 1441-44
28. Teixeira LR, Wu W, Chang D-S, Light RW, The effect of corticosteroids on pleurodesis induced by doxycycline in rabbits: Chest, 2002; 121; 216-19
29. Guo Y, Tang K, Bilaceroglu S, Iodopovidone is as effective as doxycycline in producing pleurodesis in rabbits: Respirol Carlton Vic, 2010; 15; 119-25
30. Gary Lee YC, Melkerneker D, Thompson PJ, Transforming growth factor beta induces vascular endothelial growth factor elaboration from pleural mesothelial cells in vivo and in vitro: Am J Respir Crit Care Med, 2002; 165; 88-94
31. Antony VB, Nasreen N, Mohammed KA, Talc pleurodesis: Basic fibroblast growth factor mediates pleural fibrosis: Chest, 2004; 126; 1522-28
32. Mierzejewski M, Paplinska-Goryca M, Korczynski P, Krenke R, Primary human mesothelial cell culture in the evaluation of the inflammatory response to different sclerosing agents used for pleurodesis: Physiol Rep, 2021; 9; e14846
33. Fiorelli A, Pentimalli F, D’Urso V, Antineoplastic activity of povidone-iodine on different mesothelioma cell lines: Results of in vitro study: Eur J Cardiothorac Surg, 2014; 45; 993-1000
34. Light RW, Cheng DS, Lee YC, A single intrapleural injection of transforming growth factor-beta(2) produces an excellent pleurodesis in rabbits: Am J Respir Crit Care Med, 2000; 162; 98-104
35. Lee YC, Devin CJ, Teixeira LR, Transforming growth factor beta2 induced pleurodesis is not inhibited by corticosteroids: Thorax, 2001; 56; 643-48
36. Blache U, Horton ER, Xia T, Mesenchymal stromal cell activation by breast cancer secretomes in bioengineered 3D microenvironments: Life Sci Alliance, 2019; 2(3); e201900304
37. Roberts ME, Neville E, Berrisford RGBTS Pleural Disease Guideline Group, Management of a malignant pleural effusion: British Thoracic Society Pleural Disease Guideline 2010: Thorax, 2010; 65(Suppl 2); ii32-40
38. Tomashefski JF: Dail and Hammar’s Pulmonary Pathology-Volume I: Nonneoplastic Lung Disease [Internet] [cited 2021 Feb 2]. Available from: https://www.springer.com/gp/book/9780387983950
39. Bielsa S, Hernández P, Rodriguez-Panadero F, Tumor type influences the effectiveness of pleurodesis in malignant effusions: Lung, 2011; 189; 151-55
40. Martínez-Moragón E, Aparicio J, Sanchis J, Malignant pleural effusion: Prognostic factors for survival and response to chemical pleurodesis in a series of 120 cases: Respir Int Rev Thorac Dis, 1998; 65; 108-13
41. Sabbion RO, Terra RM, Teixeira LR, Influence of the progression of pleural neoplasia on the outcome of pleurodesis in mice: Oncotarget, 2020; 11; 2002-9
Figures
 Figure 1. A flow chart presenting the general study design, including phase 1 and 2. Initially, adenocarcinoma (ADC) cells were cultured and supernatant was collected. After that, mesothelial cell cultures where incubated with or without supernatant from ADC and treated or not treated with a sclerosant. After 24 h, the levels of cytokines in cell culture supernatants and mRNA in cell lysates were measured (end of phase 1). Then, supernatants from mesothelial cell cultures were used to stimulate fibroblast culture (phase 2). The fibroblast culture was assessed for collagen secretion. The whole experiment was repeated for each of the following sclerosants: talc (20 μg/mL), PVP-I (0.001%), doxycycline (2 μg/mL), and TGF-β (0.2 μg/mL). Created with BioRender.com.
Figure 1. A flow chart presenting the general study design, including phase 1 and 2. Initially, adenocarcinoma (ADC) cells were cultured and supernatant was collected. After that, mesothelial cell cultures where incubated with or without supernatant from ADC and treated or not treated with a sclerosant. After 24 h, the levels of cytokines in cell culture supernatants and mRNA in cell lysates were measured (end of phase 1). Then, supernatants from mesothelial cell cultures were used to stimulate fibroblast culture (phase 2). The fibroblast culture was assessed for collagen secretion. The whole experiment was repeated for each of the following sclerosants: talc (20 μg/mL), PVP-I (0.001%), doxycycline (2 μg/mL), and TGF-β (0.2 μg/mL). Created with BioRender.com. Figure 2. (A–D) IL-1β, IL-6, IL-8, IL-17A mRNA expression in mesothelial cells stimulated with sclerosants (doxycycline, PVP-I, talc and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline, PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA.
Figure 2. (A–D) IL-1β, IL-6, IL-8, IL-17A mRNA expression in mesothelial cells stimulated with sclerosants (doxycycline, PVP-I, talc and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline, PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA. Figure 3. (A–C) TGF-β, MMP9, MCP-1 mRNA expression in mesothelial cells stimulated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA.
Figure 3. (A–C) TGF-β, MMP9, MCP-1 mRNA expression in mesothelial cells stimulated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA. Figure 4. (A–C) IL-6, IL-8, and TGFβ protein levels in mesothelial cell culture supernatants stimulated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA.
Figure 4. (A–C) IL-6, IL-8, and TGFβ protein levels in mesothelial cell culture supernatants stimulated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) cultivated with or without adenocarcinoma cell supernatant addition. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; IL-6, IL-8 – interleukin 6 and 8; MMP9 – matrix metalloproteinase 9; MCP-1 – monocyte chemoattractant protein; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA. Figure 5. Collagen secretion by lung fibroblasts stimulated with supernatant from mesothelial cells treated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) with or without adenocarcinoma cell supernatant addition. Collagen concentrations measured in supernatants. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA.
Figure 5. Collagen secretion by lung fibroblasts stimulated with supernatant from mesothelial cells treated with sclerosants (doxycycline, PVP-I, talc, and TGF-β) with or without adenocarcinoma cell supernatant addition. Collagen concentrations measured in supernatants. Data are presented as median with interquartile range. DOXY – doxycycline; PVP-I – iodopovidone; TGF-β – transforming growth factor β; RPMI – Roswell Park Memorial Institute medium. The figure was created using GraphPad Prism software (version 9.3.1). GraphPad Software, Inc., San Diego, CA, USA. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387