08 June 2022: Clinical Research
Association Between Variants of the Mannose-Binding Lectin 2 Gene and Susceptibility to Sepsis in the Hainan Island
Shaowen ChengABCDEFG, Junyu ZhuABCDE, Xini LiuCDEF, Jian YangAEF, Wei ZhangBCD, Zhihua HuAEF, Jiemiao OuyangADF, Huaping LiangAEFGDOI: 10.12659/MSM.936134
Med Sci Monit 2022; 28:e936134
Abstract
BACKGROUND: Sepsis has emerged as a leading cause of death in the intensive care unit. A growing number of studies have shown that genetic variants, especially single nucleotide polymorphisms, are key determinants of inter-individual variation in sepsis response. Therefore, early prediction of the onset and progression of sepsis, along with early intervention in high-risk patients, should be performed to effectively reduce the morbidity and mortality of the disease.
MATERIAL AND METHODS: A total of 581 Chinese patients were enrolled in this study, including 271 patients with sepsis and 310 patients without. We measured gene polymorphisms of MBL2 and serum levels of MBL2, tumor necrosis factor (TNF-α), interleukin (IL)-6, IL-4, and IL-10 in all patients. The effects of site mutations on the binding of MBL2 to mannose-associated serine protease 1 (MASP1) and MASP2 were also analyzed.
RESULTS: Of 3 site mutations in the MBL2 gene (rs5030737, rs1800450, and rs1800451), only rs1800450 had a mutant (G/A) genotype. The frequency of the GA genotype and A allele in the sepsis group was higher than that in the non-sepsis group. Furthermore, rs1800450G/A was associated with decreased serum MBL2 and IL-10 levels and decreased MBL2-MASP1 and MBL2-MASP2 interactions. Bioinformatics analysis showed that rs1800450G/A reduced the structural stability of the MBL2 protein and affected its function.
CONCLUSIONS: MBL2 rs1800450G/A was associated with a higher risk of sepsis, which possibly involved a decreased level of serum MBL2 that broke the balance of inflammation and weakened the binding of MBL2 to MASP1 and MASP2.
Keywords: Disease Susceptibility, Genetic Predisposition to Disease, Genetics, Genetics, Medical, Genomic Structural Variation, Sepsis, China, Genotype, Humans, Interleukin-10, Mannose-Binding Lectin, Mannose-Binding Protein-Associated Serine Proteases, Polymorphism, Single Nucleotide
Background
Sepsis causes considerable morbidity and is the main cause of death in the intensive care unit (ICU) [1]. Sepsis can change rapidly, often resulting in a poor prognosis due to the lack of a timely diagnosis and treatment. Sepsis is also complicated and progresses rapidly. Without a timely diagnosis and treatment, it is likely to develop as severe sepsis or even septic shock. When more than 3 organs or systems are involved, the case fatality rate is extremely high [2]. Therefore, the occurrence and development of sepsis should be predicted early, while early intervention for high-risk patients can effectively reduce the incidence and mortality of this disease. However, despite advances in diagnosis and treatment, the morbidity and mortality of sepsis remain high, and its mechanism is still unclear [3].
An increasing number of studies have shown that genetic variants, particularly single nucleotide polymorphisms (SNPs), are critical determinants of interindividual differences in responses to sepsis [4,5]. Many scholars have conducted genetic correlation studies on the susceptibility and genetic polymorphisms of sepsis, and a series of susceptibility genotypes of sepsis have been explored [6–9].
Many associated studies have investigated susceptibility to, or mortality from, sepsis for variants of the functionally important immune-related gene, mannose-binding lectin 2 (
To date, little is known about the association of
Material and Methods
STUDY POPULATION AND CLINICAL EVALUATION:
A total of 581 unrelated patients recruited in this study were Han Chinese from the island of Hainan in southern China. The inclusion criteria included being between 14 and 85 years of age. The patients enrolled in the sepsis group (n=271, 177 men, 94 women; mean age: 59.2±13.6 years) and the non-sepsis group (n=310, 191 men, 119 women; mean age: 64.3±12.8 years) were in the ICU between June 2017 and December 2018. This study was approved by the Medical Ethics Committee of Hainan Medical University. Approval of the local ethical committee was obtained, as was the written informed consent of patients. A diagnosis of sepsis was based on the latest diagnostic criteria, the Sequential Organ Failure Assessment (SOFA) score (>2) caused by infection [15,16]. Patients were not eligible if they had the following exclusion criteria: (1) age less than 14 years or more than 85 years; (2) were in special groups such as pregnant women and lactating women; or (3) patients who were immunosuppressed before developing sepsis were excluded, including those with known HIV infection, hematological malignancy, neutropenia (white cell count <1×109/L) or chronic liver failure, according to the APACHE II definition, and patients who had received chemotherapy, immunosuppressants, or systemic steroids in the previous 6 months.
BLOOD SAMPLE COLLECTION AND DNA EXTRACTION:
Five milliliters of peripheral venous blood was collected from each patient using ethylenediamine tetra-acetic acid anticoagulant tubes. Blood samples were collected from all patients on the first day of ICU admission. Genomic DNA was extracted from whole blood using a TIANamp Blood DNA Kit (Tiangen Biotech, Beijing, China) and stored in a freezer at −80°C for long-term storage.
GENOTYPING:
The upstream primer was 5/-CAGGCAGTTTCCTCTGGAAG-3, and the downstream primer was 5/-AGTCACGCAGTGTCACAAGG-3. The single base extension (SBE) reaction was performed in a reaction mixture with a final volume of 5 μL that contained 3 μL of purified multiplex PCR product, 1 μL of pooled extension primers, 0.5 μL of SNaPshot Mix, and 0.5 μL of dH2O. SNaPshot reaction conditions were as follows: 40 SBE cycles of initial denaturation at 95°C for 10 sec, primer annealing at 52°C for 5 sec, and primer extension at 60°C for 30 sec. Shrimp alkaline phosphatase was used for the post-SBE purification of SNaPshot reaction products. For electrophoresis, purified multiplex SBE reaction products were mixed with 0.9 μL of water, 0.5 μL of shrimp alkaline phosphatase, and 0.6 μL of 10× shrimp alkaline phosphatase buffer. The digestion reaction was incubated at 37°C for 40 min and 75°C for 15 min. A total of 2 μL of product from the minisequencing reaction was mixed with 8 μL formamide containing 0.4% 120 LIZ dye size standard (ThermoFisher Scientific, Waltham, MA, USA). The samples were denatured by incubating them at 95°C for 5 min. After cooling down to −20°C, the fluorescently labeled fragments were resolved by capillary electrophoresis on an ABI 3730XL Genetic Analyzer for allele discrimination. The resulting data were analyzed with GeneMapper v4.0 software.
SERUM MBL2, TNF-α, IL-6, IL-4 AND IL-10 MEASUREMENTS:
Aliquots of whole blood samples were collected from study participants. Blood samples were collected from sepsis patients within 24 h sepsis diagnosis. After centrifugation, the plasma was aspirated and aliquoted for storage at −80°C. The concentrations of MBL2, TNF-α, IL-6, IL-4, and IL-10 in serum were tested by enzyme-linked immunosorbent assay (ELISA) with an ELISA kit (American R&D Company) according to the manufacturer’s instructions.
MBL2-MASP1 AND MBL2-MASP2 BINDING ASSAY:
Recombinant human wild-type MBL2 protein and mutant MBL2 protein were expressed in Chinese hamster ovary cells [13,18]. Then, 10 mg/L of purified recombinant human MASP1-N and MASP2-N terminal proteins were added to an ELISA plate and coated overnight at 4°C. Different concentrations of MBL2 protein were added and reacted at 37°C for 1 h. After washing, 1: 1000 mouse anti-human MBL-CRD monoclonal antibody and 1: 5000 horse radish peroxidase (HRP)-labeled sheep anti-rat IgG were added and reacted at 37°C for 30 min. The substrate solution for HRP was tetramethylbenzidine, and the reaction was terminated by the addition of 2 M H2SO4. The absorbance at 450 nm (A450) was measured in a microplate reader.
EFFECTS OF SITE MUTATIONS ON THE STRUCTURE AND FUNCTION OF THE MBL2 PROTEIN:
The MBL2 proteins of 8 species were aligned using MAFFT v7.38, and the evolutionary tree was calculated using MEGA. Structural domains were analyzed using SMART. iStable software predicted the effect of G54D on protein structure, and Polyphen-2 and SIFT software were used to predict the effect of conservative mutations on protein function.
STATISTICAL ANALYSIS:
Allele and genotype frequencies of variants were determined by gene counting. The genotype distribution of each SNP was tested for departure from the Hardy-Weinberg equilibrium using χ2 analysis. Additionally, χ2 analysis was used to analyze the associations of
Results
OVERALL CLINICAL CHARACTERISTICS OF PATIENTS:
Two independent groups existed, sepsis and non-sepsis, which consisted of 271 and 310 individuals, respectively. The main clinical characteristics of patients are presented in Table 1. The sex and mean age of patients were not significantly different between the 2 groups. The source of infection in patients with sepsis was mainly a respiratory tract infection (31.4%, n=85). Gram-positive and gram-negative infections, fungal infections, and mixed infections accounted for approximately 20.0%, 15.5%, 2.2%, and 7.0% of cases, respectively, in the sepsis group with positive blood cultures. Negative blood cultures were identified in 55.3% of patients in the sepsis group. The common pathogens identified in this study were Staphylococcus aureus, Escherichia coli, Acinetobacter baumannii, Enterobacter cloacae, and Pseudomonas aeruginosa.
ASSOCIATION OF MBL2 GENE POLYMORPHISMS WITH SEPSIS:
Only the mutation site of rs1800450 G/A was found in the 3 site mutations of MBL2 gene polymorphisms (rs1800450, rs180045, and rs50307371). The rs1800450 genotype distributions were consistent with the Hardy-Weinberg equilibrium (χ2=1.128, P=0.288; Table 2).
The frequency of a GA genotype was significantly higher in the sepsis group than in the non-sepsis group (P=0.010). Allele frequency analysis also showed that the frequency of the A allele in the sepsis group was much higher than that in the non-sepsis group (P=0.029; Table 3). Further, χ2 analysis showed that GA genotype patients were 1.682 times more likely to develop sepsis than GG genotype patients (OR=1.682, 95% CI=1.340–2.505). Patients who carried the A allele were more likely to acquire sepsis than G allele carriers (OR=1.465, 95% CI=1.041–2.061), which means that the variation of rs1800450, G→A increased the incidence of sepsis (Table 3).
MBL2, TNF-α, IL-6, IL-4, AND IL-10 LEVELS IN SERUM:
We found the rs1800450 variant had a certain effect on the MBL2 serum levels in the sepsis and non-sepsis groups, and was more obvious in sepsis group. But there was no statistical difference among patients with GG, GA, and AA genotypes in the non-sepsis group. In the sepsis group, the MBL2 serum levels of patients with GA and AA genotypes were markedly lower than those in patients with the GG genotype (P≤0.001 and P=0.001, respectively; Figure 1).
Serum TNF-α, IL-6, IL-4, and IL-10 levels in patients with GG, GA, and AA genotypes in the non-sepsis group had no significant differences. We found that serum TNF-α levels in septic patients with GA and AA genotypes were markedly higher than those in septic patients with the GG genotype (P=0.008 and P=0.005, respectively). IL-10 levels in septic patients with GA and AA genotypes were lower than those in patients with the GG genotype. However, a significant difference in IL-6 and IL-4 levels between groups was not noted (Figure 2A–2D).
EFFECT OF SITE MUTATIONS ON THE BINDING OF MBL2 TO MBL-ASSOCIATED SERINE PROTEASES 1 AND 2:
To further examine the effect of the amino acid substitution G54D on MBL2-MASP1 and MBL2-MASP2 interactions, we also investigated the binding of wild-type and mutated MBL2 proteins to MASP1 and MASP2. As expected, the binding of mutated MBL2 protein to MASP1 and MASP2 was weaker than that of the wild-type MBL2 protein (Figure 3A, 3B).
EFFECTS OF SITE MUTATIONS ON MBL2 PROTEIN STRUCTURE AND FUNCTION:
Sequence analysis of the human MBL2 protein showed that the corresponding mutation of rs1800450G/A was G54D. Multisequence alignment of MBL2 proteins from 8 species indicated that the 54th amino acid G of the human MBL2 protein was a highly conserved site in species that was located in the middle of a highly conserved motif and might have important functions (Figures 4, 5). From domain analysis, MBL2 has a known conserved domain (127–245), a low complexity domain (61–107), and 2 unknown conserved domains (14–61 and 108–126). The G54D mutation is located in the unknown conserved region of the N-terminal segment (Figure 6).
Analysis with iStable predicted that the G54D mutation in the MBL2 protein would change the stability of the protein: The result was “decrease”, which means that the mutation was expected to reduce the structural stability of the protein and might affect the structure of conserved N-terminal regions.
Analysis with Polyphen-2 predicted that this mutation would be “probably damaging” with a score of 1.000. The predicted SIFT score was 0.02, and the result was “affect protein function”, suggesting that the mutation might have an impact on protein function
Discussion
As an important ingredient within the complement system,
We found that only mutations of codon 54 (rs1800450G/A, termed A/B) of exon 1 were found in the Chinese patients. Allele frequency analysis showed that the frequency of the A allele in the sepsis group was significantly higher than that in the non-sepsis group, and that patients who carried the A allele were more likely to acquire sepsis than the G allele carriers. Therefore, rs1800450G/A, among the
The maintenance of a protective function of MBL2 in the body is closely related to the level of MBL2 in plasma: The lower the MBL2 level is, the more susceptible patients are to infectious diseases. Serum levels of MBL2 were mainly determined by polymorphisms of the promoter and exon 1; a study found that a single nucleotide mutation of the first exon codon 54 (GGC-GAC) of exon 1 decreased MBL2 serum levels and affected the complement activation of our immune system, resulting in infectious and immune-diversified diseases [16]. In the present study, the rs1800450 variant had a certain effect on the MBL2 serum levels in the sepsis and non-sepsis groups. However, there were no statistical differences among patients with GG, GA, and AA genotypes in the non-sepsis group. MBL2 serum levels in septic patients with the GA and AA genotype were markedly lower those in patients with the GG genotype. It was suggested that the single nucleotide mutations reduced the MBL2 polymer and weakened ligand-binding capacity. In addition, it might make the MBL2 polymer more easily degraded by metalloprotease to regulate the concentration of MBL2 in plasma [16].
The level of MBL2 is closely related to the inflammatory state of the body and the level of inflammatory secretions. MBL2 can reduce the expression of the pro-inflammatory factors IL-1 alpha and IL-1 beta and increase the release of IL-10 and IL-6 in monocytes after stimulation with lipopolysaccharide [16]. Elevating the level of MBL2 could promote the release of TNF and IL-6 in monocytes, thus controlling the further aggravation of infection [17]. Zhang et al found that the miR-608 rs4919510 polymorphism was significantly associated with a higher production of proinflammatory cytokines (TNF-α, IL-6, and IL-1β) and lower production of anti-inflammatory cytokines (IL-10, IL-4) in patients with sepsis [18]. The results further indicated that the G→C variant of the rs4919510G/C polymorphism might enhance the production of proinflammatory cytokines and weaken the production of anti-inflammatory cytokines. Therefore, a single variant may impact the release of these inflammatory markers and increase the risk of sepsis. To verify that the
Binding of the MBL2 protein to serine protease (MASP-1 and MASP-2) is required to activate the lectin pathway, trigger the complement system, and resist infection [19,20]. Based on the significant clinical relevance of the rs1800450 polymorphism, we further hypothesized that amino acid substitutions might affect MBL2-MASP1 and MBL2-MASP2 interactions. As expected, the variant MBL2 protein also shows a weaker binding interaction with MASP1 and MASP2. Therefore, one can deduce that the rs1800450 polymorphism is not only a useful clinical biomarker for the prognostic assessment of sepsis but also a functional SNP, which affects MBL2 protein activities and binding interactions with MASP1 and MASP2.
Through bioinformatics analysis, we also found that the 54th amino acid G of the human MBL2 protein was a highly conserved site in species that was located in the middle of the highly conserved motif, and which may have important functions. However, site mutations can reduce the stability of the MBL2 protein structure and affect protein function, thus affecting the efficiency of MBL2 in vivo, making the body susceptible to infection.
Several potential limitations should be taken into consideration in this study. First, the sample size of patients we recruited was small, and a control group with healthy individuals was lacking. Furthermore, we predicted that the rs1800450G/A polymorphism possibly reduced the stability of the MBL2 protein structure and affected protein function according to bioinformatics analysis; more relevant experiments are needed for further verification. Finally, the exact mechanisms by which the rs1800450G/A polymorphism is involved in the pathogenesis of sepsis are unknown, and further mechanistic research must be carried out.
Conclusions
This study demonstrated that
Figures
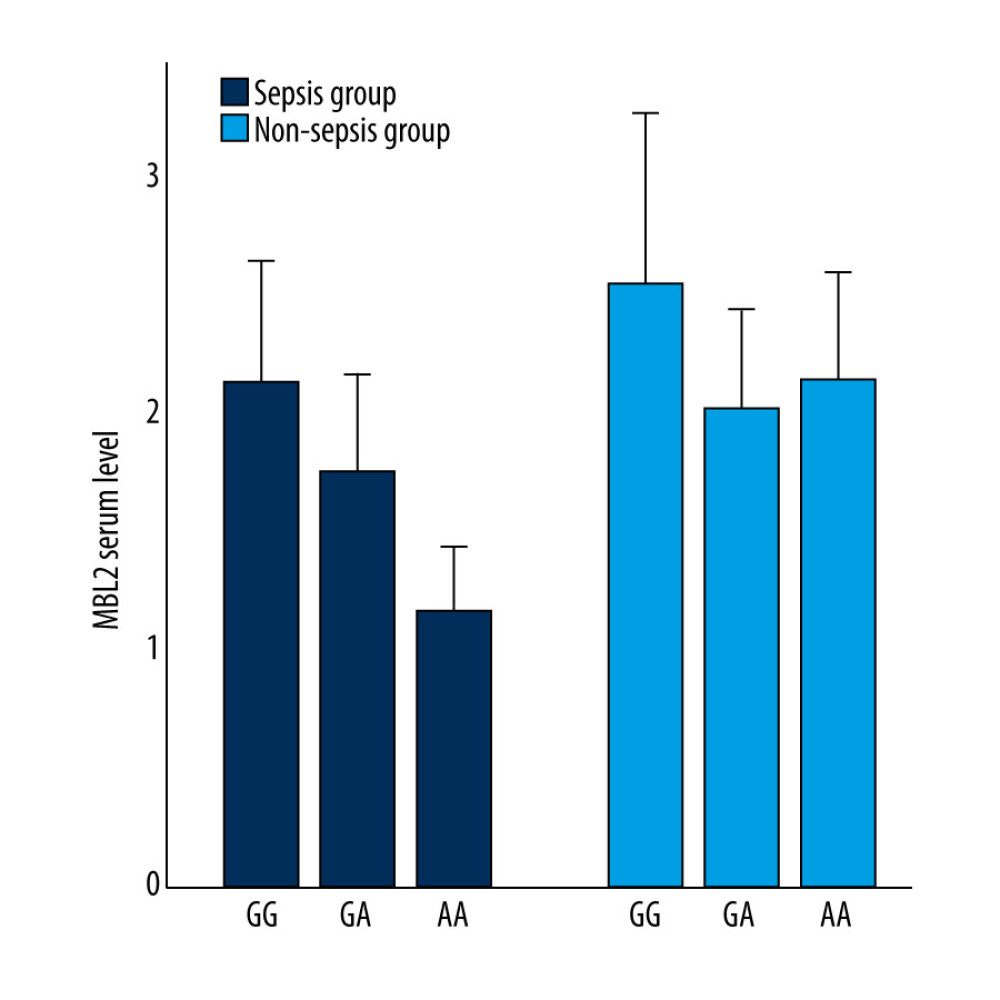 Figure 1. MBL2 serum level in different groups (n=63 for the GG genotype, n=34 for GA genotype, and n=6 for the AA genotype in the sepsis group; n=63 for the GG genotype, n=34 for GA genotype, and n=7 for the AA genotype in the non-sepsis group). In the sepsis group, GG VS GA (P=0.000), GG VS AA (P=0.001). Origin v8.0, United States OriginLab Corporation.
Figure 1. MBL2 serum level in different groups (n=63 for the GG genotype, n=34 for GA genotype, and n=6 for the AA genotype in the sepsis group; n=63 for the GG genotype, n=34 for GA genotype, and n=7 for the AA genotype in the non-sepsis group). In the sepsis group, GG VS GA (P=0.000), GG VS AA (P=0.001). Origin v8.0, United States OriginLab Corporation. 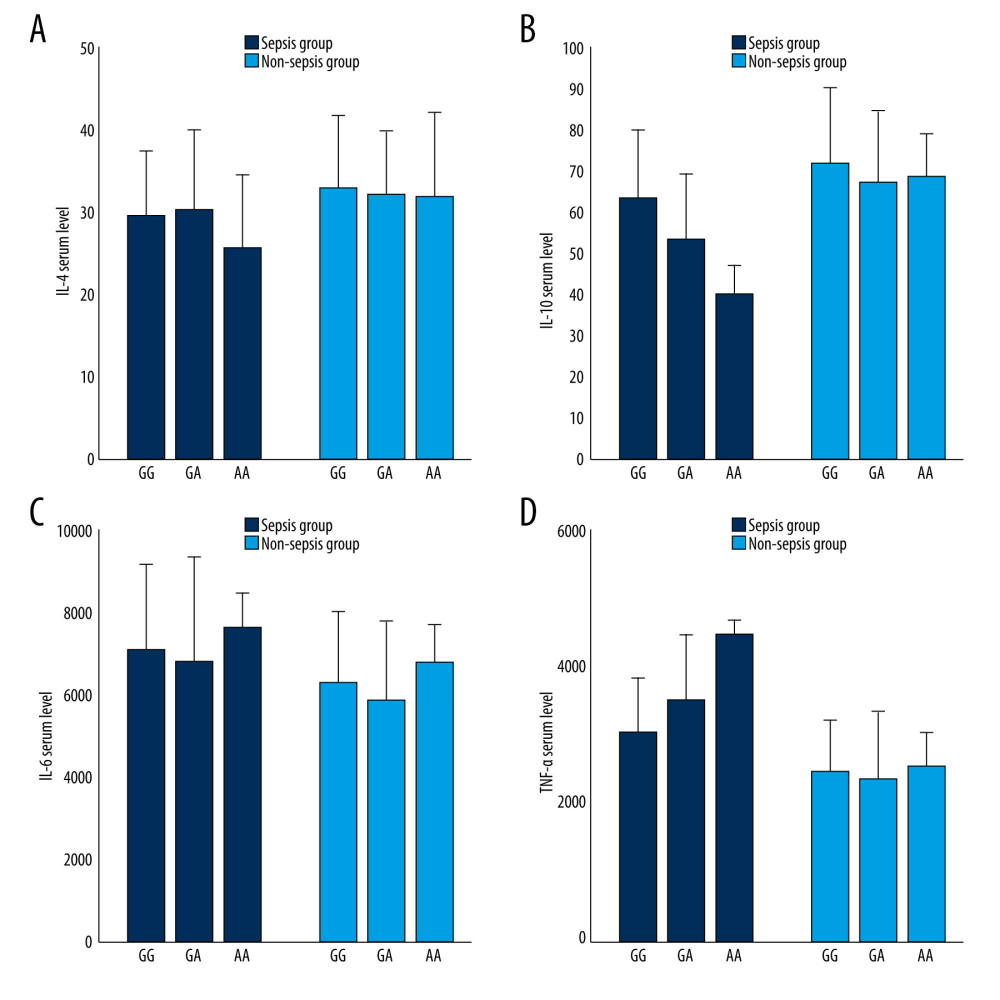 Figure 2. (A) IL-4, TNF-α, IL-6, and IL-10 serum level in different groups (n=63 for the GG genotype, n=34 for GA genotype and n=6 for the AA genotype in sepsis group; (B) n=63 for the GG genotype, n=34 for GA genotype and n=7 for the AA genotype in non-sepsis group). (C) In sepsis group, TNF-α: GG VS GA (P=0.008), GG VS AA (P=0.005); (D) IL-10: GG VS GA (P=0.005), GG VS AA (P=0.017). Origin v8.0, United States OriginLab Corporation.
Figure 2. (A) IL-4, TNF-α, IL-6, and IL-10 serum level in different groups (n=63 for the GG genotype, n=34 for GA genotype and n=6 for the AA genotype in sepsis group; (B) n=63 for the GG genotype, n=34 for GA genotype and n=7 for the AA genotype in non-sepsis group). (C) In sepsis group, TNF-α: GG VS GA (P=0.008), GG VS AA (P=0.005); (D) IL-10: GG VS GA (P=0.005), GG VS AA (P=0.017). Origin v8.0, United States OriginLab Corporation. 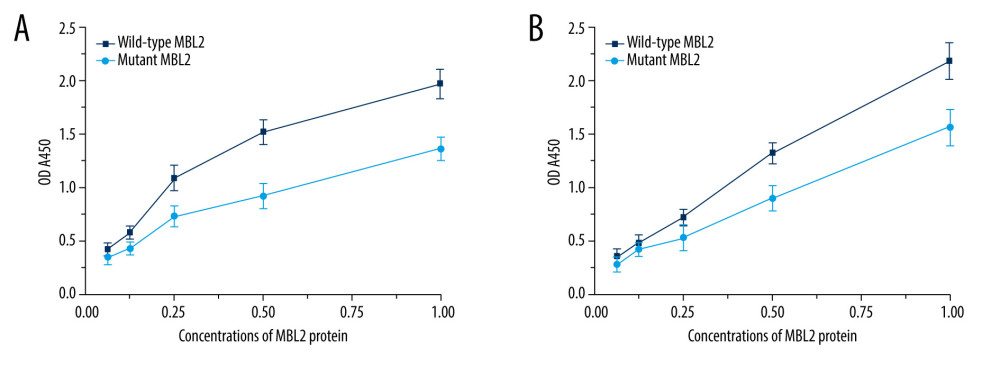 Figure 3. (A) The binding between the wild-type and the mutated MBL2 proteins to MASP1. (B) The binding between the wild-type and the mutated MBL2 proteins to MASP2. Origin v8.0, United States OriginLab Corporation.
Figure 3. (A) The binding between the wild-type and the mutated MBL2 proteins to MASP1. (B) The binding between the wild-type and the mutated MBL2 proteins to MASP2. Origin v8.0, United States OriginLab Corporation. 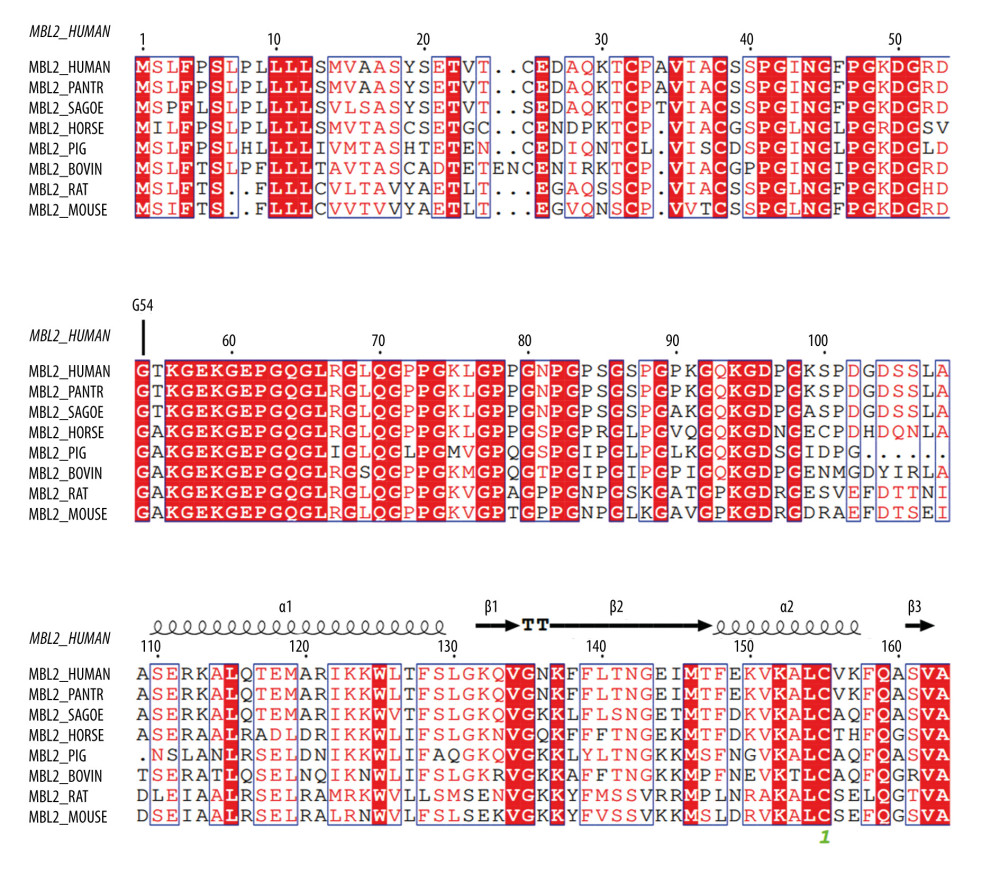 Figure 4. Multi-sequence alignment of MBL2 proteins from eight species. MAFFT v7.0 (http://mafft.cbrc.jp/alignment/server/).
Figure 4. Multi-sequence alignment of MBL2 proteins from eight species. MAFFT v7.0 (http://mafft.cbrc.jp/alignment/server/). 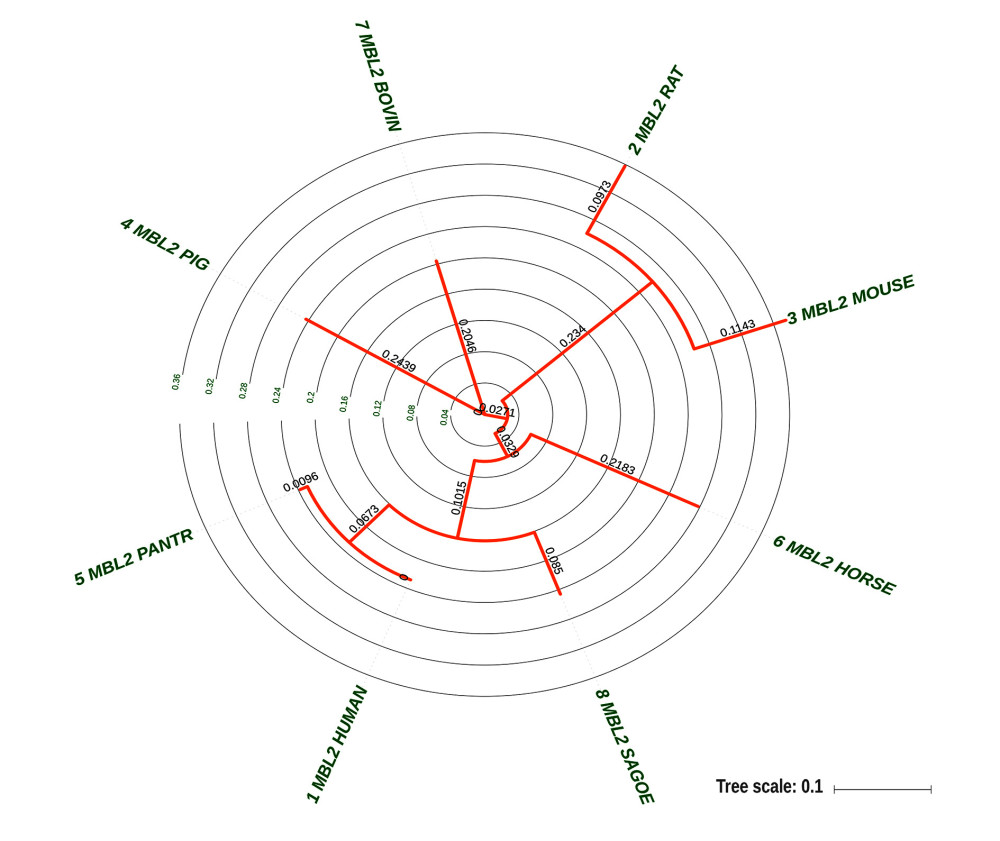 Figure 5. Evolutionary tree of 8 species used for conservative analysis. Molecular Evolutionary Genetics Analysis7.0, Mega Limited, Auckland, New Zealand.
Figure 5. Evolutionary tree of 8 species used for conservative analysis. Molecular Evolutionary Genetics Analysis7.0, Mega Limited, Auckland, New Zealand. 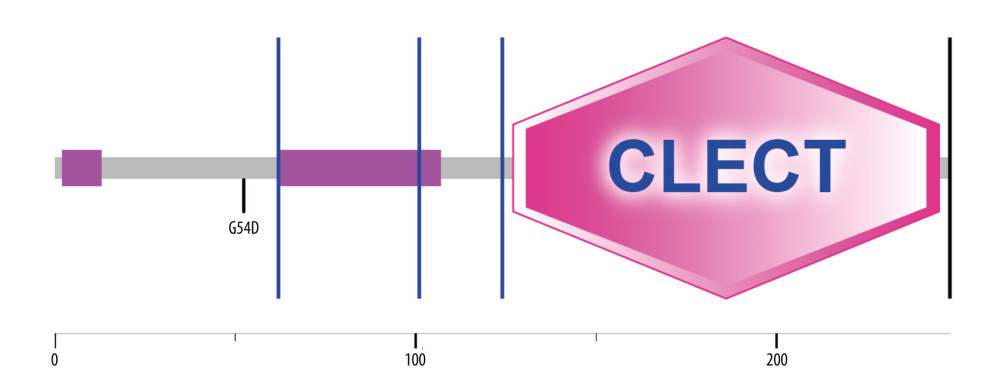 Figure 6. The relationship between G54D mutation and the known domain of MBL2 protein. SMART(http://smart.embl-heidelberg.de/).
Figure 6. The relationship between G54D mutation and the known domain of MBL2 protein. SMART(http://smart.embl-heidelberg.de/). Tables
Table 1. Overall clinical characteristics of patients in the sepsis group and non-sepsis group.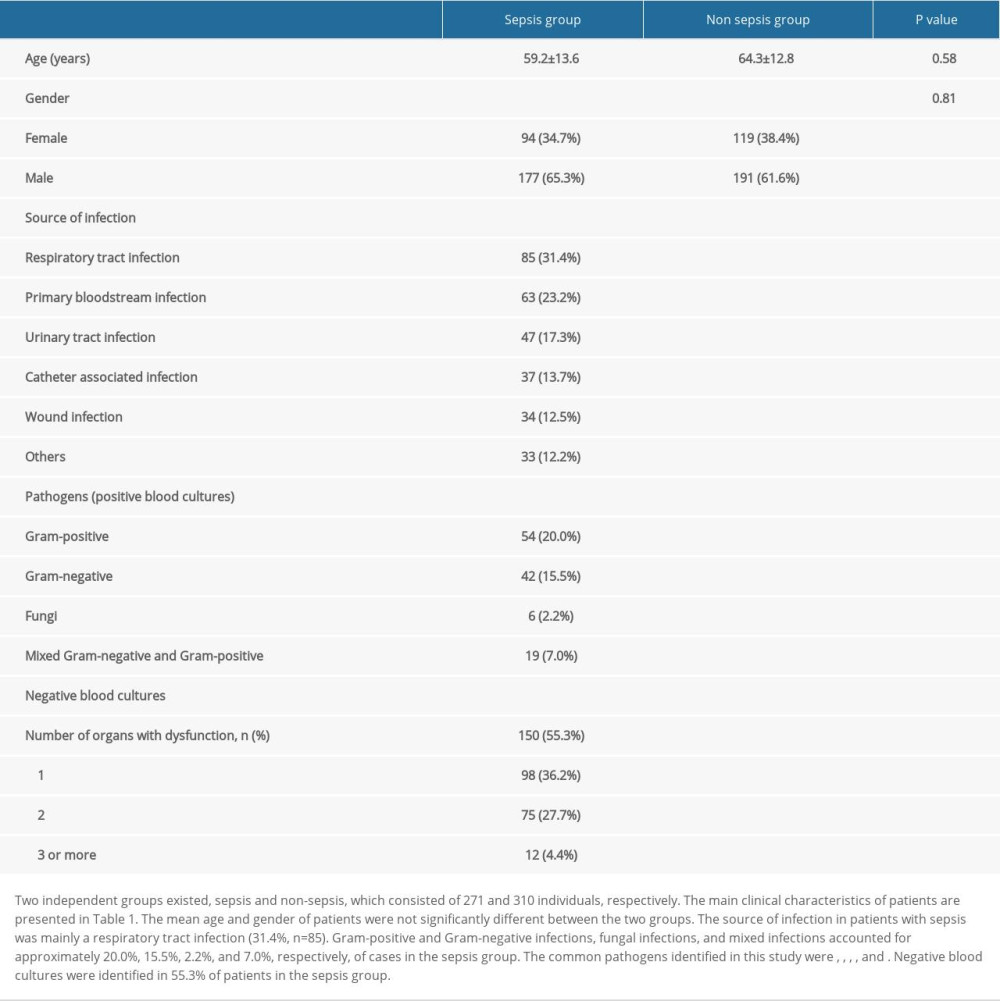 Table 2. The rs1800450 genotype distributions of Hardy-Weinberg equilibrium in total (n/%).
Table 2. The rs1800450 genotype distributions of Hardy-Weinberg equilibrium in total (n/%). Table 3. Genotypic and allelic frequency distribution of rs1800450 in the sepsis group and non-sepsis group. P=0.014 for dominant association (GA+AA vs GG). P=0.971 for recessive association (AA vs GA+GG).
Table 3. Genotypic and allelic frequency distribution of rs1800450 in the sepsis group and non-sepsis group. P=0.014 for dominant association (GA+AA vs GG). P=0.971 for recessive association (AA vs GA+GG).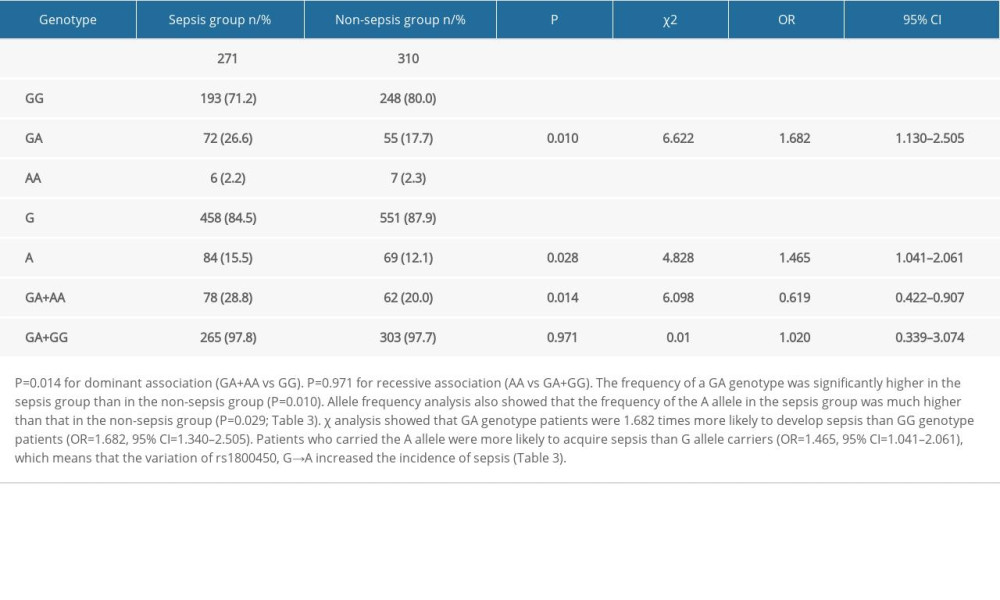
References
1. Rochwerg B, Alhazzani W, Sindi A, Fluid resuscitation in sepsis: A systematic review and network meta-analysis: Ann Intern Med, 2014; 161(5); 347-55
2. Deitch EA, Dayal SD, Intensive care unit management of the trauma patient: Crit Care Med, 2006; 34(9); 2294-301
3. Varon J, Baron RM, A current appraisal of evidence for the approach to sepsis and septic shock, 2019 2049936119856517
4. Arcaroli J, Fessler MB, Abraham E, Genetic polymorphisms and sepsis: Shock, 2005; 24(4); 300-12
5. Lin MT, Albertson TE, Genomic polymorphisms in sepsis: Crit Care Med, 2004; 32(2); 569-79
6. Zhang H, Lu Y, Sun G, The common promoter polymorphism rs11666254 downregulates FPR2/ALX expression and increases risk of sepsis in patients with severe trauma: Crit Care, 2017; 21(1); 171
7. Wang X, Li ZY, Zeng L, Neutrophil CD64 expression as a diagnostic marker for sepsis in adult patients: A meta-analysis: Crit Care, 2015; 19(1); 245
8. El-Behedy EM, Akeel N, El-Maghraby HM, Shawky A, Serum level and genetic polymorphism of mannose-binding lectin in infants with neonatal sepsis at Zagazig University Hospitals: Egypt J Immunol, 2019; 26(1); 91-99
9. Gao JW, Zeng L, Zhang AQ, Identification of haplotype tag single-nucleotide polymorphisms within the PPAR family genes and their clinical relevance in patients with major trauma: Int J Environ Res Public Health, 2016; 13(4); 374
10. Özkan H, Köksal N, Çetinkaya M, Serum mannose-binding lectin (MBL) gene polymorphism and low MBL levels are associated with neonatal sepsis and pneumonia: J Perinatol, 2012; 32(3); 210-17
11. Liu L, Ning B, The role of MBL2 gene polymorphism in sepsis incidence: Int J Clin Exp Pathol, 2015; 8(11); 15123-27
12. Hartz A, Pagel J, Humberg A, The association of mannose-binding lectin 2 polymorphisms with outcome in very low birth weight infants: PLoS One, 2017; 12(5); e0178032
13. Mandal RK, Khan MA, Hussain A, Association of MBL2 gene polymorphisms with pulmonary tuberculosis susceptibility: Trial sequence meta-analysis as evidence: Infect Drug Resist, 2019; 12; 185-210
14. Mills TC, Chapman S, Hutton P, Variants in the mannose-binding lectin gene MBL2 do not associate with sepsis susceptibility or survival in a Large European Cohort: Clin Infect Dis, 2015; 61(5); 695-703
15. Singer M, Deutschman CS, Seymour CW, The third international consensus definitions for sepsis and septic shock (Sepsis-3): JAMA, 2016; 315(8); 801-10
16. Seymour CW, Liu VX, Iwashyna TJ, Assessment of clinical criteria for sepsis: for the third international consensus definitions for sepsis and septic shock (Sepsis-3): JAMA, 2016; 315(8); 762-74
17. Zuo D, Cai X, Zhao N, Location of MBL-associated serine proteases binding motifs on human mannan-binding lectin (MBL): Protein Pept Lett, 2010; 17(1); 131-36
18. Zhang AQ, Gu W, Zeng L, Genetic variants of microRNA sequences and susceptibility to sepsis in patients with major blunt trauma: Ann Surg, 2015; 261(1); 189-96
19. Keizer MP, Kamp A, van Mierlo G, Substitution of mannan-binding lectin (MBL)-deficient serum with recombinant MBL results in the formation of new MBL/MBL-associated serine protease complexes: Front Immunol, 2018; 9; 1406
20. Swierzko AS, Michalski M, Sokolowska A, The role of complement activating collectins and associated serine proteases in patients with hematological malignancies, receiving high-dose chemotherapy, and autologous hematopoietic stem cell transplantations (auto-HSCT): Front Immunol, 2018; 9; 2153
Figures
 Figure 1. MBL2 serum level in different groups (n=63 for the GG genotype, n=34 for GA genotype, and n=6 for the AA genotype in the sepsis group; n=63 for the GG genotype, n=34 for GA genotype, and n=7 for the AA genotype in the non-sepsis group). In the sepsis group, GG VS GA (P=0.000), GG VS AA (P=0.001). Origin v8.0, United States OriginLab Corporation.
Figure 1. MBL2 serum level in different groups (n=63 for the GG genotype, n=34 for GA genotype, and n=6 for the AA genotype in the sepsis group; n=63 for the GG genotype, n=34 for GA genotype, and n=7 for the AA genotype in the non-sepsis group). In the sepsis group, GG VS GA (P=0.000), GG VS AA (P=0.001). Origin v8.0, United States OriginLab Corporation. Figure 2. (A) IL-4, TNF-α, IL-6, and IL-10 serum level in different groups (n=63 for the GG genotype, n=34 for GA genotype and n=6 for the AA genotype in sepsis group; (B) n=63 for the GG genotype, n=34 for GA genotype and n=7 for the AA genotype in non-sepsis group). (C) In sepsis group, TNF-α: GG VS GA (P=0.008), GG VS AA (P=0.005); (D) IL-10: GG VS GA (P=0.005), GG VS AA (P=0.017). Origin v8.0, United States OriginLab Corporation.
Figure 2. (A) IL-4, TNF-α, IL-6, and IL-10 serum level in different groups (n=63 for the GG genotype, n=34 for GA genotype and n=6 for the AA genotype in sepsis group; (B) n=63 for the GG genotype, n=34 for GA genotype and n=7 for the AA genotype in non-sepsis group). (C) In sepsis group, TNF-α: GG VS GA (P=0.008), GG VS AA (P=0.005); (D) IL-10: GG VS GA (P=0.005), GG VS AA (P=0.017). Origin v8.0, United States OriginLab Corporation. Figure 3. (A) The binding between the wild-type and the mutated MBL2 proteins to MASP1. (B) The binding between the wild-type and the mutated MBL2 proteins to MASP2. Origin v8.0, United States OriginLab Corporation.
Figure 3. (A) The binding between the wild-type and the mutated MBL2 proteins to MASP1. (B) The binding between the wild-type and the mutated MBL2 proteins to MASP2. Origin v8.0, United States OriginLab Corporation. Figure 4. Multi-sequence alignment of MBL2 proteins from eight species. MAFFT v7.0 (http://mafft.cbrc.jp/alignment/server/).
Figure 4. Multi-sequence alignment of MBL2 proteins from eight species. MAFFT v7.0 (http://mafft.cbrc.jp/alignment/server/). Figure 5. Evolutionary tree of 8 species used for conservative analysis. Molecular Evolutionary Genetics Analysis7.0, Mega Limited, Auckland, New Zealand.
Figure 5. Evolutionary tree of 8 species used for conservative analysis. Molecular Evolutionary Genetics Analysis7.0, Mega Limited, Auckland, New Zealand. Figure 6. The relationship between G54D mutation and the known domain of MBL2 protein. SMART(http://smart.embl-heidelberg.de/).
Figure 6. The relationship between G54D mutation and the known domain of MBL2 protein. SMART(http://smart.embl-heidelberg.de/). Tables
 Table 1. Overall clinical characteristics of patients in the sepsis group and non-sepsis group.
Table 1. Overall clinical characteristics of patients in the sepsis group and non-sepsis group. Table 2. The rs1800450 genotype distributions of Hardy-Weinberg equilibrium in total (n/%).
Table 2. The rs1800450 genotype distributions of Hardy-Weinberg equilibrium in total (n/%). Table 3. Genotypic and allelic frequency distribution of rs1800450 in the sepsis group and non-sepsis group. P=0.014 for dominant association (GA+AA vs GG). P=0.971 for recessive association (AA vs GA+GG).
Table 3. Genotypic and allelic frequency distribution of rs1800450 in the sepsis group and non-sepsis group. P=0.014 for dominant association (GA+AA vs GG). P=0.971 for recessive association (AA vs GA+GG). Table 1. Overall clinical characteristics of patients in the sepsis group and non-sepsis group.
Table 1. Overall clinical characteristics of patients in the sepsis group and non-sepsis group. Table 2. The rs1800450 genotype distributions of Hardy-Weinberg equilibrium in total (n/%).
Table 2. The rs1800450 genotype distributions of Hardy-Weinberg equilibrium in total (n/%). Table 3. Genotypic and allelic frequency distribution of rs1800450 in the sepsis group and non-sepsis group. P=0.014 for dominant association (GA+AA vs GG). P=0.971 for recessive association (AA vs GA+GG).
Table 3. Genotypic and allelic frequency distribution of rs1800450 in the sepsis group and non-sepsis group. P=0.014 for dominant association (GA+AA vs GG). P=0.971 for recessive association (AA vs GA+GG). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








