07 June 2022: Clinical Research
Clinical Application of C-TIRADS Category and Contrast-Enhanced Ultrasound in Differential Diagnosis of Solid Thyroid Nodules Measuring ≥1 cm
Zhuang Jin1ABCDEF, Yaqiong Zhu2ABCDEF, Yu Lei3BF, Xin Yu1BCD, Nan Jiang1BCD, Yue Gao1CF, Junying Cao1ACDEF*DOI: 10.12659/MSM.936368
Med Sci Monit 2022; 28:e936368
Abstract
BACKGROUND: The aim of this study was to investigate the performance of Chinese Thyroid Imaging Reporting and Data System (C-TIRADS) category combined with contrast-enhanced ultrasound (CEUS) in diagnosing thyroid cancer.
MATERIAL AND METHODS: From October 2020 to March 2021, 116 thyroid nodules from 113 patients who underwent conventional ultrasound and CEUS examinations at the General Hospital of Northern Theater Command were reviewed. In the conventional ultrasound examination, thyroid nodules were categorized by C-TIRADS. The nodules were reclassified based on CEUS scoring, then a combined diagnosis was made. The pathological results were taken as the criterion standard. To compare the diagnostic performance of the 3 methods according to the receiver operating characteristic curves produced for thyroid nodules.
RESULTS: After chi-square test, the 7 characteristics of enhancement patterns, internal homogeneity, wash-in, wash-out, ring enhancement, morphology, and contrast agent retention characteristics (CAR) were statistically different between malignant and benign tumors; A high diagnostic performance was demonstrated by C-TIRADS combined with CEUS in diagnosing thyroid cancer. The area under the curve (AUC), the sensitivity, and the specificity of C-TIRADS combined with CEUS for diagnosing thyroid cancer were 0.918 (95% CI: 0.852-0.961), 80.9% (95% CI: 69.1-89.8%), and 90.6% (95% CI: 79.3-96.9%), respectively. The AUC of C-TIRADS alone was significantly lower than that of C-TIRADS combined with CEUS (P=0.0056), while there was no significant difference between CEUS and C-TIRADS combined with CEUS (P=0.59).
CONCLUSIONS: The combined method of C-TIRADS and CEUS, with reduced rate of thyroid nodule biopsy and clinical application value, has higher diagnostic accuracy than the single diagnosis method.
Keywords: contrast-enhanced ultrasound, TI-RADS, thyroid nodules, Diagnosis, Differential, Humans, Thyroid Neoplasms, Thyroid Nodule, Ultrasonography
Background
Thyroid nodules are one of the most common endocrine diseases in the world. In the normal population, 47% are found through physical examination, while 30–67% are found through ultrasound examination [1]. The vast majority of thyroid cancer, which occurs in 7–15% of patients with thyroid nodules, is papillary cancer [2], and thyroid cancer has become the fourth most common cancer in women [3]. The number of new cases of thyroid cancer among Chinese people in 2015 was 90 000 [4], while the number of new cases of thyroid cancer in the United States in 2019 was 52070 [5]. How to accurately identify thyroid cancer before surgery remains a challenge [6,7]. Ultrasound is the most commonly used method for evaluating thyroid nodules and has become the imaging method of choice for thyroid nodule screening, preoperative diagnosis, and postoperative follow-up [8,9]. To standardize the diagnosis and classification criteria of thyroid nodules, and exclude subjective factors in the interpretation of results, Horvath et al [10] published the Thyroid Imaging Reporting and Data System (TIRADS) for the first time after drawing on the Breast Imaging Reporting and Data System (BI-RADS). In 2020, the Superficial Organ and Vascular Ultrasound Group of the Society of Ultrasound in Medicine of the Chinese Medical Association released a simple and practical risk stratification system for thyroid nodules called C-TIRADS [11]. However, the overlapping of ultrasound signs of benign and malignant thyroid nodules, especially for TIRADS 3 and 4 thyroid nodules, reduces the accuracy of conventional ultrasound in diagnosing thyroid nodules [12,13]. Fine-needle aspiration biopsy (FNA) is considered to be an accurate and effective method for preoperative diagnosis of thyroid nodules. However, at least 50% of needle biopsy nodules are benign [14], and about 20–30% of the results are uncertain [15]. Therefore, there may be overdiagnosis of benign nodules. Therefore, a new technique is urgently needed to improve the diagnostic accuracy of thyroid cancer, thereby reducing unnecessary FNA for benign nodules.
Contrast-enhanced ultrasound (CEUS) enables radiologists to make dynamic, real-time, non-invasive, and continuous observation of hemodynamics and microvascular perfusion in thyroid disease. CEUS has the ability to evaluate microvascular blood flow changes in thyroid lesions [16,17], and is an effective supplement to conventional ultrasound. Studies have confirmed that the diagnostic accuracy of thyroid cancers can been improved by conventional ultrasound combined with CEUS [18,19]. However, as far as we know, there is no research report on the differential diagnosis of C-TIRADS 3–5 nodules using C-TIRADS combined with CEUS so far. Therefore, we sought to show that CEUS can supplement the insufficiency of conventional ultrasonography in diagnosing thyroid nodules, while improving the diagnostic accuracy of thyroid benign and malignant tumors.
Material and Methods
ETHICS:
Institutional review board approval was obtained from the Ethics Committee of the General Hospital of Northern Theater Command for this retrospective study, and informed consent was waived.
RESEARCH DESIGN:
This was a retrospective study. Between October 2020 and March 2021, a total of 139 thyroid nodules from 131 patients were recruited at the General Hospital of Northern Theater Command in this study.
INCLUSION CRITERIA:
1. Solid nodules classified into 3–5 categories by conventional ultrasound; 2. Nodules with complete clinical, conventional ultrasound and contrast-enhanced ultrasound data; 3. Nodules clearly confirmed by surgery or needle biopsy; 4. The largest diameter was greater than 1.0 cm;
EXCLUSION CRITERIA: 1. Nodules with surrounding calcification or coarse calcification (Because surrounding calcification or coarse calcification affected the visualization of microbubbles inside the nodule, no real blood perfusion information could be observed); 2. Nodules without surrounding normal parenchyma for contrast. Nineteen cases were ruled out because the nodule had surrounding calcification, and 4 cases were ruled out because of its oversize. In the end, 116 thyroid nodules of 113 patients were enrolled in the study. Figure 1 shows the flow chart of this study.
INSPECTION METHOD:
GE LOGIQ E20 (E20; GE Medical Systems Ultrasound & Primary Care Diagnostics, LLC, Wauwatosa, USA) or Samsung RS80A (RS80A; Samsung Madison Co., Ltd., Seoul, Korea) ultrasound diagnostic system was used for conventional ultrasound and CEUS. Conventional ultrasound inspections used 10–15 MHz linear array transducers, and CEUS inspections used 7–9 MHz linear array transducers. The examination was performed by a radiologist with 10 years of experience in thyroid ultrasound and 3 years of experience in CEUS. The patient was in a supine position, with head tilted back, and the inspection area was fully revealed. To obtain the best image quality, the patient was asked not to swallow and breathe to calmly during the examination. The probe should be in close contact with the skin with enough coupling agent to ensure that the target thyroid nodule imaging signal is not lost. The image settings included focus, filter, gain and color gain, and adjusted until the best image was obtained. In the dual-frame contrast enhancement mode, the best section of the nodule was selected for CEUS. Five ml of normal saline was injected into the contrast medium (SonoVue, Bracco, Milan, Italy) and mixed until a homogeneously mixed suspension was produced. An intravenous access was created in the anterior cubital fossa, and 1.5 mL of microbubble suspension was rapidly injected. Then, the test tube was rinsed with 5 mL of normal saline. At the same time, the timer was started to dynamically record the process of CEUS (at least 2 minutes). During the CEUS, the patient was told to maintain posture and breathe calmly.
IMAGE ANALYSIS AND DIAGNOSTIC CRITERIA:
Blind review was performed by 2 radiologists with more than 10 years of experience in ultrasound diagnosis of thyroid diseases. All differences of opinion were resolved through discussion and negotiation. Conventional ultrasound was classified in TIRADS according to the protocol of the Superficial Organ and Vascular Ultrasound Group of the Society of Ultrasound in Medicine of the Chinese Medical Association [10]. The following ultrasound features of thyroid nodules were assessed: the composition of the nodule, internal echo, aspect ratio, morphology, and calcification. The C-TIRADS category criteria of this study are shown in Supplementary Table 1.
The qualitative indicators of CEUS analysis of thyroid target nodules included enhancement intensity (hypo-enhancement, iso-enhancement, hyperenhancement, no enhancement); patterns of enhancement (centrality, diffuse, centrifugal); internal homogeneity (homogeneity, heterogeneity); wash-in (earlier, synchronous, later); wash-out (earlier, synchronous, later); ring enhancement (yes or no), boundary (clear, unclear); morphology (regular, irregular); CAR [16] (yes, no). The CEUS features that were significant in differentiating benign and malignant thyroid nodules were scored, and the score for each thyroid nodule was calculated. One point was subtracted for each benign feature, and 1 point was added for each malignant feature. Nodules with a score of <0 were classified as benign, and with a score of >0 as malignant (Table 1).
The diagnostic criteria for C-TIRADS category combined with CEUS were as follows: Nodules with a CEUS score of 0 point were the same as TIRADS category; if nodules with a CEUS score of <0 point, the TIRADS category will be down-graded. When the nodule score was above 0 point, the TIRADS category was up-graded (for instance, if the nodule of C-TIRADS4b scored 3 points, the TIRADS was up-graded to TIRADS 4c). In this study, when the nodule was C-TIRADS 3 or 5, even if the CEUS score was less than or greater than 0, the category of the nodule remained unchanged.
STATISTICAL ANALYSIS:
Statistical analysis was performed using the SPSS 20.0 software package (SPSS, Inc, Chicago, IL) and MedCalc version 11.4.2.0 (MedCalc Software, Ostend, Belgium). Count data were expressed as means (percentage) and analyzed with the chi-square test or Fisher’s exact test. Measurement data were expressed as mean±standard deviation, and independent sample
Results
GENERAL INFORMATION ABOUT PATIENTS AND NODULES:
A total of 113 patients with an average age of 47.2±12.2 (22–84) years old were recruited in this study, including 37 males (37 cases of nodules) and 76 females (79 cases of nodules). There was a total of 116 cases of thyroid nodules, the average size of the nodules was 1.7±0.6 (1.0–5.1) cm, of which 53 cases were benign nodules, the average size was 1.6±0.7 (1.0–5.1) cm, 63 cases were malignant nodules, the average size was 1.8±0.6 (1.0–3.3) cm. All benign nodules consisted of 38 nodular goiters, 5 follicular adenomas, and 10 inflammatory lesions; while among the malignant tumors, 59 were papillary carcinoma, 2 were medullary carcinoma, 1 was follicular carcinoma, 1 was thyroid sarcoma. See Table 2 for details. Benign and malignant thyroid nodules had significant differences in markedly hypoechoic, vertical orientation, microcalcifications, regular, and C-TIRADS classification (Table 3).
ANALYSIS OF THE CHARACTERISTICS OF CEUS IN DIAGNOSING THYROID CANCER:
After chi-square testing, the 7 characteristics of patterns of enhancement, internal homogeneity, wash-in, wash-out, ring enhancement, morphology, and CAR were statistically different in benign and malignant tumors (Table 4). Please refer to Table 1 for the scoring method of CEUS differentiation of thyroid malignant and benign nodules.
ANALYSIS OF DIAGNOSTIC EFFICACY OF C-TIRADS, CEUS AND C-TIRDS COMBINED WITH CEUS IN THE DIAGNOSIS OF THYROID CANCER:
The AUC value of C-TIRADS in the diagnosis of thyroid cancer was 0.845 (95% CI: 0.766–0.905), the Se was 85.7% (95% CI: 74.6–93.3%), the Sp was 73.6% (95% CI: 59.7–84.7%), and the accuracy was 80.2%. The AUC value of CEUS in diagnosing thyroid cancer was 0.908 (95% CI: 0.840–0.954), the Se was 79.8% (95% CI: 67.3–88.3%), the Sp was 88.7% (95% CI: 77.0–95.7%), and the accuracy was 83.6%. The AUC value of C-TIRADS combined with CEUS in the diagnosis of thyroid cancer was 0.918 (95% CI: 0.852–0.961), the Se was 80.9% (95% CI: 69.1–89.8%), the Sp was 90.6% (95% CI: 79.3–96.9%), and the accuracy was 85.3%. Compared to C-TIRADS, the AUC value of C-TIRADS combined with CEUS in the diagnosis of thyroid cancer was higher(P=0.0056), while there was no significant difference between C-TIRADS combined with CEUS and CEUS in diagnosing thyroid cancer(P=0.59). See Table 5 and Figure 2.
THE INTEROBSERVER AGREEMENT ANALYSIS BETWEEN 2 RADIOLOGISTS:
The interobserver agreement of C-TIRADS and internal homogeneity, wash-in, wash-out, patterns of enhancement, ring enhancement, morphology, and CAR of CEUS between the 2 radiologists were 0.868, 0.921, 0.931, 0.947, 0.896, 0.963, 0.944, and 0.967, respectively.
Discussion
Due to its advantages of being convenient, cheap, non-invasive, and non-radiation, ultrasound examination has become the most important method clinical examination of thyroid diseases. In conventional ultrasound, the TIRADS category is a classification method that helps diagnose malignant nodules and provides guidance for clinical treatment [9,20,21]. This also helps the communication between radiologists and clinicians so as to reduce the effect of subjective factors. The TIRADS category was originally proposed and evaluated by Horvath et al [10]. In 2020, China officially released the TIRADS category based on China’s national conditions [11]. Zhou et al [22] reported a total of 2141 cases of thyroid nodules were included, and thyroid nodules were evaluated by quantitative scoring method. For classification, the AUC value for the diagnosis of thyroid cancer could reach 0.913, the Se was 92.7%, and the Sp was 71.2%. In this study, the AUC value of C-TIRADS in the diagnosis of thyroid cancer was 0.845, the Se was 85.7%, and the Sp was 73.6%. This shows that C-TIRADS has a high diagnostic value. However, in actual clinical work, the C-TIRADS category and diagnostic criteria are not perfect, and there is large variability in the AUC value, Se, and Sp of the diagnosis of thyroid cancer. Although the AUC value and Se in this study were lower than those of Zhou et al, they are similar to the results of Hu et al [23]. The diagnostic Se of thyroid nodules above 1 cm is 85.3% and the Sp is 70.6% in the study of Hu et al. The analysis may have the following reasons: (1) The proportions of malignant and benign nodules in the 2 studies are different, and the proportions of malignant and benign nodules in this study are close 1 (53/63), while the ratio of malignant and benign in the study of Zhou are close to 3 (1576/565); and (2) The ratio of inflammatory lesions in the 2 studies is also different (10/116 vs 21/2141). In addition, the natural attributes of thyroid nodules cannot be determined only by ultrasound imaging [24], and more methods are needed to reduce or prevent missed diagnosis, misdiagnosis, delayed treatment, or over-treatment.
Recently, application of CEUS in thyroid diseases has turn into a research hot spot. In China, CEUS has been applied in the differential diagnosis of benign and malignant thyroid nodules. CEUS can make up for the shortcomings of color Doppler and better display the microcirculation conditions in the nodules or tissues [16,25]. CEUS can not only display the perfusion intensity of the contrast agent, but also provide information such as the wash-in or wash-out of the contrast agent into the lesion [16,26], as well as the morphological and biological characteristics of blood vessels. Some research analyses have found that compared with conventional ultrasound, CEUS has higher accuracy in detecting and identifying thyroid nodules [18,27,28]. Our study showed that the Se, Sp, and AUC of CEUS in the diagnosis of thyroid cancer were 79.8%, 88.7%, and 0.908, respectively, which are consistent with results of the above-mentioned research. These findings demonstrate that CEUS has clinical value in diagnosing thyroid cancer. Previous studies have indicated that ring enhancement is strongly correlated with benign nodules, while hypo-enhancement and uneven enhancement are malignant signs [12,29]. However, a single CEUS feature does not seem to have good Se or Sp to discriminate malignant and benign nodules. A previous meta-analysis has concluded that CEUS qualitative indicators are better than quantitative indicators in the discrimination of thyroid nodules [30]. In this study, it was found that the 7 characteristics of patterns of enhancement, internal homogeneity, wash-in, wash-out, ring enhancement, morphology, and CAR were different in malignant and benign nodules. However, there was no difference in the enhancement intensity between malignant and benign nodules, which seemed to be inconsistent with the results of the 2 above studies. Zhang et al [31] demonstrated that as the volume of thyroid nodules increased, the CEUS of the nodules showed mostly iso-enhancement or hyperenhancement, rather than low enhancement. This was similar to the results of this study. Among nodules ≥ 1 cm, benign and malignant nodules mostly showed iso- enhancement or hyperenhancement, and the difference was not statistically significant. Liu et al [32] demonstrated that the expansion and invasion of malignant nodules outside the capsule led to irregular features in CEUS. Heterogeneity enhancement showed inhomogeneous distribution of blood vessels inside malignant nodules [33], and as the nodules grew, these 2 signs were more obvious. The blood supply of malignant nodules can be divided into central and peripheral parts. The blood vessels in the central part are relatively scarce, while the blood vessels in the peripheral part are relatively dense. This causes the signs of centripetal enhancement in most malignant nodules during CEUS. The CAR is a recently discovered characteristic malignant nodules. This may be correlated with the increase in tumor cells and volume of the malignant nodule, which oppresses the interstitial tissue, especially the compression of the microvessels, leading to the signs of microvascular congestion in the nodules [17]. There were also differences in wash-in and wash-out between benign and malignant nodules, which might be caused by differences in microvascular diameter, morphology, vascular branches, arteriovenous fistulas, microthrombosis, and fibrosis.
However, studies have found that the Se and Sp of TIRADS classification and CEUS may vary greatly [34,35]. To improve diagnostic accuracy and reduce missed diagnosis and misdiagnosis, we combined C-TIRADS and CEUS to identify thyroid nodules. We observed that the AUC value was the highest (0.918) after the combination of C-TIRADS and CEUS, which was significantly higher than the AUC value of C-TIRADS (0.845), and slightly higher than the AUC value of CUES (0.908), but there was no significant difference between the 2 methods. This was line with the results of other studies [18,19,36]. These indicate that the combined method is beneficial to reduce the occurrence of misdiagnosis and missed diagnosis. C-TIRADS alone misdiagnosed 23 cases of thyroid nodules, 16 of which were corrected by CEUS, which showed that CEUS could play a role in complementing C-TIRADS. In this study, there were 10 cases of inflammatory lesions and 8 cases of C-TIRADS were misdiagnosed, but 7 of them were corrected by CEUS, which showed that CEUS could assist in the identification of inflammatory lesions (Figure 3). However, this was not consistent with the results of Zhang et al [19]. The discrepancies may arise from the different methods used by CEUS in the 2 studies. Furthermore, the number of misdiagnosed cases of thyroid nodules by CEUS alone was 19, of which 2 cases were corrected by C-TIRADS, which showed that C-TIRADS had limited supplementary effect on CUES, which also caused there to be no significant difference in AUC value between the combined method and CEUS. (0.918 vs 0.908).
In addition, we analyzed the interobserver agreement between 2 radiologists and found that the 2 radiologists had a high degree of agreement in the characteristics of C-TIRADS and CEUS (0.868–0.967). This shows that C-TIRADS category and CEUS features are less subjectively affected by the radiologist.
Our study has the following limitations: 1. This study was a retrospective study, and the data were inevitably biased; 2. The sample size of this study was not large enough, which might have an effect on the results. In the future, the sample size should be increased, and multi-center verification should be carried out; 3. In this study, most of the malignant tumors were papillary carcinoma, and most of the benign tumors were nodular goiters. 4. The proportion of benign and malignant nodules in this study was lower than the actual proportion of benign and malignant nodules, which may have influenced the results of the study. Therefore, this result had limited diagnostic value for other types of malignant and benign nodules, and further verification is needed in the future.
Conclusions
We found that CEUS has higher accuracy than C-TIRADS in the diagnosis of thyroid cancer, and the diagnostic accuracy was further improved after the combination of the 2. Therefore, the combined method of C-TIRADS and CEUS for the diagnosis of thyroid cancer should be promoted and applied.
Figures
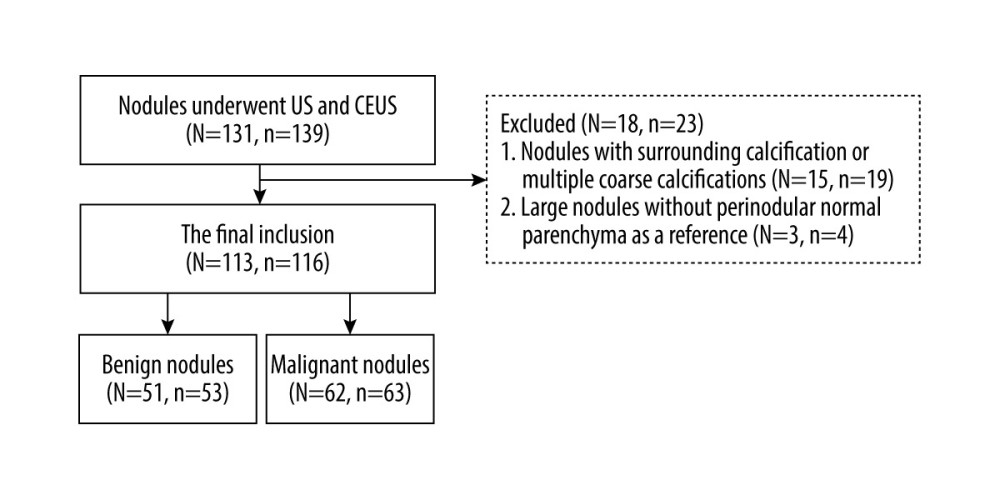 Figure 1. The flowchart of patients with thyroid nodules selection. N – number of patients; n – number of thyroid nodules. The Figure 1 was produced by PowerPoint version 2016 (Microsoft corporation, WA, USA).
Figure 1. The flowchart of patients with thyroid nodules selection. N – number of patients; n – number of thyroid nodules. The Figure 1 was produced by PowerPoint version 2016 (Microsoft corporation, WA, USA). 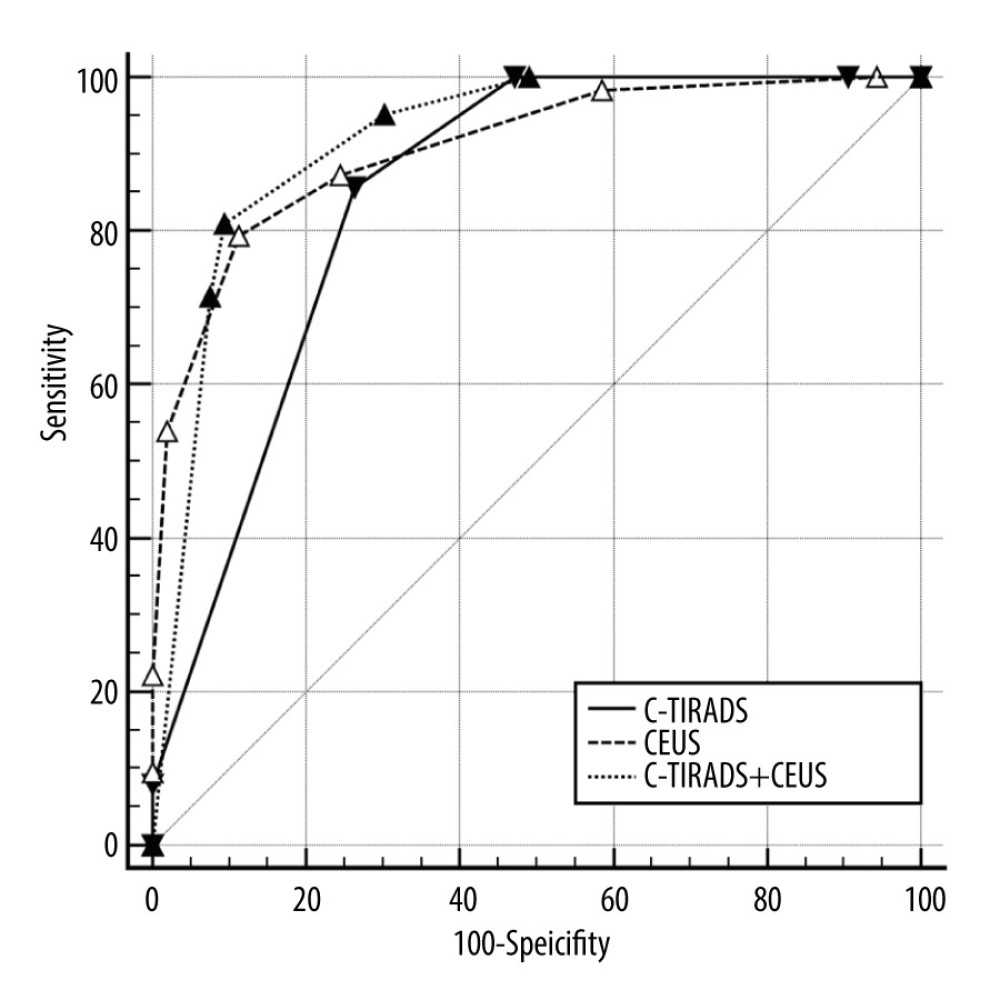 Figure 2. ROC curves for C-TIRADS, CEUS, and combined method. The Figure 2 was produced by MedCalc version 11.4.2.0 (MedCalc Software, Ostend, Belgium).
Figure 2. ROC curves for C-TIRADS, CEUS, and combined method. The Figure 2 was produced by MedCalc version 11.4.2.0 (MedCalc Software, Ostend, Belgium). 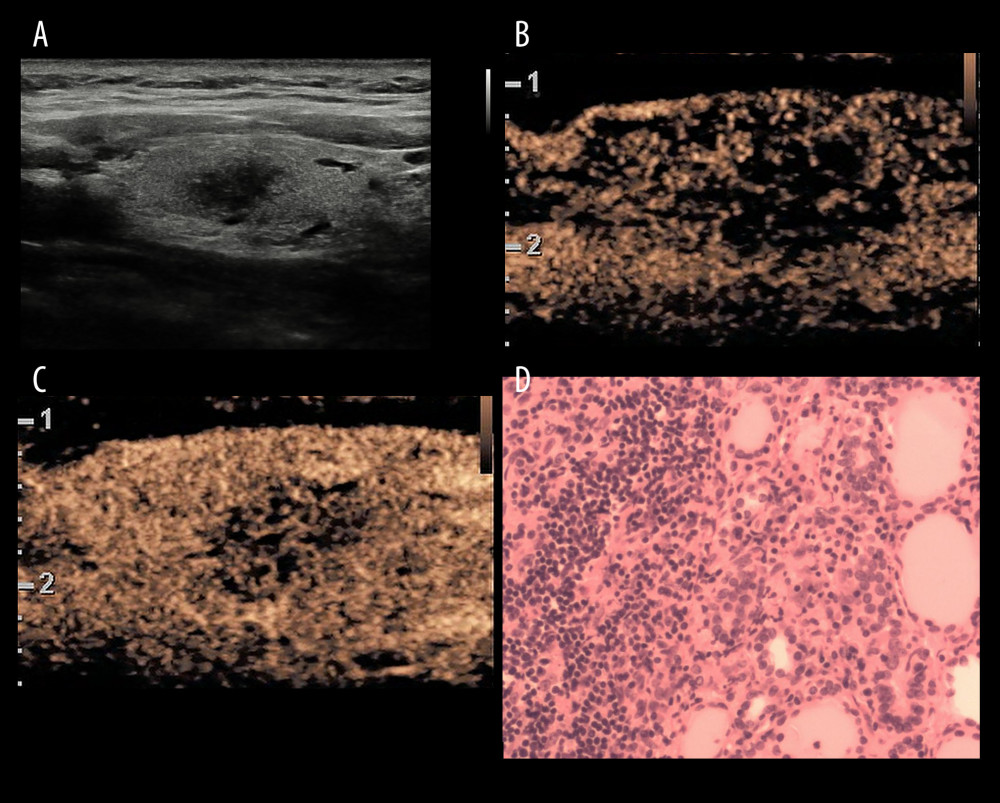 Figure 3. A inflammatory lesion in a 35-year-old woman. (A) Greyscale ultrasound showed that there was a solid very hypoechoic lesion in the left lobe of the thyroid, with irregular margin and a wider-than-tall shape. The nodule was C-TIRADS category 4c.(B) Contrast-enhanced ultrasound showed diffused and synchronous enhancement within the nodule at the time of the10th second after the injection of contrast agent. (C) Contrast-enhanced ultrasound showed hypo-enhancement and heterogeneity at peak (the 15th second after the injection of contrast agent), with irregular morphology. The CUES score of the lesion was −1. (D) The pathological image of the lesion, which was of subacute thyroiditis. Figure 3 was produced by PowerPoint version 2016(Microsoft corporation, WA, USA).
Figure 3. A inflammatory lesion in a 35-year-old woman. (A) Greyscale ultrasound showed that there was a solid very hypoechoic lesion in the left lobe of the thyroid, with irregular margin and a wider-than-tall shape. The nodule was C-TIRADS category 4c.(B) Contrast-enhanced ultrasound showed diffused and synchronous enhancement within the nodule at the time of the10th second after the injection of contrast agent. (C) Contrast-enhanced ultrasound showed hypo-enhancement and heterogeneity at peak (the 15th second after the injection of contrast agent), with irregular morphology. The CUES score of the lesion was −1. (D) The pathological image of the lesion, which was of subacute thyroiditis. Figure 3 was produced by PowerPoint version 2016(Microsoft corporation, WA, USA). Tables
Table 1. The CUES thyroid nodules scores.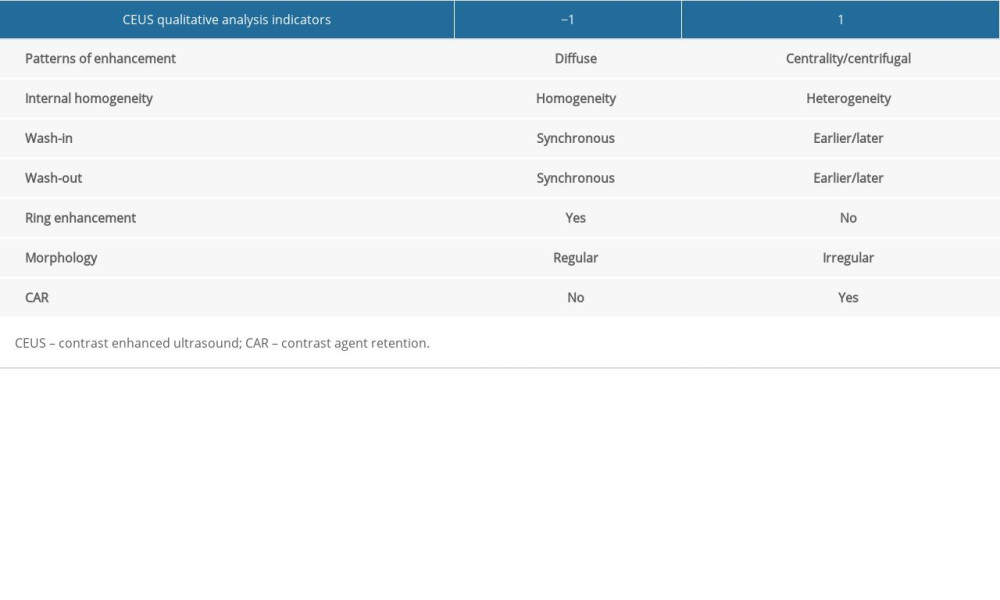 Table 2. Patient clinical data.
Table 2. Patient clinical data.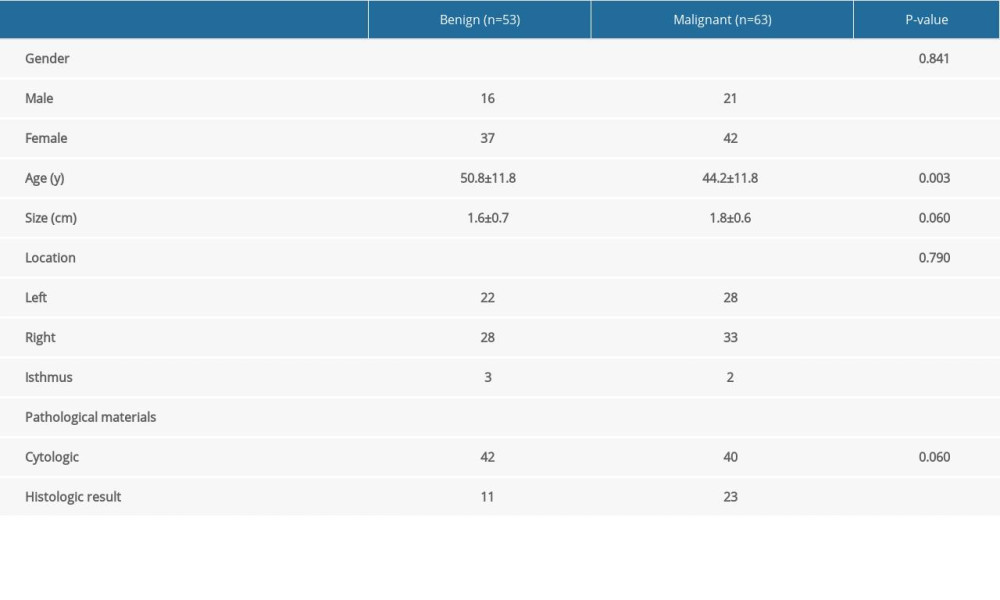 Table 3. The examination results on thyroid nodules by conventional ultrasound.
Table 3. The examination results on thyroid nodules by conventional ultrasound.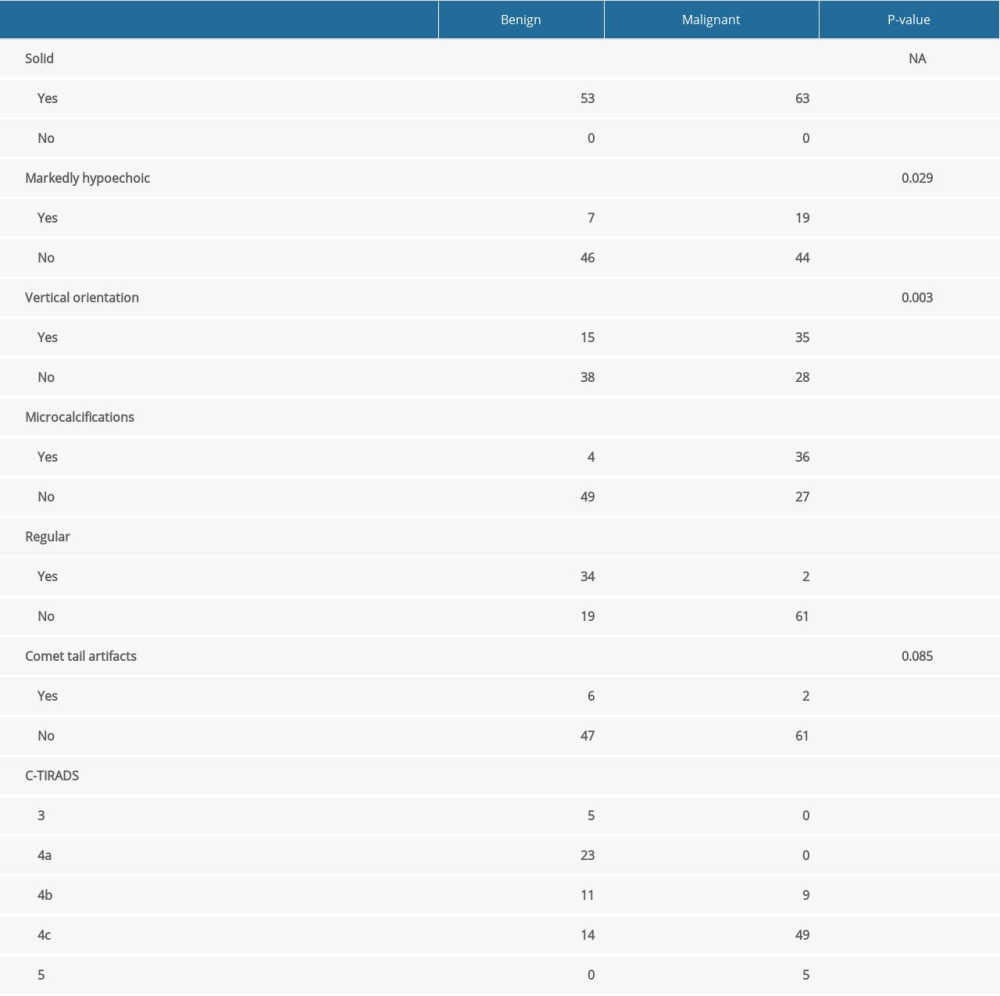 Table 4. Comparison of CEUS characteristics of thyroid nodules.
Table 4. Comparison of CEUS characteristics of thyroid nodules.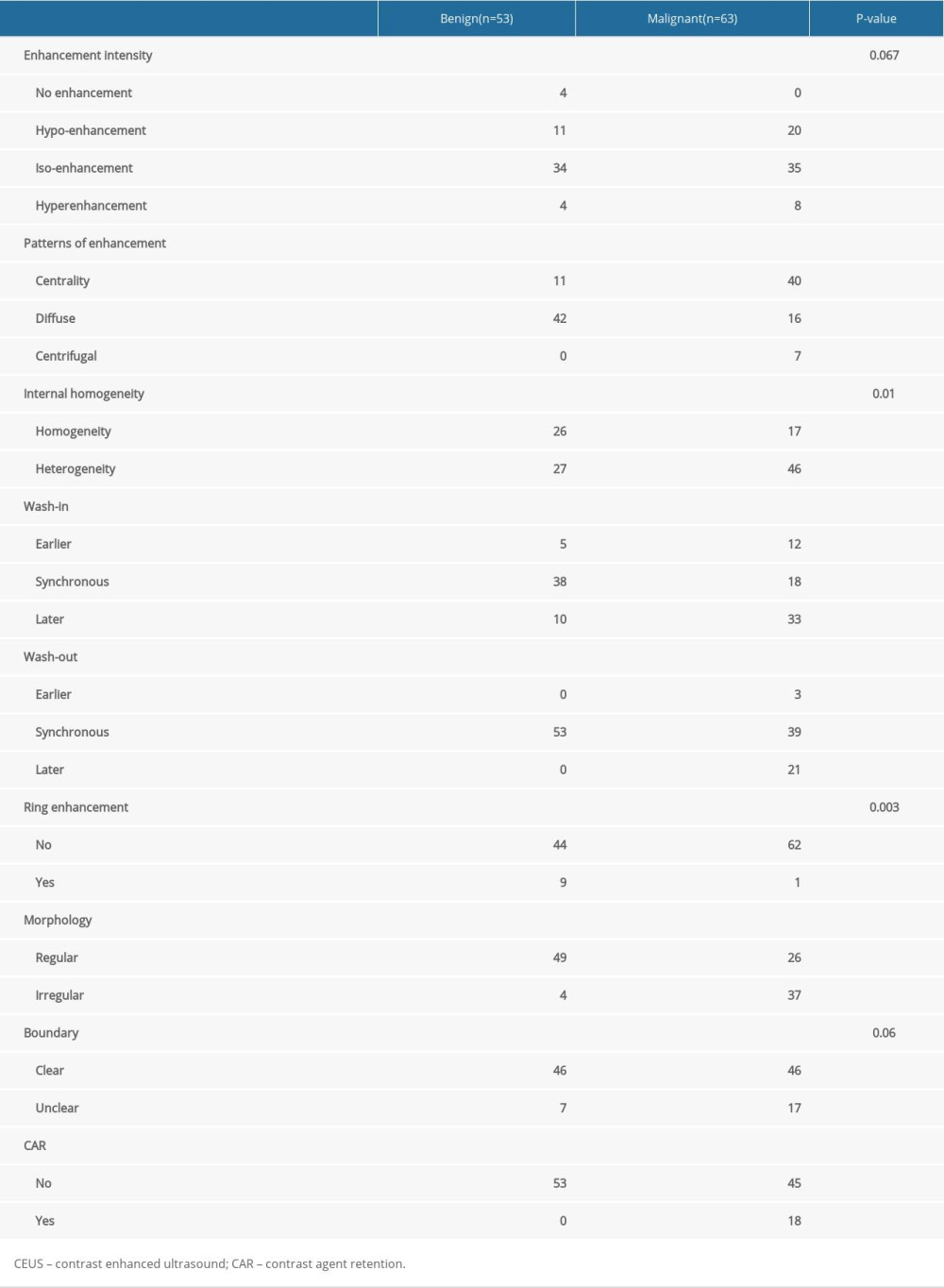 Table 5. Diagnostic performance of C-TIRADS and CEUS.
Table 5. Diagnostic performance of C-TIRADS and CEUS.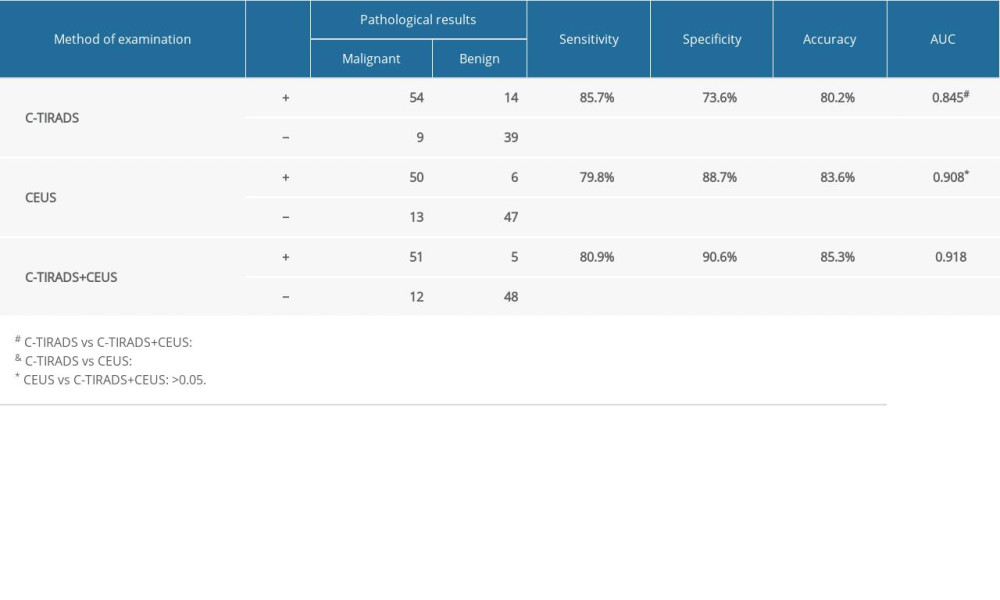 Supplementary Table 1. The C-TIRADS based on the counting method.
Supplementary Table 1. The C-TIRADS based on the counting method.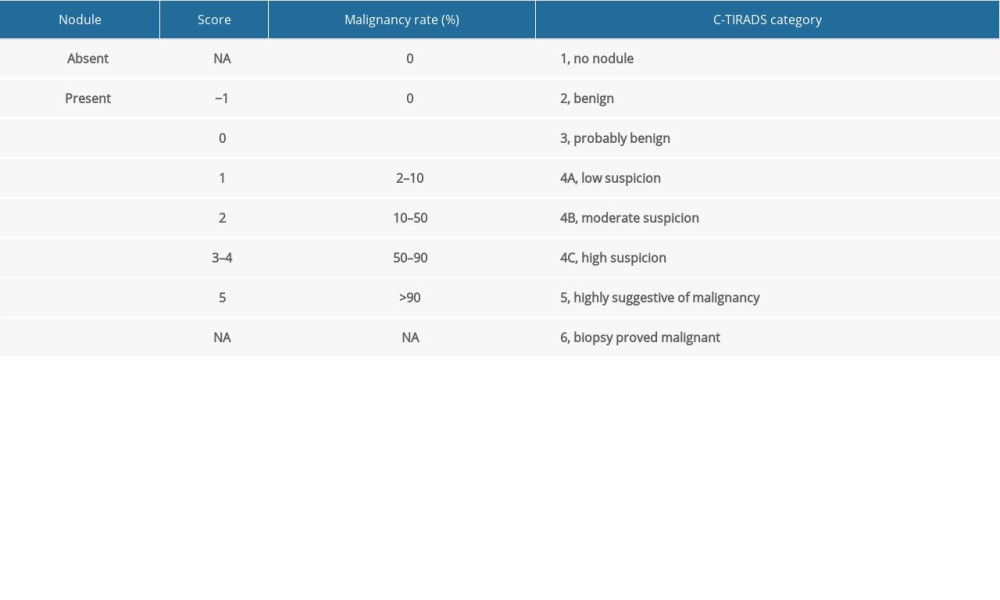
References
1. Tufano RP, Noureldine SI, Angelos P, Incidental thyroid nodules and thyroid cancer: Considerations before determining management: JAMA Otolaryngol head Neck Surg, 2015; 141(6); 566-72
2. Haugen BR, Alexander EK, Bible KC, 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines Task Force on thyroid nodules and differentiated thyroid cancer: Thyroid, 2016; 26(1); 1-133
3. Miller KD, Siegel RL, Lin CC, Cancer treatment and survivorship statistics, 2016: Cancer J Clin, 2016; 66(4); 271-89
4. Chen W, Zheng R, Baade PD, Cancer statistics in China, 2015: Cancer J Clin, 2016; 66(2); 115-32
5. Siegel RL, Miller KD, Jemal A, Cancer statistics, 2019: Cancer J Clin, 2019; 69(1); 7-34
6. Chng CL, Kurzawinski TR, Beale T, Value of sonographic features in predicting malignancy in thyroid nodules diagnosed as follicular neoplasm on cytology: Clin Endocrinol (Oxf), 2015; 83(5); 711-16
7. Pang T, Huang L, Deng Y, Logistic regression analysis of conventional ultrasonography, strain elastosonography, and contrast-enhanced ultrasound characteristics for the differentiation of benign and malignant thyroid nodules: PLoS One, 2017; 12(12); e0188987
8. Ha EJ, Baek JH, Na DG, Risk stratification of thyroid nodules on ultrasonography: Current status and perspectives: Thyroid, 2017; 27(12); 1463-68
9. Grani G, Lamartina L, Ascoli V, Ultrasonography scoring systems can rule out malignancy in cytologically indeterminate thyroid nodules: Endocrine, 2017; 57(2); 256-61
10. Horvath E, Majlis S, Rossi R, An ultrasonogram reporting system for thyroid nodules stratifying cancer risk for clinical management: J Clin Endocrinol Metab, 2009; 94(5); 1748-51
11. Zhou J, Yin L, Wei X, 2020 Chinese guidelines for ultrasound malignancy risk stratification of thyroid nodules: The C-TIRADS: Endocrine, 2020; 70(2); 256-79
12. Sidhu PS, Cantisani V, Dietrich CF, The EFSUMB guidelines and recommendations for the clinical practice of contrast-enhanced ultrasound (CEUS) in non-hepatic applications: Update 2017 (long version): Ultraschall Med, 2018; 39(2); e2-44
13. Grant EG, Tessler FN, Hoang JK, Thyroid ultrasound reporting lexicon: White paper of the ACR Thyroid Imaging, Reporting and Data System (TIRADS) Committee: J Am Coll Radiol, 2015; 12(12 Pt A); 1272-79
14. Bongiovanni M, Spitale A, Faquin WC, The Bethesda System for Reporting Thyroid Cytopathology: A meta-analysis: Acta Cytol, 2012; 56(4); 333-39
15. Durante C, Grani G, Lamartina L, The diagnosis and management of thyroid nodules: A review: JAMA, 2018; 319(9); 914-24
16. Zhou X, Zhou P, Hu Z, Diagnostic efficiency of quantitative contrast-enhanced ultrasound indicators for discriminating benign from malignant solid thyroid nodules: J Ultrasound Med, 2018; 37(2); 425-37
17. Jin Z, Zhu Y, Xie F, Contrast agent retention features in contrast-enhanced ultrasound: Diagnostic performance for the prediction of papillary thyroid carcinoma: Clin Imaging, 2021; 80; 131-38
18. Zhao H, Liu X, Lei B, Diagnostic performance of thyroid imaging reporting and data system (TI-RADS) alone and in combination with contrast-enhanced ultrasonography for the characterization of thyroid nodules: Clin Hemorheol Microcirc, 2019; 72(1); 95-106
19. Zhang Y, Zhou P, Tian SM, Usefulness of combined use of contrast-enhanced ultrasound and TI-RADS classification for the differentiation of benign from malignant lesions of thyroid nodules: Eur Radiol, 2017; 27(4); 1527-36
20. Horvath E, Silva CF, Majlis S, Prospective validation of the ultrasound based TIRADS (Thyroid Imaging Reporting And Data System) classification: results in surgically resected thyroid nodules: Eur Radiol, 2017; 27(6); 2619-28
21. Moon HJ, Kim EK, Kwak JY, Malignancy risk stratification in thyroid nodules with benign results on cytology: Combination of thyroid imaging reporting and data system and Bethesda system: Ann Surg Oncol, 2014; 21(6); 1898-903
22. Zhou J, Song Y, Zhan W, Thyroid imaging reporting and data system (TIRADS) for ultrasound features of nodules: Multicentric retrospective study in China: Endocrine, 2021; 72(1); 157-70
23. Hu L, Liu X, Pei C, Xie L, He N, Assessment of perinodular stiffness in differentiating malignant from benign thyroid nodules: Endocr Connect, 2021; 10(5); 492-501
24. Durot I, Wilson SR, Willmann JK, Contrast-enhanced ultrasound of malignant liver lesions: Abdom Radiol (NY), 2018; 43(4); 819-47
25. Friedrich-Rust M, Sperber A, Holzer K, Real-time elastography and contrast-enhanced ultrasound for the assessment of thyroid nodules: Exp Clin Endocrinol Diabetes, 2010; 118(9); 602-9
26. Jiang J, Huang L, Zhang H, Contrast-enhanced sonography of thyroid nodules: J Clin Ultrasound, 2015; 43(3); 153-56
27. Sun B, Lang L, Zhu X, Accuracy of contrast-enhanced ultrasound in the identification of thyroid nodules: A meta-analysis: Int J Clin Exp Med, 2015; 8(8); 12882-89
28. Li X, Gao F, Li F, Qualitative analysis of contrast-enhanced ultrasound in the diagnosis of small, TR3-5 benign and malignant thyroid nodules measuring ≤1 cm: Br J Radiol, 2020; 93(1111); 20190923
29. Zhang B, Jiang YX, Liu JB, Utility of contrast-enhanced ultrasound for evaluation of thyroid nodules: Thyroid, 2010; 20(1); 51-57
30. Yu D, Han Y, Chen T, Contrast-enhanced ultrasound for differentiation of benign and malignant thyroid lesions: Meta-analysis: Otolaryngol Head Neck Surg, 2014; 151(6); 909-15
31. Zhang Y, Luo YK, Zhang MB, Diagnostic accuracy of contrast-enhanced ultrasound enhancement patterns for thyroid nodules: Med Sci Monit, 2016; 22; 4755-64
32. Liu Y, Liu H, Qian CL, Utility of quantitative contrast-enhanced ultrasound for the prediction of extracapsular extension in papillary thyroid carcinoma: Sci Rep, 2017; 7(1); 1472
33. Foschini MP, Papotti M, Parmeggiani A, Three-dimensional reconstruction of vessel distribution in benign and malignant lesions of thyroid: Virchows Arch, 2004; 445(2); 189-98
34. Wei X, Li Y, Zhang S, Gao M, Thyroid imaging reporting and data system (TI-RADS) in the diagnostic value of thyroid nodules: A systematic review: Tumour Biol, 2014; 35(7); 6769-76
35. Trimboli P, Castellana M, Virili C, Performance of contrast-enhanced ultrasound (CEUS) in assessing thyroid nodules: A systematic review and meta-analysis using histological standard of reference: Radiol Med, 2020; 125(4); 406-15
36. Xu Y, Qi X, Zhao X, Clinical diagnostic value of contrast-enhanced ultrasound and TI-RADS classification for benign and malignant thyroid tumors: One comparative cohort study: Medicine (Baltimore), 2019; 98(4); e14051
Figures
 Figure 1. The flowchart of patients with thyroid nodules selection. N – number of patients; n – number of thyroid nodules. The Figure 1 was produced by PowerPoint version 2016 (Microsoft corporation, WA, USA).
Figure 1. The flowchart of patients with thyroid nodules selection. N – number of patients; n – number of thyroid nodules. The Figure 1 was produced by PowerPoint version 2016 (Microsoft corporation, WA, USA). Figure 2. ROC curves for C-TIRADS, CEUS, and combined method. The Figure 2 was produced by MedCalc version 11.4.2.0 (MedCalc Software, Ostend, Belgium).
Figure 2. ROC curves for C-TIRADS, CEUS, and combined method. The Figure 2 was produced by MedCalc version 11.4.2.0 (MedCalc Software, Ostend, Belgium). Figure 3. A inflammatory lesion in a 35-year-old woman. (A) Greyscale ultrasound showed that there was a solid very hypoechoic lesion in the left lobe of the thyroid, with irregular margin and a wider-than-tall shape. The nodule was C-TIRADS category 4c.(B) Contrast-enhanced ultrasound showed diffused and synchronous enhancement within the nodule at the time of the10th second after the injection of contrast agent. (C) Contrast-enhanced ultrasound showed hypo-enhancement and heterogeneity at peak (the 15th second after the injection of contrast agent), with irregular morphology. The CUES score of the lesion was −1. (D) The pathological image of the lesion, which was of subacute thyroiditis. Figure 3 was produced by PowerPoint version 2016(Microsoft corporation, WA, USA).
Figure 3. A inflammatory lesion in a 35-year-old woman. (A) Greyscale ultrasound showed that there was a solid very hypoechoic lesion in the left lobe of the thyroid, with irregular margin and a wider-than-tall shape. The nodule was C-TIRADS category 4c.(B) Contrast-enhanced ultrasound showed diffused and synchronous enhancement within the nodule at the time of the10th second after the injection of contrast agent. (C) Contrast-enhanced ultrasound showed hypo-enhancement and heterogeneity at peak (the 15th second after the injection of contrast agent), with irregular morphology. The CUES score of the lesion was −1. (D) The pathological image of the lesion, which was of subacute thyroiditis. Figure 3 was produced by PowerPoint version 2016(Microsoft corporation, WA, USA). Tables
 Table 1. The CUES thyroid nodules scores.
Table 1. The CUES thyroid nodules scores. Table 2. Patient clinical data.
Table 2. Patient clinical data. Table 3. The examination results on thyroid nodules by conventional ultrasound.
Table 3. The examination results on thyroid nodules by conventional ultrasound. Table 4. Comparison of CEUS characteristics of thyroid nodules.
Table 4. Comparison of CEUS characteristics of thyroid nodules. Table 5. Diagnostic performance of C-TIRADS and CEUS.
Table 5. Diagnostic performance of C-TIRADS and CEUS. Table 1. The CUES thyroid nodules scores.
Table 1. The CUES thyroid nodules scores. Table 2. Patient clinical data.
Table 2. Patient clinical data. Table 3. The examination results on thyroid nodules by conventional ultrasound.
Table 3. The examination results on thyroid nodules by conventional ultrasound. Table 4. Comparison of CEUS characteristics of thyroid nodules.
Table 4. Comparison of CEUS characteristics of thyroid nodules. Table 5. Diagnostic performance of C-TIRADS and CEUS.
Table 5. Diagnostic performance of C-TIRADS and CEUS. Supplementary Table 1. The C-TIRADS based on the counting method.
Supplementary Table 1. The C-TIRADS based on the counting method. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








