12 August 2022: Clinical Research
Clinical Value of Metagenomic Next-Generation Sequencing in Immunocompromised Patients with Sepsis
Zheng Cheng1ABCDEFG, Feng Yu1ABCDEFG*DOI: 10.12659/MSM.937041
Med Sci Monit 2022; 28:e937041
Abstract
BACKGROUND: Sepsis is a serious threat to human life, particularly in immunocompromised patients; hence, early diagnosis and targeted treatment are important. Metagenomic next-generation sequencing (NGS) has significant advantages over traditional diagnostic methods. This study investigated the clinical value of NGS for pathogen identification in immunocompromised patients with sepsis.
MATERIAL AND METHODS: From July 2020 to September 2021, 90 consecutive patients with sepsis were enrolled in this prospective study. The patients were divided into 2 groups: an immunocompromised group (n=30) and an immunocompetent group (n=60). The pathogens causing sepsis were concurrently identified using NGS and traditional diagnostic methods. The pathogen detection rates and the spectrum of pathogens identified were compared according to the method of detection and between the immunocompromised and immunocompetent groups.
RESULTS: Of the 90 patients, 77 (86%) were positive for 1 or more pathogens using NGS, and 50 (56%) were positive using traditional detection methods. The positivity rate of sputum and bronchoalveolar lavage fluid was higher than that of blood samples. Pneumocystis jirovecii and cytomegalovirus infections were more common in the immunocompromised group than in the immunocompetent group.
CONCLUSIONS: The performance of NGS in identifying pathogens for patients with sepsis is better than that of traditional detection methods, especially in immunocompromised patients. Pneumocystis jirovecii and cytomegalovirus infections are more common in immunocompromised patients.
Keywords: High-Throughput Nucleotide Sequencing, Immunocompromised Host, Pneumocystis Infections, Sepsis, Bronchoalveolar Lavage Fluid, Cytomegalovirus Infections, Humans, Pneumocystis carinii, Prospective Studies
Background
Sepsis refers to organ dysfunction caused by a defect in the host’s response to infection [1]. The incidence of sepsis and sepsis-related mortality are high worldwide, with an estimated 5.3 million deaths caused by sepsis annually [2]. Current guidelines recommend early application of targeted antibiotics to improve the prognosis of patients; however, most early antibiotic treatment is empirical. According to 1 study, approximately 46% of early empirical antibiotic treatment was inappropriate, directly leading to a sepsis-related mortality rate of almost 35%. Approximately 50% of antibiotics administered are unnecessary or too broad-spectrum, which increases the toxicity of drugs and the incidence of bacterial resistance [3]. Therefore, early identification of pathogens is particularly important to enable targeted antibiotic therapy. Currently, the common methods used to identify pathogens include microbial culture, serology, and polymerase chain reaction (PCR); however, the performance of these methods is affected by several factors and their sensitivity and specificity are low [4]. For example, culture, the criterion standard for pathogen detection, does not detect viruses and parasites; some bacterial pathogens are difficult to culture; and the culture cycle is long, so the results are delayed. In particular, traditional methods of pathogen identification are extremely unfavorable for immunocompromised patients with sepsis. As immunocompromised patients often have atypical clinical symptoms, the increase in inflammatory indices is not obvious, and most immunocompromised patients have mixed infections [5]. Although serology and PCR can compensate for these defects to some extent, their scope in detecting microorganisms is limited. The metagenomic next-generation sequencing (NGS) field has gradually developed and matured in recent years. NGS directly extracts nucleic acid fragments from samples, compares them with a constructed gene library, and identifies pathogens according to their characteristic readings and coverage. Compared to traditional detection methods, NGS has significant advantages in terms of sensitivity and specificity [6]. NGS technology has the advantages of being unaffected by the use of antibiotics and having a short detection time.
The aim of this study was to evaluate the clinical value of NGS in identifying pathogens in immunocompromised patients with sepsis.
Material and Methods
PATIENT INCLUSION CRITERIA AND GROUPS:
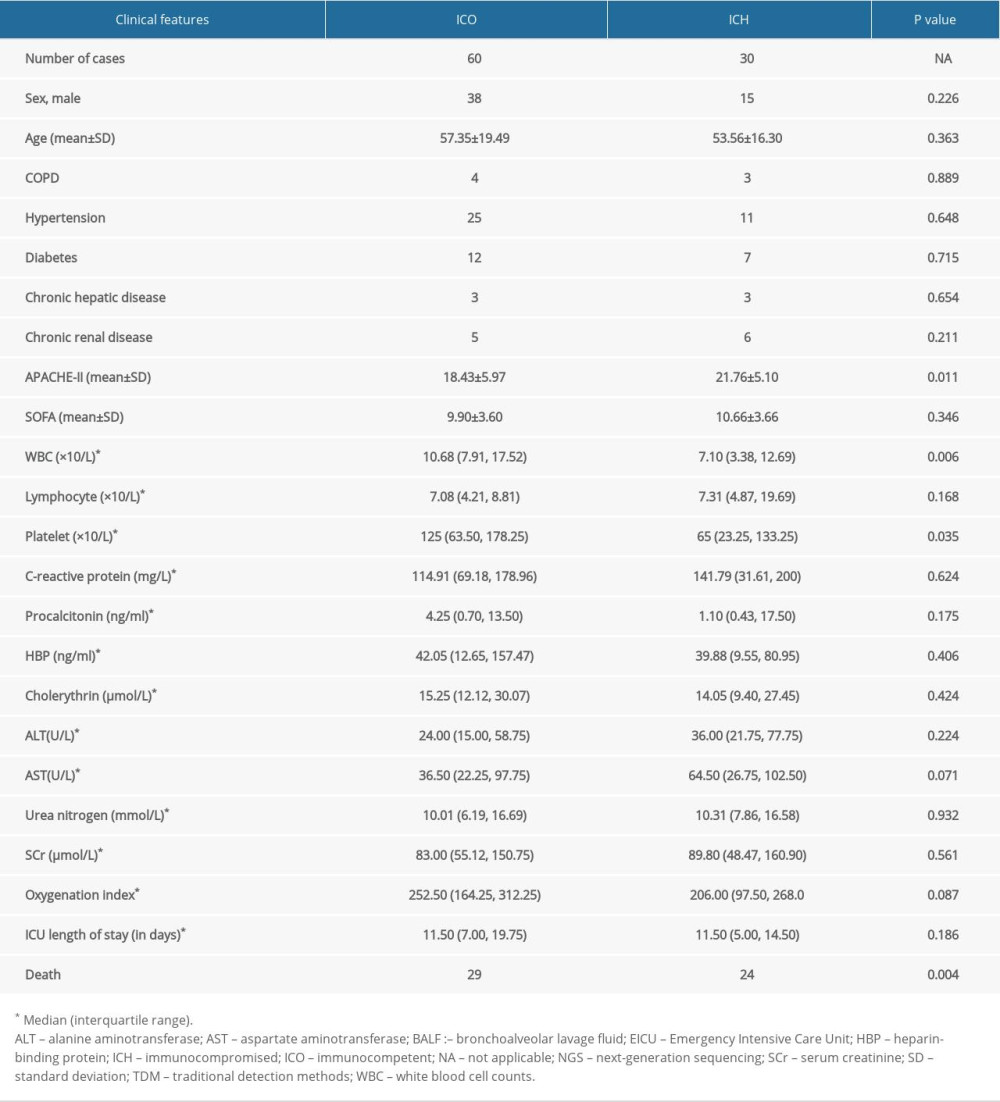
This prospective study included patients admitted to the Emergency Intensive Care Unit (EICU) of the First Affiliated Hospital of Anhui Medical University in Hefei, China, from July 2020 to September 2021, who met the diagnostic criteria for sepsis. The Clinical Ethics Committee of the First Affiliated Hospital of Anhui Medical University approved this study (No. PJ2021-03-32), and all patients or their guardians signed informed consent forms. The inclusion criteria were as follows: (1) met the Sepsis 3.0 criteria for sepsis [1]; (2) 18 years of age or older; (3) stayed in the EICU for more than 48 hours; and (4) the patient or their guardian provided written informed consent. Among patients with infection or suspected infection, sepsis can be diagnosed when the Sequential Organ Failure Assessment (SOFA) score increases by more than 2 points from the baseline value. The exclusion criteria were as follows: (1) individuals with HIV infection; (2) pregnant women; and (3) patients who withdrew from the study for any reason. The immunocompromised (ICH) group included patients with any of the following conditions before the onset of the disease: (1) use of long-term (>3 months) or high-dose (>0.5 mg/kg/day) steroids or other immunosuppressive drugs, (2) solid organ transplant, (3) solid tumor requiring chemotherapy, (4) hematologic malignancy, and (5) primary immunodeficiency [7]. The immunocompetent group (ICO) consisted of patients who did not meet any of these criteria for inclusion in the ICH group (Figure 1).
CLINICAL DATA COLLECTION:
At enrollment, baseline data, such as sex, age, and chronic diseases, were collected and participants’ Acute Physiology and Chronic Health Evaluation (APACHE II) and SOFA scores were assessed. All enrolled patients underwent laboratory tests (white blood cell, neutrophil, and platelet counts; hemoglobin, creatinine, albumin, bilirubin, alanine aminotransferase, aspartate aminotransferase, blood potassium, blood sodium, blood calcium, blood phosphorus, D-dimer, activated partial thromboplastin time, C-reactive protein, procalcitonin, arterial blood lactic acid, pH, heparin-binding protein, and arterial blood gas analysis).
SPECIMEN COLLECTION:
Blood and sputum samples were collected from all patients within 24 hours after admission to the EICU. The clinician performed fiberoptic bronchoscopy as indicated and submitted bronchoalveolar lavage fluid (BALF) samples for bacterial and fungal culture. All samples submitted for culture were collected under aseptic conditions using a special sampling tube. Samples of whole blood (3–5 mL), sputum (>3 mL), BALF (>5 mL), ascites (>5 mL), urine (>5 mL), cerebrospinal fluid (CSF) (>5 mL), and pleural effusion (>5 mL) were collected, as applicable. All blood samples were stored at 6–35°C, and all other samples were stored at −20°C. The collected samples were concurrently sent for traditional microbiological examination and second-generation sequencing. Traditional microbiological detection included bacterial and fungal culture, detection of common viruses and mycoplasmas by PCR, and serological detection of fungi. Specimens requiring NGS testing were sent to the Guangzhou Weiyuan Gene Technology Company (Guangzhou, China) for testing within 48 hours after collection.
NUCLEIC ACID EXTRACTION, LIBRARY PREPARATION, AND SEQUENCING FOR NGS:
To perform NGS, the first step was extraction of nucleic acid. The corresponding kit to extract pathogen DNA or RNA, and then Benzonase (Qiagen) and Tween 20 were used to remove the human-derived sequences [8] using the Ribo-Zero rRNA Removal Kit (Illumina, San Diego, CA, USA), which removed ribosomal RNA following RNA extraction. Complementary DNA (cDNA) was then produced using reverse transcriptase and deoxyribonucleotide triphosphate (dNTPs). DNA and cDNA were used to construct the library, using the appropriate library preparation kit [9]. The Qubit dsDNA HS Assay Kit (Thermo Fisher Scientific, Waltham, MA, USA) was used for library quality assessment. The samples were then sequenced for 75 cycles of single-end data using a sequencer, and each library eventually produced 20 million reads. In this process, blood samples from healthy people at a concentration of 105 cells/mL were used as negative controls, and sterile deionized water was used as non-template controls [10].
BIOINFORMATICS ANALYSES FOR NGS:
To conduct the bioinformatics analyses for NGS, low-quality sequences and duplicate reads were removed using Trimmomatic [11]. Next, low-complexity reads were deleted using Kolmogorov complexity. Human sequences were identified and removed using Burrows–Wheeler Aligner software [12]. Third, the representative sequence assemblies of the corresponding microorganism were selected from the database. Finally, the list of pathogens was determined according to the Johns Hopkins ABX Guide [13] and the Manual of Clinical Microbiology [14]. We obtained a database containing about 13 000 genomes, and then used SNAP V1.0 Beta to compare microbial data to the database [15]. If the number of reads per million (RPM) of a microorganism was >5, it was considered positive. If the microorganism was also present in the non-template controls, its RPM was reduced accordingly.
CLINICAL ASSESSMENT:
An expert group composed of senior clinicians reviewed the NGS reports and traditional microbiology results. Combined with the clinical characteristics of the patient, pathogens were identified according to the following criteria: If the same microorganism was identified using the traditional detection methods and NGS, it was considered a pathogen. If the microorganism was identified using NGS alone, it was required to meet the following additional criteria in order to be considered a pathogen: (1) characteristic imaging changes were present; and (2) the sequence number of the microorganism was at least twice as high as that of other microorganisms [16].
STATISTICAL ANALYSIS:
SPSS version 21 (IBM Corp., Armonk, NY, USA) was used to analyze the data. Continuous variables were expressed as the mean±standard deviation if the data distribution was normal, or as the median, range, and interquartile range if the data had a non-normal distribution. Categorical variables were reported as counts and percentages. The Shapiro-Wilk test was used to test the normality and homogeneity of variance of continuous variables. The
Results
PATIENT CHARACTERISTICS:
A total of 90 patients with sepsis were included in the study, of whom 60 (67%) and 30 (33%) were in the ICO and ICH groups, respectively. The ICH group consisted of 3 patients who had undergone solid organ transplantation, 6 patients with hematologic malignancies, 20 patients on immunosuppressive treatment, and 1 patient with a solid tumor requiring chemotherapy. The mean APACHE II score was higher (21.76 vs 18.43; P=0.011), and the median white blood cell count was significantly lower (7.10×109 cells/L vs 10.68×109 cells/L; P=0.006) in the ICH group than in the ICO group. The mortality rates were 48% (29/60) and 80% (24/30) in the ICO and ICH groups, respectively (P=0.004). The ICH and ICO groups were also compared to assess differences in sex, age, disease type, or time spent in the EICU between the ICH and ICO groups, but there were no statistically significant differences in any of these characteristics between groups (Table 1).
COMPARISON OF DETECTION PERFORMANCE OF NGS AND TRADITIONAL DETECTION METHODS:
A total of 165 specimens were sent for testing using NGS, including 86 blood, 21 sputum, 45 BALF, 3 ascites, 4 urine, 5 cerebrospinal fluid (CSF), and 1 pleural effusion sample. A total of 239 samples were tested using traditional detection methods, including 90 blood, 90 sputum, 45 BALF, 3 ascites, 5 urine, 5 CSF, and 1 pleural effusion sample. Seventy-seven patients (86%) were positive for 1 or more pathogens using NGS, and 50 patients (56%) were positive using traditional detection methods (P<0.001). In both the ICH and ICO groups, the NGS-positivity rate was significantly higher than that of traditional detection methods (Figure 2). In the ICH and ICO groups, 47% (14/30) and 27% (16/60) of patients, respectively, were positive by NGS but negative by traditional detection methods (P=0.058). Three patients in the ICO group were negative using NGS but positive using traditional detection methods. The positivity rates of blood, sputum, and BALF using NGS were 55% (47/86), 90% (19/21), and 89% (40/45), respectively, and were significantly higher with NGS than with traditional detection methods (Figure 3A). Compared with blood samples, sputum and BALF samples showed significantly higher NGS-positivity rates; however, the difference in NGS-positivity rates between the sputum and BALF samples was not statistically significant (Figure 3B). The positivity rates of blood, sputum, and BALF using traditional detection methods were 14% (13/90), 38% (34/90), 22% (10/45), respectively. The difference in the positivity rates of sputum and blood was statistically significant; however, there was no significant difference between the positivity rates of sputum and BALF samples, or of blood and BALF samples (Figure 3B). In the current study, some rare pathogens, such as Cryptococcus neoformans, Chlamydia psittaci, Ureaplasma urealyticum, and Mycoplasma hominis, were only detected using NGS and no positive results were obtained using traditional detection methods (Figure 4).
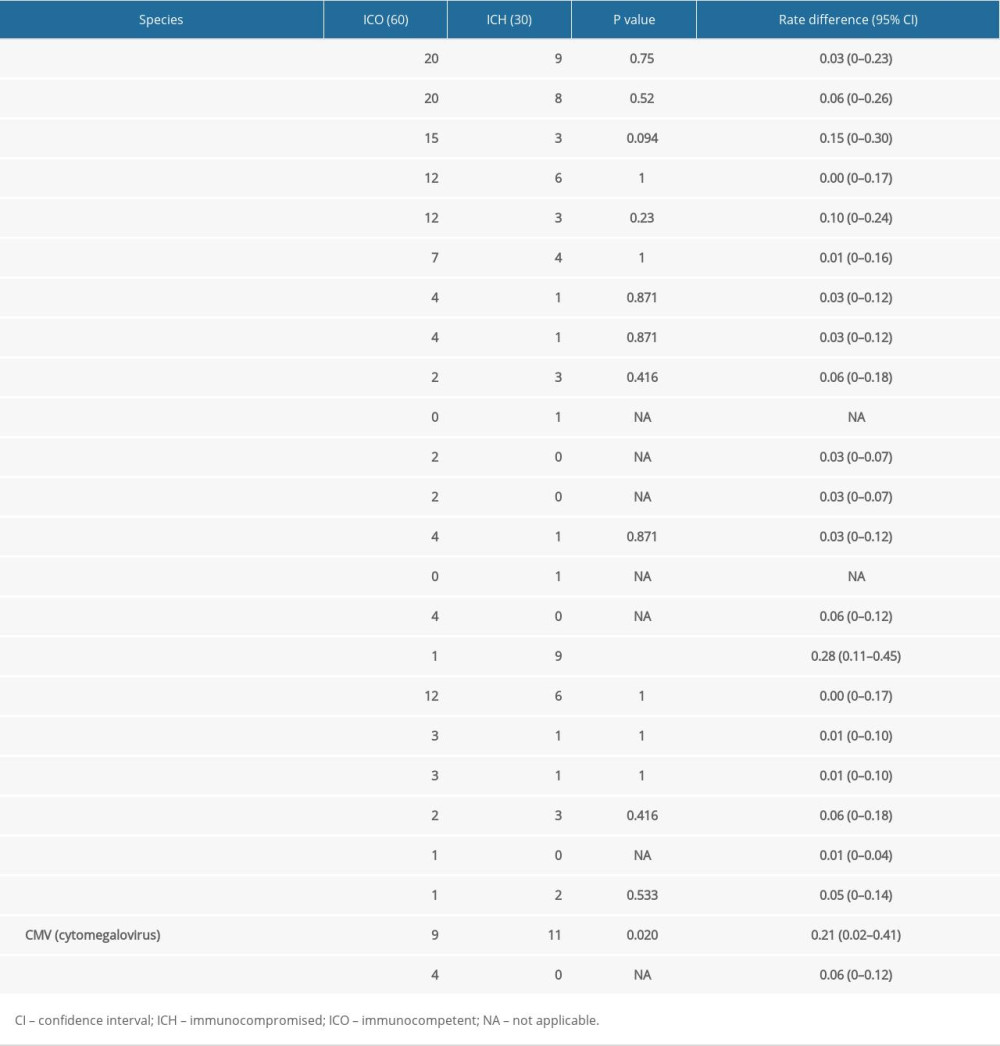
DIFFERENCES OF THE PATHOGENIC SPECTRUM BETWEEN IMMUNOCOMPROMISED AND IMMUNOCOMPETENT PATIENTS:
There were 60 patients with mixed infections of 2 or more pathogens, of which 56 (93%) were detected using NGS and 19 (32%) were detected using traditional detection methods (P<0.001). A total of 43/60 (72%) of the ICO group and 17/30 (57%) of the ICH group had mixed infections (P=0.155). The top 6 bacterial pathogens identified in the ICO group were Acinetobacter baumannii, Klebsiella pneumoniae, Escherichia coli, Pseudomonas aeruginosa, Candida albicans, and Enterococcus faecium. The top 6 bacterial pathogens in the ICH group were Acinetobacter baumannii, Klebsiella pneumoniae, Pseudomonas aeruginosa, Staphylococcus aureus, Pneumocystis jirovecii, and cytomegalovirus (CMV). The only significant difference in pathogen frequency observed between the ICH and ICO groups was Pneumocystis jirovecii infection – of the 10 cases of Pneumocystis jirovecii infection, 9 (90%) were in the patients in the ICH group (Figure 4, Table 2).
Discussion
This study showed that the NGS-positivity rate in both the ICH and the ICO groups was greater than that of traditional detection methods. Miao et al [17] reported that the sensitivity and specificity rates of NGS for the identification of pathogens in infectious diseases are 50.7% and 85.7%, respectively, which was better than that of traditional detection methods, consistent with our results. Although not all samples in this study were sent for NGS, the positivity rate was still higher than that of traditional detection methods, demonstrating the advantage of NGS. In our study, 3 patients in the ICO group had pathogens that were detected using traditional detection methods but not using NGS. This could be attributed to a low abundance of pathogenic microorganisms in the sample [18]. Moreover, NGS has unique advantages for detecting rare, difficult-to-culture pathogens. In this study,
Based on NGS, we assessed the positivity rates of various specimen types and discovered that the positivity rates of sputum and BALF samples were higher than those of blood samples; however, a significant difference in positivity rate was not observed between BALF and sputum samples, suggesting these are the best types of samples for NGS. Zhang et al [19] reported that in patients with pulmonary infection who had samples tested using NGS, BALF and sputum were the most effective pathogen detection sample types, with a positivity rate of 75%, while the positivity rate of blood samples was 35%. Another study found that sputum and BALF provided similar results in patients with respiratory infections [20], but that the sputum samples were more easily influenced than BALF by oral colonization bacteria, resulting in a higher false-positivity rate in sputum samples. NGS cannot distinguish between colonization and infection; therefore, if conditions permit, fiberoptic bronchoscopy should be actively performed and BALF should be the first choice for NGS testing. However, sputum is easier to obtain, which is more practical for patients with definite contraindications to bronchoscopy and hospitals without the ability to perform bronchoscopy.
Few studies have been conducted using NGS with regard to the pathogen spectrum among immunocompromised patients with sepsis. In the present study, we compared the pathogen spectrum of immunocompromised patients with that of the immunocompetent patients. The prevalence of
This study has some limitations. First, it was a single-center study with a small number of participants. Second, not all samples were sent for NGS, which may have reduced the NGS-positivity rate. Third, the patients included in this study were severely ill, and both groups had high APACHE II scores. Patients with high APACHE II scores have high mortality rates, thus reducing the potential positive impact of NGS on patients with sepsis. Fourth, NGS still lacks the ability to identify drug resistance. Research on drug resistance genes is ongoing, and it is likely that this limitation of NGS will be overcome in the future.
Conclusions
Patients with sepsis are more likely to have pathogens identified through NGS than by conventional detection methods, especially those who are immunocompromised. BALF is the optimum specimen type for NGS; however, sputum is a good second choice if it is not possible to obtain a BALF sample.
Figures
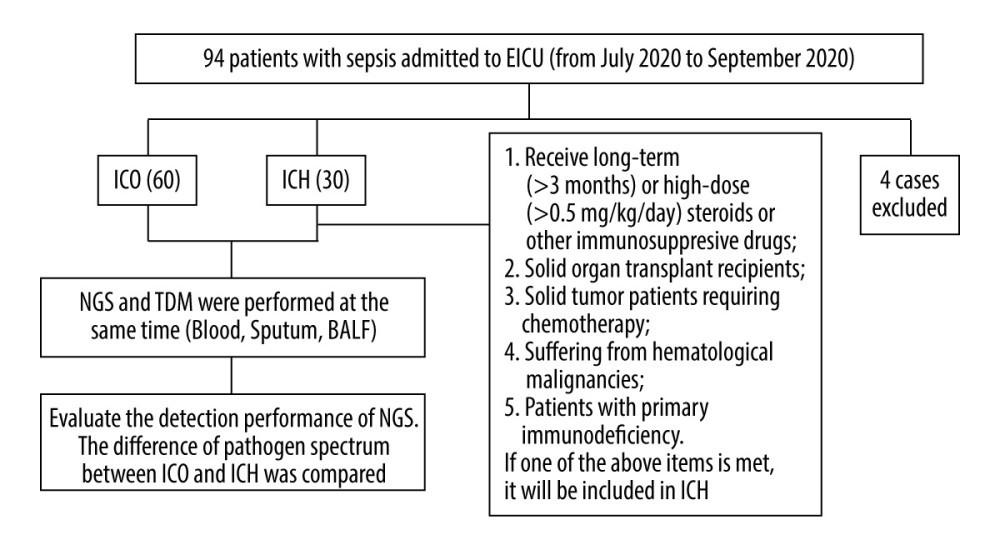 Figure 1. Research flow chart. BALF – bronchoalveolar lavage fluid; EICU – Emergency Intensive Care Unit; ICH – immunocompromised; ICO – immunocompetent; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA).
Figure 1. Research flow chart. BALF – bronchoalveolar lavage fluid; EICU – Emergency Intensive Care Unit; ICH – immunocompromised; ICO – immunocompetent; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA). 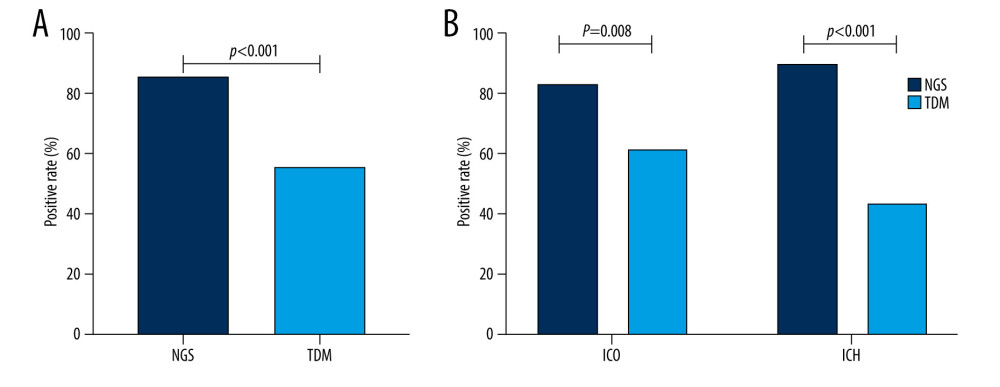 Figure 2. Comparison of the positivity rates of next-generation sequencing and traditional detection methods. (A) Comparison of the positivity rates of NGS and TDM in all enrolled patients. (B) Comparison of positivity rates of NGS and TDM according to patient immune status. ICH – immunocompromised; ICO – immunocompetent; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software San Diego, CA, USA).
Figure 2. Comparison of the positivity rates of next-generation sequencing and traditional detection methods. (A) Comparison of the positivity rates of NGS and TDM in all enrolled patients. (B) Comparison of positivity rates of NGS and TDM according to patient immune status. ICH – immunocompromised; ICO – immunocompetent; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software San Diego, CA, USA). 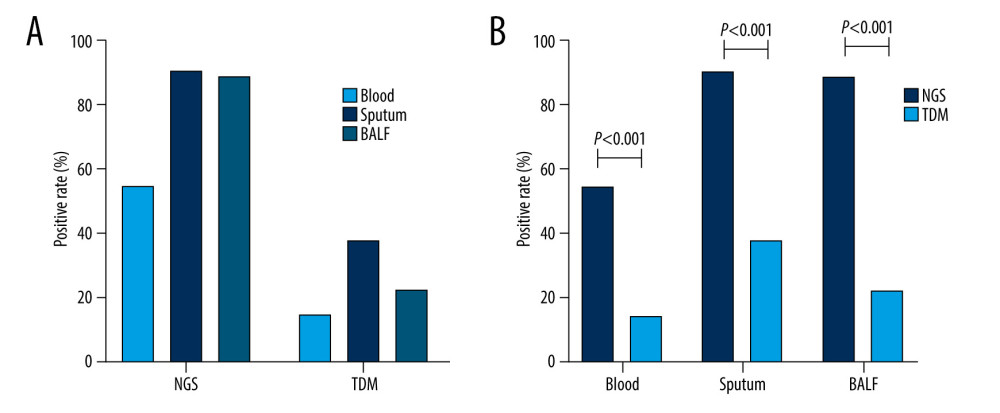 Figure 3. Comparison of the positivity rates of pathogen identification according to the detection method and the sample type. (A) Positivity rates according to the testing method. (B) Positivity rates according to the sample type. BALF – bronchoalveolar lavage fluid; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA).
Figure 3. Comparison of the positivity rates of pathogen identification according to the detection method and the sample type. (A) Positivity rates according to the testing method. (B) Positivity rates according to the sample type. BALF – bronchoalveolar lavage fluid; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA). 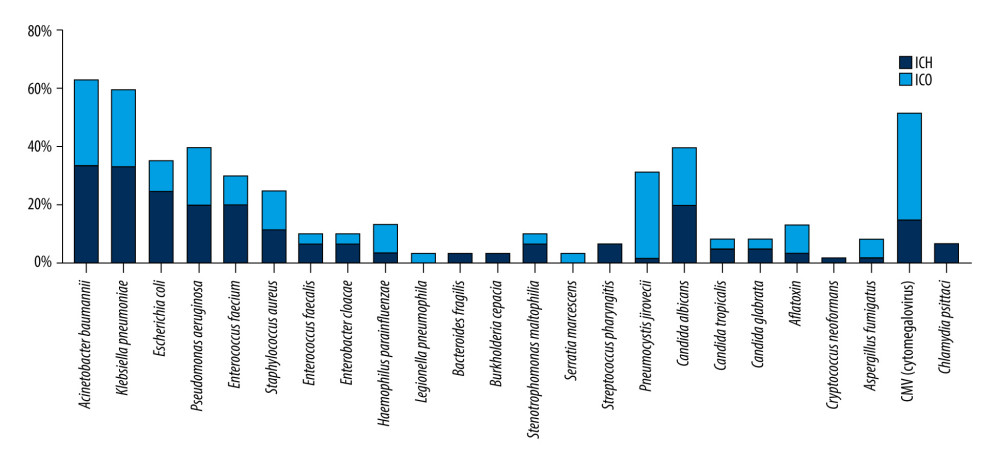 Figure 4. Frequency distribution of pathogens identified according to patient immune status. ICH – immunocompromised; ICO – immunocompetent. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA).
Figure 4. Frequency distribution of pathogens identified according to patient immune status. ICH – immunocompromised; ICO – immunocompetent. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA). References
1. Singer M, Deutschman CS, Seymour CW, The third international consensus definitions for sepsis and septic shock (Sepsis-3): JAMA, 2016; 315; 801-10
2. Fleischmann C, Scherag A, Adhikari NK, Assessment of global incidence and mortality of hospital-treated sepsis. Current estimates and limitations: Am J Respir Crit Care Med, 2016; 193; 259-72
3. Paul M, Shani V, Muchtar E, Kariv G, Systematic review and meta-analysis of the efficacy of appropriate empiric antibiotic therapy for sepsis: Antimicrob Agents Chemother, 2010; 54; 4851-63
4. Lee JH, Kim YH, Predictive factors of true bacteremia and the clinical utility of blood cultures as a prognostic tool in patients with community-onset pneumonia: Medicine (Baltimore), 2016; 95; e5058
5. McCreery RJ, Florescu DF, Kalil AC, Sepsis in immunocompromised patients without human immunodeficiency virus: J Infect Dis, 2020; 222(Suppl 2); S156-65
6. Bolger AM, Lohse M, Usadel B, Trimmomatic: A flexible trimmer for Illumina sequence data: Bioinformatics, 2014; 30; 2114-20
7. Lindell RB, Nishisaki A, Weiss SL, Risk of mortality in immunocompromised children with severe sepsis and septic shock: Crit Care Med, 2020; 48; 1026-33
8. Amar Y, Lagkouvardos I, Silva RL, Pre-digest of unprotected DNA by benzonase improves the representation of living skin bacteria and efficiently depletes host DNA: Microbiome, 2021; 9; 123
9. Miller S, Naccache SN, Samayoa E, Laboratory validation of a clinical metagenomic sequencing assay for pathogen detection in cerebrospinal fluid: Genome Res, 2019; 29; 831-42
10. Li H, Gao H, Meng H, Detection of pulmonary infectious pathogens from lung biopsy tissues by metagenomic next-generation sequencing: Front Cell Infect Microbiol, 2018; 8; 205
11. Xu Y, Kang L, Shen Z, Dynamics of severe acute respiratory syndrome coronavirus 2 genome variants in the feces during convalescence: J Genet Genomics, 2020; 47; 610-17
12. Li H, Durbin R, Fast and accurate short read alignment with Burrows-Wheeler transform: Bioinformatics, 2009; 25; 1754-60
13. Johns Hopkins University: The Johns Hopkins POC-IT ABX guide, 2017, Maryland https://www.hopkinsmedicine.org/apps/all-apps/johns-hopkins-abx-guide-2017
14. Miller JM, Binnicker MJ, Campbell S, A guide to utilization of the microbiology laboratory for diagnosis of infectious diseases: 2018 update by the Infectious Diseases Society of America and the American Society for Microbiology: Clin Infect Dis, 2018; 67; e1-e94
15. O’Connor TP, SNAP assay technology: Top Companion Anim Med, 2015; 30; 132-38
16. Piantadosi A, Mukerji SS, Ye S, Enhanced virus detection and metagenomic sequencing in patients with meningitis and encephalitis: mBio, 2021; 12; e0114321
17. Miao Q, Ma Y, Wang Q, Microbiological diagnostic performance of metagenomic next-generation sequencing when applied to clinical practice: Clin Infect Dis, 2018; 67(Suppl, 2); S231-40
18. Chiu CY, Miller SA, Clinical metagenomics: Nat Rev Genet, 2019; 20; 341-55
19. Zhang Y, Xu T, He F, Clinical evaluation of a metagenomics-based assay for pneumonia management: Front Microbiol, 2021; 12; 751073
20. Kalantar KL, Moazed F, Christenson SC, Metagenomic comparison of tracheal aspirate and mini-bronchial alveolar lavage for assessment of respiratory microbiota: Am J Physiol Lung Cell Mol Physiol, 2019; 316; L578-84
21. Langelier C, Zinter MS, Kalantar K, Metagenomic sequencing detects respiratory pathogens in hematopoietic cellular transplant patients: Am J Respir Crit Care Med, 2018; 197; 524-28
22. Ponce CA, Gallo M, Bustamante R, Vargas SL, Pneumocystis colonization is highly prevalent in the autopsied lungs of the general population: Clin Infect Dis, 2010; 50; 347-53
23. Stern A, Green H, Paul M, Vidal L, Leibovici L, Prophylaxis for Pneumocystis pneumonia (PCP) in non-HIV immunocompromised patients: Cochrane Database Syst Rev, 2014; 10; CD005590
24. Zhang Y, Ai JW, Cui P, Zhang WH, A cluster of cases of Pneumocystis pneumonia identified by shotgun metagenomics approach: J Infect, 2019; 78; 158-69
Figures
 Figure 1. Research flow chart. BALF – bronchoalveolar lavage fluid; EICU – Emergency Intensive Care Unit; ICH – immunocompromised; ICO – immunocompetent; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA).
Figure 1. Research flow chart. BALF – bronchoalveolar lavage fluid; EICU – Emergency Intensive Care Unit; ICH – immunocompromised; ICO – immunocompetent; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA). Figure 2. Comparison of the positivity rates of next-generation sequencing and traditional detection methods. (A) Comparison of the positivity rates of NGS and TDM in all enrolled patients. (B) Comparison of positivity rates of NGS and TDM according to patient immune status. ICH – immunocompromised; ICO – immunocompetent; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software San Diego, CA, USA).
Figure 2. Comparison of the positivity rates of next-generation sequencing and traditional detection methods. (A) Comparison of the positivity rates of NGS and TDM in all enrolled patients. (B) Comparison of positivity rates of NGS and TDM according to patient immune status. ICH – immunocompromised; ICO – immunocompetent; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software San Diego, CA, USA). Figure 3. Comparison of the positivity rates of pathogen identification according to the detection method and the sample type. (A) Positivity rates according to the testing method. (B) Positivity rates according to the sample type. BALF – bronchoalveolar lavage fluid; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA).
Figure 3. Comparison of the positivity rates of pathogen identification according to the detection method and the sample type. (A) Positivity rates according to the testing method. (B) Positivity rates according to the sample type. BALF – bronchoalveolar lavage fluid; NGS – next-generation sequencing; TDM – traditional detection methods. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA). Figure 4. Frequency distribution of pathogens identified according to patient immune status. ICH – immunocompromised; ICO – immunocompetent. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA).
Figure 4. Frequency distribution of pathogens identified according to patient immune status. ICH – immunocompromised; ICO – immunocompetent. The figure was created by GraphPad Prism version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387