09 July 2022: Meta-Analysis
Urodynamics in Early Diagnosis of Diabetic Bladder Dysfunction in Women: A Systematic Review and Meta-Analysis
Ágnes Rita MartonosiDOI: 10.12659/MSM.937166
Med Sci Monit 2022; 28:e937166
Abstract
BACKGROUND: Urodynamics can detect subtle voiding changes before cystopathy symptoms manifest. The aim of the present study was to assess urodynamic changes in diabetic women.
MATERIAL AND METHODS: A systematic search was performed on 04 November 2021 to identify studies reporting urodynamic parameters in diabetic women. Data were analyzed in a single-arm meta-analysis due to lack of sufficient studies with direct comparisons to healthy women. For data synthesis, a random-effects model with restricted maximum-likelihood estimation was applied. The calculated effect sizes were visualized in forest plots. Statistical heterogeneity was assessed using the I² measure and the χ² test. The risk of bias was assessed using the QUIPS tool. PROSPERO ID: CRD42021256275.
RESULTS: Out of 1750 records, 10 studies were used in the analysis (n=2342 diabetic women). Pooled event rates showed that mean voided volume was 288.21 mL [95% confidence interval (CI): 217.35–359.06, I²=98%], mean postvoid residual volume was 93.67 mL [95% CI: 31.35-155.99, I²=100%], mean Qmax was 18.80 mL/sec [95% CI: 15.27-22.33, I²=99%], mean PdetQmax is 30.13 cmH2O [95% CI: 25.53–34.73, I²=90%], mean first sensation of bladder filling was 178.66 mL [95% CI: 150.59-206.72, I²=97%], and mean cystometric capacity was 480.41 mL [95% CI: 409.32-551.50, I²=98%] in diabetic women.
CONCLUSIONS: Pooled results indicate that diabetic women tend to have a smaller voided volume, slower Qmax and PdetQmax, larger postvoid residual, and higher first sensation of bladder filling and cystometric capacity compared to the general female population.
Keywords: Diabetes Complications, Diabetes Mellitus, Diabetic Neuropathies, Meta-Analysis, systematic review, Urodynamics, Urology, Early Diagnosis, Female, Humans, urinary bladder, Urination
Background
Diabetic cystopathy (DC) is a well-recognized urological complication of diabetic autonomic neuropathy [1]. The classic triad of DC is decreased bladder sensation, increased bladder capacity, and impaired bladder emptying with postvoid residual volume [2–6]. Patients usually have overactive bladder or overflow incontinence, including urinary frequency, urgency, incontinence, and nocturia, which are listed among the lower urinary tract symptoms (LUTS) [7,8].
DC gradually progresses over time from an initial compensated [typically manifests as overactive bladder (OAB)] to a later decompensated [typically manifests as underactive bladder (UB)] phase that cause storage or voiding LUTS [9,10].
Uroflowmetry is a simple urodynamic diagnostic tool to measure voided volume and maximum flow rate. Cystometry measures pressure/volume relationship of bladder during the filling (storage) phase of the micturition cycle via a catheter. As urodynamics can detect subtle voiding changes even before the manifestation of LUTS, urodynamic evaluation can be useful in the early diagnosis of DC.
The normative reference values for bladder function by urodynamics in healthy women have been described earlier [11,12], but the literature on diabetic women is scant. Therefore, our aim was to assess urodynamic alterations in diabetic women.
Material and Methods
STUDY PROTOCOL:
The study was in line with the protocol registered on 23 May 2021, and can be found under the registration number: CRD42021256275 in PROSPERO. A review protocol was not prepared.
Our primary aim was to compare the urodynamic parameters of diabetic women to those of healthy women, but the eligible studies according to our inclusion criteria did not provide sufficient raw data on healthy women. Since a direct comparison could not be implemented in diabetic and non-diabetic patients, we conducted a single-arm meta-analysis and positive event rates were pooled for statistical analysis. Furthermore, our intent was to detect changes in voiding in diabetic women with cystopathy compared to diabetic women without peripheral neuropathy by uroflowmetry, but the eligible studies did not contain sufficient raw data to conduct the analysis. Otherwise, we fully adhered to the study protocol.
SYSTEMATIC SEARCH:
Two review authors (ÁRM and PP) independently carried out the systematic literature search in Embase, MEDLINE (via PubMed), the Cochrane Central Register of Controlled Trials (CENTRAL), and the Web of Science. The authors applied the following keywords: (urodynamic or uroflow* or uroflowmetry) and (diabetes or diab*) (Supplementary Table 1) on 04 November 2021. In each database, they applied “All text” or “All fields” in the search bar avoiding any filters or restrictions regarding publication year, language, and place of origin. They reviewed the included studies to find any articles previously missed in the original search.
SELECTION AND ELIGIBILITY:
The authors included case-control and cohort studies, as well as full-text articles and conference abstracts in the synthesis of quantitative and qualitative data to reduce selection bias. Case reports featuring single patients were excluded. In case of potentially overlapping study populations (based on authors, sites, patients and urodynamic parameters), those with better quality of data were included.
Eligible studies had to provide data on diabetic women, with or without voiding disorder, and urodynamic parameters [14], which include uroflowmetry and cystometry parameters [15,16]. Regardless of the method of measurement (with ultrasonography or via catheter), postvoid residual volume (in mL) was also included in the analysis. Studies that included both sexes were included if they performed subgroup analysis for diabetic women.
Articles examining patients after kidney transplantation or surgery affecting the genitourinary tract, patients with neurogenic bladder dysfunction and other neurological disease (eg, progressive neurological conditions such as Parkinson’s disease, dementia, multiple sclerosis) except peripheral neuropathy; pregnant women and women within first 6 months postpartum, end-stage kidney disease, or kidney transplants were excluded, since they can influence urodynamic parameters per se, causing indistinguishable confounding factors.
The records were selected via a standard three-phased process including titles, abstracts, and full-texts independently by the 2 review authors (ÁRM and PP) with EndNote X9.1.1 software (2020 Clarivate™ Analytics, Philadelphia, PA, USA). They resolved any disagreements in any phase through consensus, and calculated the Cohen’s kappa in each phase to test inter-rater reliability.
DATA EXTRACTION:
The 2 independent review authors (ÁRM and PP) extracted the data into a purpose-designed data collection table, with any disagreements resolved by consensus. The following data were extracted from each study: 1) study information (first author, year of publication, recruitment period, country of origin), 2) study design and methodology (retrospective versus prospective, inclusion and exclusion criteria, single versus multicenter study), 3) patient information (number of patients, mean age, definition of diabetes, type of diabetes, diabetes duration, body mass index (BMI), HgA1c values, diabetes treatment, diabetic complication), 4) LUTS assessment and symptoms, DC and peripheral neuropathy existence, and 5) urodynamic parameters of diabetic patients (voided volume, postvoid residual, Qmax, PdetQmax, volume at first sensation, maximal cystometry capacity).
STATISTICAL ANALYSIS:
Cohen’s kappa (κ) was calculated to test interrater reliability. It ranges from −1 to +1, where values ≤0 indicates no agreement (which are unlikely in practice), 0 represents the amount of agreement that can be expected from random chance, and 1 represents perfect agreement between the review authors. κ results should be interpreted as the followings: values ≤0 as indicating “no agreement”, 0.01–0.20 as “none to slight”, 0.21–0.40 as “fair”, 0.41–0.60 as “moderate,” 0.61–0.80 as “substantial”, and 0.81–1.00 as “almost perfect” agreement [17].
For data synthesis we used the random-effects model with restricted maximum-likelihood (REML) estimation in all cases; means and 95% confidence intervals (CIs) were calculated. The calculated effect sizes were visualized in forest plots. Heterogeneity was tested using Cochrane’s Q and the I2 statistics. I2 statistic represents the percentage of the total variability across studies: 30% to 60%, 50% to 90%, and 75% to 100% corresponded to “moderate”, “substantial”, and “considerable” degrees of heterogeneity, respectively, based on the Cochrane’s handbook for Systematic Reviews of Interventions [18]. We considered the Q test significant if P<0.1. Statistical analyses were carried out using R statistical software (version 4.0.5) and package meta (version 4.18-1). We created a single-arm meta-analysis based on urodynamic parameters and the results are graphically presented in forest plots.
RISK OF BIAS ASSESSMENT:
The risk of bias of the studies were evaluated by 2 independent review authors (ÁRM and PP) using the Quality In Prognostic Studies (QUIPS) tool [19]. The result of the assessment was graphically demonstrated; any disagreements were resolved by consensus among the review authors.
To judge overall risk, the review authors (ÁRM and PP) described studies with a low risk of bias as those in which at least 5 of the 6 important bias domains (study participants, study attrition, prognostic factor measurement, outcome measurement, study confounding, and statistical analysis) were rated as having low risk of bias. If there was at least 1 domain rated as high risk, or more than 3 domains rated as moderate risk of bias, the overall risk of bias was deemed high. All other variations were determined as moderate risk of bias.
When the study design could not be identified, study attrition was not evaluated and was thus labeled as ‘grey’. Grey means not applicable or not reported.
There were not enough studies in the analyses to evaluate publication bias by funnel plots and statistical tests.
Results
STUDY SELECTION:
Out of 1750 records (MEDLINE, n=454; Embase, n=773; CENTRAL, n=63; and Web of Science, n=460), a total of 140 articles were assessed for eligibility by full text, of which 10 studies [20–29] were used in the quantitative synthesis. κ of the title selection was 0.99 (99.7% agreement), 0.98 (99.3% agreement) of the abstract selection, and 0.99 (99.8% agreement) of the full-text selection. The flowchart and reasons for exclusions on full-text assessment is illustrated in Figure 1.
STUDY CHARACTERISTICS:
The baseline characteristics of the included studies are reported in Table 1. Studies took place in 9 different countries, and were published between 2002 and 2020. Six studies were prospective [20,21,25,26,28,29], 2 were retrospective cohorts [23,27], and 2 studies did not provide sufficient information about study design [22,24].
GENERAL CHARACTERISTICS OF DIABETIC WOMEN:
We included 10 studies to the quantitative synthesis that reported on a total of 2342 diabetic patients, including 2055 patients (87.7%) with LUTS. The majority of the patients had type 2 diabetes. In 7 studies, the type of diabetes was reported [20–24,28,29], while in 3 studies [25–27] it was not, although they had small number of patients. The mean age of the study populations ranged between 52.75±9.2 and 64.7±11.1 years, the mean duration of diabetes ranged between 8.04±0.69 and 12.42±7.3 years, the mean BMI ranged between 22.8±2.4 and 33.2±7.8 kg/m2, and the mean HgA1c ranged between 6.05±2.38 and 9.1±2.6%. The demographic characteristics of diabetic patients are presented in Table 2.
URODYNAMIC PARAMETERS OF DIABETIC WOMEN:
The urodynamic parameters of the diabetic women were assessed qualitatively. These data are represented in Table 3.
VOIDED VOLUME: The pooled event rates show that mean voided volume in diabetic women (n = 471) was 288.21 mL [95% CI: 217.35–359.06] with a considerable level of heterogeneity (I2=98%) (Figure 2 A).
POSTVOID RESIDUAL VOLUME: The pooled event rate represents that mean postvoid residual volume in diabetic women (n=1589) was 93.67 mL [95% CI: 31.35–155.99] with a considerable level of heterogeneity (I2=100%) (Figure 2B).
: The mean Qmax in diabetic women (n=1620) was 18.80 mL/sec [95% CI: 15.27–22.33] with a considerable level of heterogeneity (I2=99%) (Figure 2C).
: The mean PdetQmax in diabetic women (n=1211) was 30.13 cmH2O [95% CI: 25.53–34.73] with a considerable level of heterogeneity (I2=90%) (Figure 2D).
FIRST SENSATION OF BLADDER FILLING: The mean first sensation of bladder filling in diabetics (n=1201) was 178.66 mL [95% CI: 150.59–206.72] with a considerable level of heterogeneity (I2=97%) (Figure 2E).
CYSTOMETRIC CAPACITY: Th mean maximum cystometric capacity (MCC) in diabetic women (n=1178) was 480.41 mL [95% CI: 409.32–551.50] with a considerable level of heterogeneity (I2 = 98%) (Figure 2F).
RISK OF BIAS ASSESSMENT:
A summary of the risk of bias assessment is visually presented in Supplementary Figure 1.
In the analysis of female diabetic patients’ urodynamic parameters, the majority of the studies had a high overall risk of bias [20,22–27,29]. The main reasons include confounding factors, such as unreported 1) diabetes definition, 2) diabetes duration, 3) HgA1c, 4) diabetes treatment, 5) BMI, 6) LUTS assessment, 7) device and methodology of the uroflowmetry measurement, 8) measuring method of postvoid residual volume, and 9) statistical analysis. Existence of pyuria was a confounding factor as well. One study had moderate overall risk of bias because not all patients were included in the urodynamic analysis, and an assessed confounding factor was that BMI was not reported [21]. One study was reported as having low overall risk of bias [28].
Discussion
SUMMARY OF EVIDENCE:
Lower mean voided volume, Qmax and PdetQmax, as well as higher mean postvoid residual volume, first sensation of bladder filling, and cystometric capacity in the diabetic group was detected compared to healthy women [11,12].
VOIDED VOLUME: Lower voided volume in diabetes is a surprising finding, because higher voided volumes are generally expected in diabetic patients with higher fluid turnover; and even much higher volumes in autonomic neuropathy [1,5,30]. However, in diabetic patients, according to the literature [31,32], the residual urine is larger as the autonomic neuropathic bladder cannot completely empty the bladder, and there will be residue in it. On the other hand, if we add the emptied amount and the residue volume, there is a clearly higher bladder capacity in diabetic patients [31]. This phenomenon could be explained by the selection of different stages and duration of diabetes. This clinical heterogeneity is also indicated by the statistical heterogeneity.
: Qmax is always lower in patients with an autonomic neuropathic bladder [33] due to impaired detrusor muscle function, which agrees with our findings. A smaller voided volume always has a lower Qmax [34,35], but in this case the bladder also contains residual volume, and this should be taken into account during contraction, so the value is even worse.
At maximum flow, the detrusor pressure (PdetQmax) is lower in diabetes due to the dysfunction and abnormal innervation.
Lee at al [24] reports lower Qmax in the diabetic group without bladder dysfunction (n=135, Qmax: 19.9±0.7 mL/s), which is even more profound with bladder dysfunction (n=47, Qmax: 15.2±1.2 mL/s) compared to healthy women (n=197, Qmax: 25.8±8.4 mL/s).
POSTVOID RESIDUAL VOLUME, FIRST SENSATION OF BLADDER FILLING, AND CYSTOMETRIC CAPACITY: Autonomic neuropathy reduces the sensation of the bladder [36–38], so it is well understood that the onset of the first urge to urinate in diabetic patients occurs at higher bladder volumes.
Bladder capacity is always higher during cystometry and is markedly higher in diabetic patients [36,39].
Lee at al [24] reported that 1.6% of diabetic women had bladder capacity >500 mL and 0% in the healthy control group; 25.8% of patients had voiding dysfunction in the diabetic group and 3.5% in the healthy population; and 14.8% of diabetic patients and 1.5% of healthy people have postvoid residual >100 mL.
NON-DIABETIC WOMEN:
Haylen et al [34] found that Qmax and Qave are strongly related to voided volume. Afraa et al [40] found Qmax values ranging between 23 and 32 mL. Wyman et al [12] included 3090 healthy women ranging in age from 19 to 91 years from 24 studies in their meta-analysis of normative reference values for bladder function parameters. They found 334 mL [95% CI: 299–350] for mean voided volume, 12 mL [95% CI: 4–20] for mean postvoid residual volume, and 28 mL/s [95% CI: 27–30] for mean Qmax. Sorel et al [11] included 1416 adult patients to their systematic review. They found 338 mL [SD: 161] for mean voided volume, 23.5 mL [SD: 10] for mean Qmax, and 15.5 mL [SD: 25] for mean postvoid residual volume. Mahfouz et al [41] found 175 mL for first sensation of bladder filling, and normal maximum cystometric capacity of 300–500 mL.
These findings agree with our results.
DIABETIC ADULTS WITH AND WITHOUT LUTS:
As diabetes progresses, LUTS also appears [36,42] which suggests more pronounced bladder damage, so it is understandable that in diabetes with LUTS, higher voided volumes suggest greater bladder capacity compared to diabetics without LUTS. The same more pronounced damage is indicated by increased detrusor muscle weakness in diabetic patients with LUTS compared to those without voiding symptoms. Higher detrusor pressure at maximum flow only fits into this pattern if it indicates a LUTS outflow disturbance. The rate of residual urine is higher in diabetics who also have LUTS.
NON-INVASIVE URODYNAMIC MEASUREMENTS IN ROUTINE DIABETES FOLLOW-UPS:
Translational research takes scientific discoveries made in the laboratory and transforms them into new treatments [43]. Therefore, the sooner the early signs of DC are discovered, the earlier the therapeutic modifications can be initiated. Uroflowmetry can highlight the progressive nature of diabetes – starting with storage changes, then developing voiding dysfunction due to detrusor overdistension, to the decompensated phase. As early alterations in voiding patterns can be seen during the urodynamic examination before bothersome urinary symptoms are recognized by patients, urodynamics, mostly uroflowmetry, can contribute to early diagnosis of DC. Therefore, the inclusion of routine uroflow measurements to the current guidelines of diabetes management is crucial.
Although progression of DC is believed be related to the duration of diabetes, and poor metabolic status; animal studies raised the question of whether changes in bladder function begin soon after its onset [43,44]. Previous studies have suggested that DC is not the prime urodynamic finding in diabetics. Kaplan et al [6] found that detrusor overactivity was the most common finding. Kebapci et al [45] came to the conclusion that classic CD occurs in only 44% of women with type 2 diabetes followed for a mean of 13.85 years; more common findings are detrusor overactivity, stress, and urge incontinence.
Although, summary mean estimates of bladder function parameters for diabetic women were calculated, heterogeneity between the studies was high for all outcomes. Therefore, this precludes generalization of these estimates to all diabetic women. Further research is needed to determine reference values within specific subgroups.
STRENGTH OF THE STUDY AND LIMITATIONS:
To the best of our knowledge, this is the first meta-analysis that synthetizes quantitative data about urodynamic measurements of female patients with diabetes. Nonetheless, the strength of our meta-analysis is the use of a comprehensive and precise search strategy and data extraction.
The main limitation is that we could not directly compare diabetic and non-diabetic women, since there were insufficient studies directly comparing diabetic and non-diabetic patients. The lack of definition of diabetic cystopathy is also a limitation, and only 2 studies reported it [24,29].
Conclusions
IMPLICATION FOR PRACTICE:
Diabetes is an important independent risk factor for LUTS. Urodynamics can detect early alterations in voiding function, which might help to apply interventions to delay or prevent the onset of diabetes to limit difficulties in voiding.
Uroflowmetry may be considered in current diabetes guidelines. Regular uroflow measurements can contribute to the early recognition of DC.
IMPLICATION FOR RESEARCH:
Due to the limitations, our findings should be verified by future comparative studies in people with diabetes. To carry out more accurate analyses, it is important to compare larger number of patients with different stages and duration of diabetes, with different metabolic status as well.
QUIPS Tool – Risk of Bias Assessment
QUIPS assess risk of bias in studies of prognostic factors.
Concerns about risk of bias and applicability were rated as “low”, “moderate” or “high” in six domains 1) study participation, 2) study attrition, 3) prognostic factor measurement, 4)outcome measurement, 5) study confounding, as well as 6) statistical analysis and reporting.
To judge overall risk, the review authors (ÁRM and PP) described studies with a low risk of bias as those in which at least five of the six important bias domains were rated as having low risk of bias. If there was at least one domain rated as high risk, or more than three domains rated as moderate risk of bias, the overall risk of bias was deemed high. All other variations were determined as moderate risk of bias.
Abbreviations: QUIPS – quality in prognostic studies.
Figures
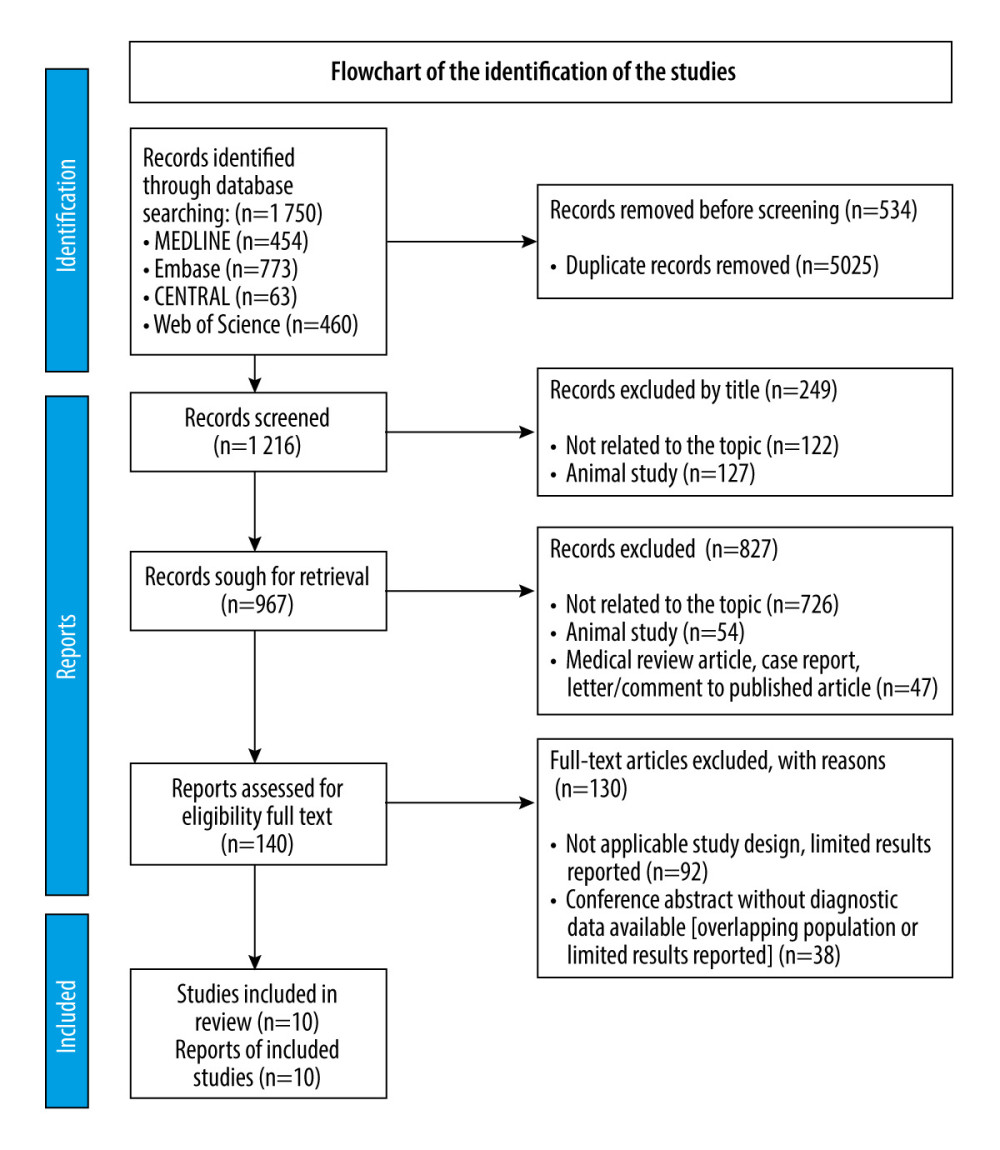 Figure 1. PRISMA flow diagram of the selection of the studies. The algorithm of the study selection; out of the 1750 records, 10 full-text articles were used in the final analysis. The figure was created using Microsoft® Word (version 16.54; 2019).
Figure 1. PRISMA flow diagram of the selection of the studies. The algorithm of the study selection; out of the 1750 records, 10 full-text articles were used in the final analysis. The figure was created using Microsoft® Word (version 16.54; 2019). 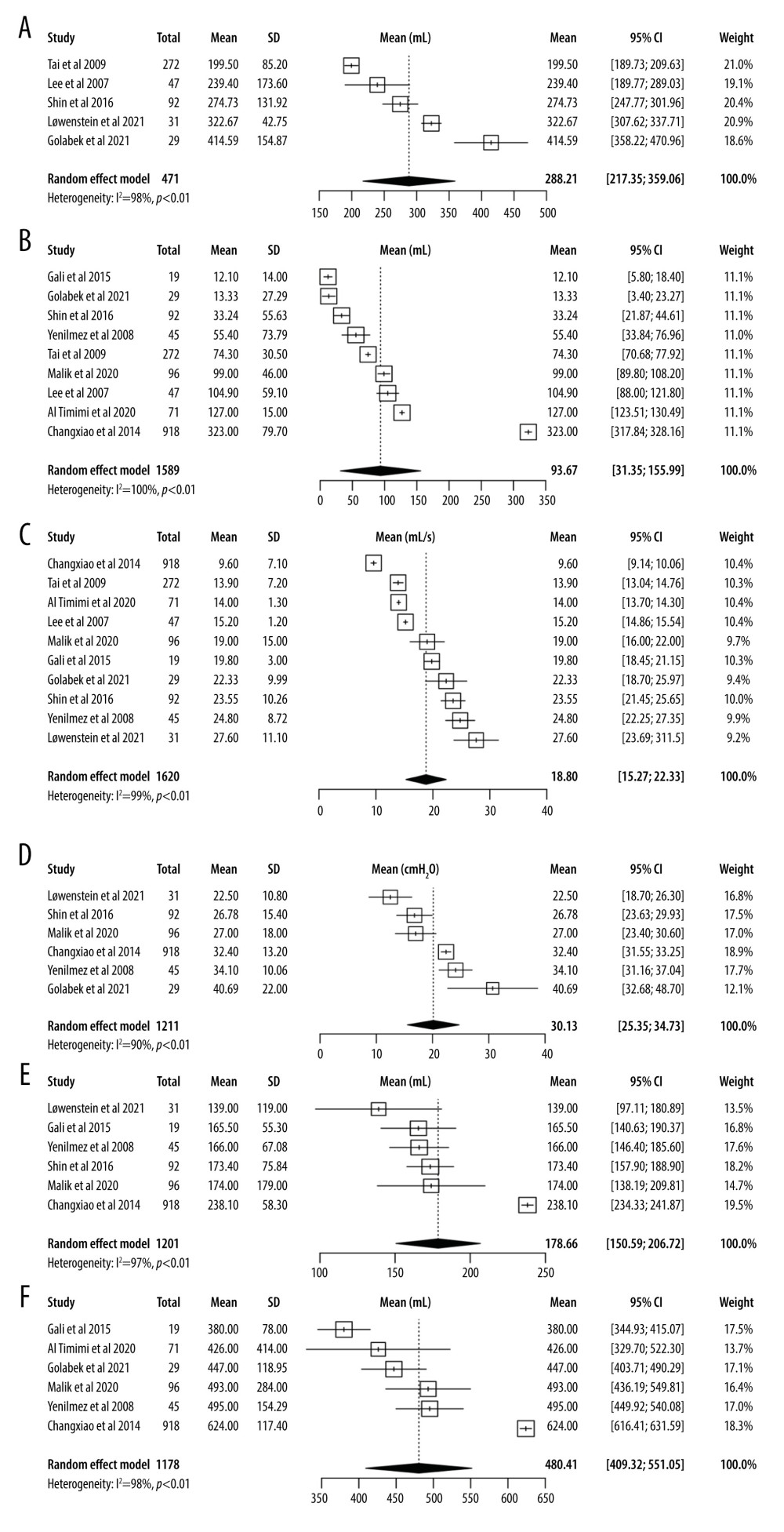 Figure 2. Forest plots of the urodynamic parameters of diabetic women. (A) Represents the pooled mean voided volume of diabetic women (n=471). (B) Shows the pooled mean postvoid residual volume of diabetic women (n=1589). (C) Demonstrates the pooled mean Qmax values in diabetic women (n=1620). (D) Presents the pooled mean PdetQmax values in diabetic women (n=1211). (E) Reveals the pooled mean first sensation of bladder filling in diabetic women (n=1201). (F) Unveils the pooled mean maximum cystometric capacity (MCC) in diabetic women (n=1178). Statistical analyses were carried out using R statistical software (version 4.0.5) and package “meta” (version 4.18-1).
Figure 2. Forest plots of the urodynamic parameters of diabetic women. (A) Represents the pooled mean voided volume of diabetic women (n=471). (B) Shows the pooled mean postvoid residual volume of diabetic women (n=1589). (C) Demonstrates the pooled mean Qmax values in diabetic women (n=1620). (D) Presents the pooled mean PdetQmax values in diabetic women (n=1211). (E) Reveals the pooled mean first sensation of bladder filling in diabetic women (n=1201). (F) Unveils the pooled mean maximum cystometric capacity (MCC) in diabetic women (n=1178). Statistical analyses were carried out using R statistical software (version 4.0.5) and package “meta” (version 4.18-1). Tables
Table 1. Baseline characteristics of the included studies.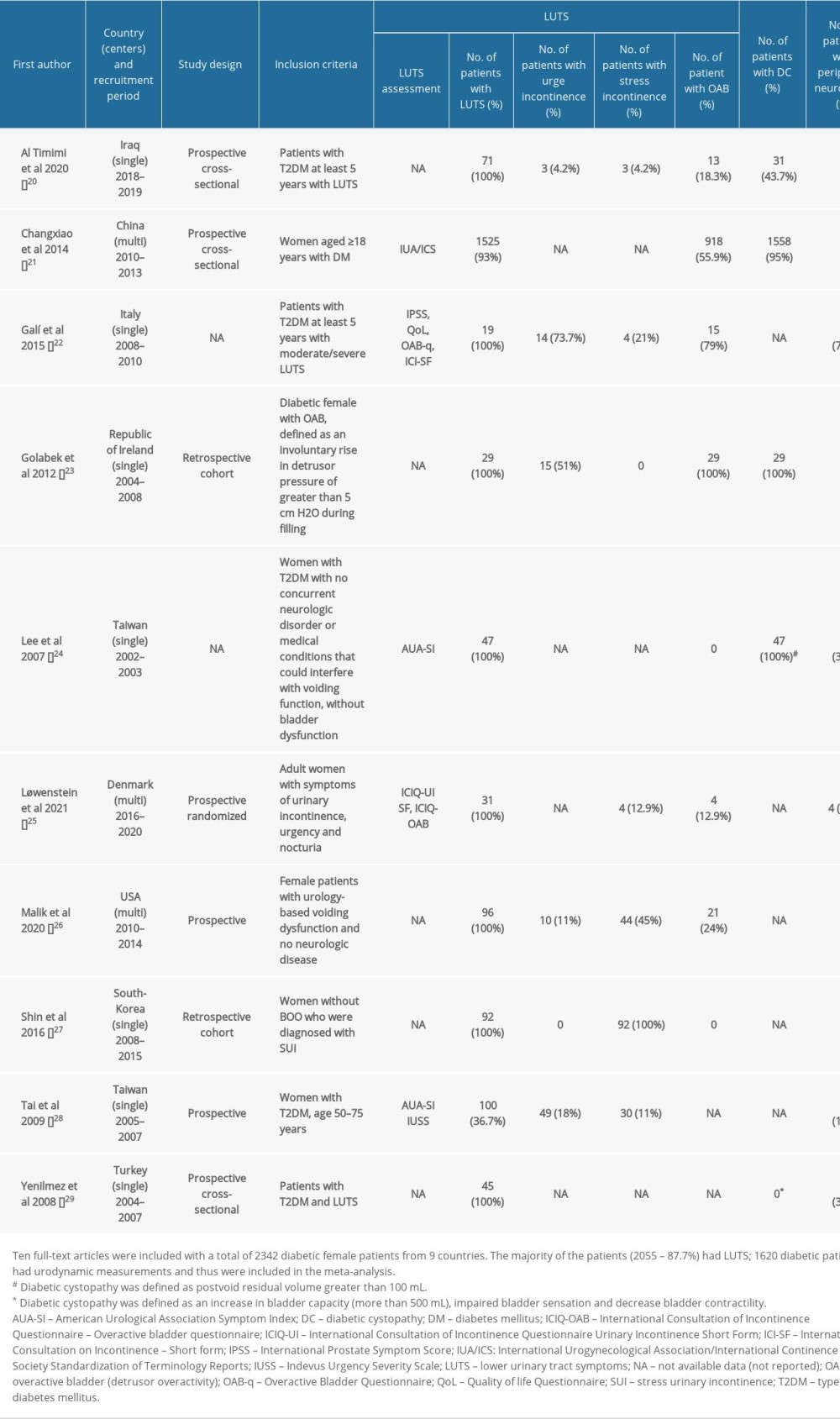 Table 2. Demographic characteristics of women with diabetes of the included studies.
Table 2. Demographic characteristics of women with diabetes of the included studies.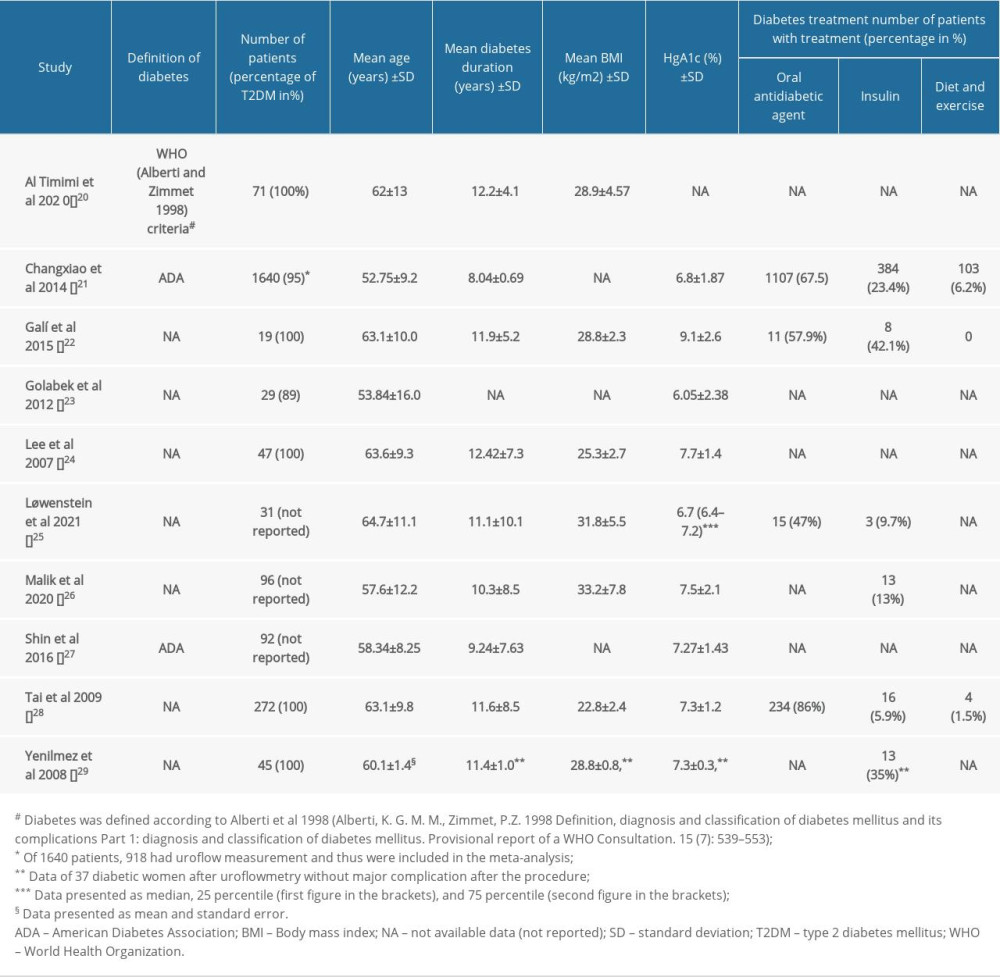 Table 3. Baseline characteristics of urodynamic parameters of diabetic women in the included studies.
Table 3. Baseline characteristics of urodynamic parameters of diabetic women in the included studies.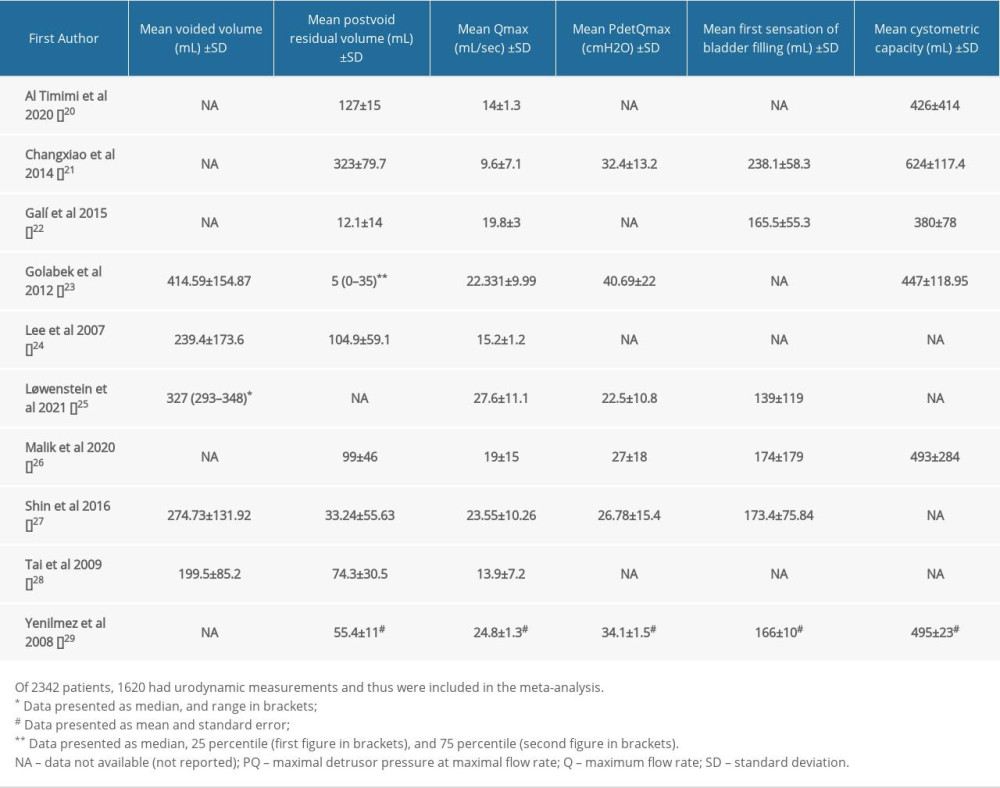 Supplementary Table 1. Search strategy of diabetes and urodynamics.
Supplementary Table 1. Search strategy of diabetes and urodynamics.
References
1. Yuan Z, Tang Z, He C, Tang W, Diabetic cystopathy: A review: J Diabetes, 2015; 7; 442-47
2. Moller CF, Diabetic cystopathy. I: A clinical study of the frequency of bladder dysfunction in diabetics: Dan Med Bull, 1976; 23; 267-78
3. Moller CF, Diabetic cystopathy.II: Relationship to some late-diabetic manifestations: Dan Med Bull, 1976; 23; 279-87
4. Moller CF, Diabetic cystopathy. III: Urinary bladder dysfunction in relation to bacteriuria: Dan Med Bull, 1976; 23; 287-91
5. Moller CF, Olesen KP, Diabetic cystopathy. IV: Micturition cystourethrography compared with urodynamic investigation: Dan Med Bull, 1976; 23; 291-94
6. Kaplan SA, Te AE, Blaivas JG, McGuire EJ, Urodynamic findings in patients with diabetic cystopathy: J Urol, 1995; 153; 342-44
7. Fayyad AM, Hill SR, Jones G, Prevalence and risk factors for bothersome lower urinary tract symptoms in women with diabetes mellitus from hospital-based diabetes clinic: Int Urogynecol J Pelvic Floor Dysfunct, 2009; 20; 1339-44
8. Brown JS, Wessells H, Chancellor MB, Urologic complications of diabetes: Diabetes Care, 2005; 28; 177-85
9. Lee WC, Wu HP, Tai TY, Yu HJ, Chiang PH, Investigation of urodynamic characteristics and bladder sensory function in the early stages of diabetic bladder dysfunction in women with type 2 diabetes: J Urol, 2009; 181; 198-203
10. Kebapci N, Yenilmez A, Efe B, Bladder dysfunction in type 2 diabetic patients: Neurourol Urodyn, 2007; 26; 814-19
11. Sorel MR, Reitsma HJB, Rosier P, Uroflowmetry in healthy women: A systematic review: Neurourol Urodyn, 2017; 36; 953-59
12. Wyman JF, Zhou J, Yvette LaCoursiere D, Normative noninvasive bladder function measurements in healthy women: A systematic review and meta-analysis: Neurourol Urodyn, 2020; 39; 507-22
13. Page MJ, McKenzie JE, Bossuyt PM, The PRISMA 2020 statement: An updated guideline for reporting systematic reviews: BMJ, 2021; 372; n71
14. Rosier P, Schaefer W, Lose G, International Continence Society Good Urodynamic Practices and Terms 2016: Urodynamics, uroflowmetry, cystometry, and pressure-flow study: Neurourol Urodyn, 2017; 36; 1243-60
15. Pessoa R, Kim FJ, Chapter 103 – Urodynamics and voiding dysfunction: Abernathy’s surgical secrets, 2018; 452-54, Elsevier
16. Henry Lai H, Smith CP, Boone TB, Chapter 11 – Urodynamics: female urology (third edition), 2008; 133-46, Philadelphia, WB Saunders
17. McHugh ML, Interrater reliability: The kappa statistic: Biochem Med (Zagreb), 2012; 22; 276-82
18. Cumpston M, Li T, Page MJ, Updated guidance for trusted systematic reviews: A new edition of the Cochrane Handbook for Systematic Reviews of Interventions: Cochrane Database Syst Rev, 2019; 10; ED000142
19. Hayden JA, van der Windt DA, Cartwright JL, Assessing bias in studies of prognostic factors: Ann Intern Med, 2013; 158; 280-86
20. Al-Timimi HFH, Ismail MB, Al-Musawi MM, Urodynamic findings of bladder dysfunction in patients with type 2 dm: International Journal of Research in Pharmaceutical Sciences, 2020; 11; 2433-37
21. Changxiao H, Zhengyong Y, Shibing Y, Clinical and urodynamic evaluation of women referred with diabetes mellitus: International Urogynecology Journal and Pelvic Floor Dysfunction, 2014; 25; 979-83
22. Galì A, Mucciardi G, Butticè S, Correlation between advanced glycation end-products, lower urinary tract symptoms and bladder dysfunctions in patients with type 2 diabetes mellitus: LUTS: Lower Urinary Tract Symptoms, 2017; 9; 15-20
23. Golabek T, Kiely E, O’Reilly B, Detrusor overactivity in diabetic and non-diabetic patients: Is there a difference?: International Braz J Urol, 2012; 38; 652-59
24. Lee WC, Wu CC, Wu HP, Tai TY, Lower urinary tract symptoms and uroflowmetry in women with type 2 diabetes mellitus with and without bladder dysfunction: Urology, 2007; 69; 685-90
25. Løwenstein EP, Andersen LL, Møller LA, Urodynamic and questionnaire findings in urinary incontinent women with and without diabetes. Data from a health study: Int Urogynecol J, 2021; 32; 2847-56
26. Malik RD, Cohn JA, Volsky J, A modern comparison of urodynamic findings in diabetic versus nondiabetic women: Female Pelvic Med Reconstr Surg, 2020; 26; 44-50
27. Shin YS, On JW, Kim MK, Clinical significance of diabetes mellitus on detrusor functionality on stress urinary incontinent women without bladder outlet obstruction: Int Urogynecol J, 2016; 27; 1557-61
28. Tai HC, Chung SD, Ho CH, Metabolic syndrome components worsen lower urinary tract symptoms in women with type 2 diabetes: J Clin Endocrinol Metab, 2010; 95; 1143-50
29. Yenilmez A, Kebapci N, Isikli B, Morbidity after urodynamic study in diabetic patients: Acta Diabetologica, 2009; 46; 197-202
30. Liu G, Daneshgari F, Diabetic bladder dysfunction: Chin Med J (Engl), 2014; 127; 1357-64
31. Appa AA, Brown JS, Creasman J, Clinical predictors and significance of postvoid residual volume in women with diabetes: Diabetes Res Clin Pract, 2013; 101; 164-69
32. Sakakibara R, Takahashi O, Nishimura H, The relationship between bladder, periarterial and somatic neuropathy in diabetes: Intern Med, 2018; 57; 2165-68
33. Jarvis TR, Chan L, Tse V, Practical uroflowmetry: BJU Int, 2012; 110(Suppl 4); 28-29
34. Haylen BT, Ashby D, Sutherst JR, Maximum and average urine flow rates in normal male and female populations – the Liverpool nomograms: Br J Urol, 1989; 64; 30-38
35. Szabo L, Fegyverneki S, Maximum and average urine flow rates in normal children – the Miskolc nomograms: Br J Urol, 1995; 76; 16-20
36. Wittig L, Carlson KV, Andrews JM, Diabetic bladder dysfunction: A review: Urology, 2019; 123; 1-6
37. Yamaguchi C, Sakakibara R, Uchiyama T, Overactive bladder in diabetes: A peripheral or central mechanism?: Neurourol Urodyn, 2007; 26; 807-13
38. Barkai L, Szabo L, Urinary bladder dysfunction in diabetic children with and without subclinical cardiovascular autonomic neuropathy: Eur J Pediatr, 1993; 152; 190-92
39. Ertberg P, Møller LA, Lose G, A comparison of three methods to evaluate maximum bladder capacity: Cystometry, uroflowmetry and a 24-h voiding diary in women with urinary incontinence: Acta Obstet Gynecol Scand, 2003; 82; 374-77
40. Al Afraa T, Mahfouz W, Campeau L, Corcos J, Normal lower urinary tract assessment in women: I. Uroflowmetry and post-void residual, pad tests, and bladder diaries: Int Urogynecol J, 2012; 23; 681-85
41. Mahfouz W, Al Afraa T, Campeau L, Corcos J, Normal urodynamic parameters in women: Part II – invasive urodynamics: Int Urogynecol J, 2012; 23; 269-77
42. Wyndaele JJ KA, Madersbacher H, Radziszewski O, Neurologic urinary and faecal incontinence (diabetes mellitus): Incontinence, 2009; 916-17, Paris, Health Publication Ltd.
43. Liu G, Li M, Vasanji A, Daneshgari F, Temporal diabetes and diuresis-induced alteration of nerves and vasculature of the urinary bladder in the rat: BJU Int, 2011; 107; 1988-93
44. Beshay E, Carrier S, Oxidative stress plays a role in diabetes-induced bladder dysfunction in a rat model: Urology, 2004; 64; 1062-67
45. Kebapci N, Yenilmez A, Efe B, Bladder dysfunction in type 2 diabetic patients: Neurourol Urodyn, 2007; 26; 814-19
Figures
 Figure 1. PRISMA flow diagram of the selection of the studies. The algorithm of the study selection; out of the 1750 records, 10 full-text articles were used in the final analysis. The figure was created using Microsoft® Word (version 16.54; 2019).
Figure 1. PRISMA flow diagram of the selection of the studies. The algorithm of the study selection; out of the 1750 records, 10 full-text articles were used in the final analysis. The figure was created using Microsoft® Word (version 16.54; 2019). Figure 2. Forest plots of the urodynamic parameters of diabetic women. (A) Represents the pooled mean voided volume of diabetic women (n=471). (B) Shows the pooled mean postvoid residual volume of diabetic women (n=1589). (C) Demonstrates the pooled mean Qmax values in diabetic women (n=1620). (D) Presents the pooled mean PdetQmax values in diabetic women (n=1211). (E) Reveals the pooled mean first sensation of bladder filling in diabetic women (n=1201). (F) Unveils the pooled mean maximum cystometric capacity (MCC) in diabetic women (n=1178). Statistical analyses were carried out using R statistical software (version 4.0.5) and package “meta” (version 4.18-1).
Figure 2. Forest plots of the urodynamic parameters of diabetic women. (A) Represents the pooled mean voided volume of diabetic women (n=471). (B) Shows the pooled mean postvoid residual volume of diabetic women (n=1589). (C) Demonstrates the pooled mean Qmax values in diabetic women (n=1620). (D) Presents the pooled mean PdetQmax values in diabetic women (n=1211). (E) Reveals the pooled mean first sensation of bladder filling in diabetic women (n=1201). (F) Unveils the pooled mean maximum cystometric capacity (MCC) in diabetic women (n=1178). Statistical analyses were carried out using R statistical software (version 4.0.5) and package “meta” (version 4.18-1). Tables
 Table 1. Baseline characteristics of the included studies.
Table 1. Baseline characteristics of the included studies. Table 2. Demographic characteristics of women with diabetes of the included studies.
Table 2. Demographic characteristics of women with diabetes of the included studies. Table 3. Baseline characteristics of urodynamic parameters of diabetic women in the included studies.
Table 3. Baseline characteristics of urodynamic parameters of diabetic women in the included studies. Table 1. Baseline characteristics of the included studies.
Table 1. Baseline characteristics of the included studies. Table 2. Demographic characteristics of women with diabetes of the included studies.
Table 2. Demographic characteristics of women with diabetes of the included studies. Table 3. Baseline characteristics of urodynamic parameters of diabetic women in the included studies.
Table 3. Baseline characteristics of urodynamic parameters of diabetic women in the included studies. Supplementary Table 1. Search strategy of diabetes and urodynamics.
Supplementary Table 1. Search strategy of diabetes and urodynamics. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








