04 June 2022: Editorial
Editorial: Acute Hepatitis of Unknown Origin in Children. Is Autoimmunity at Play?
Nahum Mendez-Sanchez12CDEF*, Shreya C. Pal1CDEFDOI: 10.12659/MSM.937371
Med Sci Monit 2022; 28:e937371
Abstract
ABSTRACT: A recent global outbreak of cases of acute hepatitis of unknown origin in children has raised health alerts. Increasing numbers of cases are being reported in most countries, mainly in the United Kingdom (UK). Although the cause remains unknown, several viruses have been isolated from affected children, including adenovirus, severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2), Epstein-Barr virus (EBV), and rhinovirus. Notably, the cause is not from common hepatitis viruses, as serology for hepatitis viruses A, B, C, D, and E has been negative. Current causal hypotheses include possible infection with a new adenovirus variant that affects immunocompetent children, a new pediatric manifestation of COVID-19, or coinfection with enteric adenovirus type F41. This Editorial aims to present current hypotheses regarding the etiology of acute hepatitis of unknown origin in children, including the role of autoimmune hepatitis secondary to viral infection.
Keywords: Editorial, Hepatitis, Adenoviridae, COVID-19, Hepatitis, Autoimmune, Acute Disease, Autoimmunity, COVID-19, Child, Epstein-Barr Virus Infections, Herpesvirus 4, Human, Humans, SARS-CoV-2
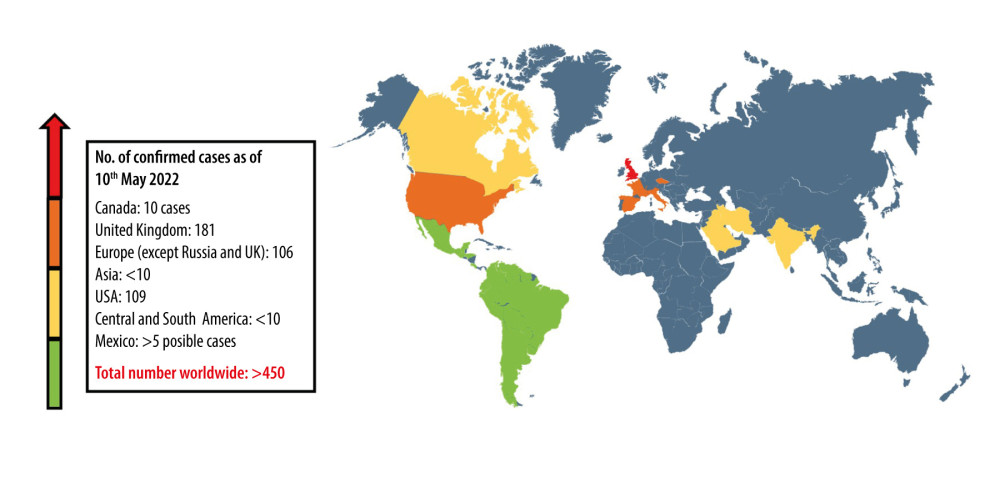
A recent global outbreak of cases of acute hepatitis of unknown origin in children has raised health alerts. Increasing numbers of cases are being reported in most countries. Since this condition was identified, studies in many centers have begun investigating the cause or causes. By 20th May 2022, more than 450 cases of acute hepatitis of unknown origin in children were reported worldwide, although there is a lack of data from some countries, mainly in Africa. However, in the United Kingdom (UK), more than 181 cases of acute hepatitis of unknown origin in children were reported [1–4] (see Figure 1).
This type of acute hepatitis affects children under 16 years of age and has a significantly higher incidence in children aged less than 5 years of age. Although statistical and epidemiological data are still lacking from several countries, mortality for cases reported in Europe and the USA is estimated to be <3% (with 11 total deaths) [5]. As of 20th May 2022, 31 children with this form of acute hepatitis have required liver transplants. Clinical presentation varies among children, with more frequent gastrointestinal symptoms than upper respiratory tract symptoms on presentation. Children with gastrointestinal symptoms most commonly present with jaundice, and nausea, vomiting, and diarrhea are less common. Constitutional symptoms include fatigue and fever in some children [5].
Acute hepatitis of unknown origin in children is associated with altered laboratory biochemical findings that support liver disease, including raised transaminases, which may reach values exceeding 4000 U/L [3] (see Table 1). Recent studies have focussed on investigating a variant of the DNA virus, adenovirus F type 41 [1–6]. However, there is still the possibility that infection with some commonly circulating viruses may be causing hepatitis in immunocompetent children. The new adenovirus F type 4 variant may have different pathogenesis in some children. There may be an altered viral immune response due to a lack of childhood exposure during the COVID-19 pandemic restrictions. Also, altered immune responses to adenovirus may have developed from past infections or co-infection with SARS-CoV-2.
Although adenovirus has been isolated from many children with acute hepatitis of unknown origin, it is essential to note that this association does not prove causation. Adenovirus is a ubiquitous virus in children, and in children with hepatitis, it may be an incidental finding [6]. There should be caution in attributing a new disease in children during the COVID-19 pandemic to infection with the SARS-CoV-2 virus, either directly or as a co-infection. However, at this time, it is unavoidable to consider that SARS-CoV-2 may have a direct role in the cause of cases of acute hepatitis of unknown origin in children.
Acute hepatitis of unknown origin in children is assumed to affect immunocompetent children. However, because there still have been no studies to investigate whether autoantibodies are present, the use of the term, immunocompetent may be of concern. Given the high detection rate of adenovirus in affected children and what is known about the pathophysiology of hepatitis, there are three main possibilities regarding the cause of acute hepatitis of unknown origin in children: infection with an adenovirus variant; autoimmune hepatitis; and an autoimmune childhood form of COVID-19.
Firstly, adenovirus is a DNA virus responsible for many upper and lower respiratory tract infections, especially in the pediatric population with humoral immune deficiencies. Uncommon but possible presentations can include hemorrhagic cystitis, hemorrhagic colitis, meningoencephalitis, and acute hepatitis [6]. Serotypes 40–41 have a greater affinity for the gastrointestinal tract within the adenovirus species F [6,7]. Recently identified cases of acute hepatitis of unknown origin in children have been associated with the isolation of adenovirus F type 41 in the blood, feces, and respiratory exudates [6,7]. Other isolated pathogens have included SARS-CoV-2 (in 18% of cases), rhinovirus, enterovirus and metapneumovirus [7,8].
Multiple case reports have described adenovirus-related hepatitis with high mortality rates of up to 75% in patients undergoing allogenic hematopoietic stem cell transplantation [7,8]. Adenovirus-related hepatitis mainly presents in immunosuppressed patients with deficient adaptive cellular immune responses, including T cell-mediated immunity. This finding links the immunodeficient state in COVID-19 patients and the gastrointestinal presentation of adenovirus infection. Therefore, co-infection could be a possible cause, as adenovirus infection in up to 70% of patients and SARS-CoV-2 infection in up to 20% of patients has been reported [1,6,8].
The second hypothesis for the cause of acute hepatitis of unknown origin in children is that it is an autoimmune disease. Autoimmune hepatitis (AIH) is a progressive liver disease with three diagnostic features: hypergammaglobulinemia; identification of specific autoantibodies; and histopathology that shows an ‘interface hepatitis’ [9,10]. AIH is subclassified into two major groups: AIH-1 and AIH-2 [9,10]. AIH-1 is associated with antinuclear antibodies (ANA), anti-smooth muscle antibodies (SMA), and anti-neutrophil cytoplasmic antibodies (ANCA), mainly with a perinuclear distribution (p-ANCA) [9]. AIH-2 presents with autoantibodies against anti-liver/kidney microsomal antibody type 1 (anti-LKM1) [6]. There are several examples of drug-induced AIH and post-infectious AIH, especially after hepatitis A infection (HAV). HAV may rarely act as a trigger for autoimmune hepatitis, owing to immune mimicry [11]. It is possible that other pathogens, including SARS-CoV-2, could induce AIH by initiating self-perpetuating immune-mediated liver inflammation. Therefore, acute viral hepatitis and post-infectious AIH, by either SARS-CoV-2 or adenovirus, are two possible hypotheses for the current global acute hepatitis outbreak in children.
The third hypothesis for the cause of acute hepatitis of unknown origin in children is that this is an autoimmune pediatric manifestation of COVID-19. COVID-19 has been reported to present with acute non-icteric jaundice even before the onset of respiratory symptoms are reported [12]. Reports of COVID-19 mRNA vaccines have shown an association with the onset of AIH, possibly by altering self-tolerance. Immunological studies have shown positive ANA, anti-mitochondrial, and smooth muscle antibodies in reported cases [13,14]. These autoantibodies are characteristic of AIH-1. Even though these clinical studies have shown that most affected children were not yet vaccinated, this mechanism may also be extrapolated to SARS-CoV-2 as a cause of acute hepatitis. However, seemingly immunocompetent pediatric patients are yet to be tested for specific autoantibodies against hepatic antigens. Even though testing for all autoantibody types may be impractical, the previous cases of post-infectious AIH make the possibility of this disease following the same course as adult AIH feasible. Analyzing pediatric patient serum for autoantibodies associated with AIH may bring a clearer understanding of the cause of acute hepatitis of unknown origin in children.
Conclusions
Recent recognition of acute hepatitis of unknown origin in children has resulted in a search for the cause. Adenovirus infection may play an important role in the etiology. However, future studies should include the investigation of autoantibodies and autoimmune responses in affected children.
References
1. Joint European Centre for Disease Prevention and Control (ECDC): World Health Organisation (WHO) Regional Office for Europe Hepatitis of Unknown Origin in Children Surveillance Bulletin May 20, 2022 Available from https://www.ecdc.europa.eu/en/hepatitis/joint-weekly-hepatitis-unknown-origin-children-surveillance-bulletin
2. SINAVE, CONAVE: Boletín Epidemiológico; Sistema Nacional de Vigilancia Epidemiológica Semana 19, 2022, Sistema Único de Información [in Spanish]
3. Baker JM, Buchfellner M, Britt W, Acute hepatitis and adenovirus infection among children – Alabama, October 2021–February 2022: MMWR Morb Mortal Wkly Rep, 2022; 71(18); 638-40
4. Government of Canada: Acute severe hepatitis in children https://www.canada.ca/en/public-health/services/diseases/acute-hepatitis-children.html
5. Cevik M, Rasmussen AL, Bogoch II, Kindrachuk J, Acute hepatitis of unknown origin in children: BMJ, 2022; 377; o1197
6. Lynch JP, Kajon AE, Adenovirus: Epidemiology, global spread of novel serotypes, and advances in treatment and prevention: Semin Respir Crit Care Med, 2016; 37(4); 586-602
7. Symeonidis N, Jakubowski A, Pierre-Louis S, Invasive adenoviral infections in T-cell-depleted allogeneic hematopoietic stem cell transplantation: High mortality in the era of cidofovir: Transpl Infect Dis, 2007; 9(2); 108-13
8. Wang WH, Wang HL, Fulminant adenovirus hepatitis following bone marrow transplantation. A case report and brief review of the literature: Arch Pathol Lab Med, 2003; 127(5); e246-48
9. Schmeltzer PA, Russo MW, Clinical narrative: autoimmune hepatitis: Am J Gastroenterol, 2018; 113(7); 951-58
10. Gatselis NK, Zachou K, Koukoulis GK, Dalekos GN, Autoimmune hepatitis, one disease with many faces: Etiopathogenetic, clinico-laboratory and histological characteristics: World J Gastroenterol, 2015; 21(1); 60-83
11. Subramanian SK, Patel JM, Younes M, Nevah Rubin MI, Postinfectious autoimmune hepatitis-induced liver failure: A consequence of hepatitis A virus infection: ACG Case Rep J, 2020; 7(8); e00441
12. Wander P, Epstein M, Bernstein D, COVID-19 presenting as acute hepatitis: Am J Gastroenterol, 2020; 115(6); 941-42
13. Tan CK, Wong YJ, Wang LM, Autoimmune hepatitis following COVID-19 vaccination: True causality or mere association?: J Hepatol, 2021; 75(5); 1250-52
14. Ghielmetti M, Schaufelberger HD, Mieli-Vergani G, Acute autoimmune-like hepatitis with atypical anti-mitochondrial antibody after mRNA COVID-19 vaccination: A novel clinical entity?: J Autoimmun, 2021; 123; 102706
Tables
 Table 1. The main clinical characteristics, patient presentation, biochemical and viral serology findings in children presenting with acute hepatitis of unknown origin.
Table 1. The main clinical characteristics, patient presentation, biochemical and viral serology findings in children presenting with acute hepatitis of unknown origin. Table 1. The main clinical characteristics, patient presentation, biochemical and viral serology findings in children presenting with acute hepatitis of unknown origin.
Table 1. The main clinical characteristics, patient presentation, biochemical and viral serology findings in children presenting with acute hepatitis of unknown origin. In Press
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
11 Mar 2024 : Clinical Research
Comparison of Effects of Sugammadex and Neostigmine on Postoperative Neuromuscular Blockade Recovery in Pat...Med Sci Monit In Press; DOI: 10.12659/MSM.942773
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952