07 December 2022: Clinical Research
A Prospective Questionnaire-Based Study to Evaluate Factors Affecting the Decision to Receive COVID-19 Vaccination in 267 Patients with Inflammatory Bowel Disease in Poland
Michał ŁodygaDOI: 10.12659/MSM.938665
Med Sci Monit 2022; 28:e938665
Abstract
BACKGROUND: The aim of the study was to assess the rate of COVID-19 vaccination and the attitudes toward receiving COVID-19 vaccination among patients with inflammatory bowel disease (IBD) in Poland. An important aim of the study was to determine why some people get vaccinated and others refuse to do so.
MATERIAL AND METHODS: This was a single-center, prospective survey. The study included 267 IBD patients who agreed to complete an anonymous questionnaire comprising 31 questions.
RESULTS: We found that 71.2% of the IBD patients had been vaccinated. The history of COVID-19 was associated with a lower vaccination rate (16.9% vs 36.8%; P=0.001), regardless of IBD severity. In the vaccinated group, there were more vaccinated people among household members (90.4% vs 43.4%; p<0.001) and friends (52.9% vs 22.4%; P<0.001). Family safety (71.1%), the desire to avoid COVID-19 (67.9%), social responsibility (60.5%), the desire to return to normal life (51.6%), and faith in vaccination as such (43.2%) were the most common reasons for vaccination. The most common cause of non-vaccination was concern about adverse effects (50.0%), including long-term adverse effects (36.8%), and about the possible exacerbation of gastroenterological disease (34.2%).
CONCLUSIONS: IBD patients are more likely to be vaccinated against SARS-CoV-2 than the rest of the population in Poland. Young age, low socioeconomic status, low education, and living in the countryside were factors associated with lower vaccination rates. Family and friends had the greatest influence on the decision to vaccinate, but the influence of the mass media was very small.
Keywords: COVID-19, Inflammatory Bowel Diseases, Vaccination, Humans, Prospective Studies, COVID-19 Vaccines, SARS-CoV-2, COVID-19
Background
The invention of the SARS-CoV-2 virus vaccine is a milestone in the fight against the COVID-19 pandemic. It is worth noting that the most effective vaccines are based on mRNA technology, which is being used for the first time. This makes the invention of the vaccine even more impressive. Another, no less important, challenge was to vaccinate as many people as possible. This seems to be the only way to stop the spread of the virus and end the pandemic. This task turned out to be difficult not just for logistical reasons, but also, more importantly, in convincing the vast majority of the population to get vaccinated [1].
Significant geographic variations in vaccination rates were apparent from the outset. Initially, these were mainly due to the availability of vaccines, and, over time, the main difference was the percentage of people skeptical about vaccination in a given society. Skepticism usually builds up over time. There were several reasons for this. With time, data on the effectiveness of vaccinations began to appear, which clearly indicated that they are not able to completely eliminate the transmission of the virus, and their role is primarily to reduce the risk of a severe course of the disease and death [2]. It was found that vaccinations have to be repeated [3]. In addition, subsequent mutations of the virus that began to dominate were admittedly more infectious, but the disease caused a severe course and death for a smaller percentage [4]. With the increasing numbers of people vaccinated, concerns about the safety of the vaccines themselves arose periodically, which, although dispelled by the world of science, often still existed in the virtual world [5]. Taken together, these facts reduced the feeling of the need to be vaccinated in many people.
According to current population data from the Polish Ministry of Health, 60% of the general population have been fully vaccinated for COVID-19 [6], which is less than in some other countries in Europe (for example, 81% in France and 96% in Portugal (data as of 2 July 2022) [7]).
This fact could be expected in the context of the results of studies assessing the willingness to be vaccinated at the beginning of the vaccination campaign in the Polish general population, in which a high level of skepticism regarding vaccinations was observed (eg, in the study by Sowa et al [8] only 50.8% of the population stated they had been vaccinated).
Vaccinations, although recommended for everyone, are particularly important for people at risk of both infection and severe course of the disease. These groups include, among others, people with chronic diseases and those who are immunosuppressed (eg, as a result of the use of immunosuppressive drugs). Therefore, this group includes patients with inflammatory bowel disease (IBD) [9,10].
For this reason, the progress of vaccination in this group of patients has been studied. The available data are inconclusive. For example, a study in France found a similar proportion to the general population planning to be vaccinated in that country [11]. Similar conclusions can be drawn from studies carried out in Italy [12] and the USA [13], where a very high percentage of IBD patients stated they had been vaccinated (80.3% and 80.9%, respectively). In contrast, in 2 studies from China, the percentage of IBD patients who declared their willingness to be vaccinated and who had already been vaccinated was small [14,15]. Similar data come from Germany [16].
Therefore, this prospective questionnaire-based study aimed to evaluate factors affecting the decision to receive COVID-19 vaccination in 267 patients with inflammatory bowel disease in Poland.
Material and Methods
ETHICS APPROVAL:
This study was approved by the Ethics and Supervision Committee for Human and Animal Research at the Central Clinical Hospital of the Ministry of the Interior and Administration in Warsaw, Poland (no. 108/2021). All patients provided written informed consent to participate in the study.
ANONYMITY:
The consent to the study and the questionnaire were separate from each other. They were also processed and entered into the database separately to maintain the anonymity of the data provided by the patient in the questionnaire. The questionnaire did not contain data by which it would be possible to identify the person completing the survey.
STUDY POPULATION:
This was a single-center, prospective survey. The study included 267 IBD adult patients – 160 with Crohn`s disease (CD) and 107 with ulcerative colitis (UC) – regardless of the activity, form of the disease, and medications taken, who were visiting the center for various reasons (eg, administration of a biological drug, diagnostic tests, a visit to the outpatient clinic) between 1 September and 30 November 2021, and agreed to complete an anonymous questionnaire. The study did not include a control group.
QUESTIONNAIRE:
The questionnaire was original. The primary aim was to assess whether the patient had been vaccinated against SARS-CoV-2, and then to establish the main reasons for the decision to be vaccinated or not. The arguments most frequently appearing in the public debate were selected as possible answers, but the respondents also had the opportunity to describe their own reasons in an open question. The questionnaire was not based on another known, validated questionnaire. It was prepared in Polish and contained the following groups of questions: the history and treatment of IBD, the patient’s experience with the SARS-CoV-2 virus (symptomatic or asymptomatic infection in the patient or their relatives, vaccination among relatives and household members), demographic data (age, place of residence, assessment of socioeconomic status) and other health behaviors (eg, other recommended vaccinations, smoking). The questions asked are presented in Table 1.
STATISTICAL ANALYSIS:
The chi-square test (with Yate’s correction for continuity for 2×2 tables) or Fisher’s exact test were used to analyse the relationship between 2 qualitative variables (n and% were given), and the Mann-Whitney U test was used to compare the values of quantitative variables (the median with quartiles 1 and 3 were given). The median differences and odds ratios were calculated with 95% confidence intervals. The normality of the distributions of quantitative variables was checked with the Shapiro-Wilk test. The analysis was performed using R statistical software, version 4.2.1 (with α=0.05).
Results
STUDY POPULATION:
Of the 267 patients who completed the questionnaire, 50.6% were women. The average age was 34 years. Current smoking was declared by14.6% and 36.7% stated they had been smokers in the past. Alcohol consumption was declared by 24.4%. Most of the responders were residents of very large cities with over 100 000 inhabitants (55.1%) and most had higher education (62.8%). Most of them were working people (54.0%), with middle income.
CD was diagnosed in 59.9% of patients, and UC in 40.1%. The mean duration of IBD was 11 years. Mesalamine had been received by 77.8% of the patients, 54.4% had received thiopurines, 23.8% had received steroids, and 60.2% had received biologics. Surgery for IBD had been undergone by 34.1%, with an average of 2 operations.
The detailed characteristics of the group are presented in Tables 2 and 3.
HISTORY OF COVID-19:
Of the study participants, 22.6% reported a history of COVID-19, mostly mild or moderate (43.3% and 51.7%, respectively). A history of SARS-CoV-2 infection among the family was reported by 29.2%. Similarly, most infections were moderate and mild (53.3% and 33.3%, respectively).
COVID-19 VACCINATION:
Vaccination had been received by 71.2% of the patients. The majority (72.3%) decided by themselves to get vaccinated, while for 26.6%, the vaccination was prescribed by a doctor. Most had no concerns about vaccination (52.4%). Of the patients who got vaccinated despite their fears, none mentioned the mass media as being the factor that convinced them to do so. The vast majority of patients were not vaccinated against influenza (83.3%) or pneumococci (87.5%), while 71.4% were vaccinated against hepatitis B.
The detailed history of COVID-19 is presented in Table 4.
The influence of various factors on vaccination was studied in detail. Among IBD-dependent factors, only steroid use was found; significantly fewer vaccinated patients were steroid users compared to the unvaccinated groups (19.8% vs 33.8%;
Regarding demographic factors, there was no relationship between vaccination and sex, and in the vaccinated group the mean age was significantly higher (36 years vs 32.5 years in the unvaccinated group;
No influence of other health behaviors (eg, alcohol consumption, smoking cessation) was found. In the vaccinated group there were more people with higher education (68.4% vs 47.4%;
A history of COVID-19 disease was associated with a lower vaccination rate (16.9% vs 36.8%; P=0.001), regardless of disease severity, while family history of COVID-19 was found to have no effect. The impact of vaccinations among relatives and friends on the decision to be vaccinated was significant. In the vaccinated group, there were significantly more vaccinated people among household members (90.4% vs 43.4%; P<0.001) and friends (52.9% vs 22.4%; P<0.001). Influenza vaccination was also more common among those vaccinated against COVID-19 (19.6% vs 9.5%; P=0.008), while other vaccinations did not have such an effect. The detailed results are presented in Table 5.
REASONS FOR COVID-19 VACCINATION:
Family safety was mentioned as the most common reason for vaccination among those vaccinated (71.1%). The most frequently mentioned reasons were the desire to avoid COVID-19 (67.9%), social responsibility (60.5%), the desire to return to normal life (51.6%), and faith in vaccination as such (43.2%).
Among unvaccinated people, the most common reason for non-vaccination was concern about adverse effects (50.0%), including long-term adverse effects (36.8%), and about the possible exacerbation of gastroenterological disease after vaccination (34.2%). Lack of confidence in the vaccine development process was mentioned by 27.6%, including the short time it took to invent it (23.7%). The percentage of patients whose reasons were concerns raised by doctors (of various specialties) was significant (23.7%). The percentage of patients who self-diagnosed contraindications to vaccination was 22.4%. A small number of patients cited various conspiracy theories as the cause of non-vaccination (eg, there is no pandemic, vaccination does not work – both 2.6%).
The reasons for receiving or refusing the vaccination are presented in Table 6.
Discussion
LIMITATIONS:
Our study has several limitations. Our sample size was small and included a large percentage of patients with a more severe course of IBD and those whose condition requires a personal visit to the medical center. This is seen in the very high percentage of biologically treated patients. Therefore, patients with a severe course of the IBD, who have more contact with the health service during a pandemic, were qualified for our study. In our study it was not possible to select a control group consisting of people without IBD because, at the time of the study, there were significant restrictions on access of unvaccinated people to the hospital. The strengths of our work include the fact that it assessed vaccinations already performed, rather than intention to get vaccinated, as in most previous studies. Nevertheless, our study assessed the problems that result in a relatively high percentage of unvaccinated people in the Polish population. As shown in the discussion, there are large cultural and geographic differences in the approach to vaccination, which is why it is so important to understand the mechanisms responsible for the non-acceptance of COVID-19 vaccination in individual populations and in specific groups of patients. The presented data may help better planning for future vaccination campaigns for IBD patients and for the entire population.
Conclusions
The findings from this study showed that patients with a diagnosis of IBD were more likely to be vaccine compliant (71.2%) than the general population in Poland (60%). As in the general population, patients with younger age, low socioeconomic status, low levels of education, and those living in rural areas had lower vaccination rates. Factors that positively influenced the decision to be vaccinated included support from family and friends, but information from the media had no effect on the decision to become vaccinated.
Tables
Table 1. Vaccination questionnaire. Table 2. Group characteristics – demographic data.
Table 2. Group characteristics – demographic data.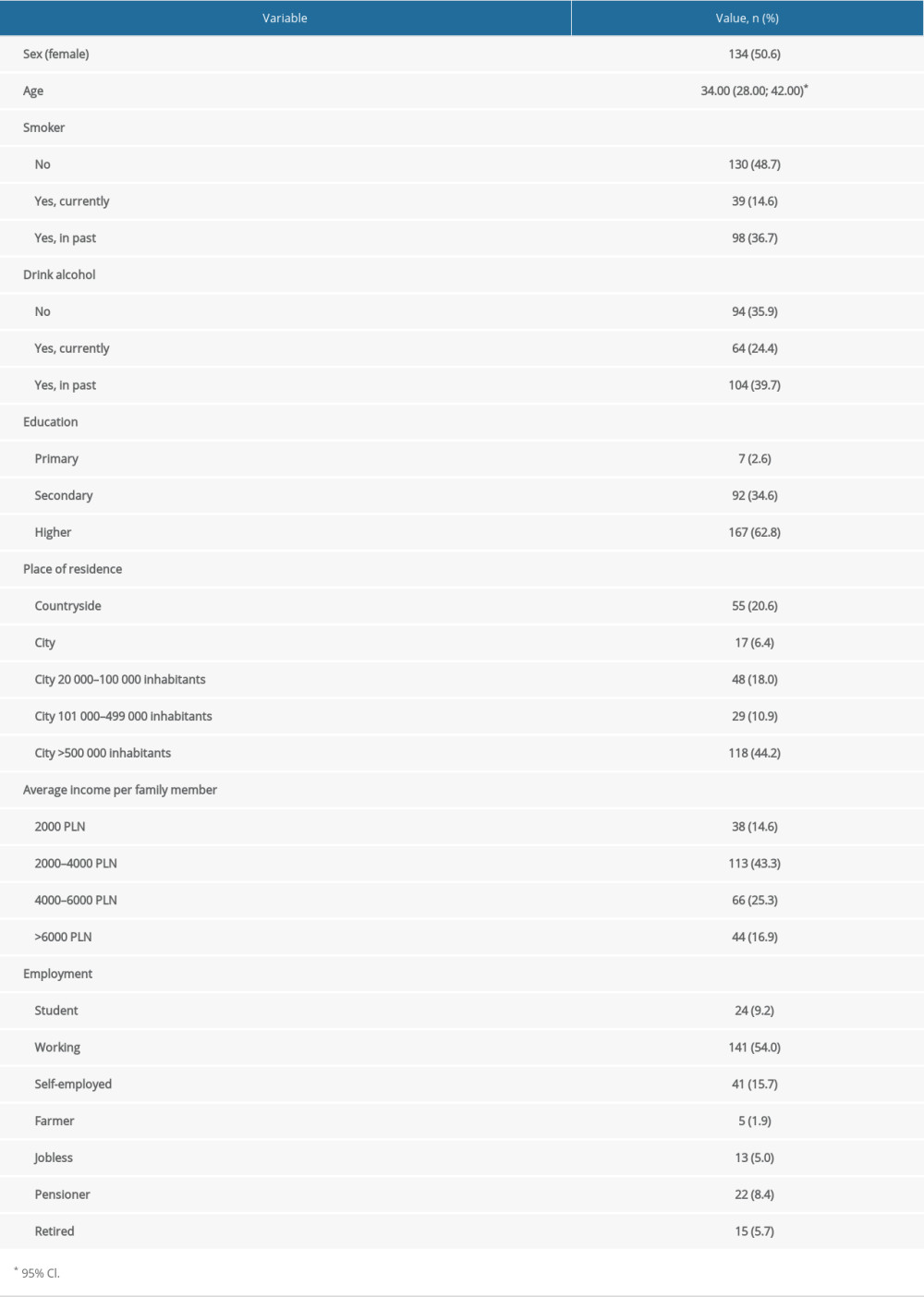 Table 3. Group characteristics – history of IBD.
Table 3. Group characteristics – history of IBD.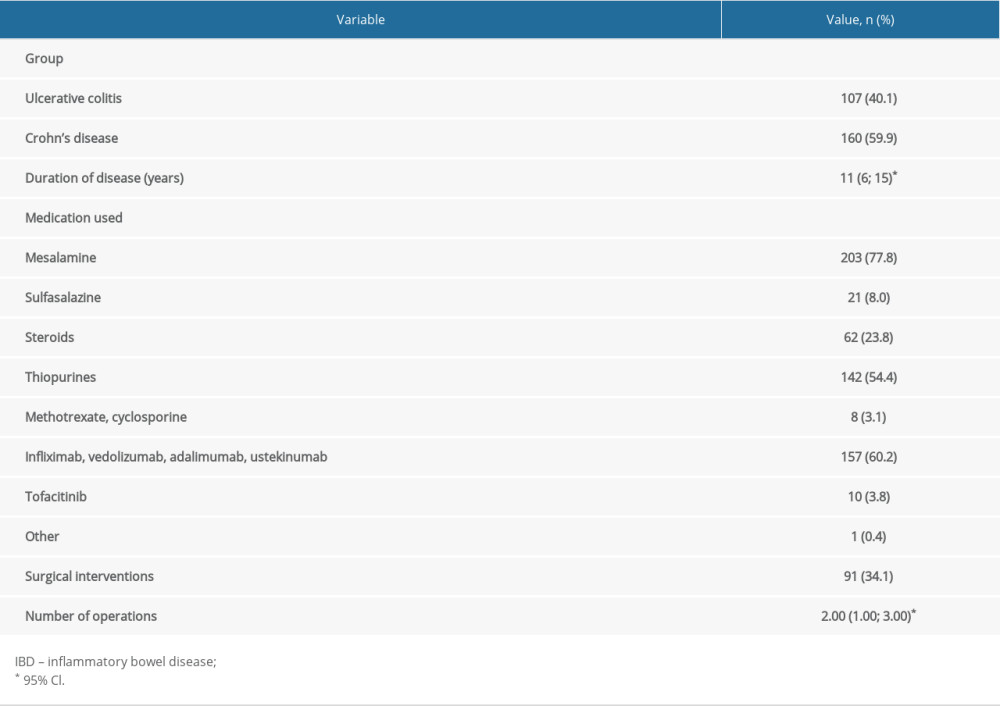 Table 4. History of COVID-19.
Table 4. History of COVID-19.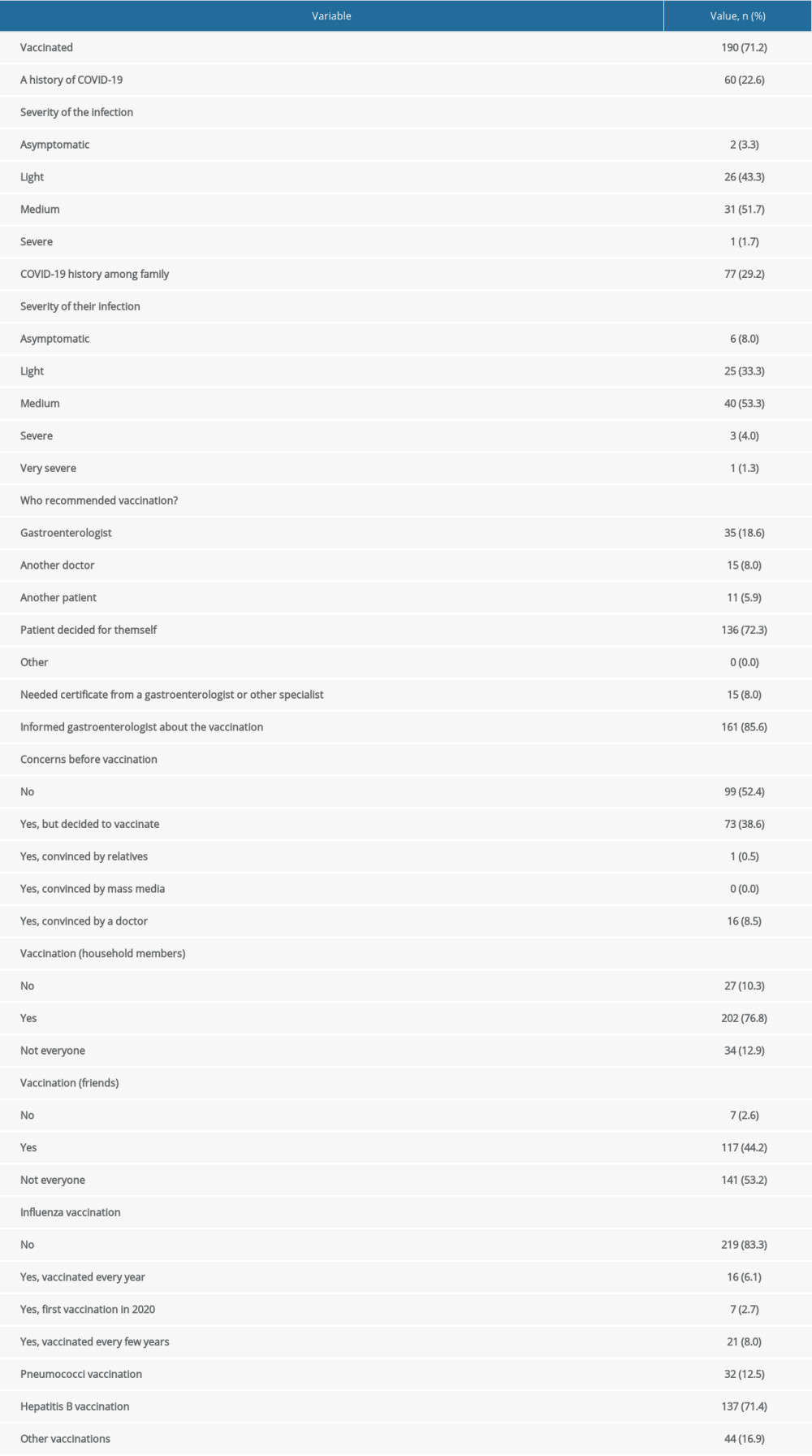 Table 5. Analyses of relationships between vaccination and selected variables.
Table 5. Analyses of relationships between vaccination and selected variables.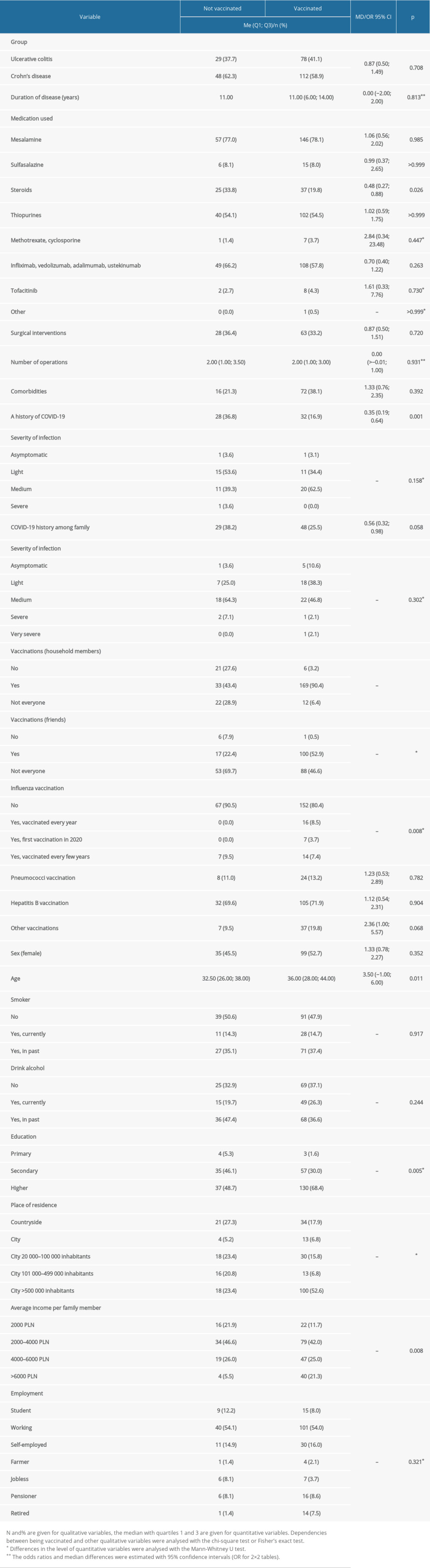 Table 6. Reasons for receiving or refusing the vaccination.
Table 6. Reasons for receiving or refusing the vaccination.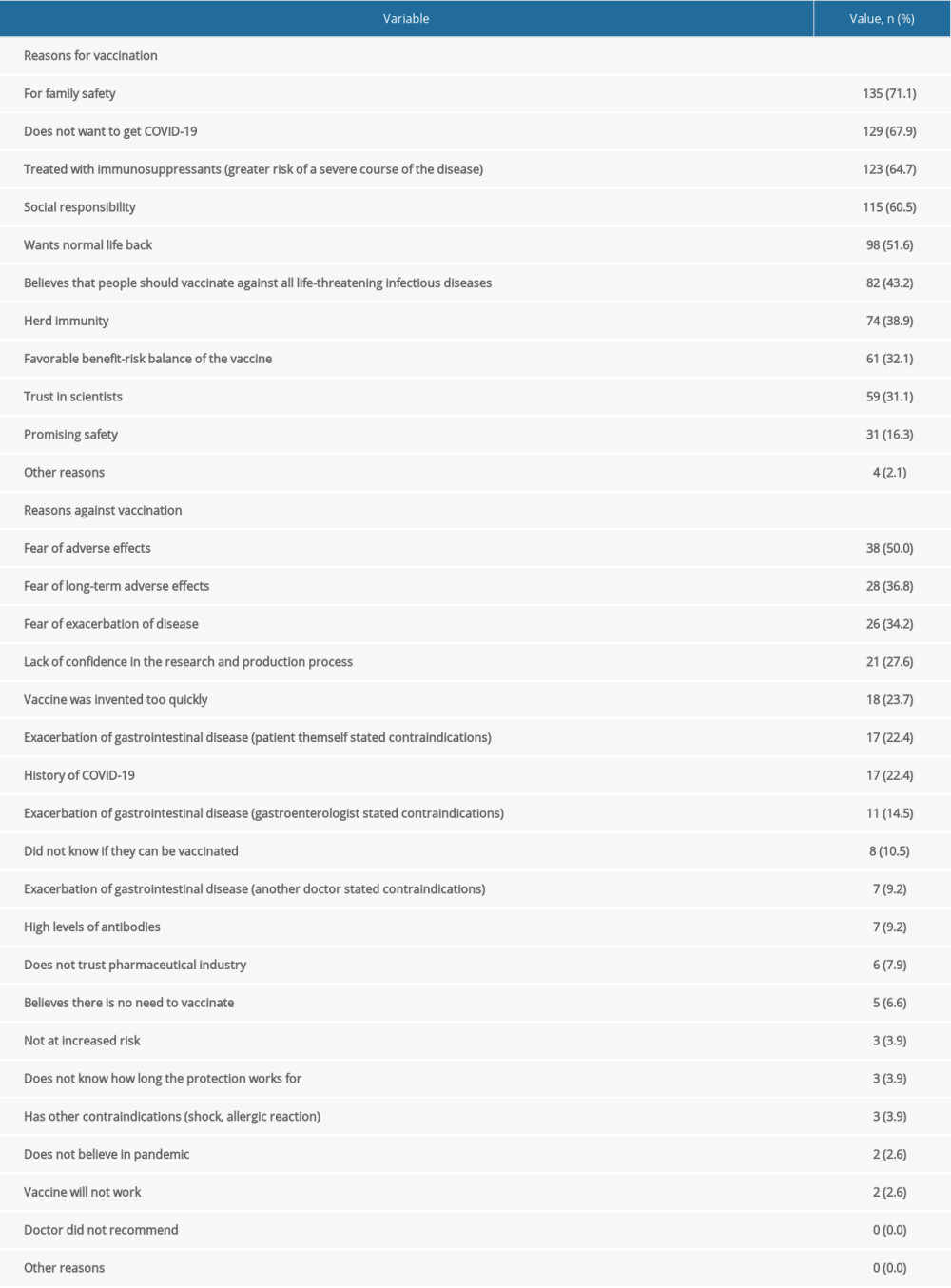
References
1. Rahman MM, Masum MHU, Wajed S, Talukder A, A comprehensive review on COVID-19 vaccines: Development, effectiveness, adverse effects, distribution and challenges: Virusdisease, 2022; 33(1); 1-22
2. Haas EJ, Angulo FJ, McLaughlin JM, Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: An observational study using national surveillance data: Lancet, 2021; 397(10287); 1819-29
3. Barda N, Dagan N, Cohen C, Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: An observational study: Lancet, 2021; 398(10316); 2093-100
4. Martínez-Flores D, Zepeda-Cervantes J, SARS-CoV-2 vaccines based on the spike glycoprotein and implications of new viral variants: Front Immunol, 2021; 12; 701501
5. Wu Q, Dudley MZ, Chen X, Evaluation of the safety profile of COVID-19 vaccines: A rapid review: BMC Med, 2021; 19(1); 173
6. Polish Ministry of Health, Online statistical data: COVID-19 Available at: https://www.gov.pl/web/szczepimysie/raport-szczepien-paniemko-covid-19
7. Oxford Martin School. University of Oxford, Our World in Data: COVID-19 Vaccinations by Country Available at: https://ourworldindata.org/covid-vaccinations#source-information-country-by-country
8. Sowa P, Kiszkiel Ł, Laskowski PP, COVID-19 vaccine hesitancy in Poland-multifactorial impact trajectories: Vaccines (Basel), 2021; 9(8); 876
9. Monteleone G, Ardizzone S, Are patients with inflammatory bowel disease at increased risk for COVID-19 infection?: J Crohns Colitis, 2020; 14(9); 1334-36
10. Zhou F, Yu T, Du R, Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study: Lancet, 2020; 395(10229); 1054-62 Erratum in: Lancet. 2020:395(10229):1038
11. Caron B, Neuville E, Peyrin-Biroulet L, Inflammatory bowel sisease and COVID-19 vaccination: A patients’ survey: Dig Dis Sci, 2022; 67(6); 2067-73
12. Costantino A, Noviello D, Conforti FS, COVID-19 vaccination willingness and hesitancy in patients with inflammatory bowel diseases: Analysis of determinants in a national survey of the Italian IBD Patients’ Association: Inflamm Bowel Dis, 2022; 28(3); 474-78
13. Dalal RS, McClure E, Marcus J, COVID-19 vaccination intent and perceptions among patients with inflammatory bowel diseases: Clin Gastroenterol Hepatol, 2021; 19(8); 1730-32e2
14. Dai C, Jiang M, Huang YH, COVID-19 vaccination in patients with inflammatory bowel disease: A survey from China: Dig Dis Sci, 2022; 67(7); 3468-70
15. Wu X, Lin J, Buch H, The COVID-19 vaccination hesitancy among the people with inflammatory bowel disease in China: A questionnaire study: Front Public Health, 2021; 9; 731578
16. Walldorf J, von Arnim U, Schmelz R, SARS-CoV-2 vaccination in patients with inflammatory bowel disease- fear and desire: Inflamm Bowel Dis, 2021; 27(11); 1858-61
17. Loftus EV, Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences: Gastroenterology, 2004; 126(6); 1504-17
18. Burisch J, Munkholm P, The epidemiology of inflammatory bowel disease: Scand J Gastroenterol, 2015; 50(8); 942-51
19. Norsa L, Indriolo A, Sansotta N, Uneventful course in IBD patients during SARS-CoV-2 outbreak in northern Italy: Gastroenterology, 2020; 159(1); 371-72
20. Brenner EJ, Ungaro RC, Gearry RB, Corticosteroids, but not TNF antagonists, are associated with adverse COVID-19 outcomes in patients with inflammatory bowel diseases: results from an international registry: Gastroenterology, 2020; 159; 481-91
21. Łodyga M, Eder P, Dobrowolska A, The position statement of the Polish Society of Gastroenterology and the Polish National Consultant in Gastroenterology regarding the management of patients with inflammatory bowel disease during the COVID-19 pandemic: Gastroenterology Review, 2020; 15(2); 85-88
22. Ungaro RC, Brenner EJ, Gearry RB, Effect of IBD medications on COVID-19 outcomes: Results from an international registry: Gut, 2021; 70(4); 725-32
23. Eder P, Łodyga M, Dobrowolska A, Position statement of the Polish Society of Gastroenterology and the National Gastroenterology Consultant on vaccination against COVID-19 among patients. with inflammatory bowel diseases: Prz Gastroenterol, 2021; 16(1); 2-4
24. Siegel CA, Melmed GY, McGovern DPInternational Organization for the Study of Inflammatory Bowel Disease (IOIBD), International Organization for the Study of Inflammatory Bowel Diseases (IOIBD) SARS-CoV-2 Vaccination for Patients with Inflammatory Bowel Diseases: recommendations from an International Consensus Meeting: Gut, 2021; 70; 635-40
25. Kucharzik T, Ellul P, Greuter T, ECCO guidelines on the prevention, diagnosis, and management of infections in inflammatory bowel disease: J Crohn’s Colitis, 2021; 15; 879-913
26. Alexander JL, Moran GW, Gaya DR, SARS-CoV-2 vaccination for patients with inflammatory bowel disease: A British Society of Gastroenterology Inflammatory Bowel Disease Section and IBD Clinical Research Group Position Statement: Gastroenterol Lancet Hepatol, 2021; 6; 218-24
27. Wisniak A, Baysson H, Pullen NSpecchio-COVID19 study group, COVID-19 vaccination acceptance in the canton of Geneva: A cross-sectional population-based study: Swiss Med Wkly, 2021; 151; w30080
28. Detoc M, Bruel S, Frappe P, Intention to participate in a COVID-19 vaccine clinical trial and to get vaccinated against COVID-19 in France during the pandemic: Vaccine, 2020; 38(45); 7002-6
Tables
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








