05 September 2023: Clinical Research
The Diagnostic Accuracy of N-Terminal Pro-B-Type Natriuretic Peptide and Soluble ST2 for Heart Failure in Chronic Kidney Disease Patients: A Comparative Analysis
Hongzhen Ma1BCDE, Jun Zhou2BCD, Meng Zhang1BF, Chun Shen1BF, Zhifan Jiang1BF, Tao ZhangDOI: 10.12659/MSM.940641
Med Sci Monit 2023; 29:e940641
Abstract
BACKGROUND: N-terminal proatrial natriuretic peptide (NT-proBNP) levels are often markedly elevated in patients with chronic kidney disease (CKD). Identifying novel biomarkers is an important step toward effective diagnosis. Interleukin-1 receptor-like 1 (IL1RL1) protein and human/Soluble suppression of tumorigenesis-2 (sST2) are promising biomarkers for heart failure (HF). This study aimed to assess the trend of NT-proBNP and sST2 in chronic kidney disease and their diagnostic value for HF.
MATERIAL AND METHODS: This study was carried out on 420 patients who were divided into a no heart failure group (N=182) and a heart failure group (N=238). Spearman correlation analysis was used to test the association of sST2 and NT-proBNP with renal function. The diagnostic value of each biomarker was assessed using receiver operating characteristic (ROC) curves according to 3 different forms: Total group (n=420), non-CKD group (n=217), and CKD group (n=203).
RESULTS: A striking correlation between eGFR and NT-proBNP (r=-0.525; P<0.001) seemed to be far stronger than that with sST2 (r=-0.147; P<0.05). The optimum cutoff points for sST2 and NT-proBNP to detect HF were 28.960 ng/mL and 1280 pg/mL, respectively, in total, 28.71 ng/mL and 481 pg/mL, respectively, in non-CKD patients, and 30.55 ng/mL and 3314 pg/mL, respectively, in CKD patients. The combined model of sST2 and NT-proBNP was superior to the model of sST2 or NT-proBNP alone, and the difference was statistically significant (P<0.05).
CONCLUSIONS: The diagnostic value of sST2 is less affected by decreased renal function. sST2 combined with NT-proBNP may improve the diagnostic accuracy of HF.
Keywords: Heart Failure, N-Terminal Proatrial Natriuretic Peptide, IL1RL1 Protein, Human, Renal Insufficiency, Chronic, Humans, Natriuretic Peptide, Brain, Interleukin-1 Receptor-Like 1 Protein, carcinogenesis, Cell Transformation, Neoplastic
Background
Heart failure (HF) is a public health epidemic consisting of symptoms and signs that result from inadequate cardiac output or cardiac filling to meet systemic demands. Currently, the overall incidence of HF is increasing globally [1], largely due to the aging of the population [2]. The reported prevalence of heart failure in 2012 was 4.3% among individuals 65–70 years old and is expected to rise steadily until 2030, when it could reach 8.5% [3, 4]. Unfortunately, HF is often complicated by multiple comorbidities, of which chronic kidney disease (CKD) is of great importance. Chronic renal insufficiency is a powerful independent risk factor for the progression of CVD [5,6]. Data have shown that approximately 40–50% of HF patients have coexistence of CKD [7]. Similarly, patients with decreased renal function are at high risk of developing HF. CVD is the leading cause of mortality in patients with CKD, and over 50% of deaths in CKD patients are from CVD [8,9].
NT-proBNP is a well-established biomarker of HF, with excellent diagnostic and prognostic power, but its general serum concentrations increase with the progression of CKD and end-stage renal disease (ESRD), which can confound their interpretation in the context of worsening cardiac function.
Interleukin-1 receptor-like 1 (IL1RL1) protein, human (also known as ST2) is a member of the interleukin-1 receptor superfamily with the specific ligand Interleukin-33. sST2, a soluble isoform of ST2, was included in the ACA/AHA guidelines in 2013 as a novel biomarker for assessing cardiac function and prognosis in patients with heart failure. The association between circulating sST2 concentration and CVD has attracted much attention in recent years. Serum sST2, a marker of cardiac stress, is upregulated in response to cardiomyocyte stretch. In 3428 Framingham Heart Study participants, elevations of sST2 were associated with predicting greater risks of death, incident HF, and overall cardiovascular events [10].
The aim of our study was to explore the performance of NT-proBNP and sST2 in patients with and without CKD, and to provide a valuable basis for the diagnosis of HF.
Material and Methods
STUDY POPULATION:
Our study recruited 420 patients admitted to the First Affiliated Hospital of Nanjing Medical University from 2018 to 2022. Among them, the reasons for hospitalization included: coronary atherosclerotic heart disease, arrhythmias, myocarditis, congenital heart disease, and others. According to the European Society of Cardiology (ESC) Guideline on Chronic Heart Failure 2021 [11], patients were divided into 2 subgroups: the no heart failure group (No-HF) and the heart failure group (HF). Renal function is routinely measured via glomerular filtration rate (GFR) [12], with CKD defined by the Kidney Disease Improving Global Outcomes (KDIGO) 2012 guidelines as GFR less than 60 mL/min/1.73 m2 for 3 months or more [13,14]. The GFR was estimated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation [15]. The exclusion criteria were as follows: (1) severe, or active infection and malignancies; (2) significant valvular heart disease; (3) mental or cognitive disorders; (4) autoimmune disease and malignant tumors.
BLOOD SAMPLES AND BIOMARKER ANALYSIS:
All participants provided a morning fasting blood sample that was collected in tubes containing heparin and centrifuged at 4000 r/min for 15 min to separate plasma. The separated plasma was packed and stored at −80°C for further analyses. NT-proBNP and sST2 were measured using enzyme-linked immunosorbent assay (ELISA) (Xuanke Biological Co., Shanghai, China) kits and performed according to the manufacturer’s instructions. Preparation of reagents and measurements were performed according to the manufacturer’s instructions. Baseline data, including biochemical parameters and clinical information, were recorded.
ECHOCARDIOGRAPHY MEASUREMENT:
Echocardiography was performed for all enrolled patients using the model VividE9 (GE Healthcare Systems, Norway). We chose to use 4 measurements of cardiac structure: left atrium dimension (LAD), left ventricular end-diastolic dimension (LVEDD), left ventricular end-systolic dimension (LVESD), and left ventricular ejection fraction (LVEF). The examinations were run by an experienced technician in a quiet room at constant temperature, who was blinded to all the other clinical data analyzed in the present study.
STATISTICAL ANALYSIS:
All statistical analyses were performed using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA) and MedCalc Software (version 20.1, Ostend, Belgium). Baseline continuous variables are expressed as the median and interquartile range (IQR), and comparisons between 2 groups were performed by the Mann-Whitney test. Categorical variables are expressed as numbers and percentages and compared using the χ2 (chi-square) test. Correlations were calculated by Spearman rank correlation. Receiver operating characteristic (ROC) curves were used to analyze the diagnostic ability of sST2, NT-proBNP, and sST2 combined with NT-proBNP to distinguish patients with HF. The most appropriate cutoff values for sST2 and NT-proBNP were obtained by calculating Youden’s index, and Youden’s index=sensitivity – (1-specificity). The longitudinal axis of the ROC curve is sensitivity, and the transverse axis is (1-specificity). Positive predictive values (PPV) and negative predictive values (NPV) were calculated to further evaluate the test performance. ROC curves were analyzed using the method proposed by DeLong et al [16]. A 2-sided P<.05 was considered statistically significant for all analyses.
Results
BASELINE CHARACTERISTICS OF THE STUDY POPULATION:
The characteristics of the study population are shown in Table 1. This study included a total of 420 patients in 2 groups: the no heart failure (N=182) and heart failure (N=238) groups. HF was significantly associated with older age, lower eGFR, higher plasma biomarker concentrations, hypertension, diabetes, worse LVEF, and greater LAD, LVEDD, and LVESD.
RELATIONSHIPS BETWEEN NT-PROBNP, SST2, AND HF:
sST2 levels were significantly higher in patients with HF [median 37.59, IQR (26.25, 58.50) ng/mL] than in those without HF [median 20.62, IQR (13.99, 27.79) ng/mL]. Similar associations were observed for NT-proBNP [median 5086.00, IQR (1589.00, 9000.00) pg/mL] for HF and [median 329.50, IQR (131.25, 1401.50) pg/mL] for No-HF. Both were statistically significant (P<0.001) (Figure 1).
RELATIONSHIPS BETWEEN NT-PROBNP, SST2 AND RNAL FUNCTION:
We found that plasma concentrations of NT-proBNP in our patients were inversely and strongly correlated with eGFR (r=−0.525, P<0.001). There was a statistically significant but weak association between sST2 and eGFR (r=−0.147, P<0.05) (Figure 2).
DIAGNOSTIC VALUE OF PLASMA NT-PROBNP AND SST2 FOR HF:
The area under the curve (AUC) and optimal cutoffs are given in Table 2. The AUCs of sST2 and NT-proBNP in Total group, No-CKD group, and CKD group were as follows: AUC: 0.797 (95% CI, 0.756–0.835, P<0.001), AUC: 0.788 (95% CI, 0.726–0.842, P<0.001), AUC: 0.802 (95%CI, 0.742–0.853, P<0.001) and AUC: 0.813 (95% CI, 0.772–0.849, P<0.001), AUC: 0.805 (95% CI, 0.744–0.858, P<0.001), AUC: 0.807 (95% CI, 0.748–0.857, P<0.001). The optimal cutoff values for sST2 and NT-proBNP to detect HF were 28.96 ng/mL and 1280 pg/mL, respectively, in total, 28.71 ng/mL and 481 pg/mL, respectively, in non-CKD patients, and 30.55 ng/mL and 3314 pg/mL, respectively, in CKD patients. The ROC curves for sST2 plus NT-proBNP were also constructed according to 3 forms. The AUCs were 0.857 (95% CI, 0.820–0.889, P<0.001), 0.861 (95% CI, 0.805–0.905, P<0.001) and 0.854 (95% CI, 0.800–0.899, P<0.001), respectively. The test performance of each model showed beneficial results, with a high PPV (range 0.767 to 0.886) and a moderate NPV (range 0.656 to 0.781), especially when the sST2 and NT-proBNP were combined. The results of pairwise comparison of ROC curves are shown in Table 3. Clearly, the AUC for the combined biomarkers was superior to the AUC of NT-proBNP and sST2 alone in diagnosing HF, and the difference was statistically significant (Figure 3).
Discussion
Cardiovascular biomarkers play an essential role in the diagnosis and assessment of HF. Given the widespread availability, high sensitivity, and low expenses, NT-proBNP may be a better biomarker for the evaluation of patients with suspected heart failure. Today, the measurement of NT-proBNP has been included in clinical guidelines, with class I recommendations in both the HF guidelines of the ESC and the joint HF guidelines of the American College of Cardiology Foundation and the American Heart Association (ACCF/AHA) [11,17]. However, there remains controversy regarding the diagnostic utility of NT-proBNP in CVDs. Several studies revealed that these peptides are susceptible to many factors, such as age, sex, and body mass index (BMI) [18], among which kidney function may be particularly important. Impaired renal clearance may affect circulating levels of biomarkers [19], and the upper reference limit was originally derived from patients without CKD. Accordingly, it has been proposed that the optimum cutoff points of NT-proBNP to diagnose HF were much higher in CKD patients than in non-CKD patients, and these values increased progressively with declining kidney function. Therefore, it is absolutely necessary to ascertain new biomarkers assisting NT-proBNP in the diagnosis of HF.
Recently, several promising biomarkers have emerged, including sST2 [20], which can add new clinical information in patients with heart failure. sST2 is a member of the interleukin (IL) 1 receptor family, with 2 main isoforms: a membrane-bound (ST2 L) and a soluble form (sST2) [21]. When IL-33 binds ST2 L in the heart, downstream signaling is believed to protect against hypertrophy and fibrosis [22]. As a decoy receptor, sST2 binds to IL33 to attenuate the cardioprotective effect [23]. Unlike NT-proBNP, sST2 is neither predominantly from the heart nor cardiac-specific [24]. Pascual-figal et al [25] suggested that the lungs, especially alveolar epithelial type II lung cells, are a relevant source of sST2. Inflammation, fibrosis, and pulmonary congestion are hallmarks of HF syndrome, which may help to explain the role of sST2 in the pathogenesis of HF [26]. On this basis, a large number of clinical studies have shown that sST2 has important value in the diagnosis and prognosis of heart failure, and can be used as a supplementary means besides NT-proBNP and echocardiography. Compared with NT-proBNP, sST2 was not affected by age, kidney function, or body mass index. In a cohort study of 876 patients with heart failure whose primary endpoints were all-cause death, cardiovascular death, and heart failure re-hospitalization, followed for an average of 4.2 years, only sST2 was independently associated with cardiovascular death. Moreover, the long-term independent prognostic value of sST2 in heart failure exceeded 11 traditional risk factors such as age, sex, eGFR and left ventricular ejection fraction [27]. Indeed, sST2 is likely to be the biomarker whose diagnostic performance is least affected by kidney function, implying potential viability in the management of patients with CVD and concomitant chronic renal dysfunction. However, sST2, as a substance related to inflammation and immune response, exists in a variety of cells throughout the body, and its specificity in diagnosing heart failure is not ideal. In a study recruiting patients with HF and other extracardiac pathologies, sST2 could not distinguish HF from pneumonia, chronic obstructive pulmonary disease (COPD), or sepsis [28]. Therefore, sST2 and NT-proBNP can complement each other, and the combined detection of 2 biomarkers can improve the clinical value in patients with HF.
Among the 420 participants in our cohort, we found that circulating levels of sST2 and NT-proBNP were robustly influenced by the presence or absence of HF, with statistically significant differences. They may be indicative of distinct biologic pathways for HF. Progressively higher NT-proBNP concentrations seem to more closely reflect left ventricular wall stress. While sST2 is stimulated by myocardial strain, myocardial fibrosis and ventricular remodeling involved in sST2 may be related to the decline in cardiac function in CKD patients [29,30].
Our study also showed an association between renal function and circulating levels of biomarkers in the analysis. As expected, NT-proBNP increased with deteriorating renal function and was inversely correlated with eGFR (r=−0.525,
Receiver operating characteristic analysis of the ability of each biomarker to detect HF was performed by different forms. The results showed that NT-proBNP had good diagnostic performance (AUC=0.805) and that the 481 pg/mL decision cutoff was suitable for detection in HF patients with normal kidney function, whereas the decision cutoff may need to be increased to 3314 pg/mL in patients with renal dysfunction to maintain discriminability. We thus conclude that the optimum cut points of NT-proBNP should be stratified according to renal function. Similar to our findings, prior studies have also confirmed that the optimal cutoff of NT-proBNP varies quite widely based on renal function [33]. A meta-analysis reported that the AUC for NT-proBNP ranged from 0.66 to 0.89 with a median cutoff point of 1980 pg/mL in patients with renal dysfunction, while the AUC ranged from 0.72 to 0.95 with a cutoff point of 450 pg/mL in patients with preserved renal function [34]. In contrast, we have also shown that the diagnostic cutoff values of sST2 did not change much and that sST2 as a potential diagnostic marker yielded independent diagnostic performance for the diagnosis of HF. Aldous et al [35] first tested ST2 as a diagnostic marker and noted that the sensitivity and specificity of ST2 in the diagnosis of acute heart failure were 73.5% (55.8–86.4%) and 79.6% (79.0–80.1%), respectively. Both this research and ours suggest that ST2 is an independent predictor of heart failure and has incremental utility in combination with BNP. In further analysis, we found that combined sST2 and NT-proBNP showed a higher AUC and better overall sensitivity and specificity in diagnosing HF than sST2 or NT-proBNP alone. The diagnostic ability of the sST2 and NT-proBNP biomarker model was better than that of the single model and obtained greater positive predictive values (PPV) and negative predictive values (NPV). These results mean that the combination of sST2 plus NT-proBNP has higher diagnostic accuracy.
There are some limitations to this study. We studied a relatively small cohort of patients from a single medical center. After our preliminary analysis, the relevant data could not distinguish between heart failure with ejection fraction retention and heart failure with ejection fraction reduction, so the types of heart failure were not classified. The clinical combination of our indicators aims to improve the scientific nature of clinical qualitative judgment. In addition, the subjects included in this study were hospitalized patients who underwent sST2 testing in some departments, so our results may not be generalizable to all patients, and the experimental results may be biased.
Conclusions
In conclusion, our research clearly demonstrated that NT-proBNP had a significant upward trend as eGFR declined, while sST2 was much less affected by renal function. Moreover, it is recommended to use NT-proBNP combined with sST2 for cardiac function assessment in CKD patients, and different diagnostic thresholds of NT-proBNP should be considered based on eGFR.
Figures
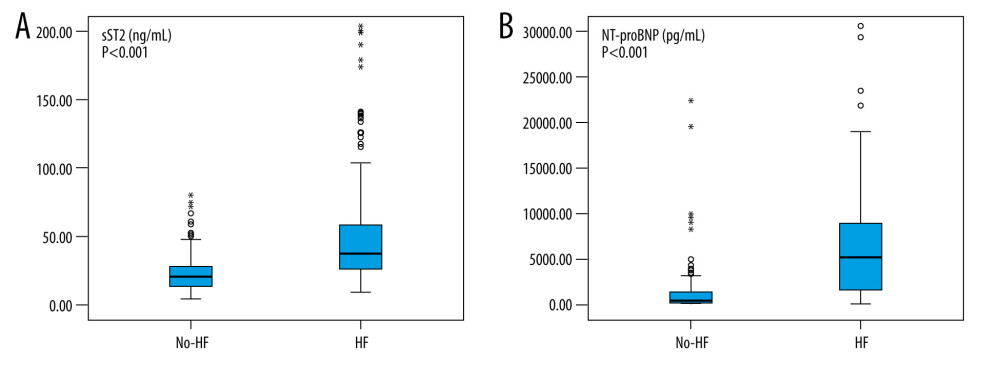 Figure 1. Box plot of plasma concentrations of sST2 (A) and NT-proBNP (B) in patients with and without heart failure. Data are presented as medians with 25th and 75th percentiles. P<0.05 was considered statistically significant. This figure was created using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA).
Figure 1. Box plot of plasma concentrations of sST2 (A) and NT-proBNP (B) in patients with and without heart failure. Data are presented as medians with 25th and 75th percentiles. P<0.05 was considered statistically significant. This figure was created using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA). 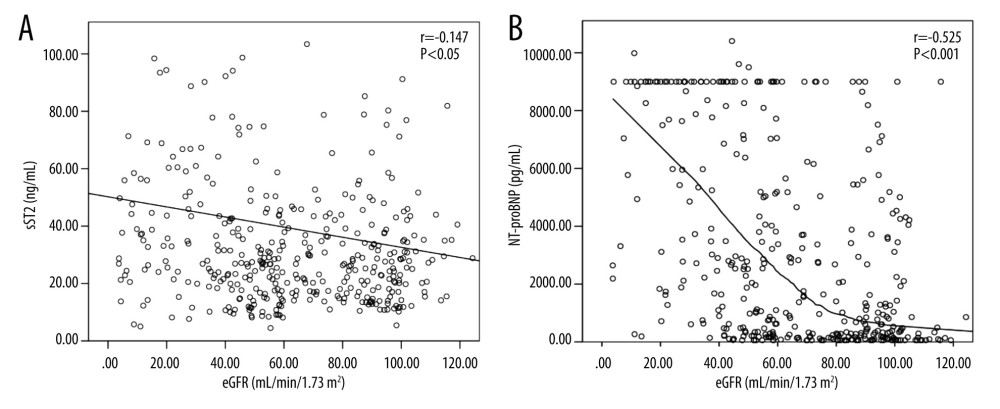 Figure 2. Scatter plots of sST2 (A) and NT-proBNP (B) serum concentrations according to estimated glomerular filtration rate (eGFR) values. Black curve line performed by Loess adjustment. This figure was created using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA).
Figure 2. Scatter plots of sST2 (A) and NT-proBNP (B) serum concentrations according to estimated glomerular filtration rate (eGFR) values. Black curve line performed by Loess adjustment. This figure was created using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA). 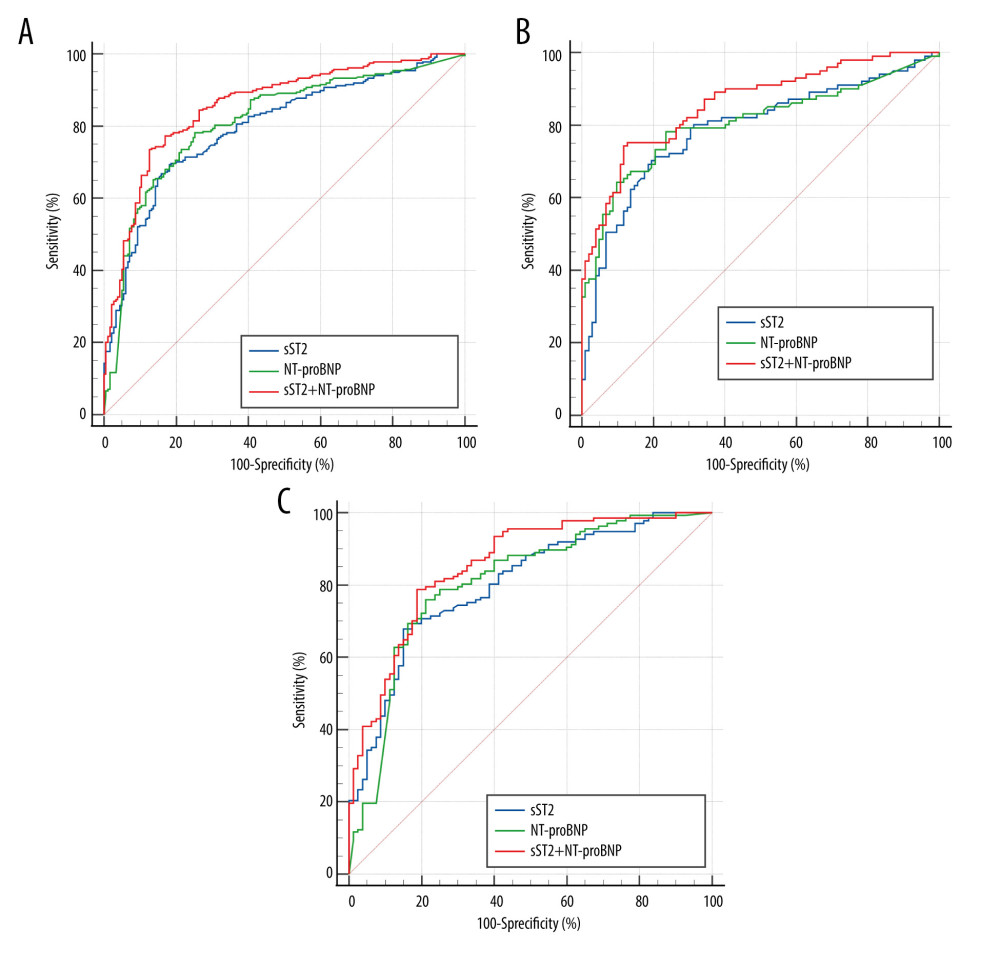 Figure 3. Receiver operating characteristic (ROC) curves of sST2 and NT-proBNP to diagnose HF in the Total (A), No-CKD (B), and CKD (C) groups. This figure was created using MedCalc Software (version 20.1, Ostend, Belgium).
Figure 3. Receiver operating characteristic (ROC) curves of sST2 and NT-proBNP to diagnose HF in the Total (A), No-CKD (B), and CKD (C) groups. This figure was created using MedCalc Software (version 20.1, Ostend, Belgium). References
1. Roger V, Epidemiology of heart failure: a contemporary perspective: Circ Res, 2021; 128(10); 1421-34
2. Chamberlain A, Boyd C, Manemann S, Risk factors for heart failure in the community: differences by age and ejection fraction: Am J Med, 2020; 133(6); e237-e48
3. Tedeschi A, Agostoni P, Pezzuto B, Role of comorbidities in heart failure prognosis Part 2: Chronic kidney disease, elevated serum uric acid: Eur J Prev Cardiol, 2020; 27(2 Suppl); 35-45
4. Van Nuys K, Xie Z, Tysinger B, Innovation in heart failure treatment: Life expectancy, disability, and health disparities: JACC Heart failure, 2018; 6(5); 401-9
5. Sud M, Tangri N, Pintilie M, Levey AS, Naimark D, Risk of end-stage renal disease and death after cardiovascular events in chronic kidney disease: Circulation, 2014; 130(6); 458-65
6. Gansevoort RT, Correa-Rotter R, Hemmelgarn BR, Chronic kidney disease and cardiovascular risk: Epidemiology, mechanisms, and prevention: Lancet, 2013; 382(9889); 339-52
7. Schefold JC, Filippatos G, Hasenfuss G, Heart failure and kidney dysfunction: Epidemiology, mechanisms and management: Nat Rev Nephrol, 2016; 12(10); 610-23
8. Isakova T, Nickolas T, Denburg M, KDOQI US Commentary on the 2017 KDIGO clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD): Am J Kidney Dis, 2017; 70(6); 737-51
9. Porter AC, Lash JP, Xie D, Predictors and outcomes of health-related quality of life in adults with CKD: Clin J Am Soc Nephrol, 2016; 11(7); 1154-62
10. Wang TJ, Wollert KC, Larson MG, Prognostic utility of novel biomarkers of cardiovascular stress: The Framingham Heart Study: Circulation, 2012; 126(13); 1596-604
11. McDonagh TA, Metra M, Adamo M, 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Eur Heart J, 2021; 42(36); 3599-726
12. Murton M, Goff-Leggett D, Bobrowska A, Burden of chronic kidney disease by KDIGO categories of glomerular filtration rate and albuminuria: A systematic review: Adv Ther, 2021; 38(1); 180-200
13. Chen T, Knicely D, Grams M, Chronic kidney disease diagnosis and management: A review: JAMA, 2019; 322(13); 1294-304
14. Stevens P, Levin A, Evaluation and management of chronic kidney disease: Synopsis of the kidney disease: Improving global outcomes 2012 clinical practice guideline: Ann Intern Med, 2013; 158(11); 825-30
15. Levey A, Stevens L, Schmid C, A new equation to estimate glomerular filtration rate: Ann Intern Med, 2009; 150(9); 604-12
16. DeLong ER, DeLong DM, Clarke-Pearson DL, Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach: Biometrics, 1988; 44(3); 837-45
17. Heidenreich PA, Bozkurt B, Aguilar D, 2022 AHA/ACC/HFSA Guideline for the management of heart failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines: Circulation, 2022; 145(18); e895-e1032
18. Mueller C, McDonald K, de Boer RA, Heart Failure Association of the European Society of Cardiology practical guidance on the use of natriuretic peptide concentrations: Eur J Heart Fail, 2019; 21(6); 715-31
19. Takase H, Dohi Y, Kidney function crucially affects B-type natriuretic peptide (BNP), N-terminal proBNP and their relationship: Eur J Clin Invest, 2014; 44(3); 303-8
20. Emdin M, Mirizzi G, Pastormerlo L, The search for efficient diagnostic and prognostic biomarkers of heart failure: Future Cardiol, 2016; 12(3); 327-37
21. Pascual-Figal D, Januzzi J, The biology of ST2: The International ST2 Consensus Panel: Am J Cardiol, 2015; 115; 3B-7B
22. Daniels L, Bayes-Genis A, Using ST2 in cardiovascular patients: A review: Future Cardiol, 2014; 10(4); 525-39
23. Wettersten N, Maisel A, Biomarker developments in heart failure: 2016 and beyond: Curr Opin Cardiol, 2019; 34(2); 218-24
24. Du W, Piek A, Schouten E, Plasma levels of heart failure biomarkers are primarily a reflection of extracardiac production: Theranostics, 2018; 8(15); 4155-69
25. Pascual-Figal D, Pérez-Martínez M, Asensio-Lopez M, Pulmonary production of soluble ST2 in heart failure: Circ Heart Fail, 2018; 11(12); e005488
26. Meijers W, Bayes-Genis A, Mebazaa A, Circulating heart failure biomarkers beyond natriuretic peptides: Review from the Biomarker Study Group of the Heart Failure Association (HFA), European Society of Cardiology (ESC): Eur J Heart Fail, 2021; 23(10); 1610-32
27. DeVeale B, Bausch-Fluck D, Seaberg R, Surfaceome profiling reveals regulators of neural stem cell function: Stem Cells, 2014; 32(1); 258-68
28. Lotierzo M, Dupuy AM, Kalmanovich E, sST2 as a value-added biomarker in heart failure: Clin Chim Acta, 2020; 501; 120-30
29. Emdin M, Aimo A, Vergaro G, sST2 predicts outcome in chronic heart failure beyond NT-proBNP and high-sensitivity troponin T: J Am Coll Cardiol, 2018; 72(19); 2309-20
30. Stein NR, Zelnick LR, Anderson AH, Associations between cardiac biomarkers and cardiac structure and function in CKD: Kidney Int Rep, 2020; 5(7); 1052-60
31. Bayes-Genis A, Zamora E, de Antonio M, Soluble ST2 serum concentration and renal function in heart failure: J Card Fail, 2013; 19(11); 768-75
32. Plawecki M, Morena M, Kuster N, sST2 as a new biomarker of chronic kidney disease-induced cardiac remodeling: Impact on risk prediction: Mediators Inflamm, 2018; 2018; 3952526
33. Sato Y, Diagnostic and prognostic property of NT-proBNP in patients with renal dysfunction: J Cardiol, 2013; 61(6); 446-47
34. Schaub JA, Coca SG, Moledina DG, Amino-terminal Pro-B-Type natriuretic peptide for diagnosis and prognosis in patients with renal dysfunction: A systematic review and meta-analysis: JACC Heart Fail, 2015; 3(12); 977-89
35. Aldous SJ, Richards AM, Troughton R, Than M, ST2 has diagnostic and prognostic utility for all-cause mortality and heart failure in patients presenting to the emergency department with chest pain: J Card Fail, 2012; 18(4); 304-10
Figures
 Figure 1. Box plot of plasma concentrations of sST2 (A) and NT-proBNP (B) in patients with and without heart failure. Data are presented as medians with 25th and 75th percentiles. P<0.05 was considered statistically significant. This figure was created using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA).
Figure 1. Box plot of plasma concentrations of sST2 (A) and NT-proBNP (B) in patients with and without heart failure. Data are presented as medians with 25th and 75th percentiles. P<0.05 was considered statistically significant. This figure was created using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA). Figure 2. Scatter plots of sST2 (A) and NT-proBNP (B) serum concentrations according to estimated glomerular filtration rate (eGFR) values. Black curve line performed by Loess adjustment. This figure was created using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA).
Figure 2. Scatter plots of sST2 (A) and NT-proBNP (B) serum concentrations according to estimated glomerular filtration rate (eGFR) values. Black curve line performed by Loess adjustment. This figure was created using SPSS software (version 23.0; IBM SPSS, Armonk, NY, USA). Figure 3. Receiver operating characteristic (ROC) curves of sST2 and NT-proBNP to diagnose HF in the Total (A), No-CKD (B), and CKD (C) groups. This figure was created using MedCalc Software (version 20.1, Ostend, Belgium).
Figure 3. Receiver operating characteristic (ROC) curves of sST2 and NT-proBNP to diagnose HF in the Total (A), No-CKD (B), and CKD (C) groups. This figure was created using MedCalc Software (version 20.1, Ostend, Belgium). Tables
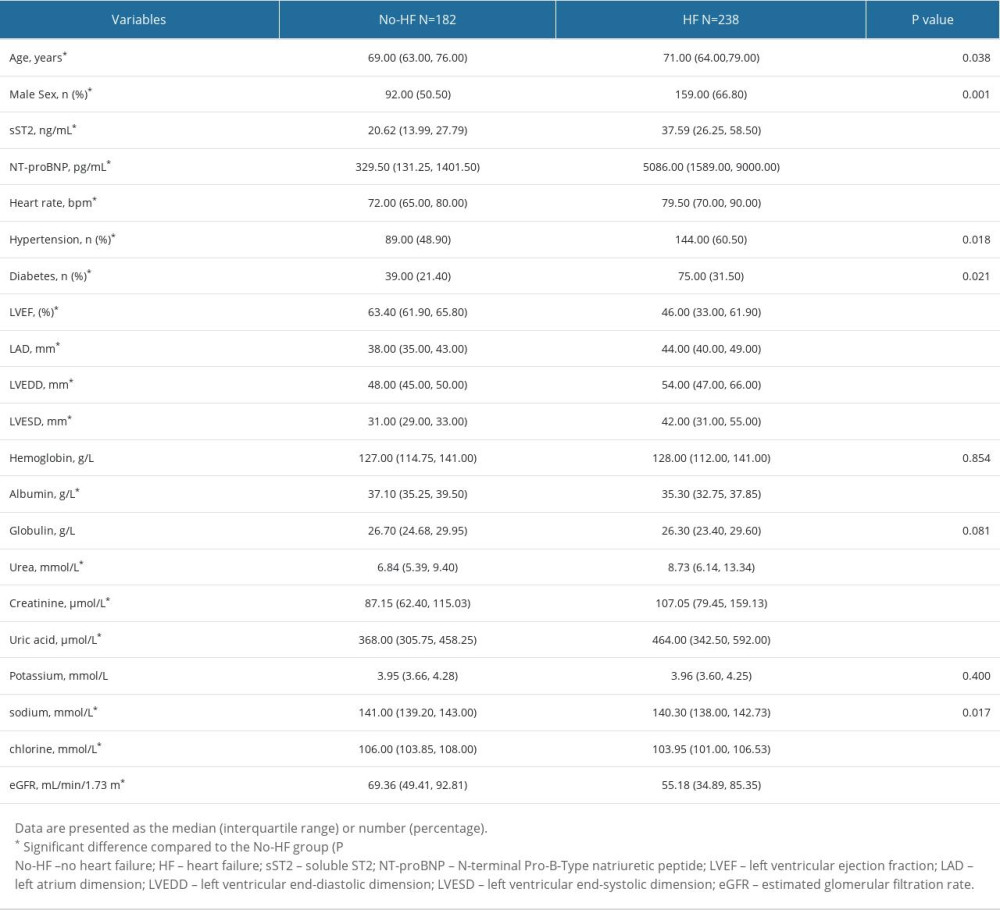 Table 1. Baseline characteristics.
Table 1. Baseline characteristics.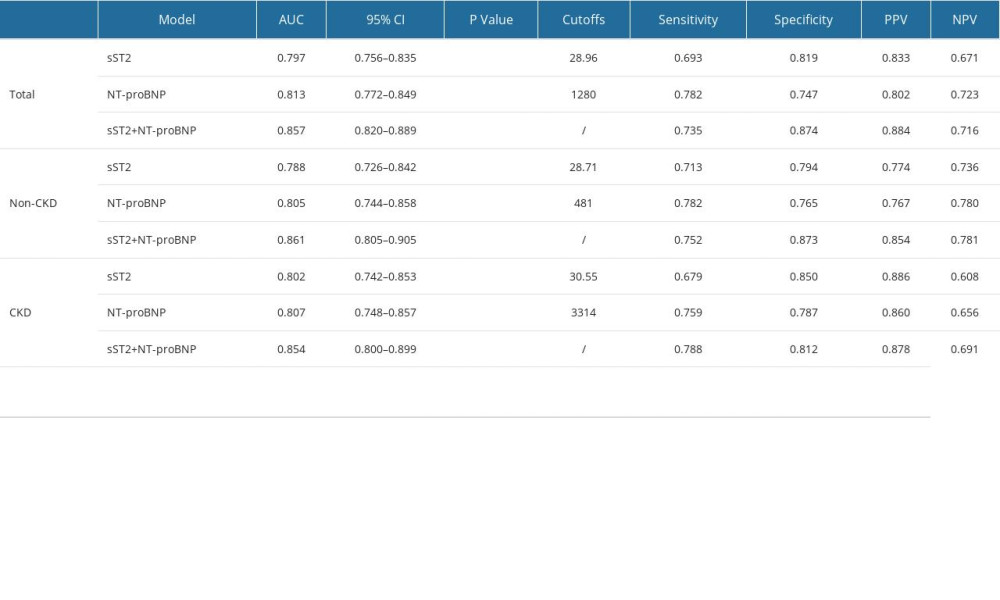 Table 2. The AUCs and optimal cutoffs of sST2, NT-proBNP and 2 combinations in the Total, No-CKD, and CKD groups.
Table 2. The AUCs and optimal cutoffs of sST2, NT-proBNP and 2 combinations in the Total, No-CKD, and CKD groups.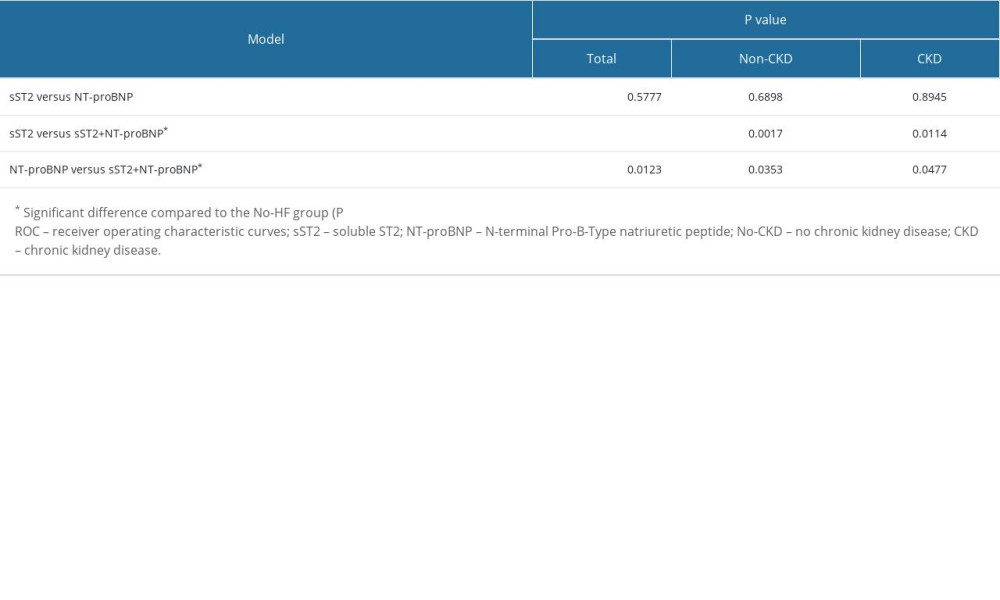 Table 3. Pairwise comparison of ROC curves (DeLong test).
Table 3. Pairwise comparison of ROC curves (DeLong test). Table 1. Baseline characteristics.
Table 1. Baseline characteristics. Table 2. The AUCs and optimal cutoffs of sST2, NT-proBNP and 2 combinations in the Total, No-CKD, and CKD groups.
Table 2. The AUCs and optimal cutoffs of sST2, NT-proBNP and 2 combinations in the Total, No-CKD, and CKD groups. Table 3. Pairwise comparison of ROC curves (DeLong test).
Table 3. Pairwise comparison of ROC curves (DeLong test). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








