20 February 2024: Lab/In Vitro Research
Influence of Chairside Simulated Adjustment (Finishing and Polishing) Protocol and Chlorhexidine Mouthwash Immersion on Color Stability and Translucency of 2 and 3 Preshaded Multilayered Monolithic Zirconia
Mohammed E. SayedDOI: 10.12659/MSM.943404
Med Sci Monit 2024; 30:e943404
Abstract
BACKGROUND: Preshaded monolithic zirconia (MLZ) is reported to have high translucency. This study aimed to assess the effect of chlorhexidine gluconate (ChG) mouthwash on color and translucency parameter (TP) of 2 different preshaded MLZ dental ceramics after clinical adjustment.
MATERIAL AND METHODS: Two MLZ disk-shaped specimens [NPM (Nacera Pearl Multi-Shade) (n=72) and CZM (Ceramill Zolid FX Multilayer)] (n=72) were simulated for clinical adjustment, finished, and polished using 2 adjustment kits [recommended kit, third-party kit: Diasynt Plus and SUN (n=12 each)] and later immersed in ChG mouthwash (Avohex) for 2 weeks. Difference in color (ΔE) and TP (Y) were calculated using the CIELab formula after measuring the coordinates (Lab) with a colorimeter. Individual changes in color and TP were assessed on the Clinical acceptance (perceptible) threshold (CAT/CPT) and Translucency perception threshold (TPT), respectively. Differences between the 2 ceramics were assessed using one-way ANOVA and post hoc tests, with all differences considered significant at P<0.05.
RESULTS: NPM and CZM differed in color at baseline despite having the same Vita shade combination. Between the 2 preshaded MLZ ceramics, NPM showed significant changes in color when adjusted with a third-party kit. Chlorhexidine produced changes in color and TP that were designated as clinically perceptible (ΔE=1.0 to 3.3) on the CAT/CPT and TPT scales, irrespective of the adjustment kit used. ChG produced the least or no changes in glazed MLZ specimens.
CONCLUSIONS: ChG mouthwash, whenever prescribed for preshaded MLZ restoration, should be adjusted prior to final glazing to avoid clinical adjustments that adversely affects color and translucency of the restoration.
Keywords: Computer-Aided Design, Dental Porcelain, Prosthesis Coloring, Tooth Crown, Yttria Stabilized Tetragonal Zirconia, Color, Chlorhexidine, Mouthwashes, Immersion, Materials Testing, Surface Properties, Ceramics, Zirconium
Background
Prosthetic dentistry is a sizable and profitable health industry, where minor developments have large and lucrative consequences for providers and users. Despite being prone to brittleness [1] and having limited use for small anterior/posterior restorations [2], dental ceramics have been extensively studied in the past few decades. Zirconia, with its polymorphic crystalline structure (monoclinic, tetragonal, and cubic), which stabilizes at various temperatures through transformation toughening, are fast replacing the conventional alloy-backed metal ceramic restorations [3]. Introduction of digital manufacturing [computer-aided diagnosis, computer-assisted machining (CADCAM)], and novel fabrication materials [disilicate ceramics, resin nanoceramics and polymer infiltrated ceramic network] have further enhanced their commercial potential due to shorter processing time and economical maintenance [3–5]. When fully contoured, conventional zirconia results in opaque restorations (inconsistent refractive indices of matrix and filler) despite being strong and having excellent biocompatibility and mechanical properties (flexural strength, bending strength) [6]. To use its strength and yet match clinical aesthetic outcomes, zirconia restorations were veneered with translucent porcelain, which solves the problem of chipping/fracture of exposed unglazed porcelain, resulting in antagonistic tooth wear [7]. Monolithic restorations made of silicate ceramics with high translucency and etching ability offered clinical advantages over veneered zirconia [8]. Simultaneously, CAD/CAM technology made monolithic zirconia (MLZ) restorations feasible, overcoming the chipping/fracture problem [8]. Reduction/exclusion of alumina (Al2O3) with concomitant increase of stabilizing oxides like yttrium oxide (Y2O3) resulted in development of partially stabilized zirconia (PSZ – Y2O3 between 4 to 6 mol%) and fully cubic stabilized zirconia (FSZ – Y2O3 more than 8 mol%) [9,10]. Stabilizing oxides, besides converting zirconia volume contraction to expansion [4,7], additionally improved zirconia translucency [4,6,9,10]. Novel processing techniques (brush, immersion, staining, cutback, and layering) and high flexural strength for MLZ [11] shifted the trend from translucent monolithic disilicate ceramics, which required more ceramic thickness (1.5 to 2 mm occlusal) in terms of conservation of natural tooth structure [12]. Color-matching ability and long-lasting color stability influence restorative material choices. In addition to color (shade), translucency imparts a life-like appearance to an artificial restoration. Characterization and opalescence are additional requirements that clinicians desire to have while selecting a restorative material [13]. Translucency of a material is based on composition, microstructure, component individual characteristics like particle size, and refractive index [3,14]. Evaluating translucency clinically using eye vision is extremely complex and difficult, hence one needs in vitro assessment under simulated clinical conditions. Translucency of a restoration decreases with aging and staining (beverage consumption) [15]. Surface porosity and defects that increase incident light scattering on the surface diminish translucency more than other factors [16]. The aging-related surface deterioration in 3Y-TZP (low-temperature degradation [LTD]) [14,17] is slowed by the addition of a small amount of alumina. LTD gradually transforms the stable tetragonal phase to the unstable monoclinic phase once zirconia surface comes into contact with water or body fluids at body temperature [14,18]. Kurt et al compared lithium disilicate (IPS e.max Press) and MLZ (Zirkonzahn Pretta) subjected to accelerated aging (water spray and ultraviolet light for 300 h) and reported MLZ to be inferior to lithium disilicate ceramic in terms of translucency and color stability [19]. Fathy et al found MLZ to be at higher risk of LTD than core zirconia [20]. The non-conservative nature of lithium disilicates (monolithic) compelled development of multilayered (multishaded) MLZ CADCAM restorations. Multi-preshaded MLZ (3Y-TZP-A or 3Y-TZP) eliminates zirconia coloring/staining after sintering for translucency, besides representing a wide shade spectrum of Vita shade guides (16 VITA and 2 bleach shades). Industrial production allows smooth transition in translucency from incisal to cervical, and introduced 2 or more shades in a blank [21]. The translucency of multilayered MLZ (Ceramill Zolid FX Multilayer) has also been reported to have higher translucency with lower contrast ratio when compared to other MLZ types (Prettau Anterior, Zenostar T) [22].
MLZ processing is associated with volumetric changes (linear sintering shrinkage) that approximates 15% to 30%, which requires them to be designed to be larger before sintering [23,24]. Clinically, these errors are reflected as high occlusion and bulky/missing contact points that need to be adjusted [25]. Surface modification kits provided by manufacturers are specific for the product they market. Separate kits with specific abrasives for finishing and polishing of MLZ have been investigated. Hatanka et al investigated surface roughness and flexural strength after adjusting MLZ and concluded that unlike other zirconia-based ceramics, glazing after finishing must be avoided, thus stressing the importance of polishing zirconia after clinical adjustment [26]. Chun et al found that after clinical adjustment of MLZ (diamond bur/glazing/polishing), glazing and polishing improve survival chances more than sintered zirconia [27]. Additionally, zirconia is polycrystalline, and surface alteration intensifies its opacity with resultant rough surface, introducing scattering, which diminishes translucency [28]. Surface adjustments indirectly affect color and translucency after bacterial adhesion [29] and consumption of dark-colored beverages (tea, coffee, wine, soft drinks) [30]. Pre- and post-sintering polishing pastes decrease translucency due to their abrasive content (silicium carbide, alumnia oxide, zirconia) [31] and binders [32]. Changes in color and translucency thus are both intrinsic and extrinsic in nature, affecting MLZ either short or long term.
In conventional fixed partial denture treatments and implant-supported crowns, clinicians prescribe additional mandatory oral hygiene aids to maintain periodontium health. Chlorhexidine gluconate (ChG) mouthwash restricts bacteria-associated periodontal disease and caries causing microorganisms. It is recommended for a short period of 1 to 2 weeks [33], as long-term use develops extrinsic staining in natural teeth and restorations [34]. Alnassar explored the consequence of chlorhexidine on MLZ (Cercon) that were colored to VITA A2 shade before and after sintering with abrasion being simulated using toothbrush and toothpaste. Results showed color changes after 2 weeks to be clinically perceptible (DE: 3.60) [35]. Derafshi et al, while investigating color stability of feldspathic-layered MLZ, found significantly higher color changes with chlorhexidine and Listerine mouth rinses [36]. Layered MLZ (milled), when compared to lithium disilicate (pressable), tends to be inferior in color stability in the presence of chlorhexidine and coffee [37]. Multi-shade MLZ does not involve layering of porcelain and is not colored at any stage. To the best of our knowledge, the influence of chlorhexidine mouthwash on multi-shade MLZ after clinical adjustment has never been reported. This study therefore investigated the influence of chlorhexidine mouthwash on the color and translucency of 2 different preshaded MLZ (Nacera Pearl Multi-Shade [NPM] and Ceramill Zolid FX Multilayer [CZM]) after in vitro simulation of clinical chairside adjustment with respective adjustment kits (Diasynt Plus, Diacera, and SUN). Additionally, the study design allowed us to assess the impact of the third-party clinical adjustment kit (not the first recommendation) on the same parameters. The objectives of the study were to establish the correct chairside adjustment technique for these 2 ceramics, recommendation of chlorhexidine mouthwash, and the effects of using adjustment kits recommended by the manufacturers. For baseline color and TP between 2 independent preshaded MLZ, we hypothesized there would be differences between the ceramics (NPM being PSZ while CZM is FSZ). For differences between manufacturer-recommended and third-party adjustment kits, we hypothesized that there would be no differences in color and TP. For influence of chlorhexidine immersion on color/TP for 2 weeks, we hypothesized there would be changes in both parameters because of the nature of the coloring immersion. For determining the best technique that produces little of no changes in color and TP, we hypothesized that glazing would have the least changes. Alternately, the null hypothesis was that there would be that no differences in color and TP at baseline and after adjustment with either the adjustment kit or chlorhexidine immersion.
Material and Methods
ETHICS:
This in vitro research was duly permitted by the research review committee at College of Dentistry, Jazan University (Reference number: CODJU-2207F). The materials during the course of this study fall in the category that are approved by the regulations of the Saudi food and drug authority (SFDA).
STUDY DESIGN:
The research was designed to follow a comparative experimental approach that included 2 dependant variables (color and translucency) and 2 distinct independent variables (surface modification and chlorhexidine immersion) (Figure 1). The bulk of the experiment was completed in the second quarter of year 2023. The 2 distinct stages were fabrication of specimens/planned interventions and measuring color attributes.
OPERATIONAL DEFINITIONS:
The term “color” was defined as the quality of the study specimen with respect to the light reflected and/or transmitted [38]. “Shade” was contextually defined as a particular hue (basic color like yellow, blue) or any of its variation (like bluish green). “Chroma” represents the intensity of the hue and “Value” indicates brightness or darkness [38]. “Contrast ratio” was defined as the ratio obtained for the luminous reflectance of translucent material against the white background to the luminous reflectance of the same material against the black background (Y tristimulus value of CIE) [39,40].
SAMPLE SIZE ESTIMATION: Determining the required sample size for each group was performed using statistical software (nQuery Advisor v7.0; Informer Technologies, Inc). An effect size (D2=.28), type 1 error rate (a=0.05), and assumption study power (80%) based on an earlier study [15], determined the smallest sample size for each group (including control) to be 12 specimens. An extra 2 samples for each cluster were kept as safeguard in case of laboratory processing errors.
: Manufacturer details and their respective codes, specifications, features, and ancillary materials used for the study are presented in Table 1.
: Two multi-shade (multilayer) MLZ: NPM (PSZ) and CZM (FSZ) were investigated for their color and translucency after in vitro simulation of clinical adjustment (finishing and polishing) using commercially available adjustment kits before immersion in artificial saliva and chlorhexidine. The absolute dimensions of the disc-shaped specimen after sintering were kept as per the guidelines (ISO 6872-2015), which was 1.2±0.2 mm in thickness and 14 mm in diameter. Oversized specimens (20%) to compensate sintering shrinkage were milled from respective zirconia block of each manufacturer (Figure 2A). A total of 5 specimens per blank conformed to the study design.
NPM (PSZ) SPECIMENS:
NPM ceramic is available in 2 bleach shades and 16 VITA (A, B, C, D) with each category having a light and dark shade [41]. The shade chosen was NPM-A (light), which represents a combination of VITA A1 and A2. For NPM ceramic, calibration for sintering temperature and specimen dimensions was first done using the calibrating tool (Nacera Calibrate, Doceram Medical GmbH, Germany). A Nacera Pearl shell (Doceram Medical GmbH, Germany) (pre-sintered white fired condition) was placed in the sintering tray and, after running the sintering program, the sintered shell was removed and measured on the gauge and compared with the gray calibration body provided with the calibrate toolkit [41]. The gray calibration body was set to the materials specific value given in the ceramic pack (calibrate to either 64 or 32). After placing the previously sintered shell on the same gauge, the differences between the gray calibration body and the sintered specimen were noted. Correct sintering temperature is considered if the dial gauge is on zero, whereas if the gauge indicates above or below zero it indicates that the sintering temperature is either high or low (0+ = high; 0− = low), which was adjusted accordingly. Each mark on the gauge indicates a difference of 2°C. During sintering of specimens, a cleansing powder (Nacera Clean LT, Doceram Medical GmbH, Germany) was placed under the support in an even layer. The cleansing powder ensures specified color and translucency by binding the residues of coloring liquids and other impurities (metal oxides) that might be present from previous sintering programs using coloring liquids. The powder binds the atmospheric crystal particles while leaving the silicon crystals on the heat conductors, which are transferred into the vitreous glass phase, thus forming a protective layer. The test is verified when the powder turns white at the end of the sintering cycle. This ensures full translucency and non-contamination of the specimens [41]. The designing of the specimens was achieved using recommended software (SolidWorks, Dassault Systems SolidWorks Corp., Waltham, MA) which was later transferred for positioning the samples in each block using other software (3Shape, Copenhagen, Denmark). NPM pre-sintered blocks were milled using manufacturer-recommended systems (imes-icore GmbH lm, Leibolzgraben 16 D-38132 Eiterfeld, Germany) [41]. NPM blocks were milled dry without any coolant and compressed air to ensure there was no reduction in translucency. The positioning of blocks for both ceramics was decided to be in the center of the block, since both color intensity and translucency were desired. The milling strategy for NPM on the CAM software was selected as “highly translucent.” The printed side on the NPM block was kept as the occlusal side (Figure 2A). The milled specimens were separated from the NPM ceramic blanks using suitable recommended tools. The final sintering was conducted by slowly heating the mounts to desired temperatures and cooling them slowly. the NPM specimens were fired from 20 to 1000 degrees with rise of 60 degrees per minute, then to 1530 degrees at 40 degrees per minute followed by holding period for 30 minutes (1530 degrees). The same pattern was followed for cooling. The final color of the sintered specimens was achieved using recommended glaze firing.
CZM (FSZ) SPECIMENS:
Ceramic specimens made of CZM (Ceramill Zolid FX Multilayer, C20 – HT) were also milled dry using open machine (Ceramill Motion 2/5x, #179250S, Amann Girrbach GmbH, Germany). Each blank had the dimensions of 14 mm by 16 mm by 20 mm. although the ceramic uses the intelligent nesting concept to increase the translucency, the central part on the blank was chosen to ensure standardized criteria for both ceramics. Three sintering times (conventional, speed, and high speed) are available in the sintering furnace (Ceramill Therm S, Amann Girrbach, Koblach, Austria), and the mode chosen was conventional for this study. The sintering temperature included raising the temperature from room temperature to final temperature of 1450°C at the rate of 5 to 10 kelvin/minute followed by a dwell time of 2 hours. The cooling phase included bringing down the temperature from 1450 degrees to room temperature at the rate of 5K/minute, which takes approximately 5 hours. Volumetric shrinkage for CZM specimens during sintering was calibrated by adjusting magnification in the software (V1.233, S18.94 and F10.27) that are dependent on the blocks (third-party). For this study, the magnification was set at V1.233, as recommended. Finishing before sintering was accomplished using the green-state finishing kit, and after sintering the sinter-state polishing kit was used. Presintering finish was done first with coarse and then with fine tools. Post-sintering finishing was done with gentle pressure using swivels and cups. Polishing was done using polishing paste (Zolid Allbright). All specimens for both ceramics were visually inspected, and any specimen with shinning area, discoloration, cracks, or material spalling were discarded and replaced. The definitive thickness of each specimen was evaluated before polishing using a micrometer (digital; Accurate up to 0.0001 mm). A thin layer of glaze for the ceramics was applied to the specimen surface with a thin brush and fired in the sintering furnace as per manufacturer recommendations [42]. All specimens for each respective ceramic were glazed to simulate the clinical setting.
:
For all specimens, the surface was ground using a diamond bur (number 40 grit) followed by finishing and polishing with respective manufacturer-recommended chairside clinical adjustment kits (Diasynt Plus, Diacera and SUN). Specifications of each finishing and polishing tool kit (size, number, function) are presented in Table 1. Forty-eight glazed specimens (24 for each ceramic type) served as controls for baseline comparisons. Ninety-six test specimens were subjected to chairside adjustment with finishing and polishing procedure performed by the single blinded operator. To standardize the chairside adjustment the surface of each specimen was adjusted by diamond bur with each application performed at 160 000 rpm with unidirectional motion. Polishing for each specimen was then carried out separately according to manufacturers recommendations. For NPM specimens, polishing was aided with polishing paste (Nacera Shine Zr, Doceram Medical Ceramics Gmbh, Dortmund, Germany). CZM were polished using the polishing kit in which diamond polishers at a speed of 10 000 to 12 000 rpm for swivel and 7000 to 12 000 rpm when a flame-shaped polishing tool was used. For both specimens, polishing was done for 60 seconds. Both ceramic manufacturers recommend a two-stage polishing technique. The polishing technique applied was in the same direction for 30 seconds followed by rotation of the specimen by 90 degrees with sweeping movements continued perpendicular to the previous direction [43]. After polishing, all specimens were rinsed using a combination of air–water spray for 30 seconds and then cleaned ultrasonically (TUC-150, Telsonic AG; Bronschhofen) for 1 minute in triple-distilled water (100%) followed by air drying.
:
After required procedures, 144 specimens were then categorized into 12 different groups that coincided with the study design. The subdivision was based on the type of immersion media (saliva and chlorhexidine). An earlier study guided the preparation of artificial saliva [44], while staining solution (chlorhexidine gluconate solution [Avohex mouthwash, 2% chlorhexidine]) was prepared as per manufacturer recommendations for use in patients. Specimens in each group were immersed in respective solutions for 10 minutes twice daily for 2 weeks (14 days). Based on the ceramic type, surface preparation (glazed, adjusted by kit 1 and 2) and immersion liquid (saliva, chlorhexidine), 12 distinct groups were formed (Figure 1). For NPM, the groups (Gp) were coded as Gp NGS (Control), Gp NGCh (Control), Gp NDS, Gp NDCh, Gp NSS, Gp NSCh and for CZM the groups were coded as Gp CGS (Control), Gp CGCh (Control), Gp CDS, Gp CDCh, Gp CSS, and Gp CSCh. Each letter of the group designates the ceramic type (C – Ceramill, N – Nacera), surface type (G – glazed), adjustment kit (D – Diasynt Plus, S – Sun), and immersion liquid (S – saliva, Ch – chlorhexidine). Therefore, Gp NDCh as an example indicates the ceramic Nacera adjusted by Diasynt Plus and immersed in chlorhexidine.
MEASURES, DATA COLLECTION, EVALUATION, AND ANALYSIS:
A portable colorimeter (Hangzhou CHNSpec Technology Co, Ltd., Hangzhou, China) was used to measure color (Lab), translucency, and contrast ratio parameter. To minimize the effect of light (external, illuminants) on color values, measurements were made in a calibration box (0 calibration standard) against a white background [45]. Calibration of the box was done by first measuring a sample repeatedly 30 times to establish the average. Change in position of colorimeter was standardized by preparing an index that was customized to the colorimeter tip. The index was prepared from silicone elastomer (Speedex, Coltène, Switzerland). Specifications for colorimeter are presented in Table 1, while other specifications included an 8-mm tip diameter, 94.38% accuracy, and 0.877 repeatability. For each group, calibration of colorimeter was performed using the manufacturer-provided tool. For all measurements, the colorimeter tip was placed at the predetermined center of each specimen. Positional uniformity for each specimen was ensured by placing the specimen on the table in the calibration box. To evaluate the color, individual measurements of each CIELAB coordinates (L*, a* and b*) were recorded, where L indicates the value [light=100 (completely white), dark=0 (completely black)], a indicates the chroma (green–red) and b indicates the hue (yellow-blue). For the latter 2 chromatic coordinates, the positive (+) and a negative sign (−) denotes the direction of the color [example C*=green (−C), and red (+C) axis, and H*=blue (−H), and yellow (+H) axis] [46]. Average of 3 readings on a specimen within each group was taken as the mean value for each specimen. Translucency has 3 measurable parameters: contrast ratio, transmittance, and translucency parameter. Since the contrast ratio is a parameter of translucency, its measurement in this study was selected primarily to draw a comparison with other studies. For CR, the range is from 0 to 1, with values near zero representing translucency and near 1 representing opacity. On the translucency perceptible threshold (TPT), a CR (Y) value of 0.07 or less in CR is considered to be invisible to the human eye [47]. CR value is linearly related to translucency parameter, and a larger CR difference means less translucency and vice versa.
STATISTICAL ANALYSIS:
Mean values for each group (n=12) with their standard deviations were obtained at baseline and at 14 days. Color difference was calculated using the CIELAB mean values with difference in color (DE00 or ΔE) using CIELab equation ΔE=[(L1–L2) 2 + (a1 – a2) 2 + (b1–b2) 2]1/2, which yields ΔE= [(ΔL)2 + (Δa)2 + (Δb)2] ½ [39,46], where different numerical values under letter represents the values for each coordinate at baseline and at 14 days. Mean values for each coordinate obtained at baseline were deducted from the mean value at 14 days to denote the color change [39,45,46]. Color change acceptability/perceptibility (clinical 50: 50% acceptability threshold) was considered to be significant if the difference in color ΔE ≥3.3 [45,48]. Translucency parameter (TP) determination involved calculating the color difference between the 2 specimens (baseline, 14 days) recorded using a black and white backing according to the equation [TP=√ (LB*-LW*)2 + (aB*-aW*)2 + (bB*-bW*) [2,3,9]. The subscripts for each color coordinate (B, W) refers to the color coordinate against 2 different backgrounds (black, white). For measurement of contrast ratio, the spectral reflectance of the specimen’s light was computed over a black and white background using the equation [CR=Yb/Yw], where Y denotes spectral reflectance of light and subscripts B and W indicate 2 separate backings (black, white). Differences in color and translucency between groups (n=12) from baseline and within groups from control were assessed using SPSS for Windows Version 25 (IBM Corp., Armonk, NY, USA). The Shapiro-Wilk test determined the normality data distribution, and one-way ANOVA detected the significance of these differences between the test groups and the control group. To observe the differences within each subgroup, the post hoc least significant different (LSD) test was employed for each dependant variable (color, TP, and CR). For all comparative statistical tests, the probability value of <0.05 was considered as statistically significant.
Results
DIFFERENCES BETWEEN CERAMICS/IMMERSIONS:
The ΔE (differences in color) between 2 ceramic types in the studied groups (Gp NGCh, Gp CGCh, Gp NDCh, Gp CDCh, Gp NSCh, Gp CSCh) showed color changes immersed in chlorhexidine, which on the Clinical acceptability/perceptible threshold (CAT/CPT) could be perceived only by skilled operators and therefore were clinically acceptable. On the contrary, saliva-immersed groups (Gp NGS, Gp CGS, Gp NDS, Gp CDS, Gp NSS, Gp CSS) showed color changes that were not clinically appreciable (ΔE ≤1.00). One-way ANOVA test results show that the differences between subgroups (Gp NGS, Gp CGS) and (Gp NSS, Gp CSS) differed significantly (P<0.05), indicating that ceramic type and adjustment kit influence color differences. CR mean values showed significant differences in 2 subgroups (Gp NDS/Gp CDS, Gp NDCh/Gp CDCh) (P<0.05), although 2 subgroups of NPM (Gp NDS, Gp NDCh) and 1 subgroup of CZM (Gp CSCh) showed higher CR values (Table 2) than the CR threshold (Y ≥0.07). Interpretation of these results reveals that the NPM ceramic is more compatible with its recommended adjustment kit and showed higher CR values even when a third-party adjustment kit was used after immersion in saliva. On the other hand, the CZM ceramic is compatible with a third-party adjustment kit in the presence of saliva. However, both ceramics showed higher CR values when immersed in chlorhexidine, with CZM ceramic showing changes when adjusted with the SUN adjustment kit. For translucency, 2 subgroup comparisons (Gp NDCh/Gp CDCh; Gp NSCh/Gp CSCh) showed statistically significant differences, indicating that chlorhexidine reduced translucency of both ceramics irrespective of the adjustment kit used. Post hoc (Tukey least significant difference test) results for within-group differences for NPM and CZM ceramic are presented in Tables 3 and 4, respectively. There were 6 subgroups for each ceramic, depending upon the surface adjustment type and immersion liquid used.
INFLUENCE ON COLOR STABILITY AND TRANSLUCENCY:
Each subgroup for ceramic NPM differed in color parameter with other subgroups except for subgroups that were immersed in the same immersion media [saliva – Gp NDS and Gp NSS: Chlorhexidine – Gp NDCh and Gp NSCh] (Table 3). For CR and translucency, all subgroups differed significantly from other subgroups. Table 4 presents the multiple comparison post hoc test results for the CZM ceramic. For all dependent variables (color, contrast ratio, translucency), differences were statistically significant (P<0.05). Post hoc test results for CZM ceramic shows which groups were different. For color and contrast ratio, Gp CDS did not show difference from Gp CSS, while for translucency, Gp CGCh did not differ from Gp CDCh and vice versa. Therefore, the interpretation of these results shows that for color and contrast ratio, the CZM ceramic was compatible with both adjustment kits in the presence of saliva. When immersed in chlorhexidine, the CZM ceramic showed no differences when glazed or when adjusted with Diasynt Plus and Diacera adjustment kit. Details of least significant difference (LSD) and their respective subgroup comparisons with other subgroups for ceramic NPM and CZM are presented in Tables 5 and 6, respectively.
Discussion
STRENGTH AND LIMITATIONS OF THE STUDY:
This study investigated multiple independent variables in a single study design covering multiple clinical case scenarios. The study also generates 2 new hypotheses (preshaded ceramics match respective Vita shade guides and shade distribution for FDP or bridges) for future studies. The study is limited by its in vitro nature, which may not reflect the clinical case scenario. The measurement of color using CIELab is another limitation, as CIEDE2000 is a more comprehensive tool to measure color differences.
Conclusions
The findings of this in vitro study apply to the corresponding experimental conditions mentioned in this study. Since only 2 MLZ, 1 mouthwash, and 2 adjustment kits were used, general inference of these results cannot be made. However, within the scope and limitations of the present study, one may conclude that despite being prepared from the same Vita shades, there was a significant color difference between NPM and CZM and MLZ ceramics. NPM also showed more changes in color and translucency when adjusted with a third-party adjustment kit, while CZM showed less change when adjusted with third-party kits. ChG immersion for 14 days brings clinically perceptible changes in color and affects translucency irrespective of the adjustment kit used for both MLZ ceramics. Glazed MLZ showed the least changes in color and translucency; therefore, one must perform all clinical adjustments on a MLZ crown prior to glazing of the crown, which may add another clinical step but does not affect the aesthetics of the crown.
Figures
![Flowchart showing study sampling and group distribution. [Basis of Grouping – First alphabet represents type of ceramic (N – Nacera Pearl Multi-Shade, C – Ceramill Zolid FX Multilayer), Second alphabet represents surface treatment/adjustment (G – glazed, D – with Diasynt Plus, Diacera, S – with SUN) for finishing/polishing, third alphabet represents the medium used (S – saliva and Ch – chlorhexidine)]. [Example Gp NDCh represents Nacera ceramic adjusted with Diasynt Plus and immersed in chlorhexidine]. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), windows 11 Pro, Microsoft corporation).](https://jours.isi-science.com/imageXml.php?i=medscimonit-30-e943404-g001.jpg&idArt=943404&w=1000) Figure 1. Flowchart showing study sampling and group distribution. [Basis of Grouping – First alphabet represents type of ceramic (N – Nacera Pearl Multi-Shade, C – Ceramill Zolid FX Multilayer), Second alphabet represents surface treatment/adjustment (G – glazed, D – with Diasynt Plus, Diacera, S – with SUN) for finishing/polishing, third alphabet represents the medium used (S – saliva and Ch – chlorhexidine)]. [Example Gp NDCh represents Nacera ceramic adjusted with Diasynt Plus and immersed in chlorhexidine]. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), windows 11 Pro, Microsoft corporation).
Figure 1. Flowchart showing study sampling and group distribution. [Basis of Grouping – First alphabet represents type of ceramic (N – Nacera Pearl Multi-Shade, C – Ceramill Zolid FX Multilayer), Second alphabet represents surface treatment/adjustment (G – glazed, D – with Diasynt Plus, Diacera, S – with SUN) for finishing/polishing, third alphabet represents the medium used (S – saliva and Ch – chlorhexidine)]. [Example Gp NDCh represents Nacera ceramic adjusted with Diasynt Plus and immersed in chlorhexidine]. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), windows 11 Pro, Microsoft corporation). 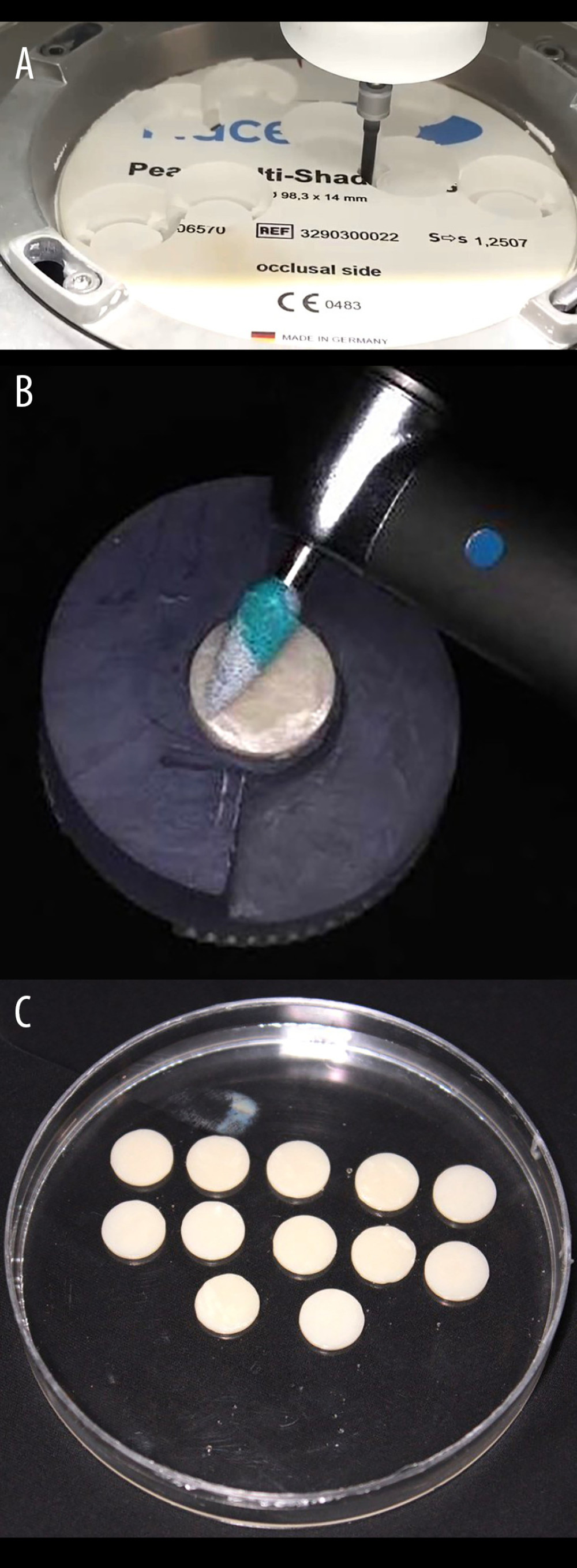 Figure 2. (A) Exemplary disk-shaped specimen preparation from a block of concerned monolithic zirconia ceramic prior to sintering. (B) Exemplary surface adjustment of the adjustment kit on the surface of the specimen prior to polishing with the respective adjustment kit. (C) Finished and polished specimens of a particular individual group before measurement of color and translucency parameter. Photograph taken using a digital single-lens reflex (DSLR) camera (Canon EOS 700D) with 100 mm macro lens) with/without ring flash.
Figure 2. (A) Exemplary disk-shaped specimen preparation from a block of concerned monolithic zirconia ceramic prior to sintering. (B) Exemplary surface adjustment of the adjustment kit on the surface of the specimen prior to polishing with the respective adjustment kit. (C) Finished and polished specimens of a particular individual group before measurement of color and translucency parameter. Photograph taken using a digital single-lens reflex (DSLR) camera (Canon EOS 700D) with 100 mm macro lens) with/without ring flash. Tables
Table 1. List of materials, instrumentation, and manufacturer.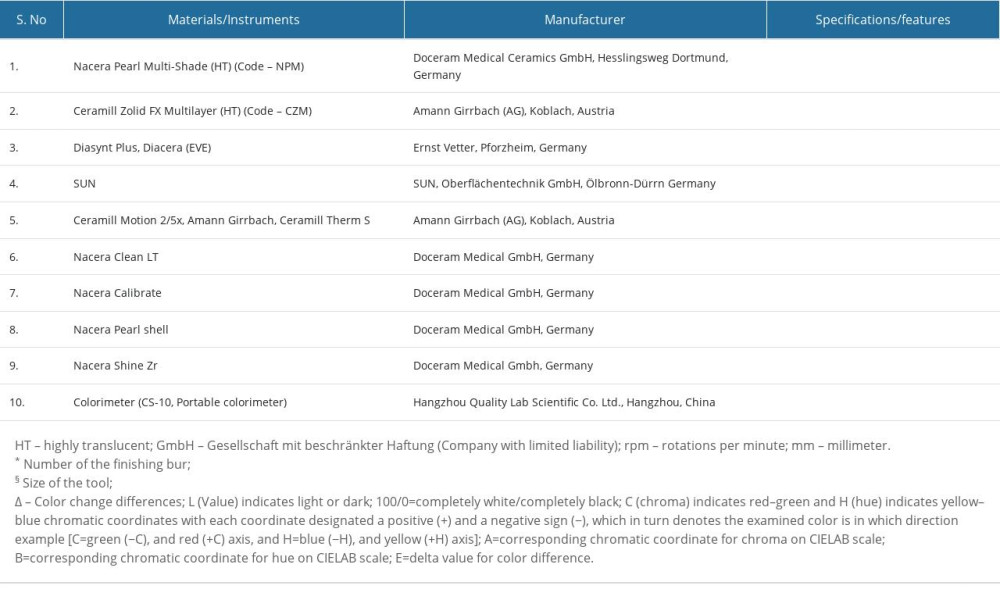 Table 2. One-way ANOVA test results for comparison of color, contrast ratio and translucency between studied ceramics (Nacera Pearl Multi-Shade/Ceramill Zolid FX multilayer) after surface alteration (finishing/polishing) with 2 different commercial kits and immersion in media (saliva/chlorhexidine).
Table 2. One-way ANOVA test results for comparison of color, contrast ratio and translucency between studied ceramics (Nacera Pearl Multi-Shade/Ceramill Zolid FX multilayer) after surface alteration (finishing/polishing) with 2 different commercial kits and immersion in media (saliva/chlorhexidine).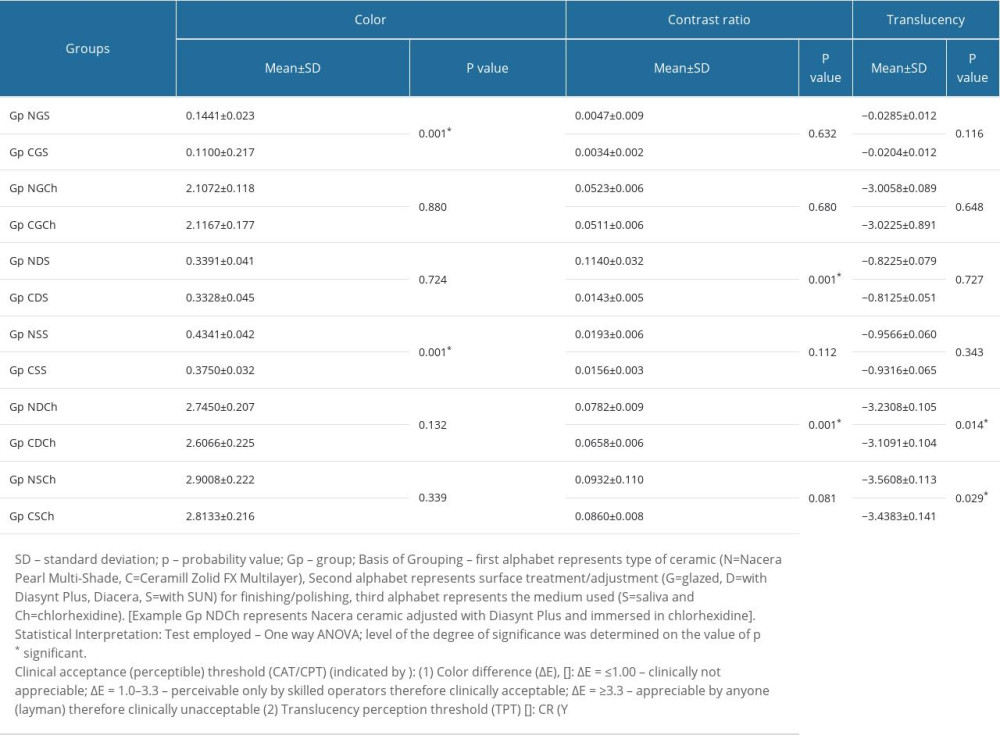 Table 3. Post hoc (Tukey least significant difference) test results for intra (multiple) group comparison of mean values in color, contrast ratio and translucency for nacera pearl multi-shade ceramic after finishing/polishing and immersion (saliva/chlorhexidine).
Table 3. Post hoc (Tukey least significant difference) test results for intra (multiple) group comparison of mean values in color, contrast ratio and translucency for nacera pearl multi-shade ceramic after finishing/polishing and immersion (saliva/chlorhexidine).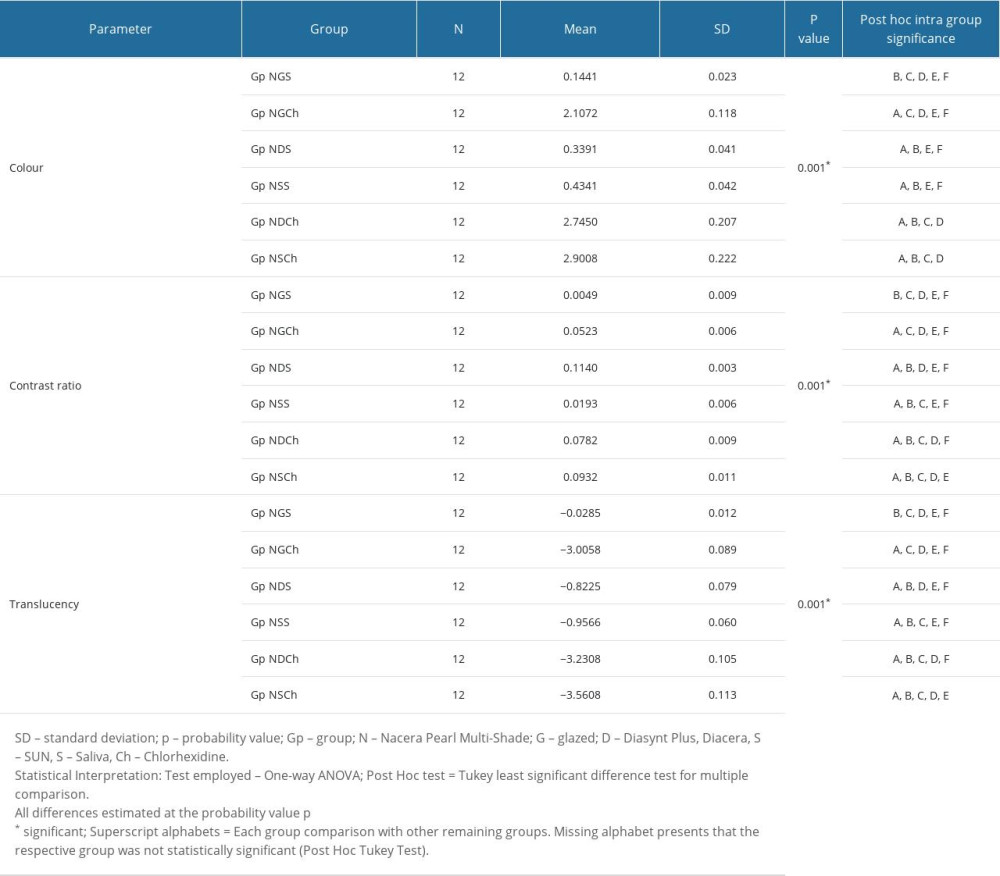 Table 4. Post hoc (Tukey least significant difference) test results for intra group comparison of mean values in color, contrast ratio and translucency for Ceramill Zolid FX multilayer ceramic after finishing/polishing and immersion (saliva/chlorhexidine).
Table 4. Post hoc (Tukey least significant difference) test results for intra group comparison of mean values in color, contrast ratio and translucency for Ceramill Zolid FX multilayer ceramic after finishing/polishing and immersion (saliva/chlorhexidine).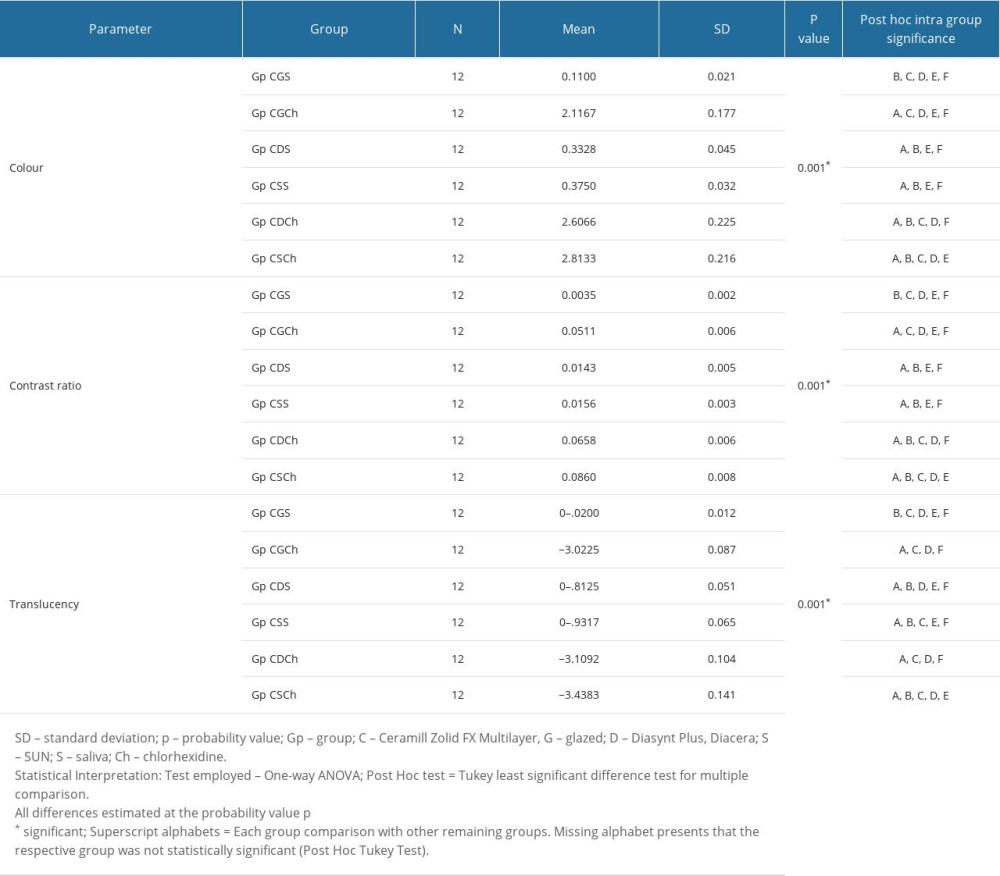 Table 5. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Nacera Ceramic type.
Table 5. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Nacera Ceramic type.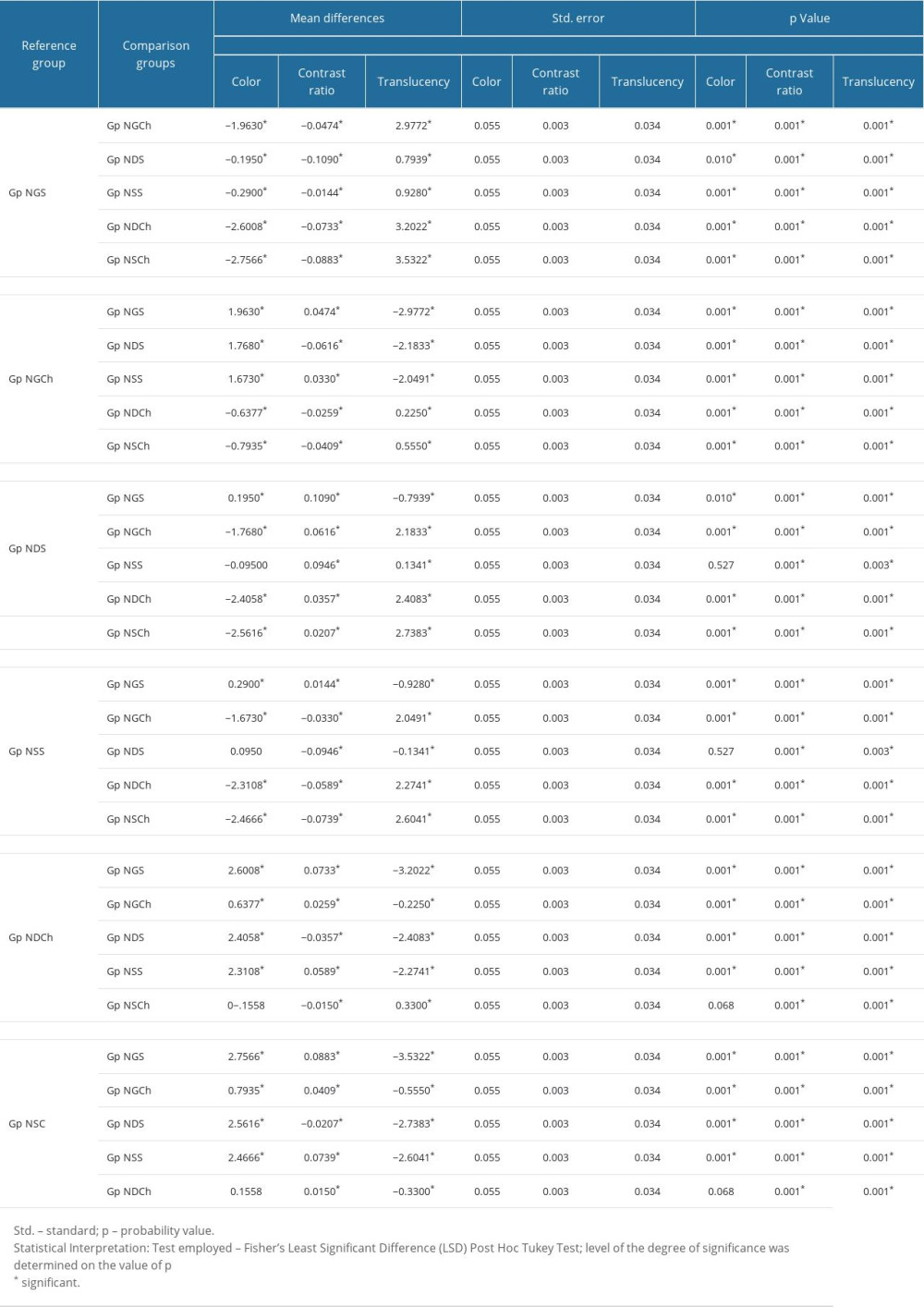 Table 6. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Ceramill Ceramic type.
Table 6. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Ceramill Ceramic type.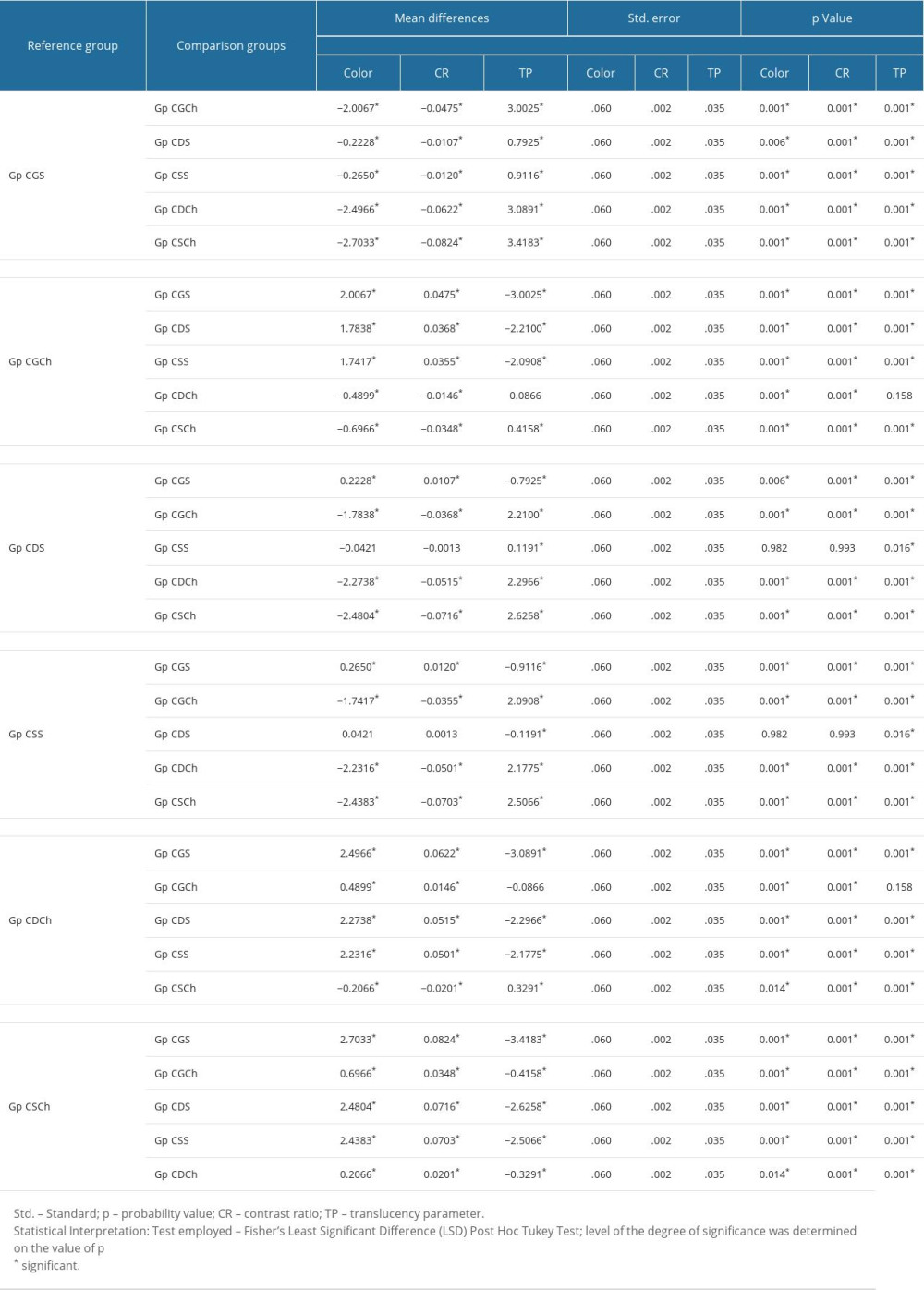
References
1. Lawn BR, Marshall DB, Brittle solids: From physics and chemistry to materials applications: Annu Rev Mater Res, 2022; 52; 441-71
2. Mattoo K, Etoude HS, Hothan HM, Failure of an all-ceramic posterior fixed partial denture as a consequence of poor clinical application of diagnostic data: Journal of Advanced Medical and Dental Science Research, 2023; 11(3); 44-48
3. Della Bona A, Corazza PH, Zhang Y, Characterization of a polymer-infiltrated ceramic-network material: Dent Mater, 2014; 30(5); 564-69
4. De Araújo-Júnior EN, Bergamo ET, Bastos TM, Ultra-translucent zirconia processing and aging effect on microstructural, optical, and mechanical properties: Dent Mater, 2022; 38(4); 587-600
5. Tian Y, Chen C, Xu X, A review of 3D printing in dentistry: Technologies, affecting factors, and applications: Scanning, 2021; 2021; 9950131
6. Kontonasaki E, Rigos AE, Ilia C, Istantsos T, Monolithic zirconia: An update to current knowledge. Optical properties, wear, and clinical performance. Dent: J, 2019; 7; 90
7. Yener ES, Ozcan M, Kazazoglu E, The effect of glazing on the biaxial flexural strength of different zirconia core materials: Acta Odontol Latinoam, 2011; 24; 133-40
8. Pieger S, Salman A, Bidra AS, Clinical outcomes of lithium disilicate single crowns and partial fixed dental prostheses: A systematic review: J Prosthet Dent, 2014; 112(1); 22-30
9. Zadeh PN, Lümkemann N, Sener B, Flexural strength, fracture toughness, and translucency of cubic/tetragonal zirconia materials. J. Prosthet: Dent, 2018; 120(6); 948-54
10. Sulaiman TA, Abdulmajeed AA, Donovan TE, The effect of staining and vacuum sintering on optical and mechanical properties of partially and fully stabilized monolithic zirconia: Dent Mater J, 2015; 34; 605-10
11. Tabatabaian F, Color in zirconia based restorations and related factors: A literature review: J Prosthodont, 2018; 27(2); 201-11
12. Gehrt M, Wolfart S, Rafai N, Clinical results of lithium disilicate crowns after up to 9 years of service: Clin Oral Investig, 2013; 17; 275-84
13. Kumar S, Mattoo KA, Staining of a fixed partial denture to restore pre-extraction self image – a case report: Journal of Indian Dental Association, 2010; 4(12); 573-74
14. Zhang HB, Li ZP, Kim BN, Effect of alumina dopant on transparency of tetragonal zirconia: J Nanomater, 2012; 2012; 269064
15. Tango RN, Todorović A, Stamenković D, Effect of staining and aging on translucency parameter of CAD-CAM materials: Acta Stomatol Croat, 2021; 55(1); 2-9
16. Baldissara P, Llukacej A, Ciocca L, Translucency of zirconia copings made with different CAD/CAM systems: J Prosthet Dent, 2010; 104; 6-12
17. Zhang F, Vanmeensel K, Inokoshi M, 3Y-TZP ceramics with improved hydrothermal degradation resistance and fracture toughness: J Eur Ceram Soc, 2014; 34; 2453-63
18. Harada K, Raigrodski AJ, Chung KH, A comparative evaluation of the translucency of zirconias and lithium disilicate for monolithic restorations: J Prosthet Dent, 2016; 116(2); 257-63
19. Kurt M, Bal BT, Effects of accelerated artificial aging on the translucency and color stability of monolithic ceramics with different surface treatments: J Prosthet Dent, 2019; 121(4); 712-e1
20. Fathy SM, El-Fallal AA, El-Negoly SA, El Bedawy AB, Translucency of monolithic and core zirconia after hydrothermal aging: Acta Biomater Odontol Scand, 2015; 1; 86-92
21. : Amann Girrbach AG Available at https://www.amanngirrbach.com/en-gb/material/zolid-fx-ml
22. Elsaka SE, Optical and mechanical properties of newly developed monolithic multilayer zirconia: J Prosthodont, 2019; 28(1); e279-84
23. Stawarczyk B, Keul C, Eichberger M, Three generations of zirconia: From veneered to monolithic. Part I: Quintessence Int, 2017; 48(5); 369-80
24. Juntavee N, Attashu S, Effect of different sintering process on flexural strength of translucency monolithic zirconia: J Clin Exp Dent, 2018; 10(8); e821
25. Stober T, Bermejo JL, Schwindling FS, Schmitter M, Clinical assessment of enamel wear caused by monolithic zirconia crowns: J Oral Rehabil, 2016; 43(8); 621-29
26. Hatanaka GR, Polli GS, Adabo GL, The mechanical behavior of high-translucent monolithic zirconia after adjustment and finishing procedures and artificial aging: J Prosthet Dent, 2020; 123(2); 330-37
27. Chun EP, Anami LC, Bonfante EA, Bottino MA, Microstructural analysis and reliability of monolithic zirconia after simulated adjustment protocols: Dent Mater, 2017; 33(8); 934-43
28. Kim HK, Kim SH, Lee JB, Effect of polishing and glazing on the color and spectral distribution of monolithic zirconia: J Adv Prosthodont, 2013; 5; 296-304
29. Azevedo SM, Kantorski KZ, Valandro LF, Effect of brushing with conventional versus whitening dentifrices on surface roughness and biofilm formation of dental ceramics: Gen Dent, 2012; 60(3); e123-e30
30. Barutçugil Ç, Bilgili D, Barutcigil K, Discoloration and translucency changes of CAD-CAM materials after exposure to beverages: J Prosthet Dent, 2019; 122(3); 325-31
31. Chun KJ, Lee JY, Comparative study of mechanical properties of dental restorative materials and dental hard tissues in compressive loads: J Dent Biomech, 2014; 5; 1758736014555246
32. Pfefferle R, Lümkemann N, Wiedenmann F, Stawarczyk B, Different polishing methods for zirconia: Impact on surface, optical, and mechanical properties: Clin Oral Investig, 2020; 24; 395-403
33. Brookes ZL, Bescos R, Belfield LA, Current uses of chlorhexidine for management of oral disease: A narrative review: J Dent, 2020; 103; 103497
34. Al Tannir MA, Goodman HS, A review of chlorhexidine and its use in special populations: Spec Care Dentist, 1994; 14(3); 116-22
35. Alnassar TM, Color stability of monolithic zirconia in various staining liquids: An in vitro study: Appl Sci, 2022; 12; 9752
36. Derafshi R, Khorshidi H, Kalantari M, Ghaffarlou I, Effect of mouth rinses on color stability of monolithic zirconia and feldspathic ceramic: An in vitro study: BMC Oral Health, 2017; 17; 129
37. Haralur SB, Raqe S, Alqahtani N, Alhassan Mujayri F, Effect of hydrothermal aging and beverages on color stability of lithium disilicate and zirconia-based ceramics: Medicina, 2019; 55(11); 749
38. The Academy of Prosthodontics., The glossary of prosthodontic terms: ninth edition: J Prosthet Dent, 2017; 117; e1-e105
39. Commission Internationale de la’Eclairage (CIE, The International Commission on Illumination): Colorimetry third edition. CIE 015, 2004, 2004, Vienna, Austria, CIE
40. Johnston WM, Review of translucency determinations and applications to dental materials: J Esthet Restor Dent, 2014; 26(4); 217-23
41. Dentaurum GmbH & Co. KG. www.dentaurum.de/eng/nacera-pearl-multi-shade-32303.aspx
42. : Amann Girrbach https://www.amanngirrbach.com/en-gb/media/instructions
43. Traini T, Gherlone E, Parabita SF, Fracture toughness and hardness of a Y-TZP dental ceramic after mechanical surface treatments: Clin Oral Invest, 2014; 18; 707-14
44. Farooq IMA, AlShwaimi E, Almas K, Efficacy of a novel fluoride containing bioactive glass-based dentifrice in remineralizing artificially induced demineralization in human enamel: Fluoride, 2019; 52; 447-55
45. Gupta S, Sayed ME, Gupta B, Comparison of composite resin (duo-shade) shade guide with vita ceramic shades before and after chemical and autoclave sterilization: Med Sci Monit, 2023; 29; e940949
46. CIE (Commission Internationale de l’Eclairage): Technical report: colorimetry, 2018; 16-20, Vienna, Austria, CIE Central Bureau CIE Pub no 15-4
47. Liu MC, Aquilino SA, Lund PS, Human perception of dental porcelain translucency correlated to spectrophotometric measurements: J Prosthodont, 2010; 19(3); 187-93
48. Iyer RS, Babani VR, Yaman P, Dennison J, Color match using instrumental and visual methods for single, group, and multi shade composite resins: J Esthet Restor Dent, 2021; 33(2); 394-400
49. Manziuc MM, Gasparik C, Burde AV, Effect of glazing on translucency, color, and surface roughness of monolithic zirconia materials: J Esthet Restor Dent, 2019; 31(5); 478-85
50. Preis V, Schmalzbauer M, Bougeard D, Surface properties of monolithic zirconia after dental adjustment treatments and in vitro wear simulation: J Dent, 2015; 43; 133-39
51. Sarac D, Sarac YS, Yuzbasioglu E, Bal S, The effects of porcelain polishing systems on the color and surface texture of feldspathic porcelain: J Prosthet Dent, 2006; 96; 122-28
52. Akar GC, Pekkan G, Çal E, Effects of surface finishing protocols on the roughness, color change, and translucency of different ceramic systems: J Prosthet Dent, 2014; 112; 314-21
53. Incesu E, Yanikoglu N, Evaluation of the effect of different polishing systems on the surface roughness of dental ceramics: J Prosthet Dent, 2020; 124(1); 100-9
54. Abullais SS, Patel SI, Asiri EA, Comparative evaluation of 3 commercial mouthwash formulations on clinical parameters of chronic gingivitis: Med Sci Monit, 2022; 28; e937111
55. Pekkan G, Pekkan K, Bayindir BÇ, Factors affecting the translucency of monolithic zirconia ceramics: A review from materials science perspective: Dent Mater J, 2020; 39(1); 1-8
56. Subaşı MG, Alp G, Johnston WM, Yilmaz B, Effect of thickness on optical properties of monolithic CAD-CAM ceramics: J Dent, 2018; 71; 38-42
57. Sabrah AH, Cook NB, Luangruangrong P, Full-contour Y-TZP ceramic surface roughness effect on synthetic hydroxyapatite wear: Dent Mater, 2013; 29; 666673
58. Janyavula S, Lawson N, Cakir D, The wear of polished and glazed zirconia against enamel: J Prosthet Dent, 2013; 109; 2229
59. Giti R, Jebal R, How could mouthwashes affect the color stability and translucency of various types of monolithic zirconia? An in-vitro study: PLoS One, 2023; 18(12); e0295420
60. Beuer F, Stimmelmayr M, Gueth J, In vitro performance of full-contour zirconia single crowns: Dent Mater, 2012; 28; 449-56
Figures
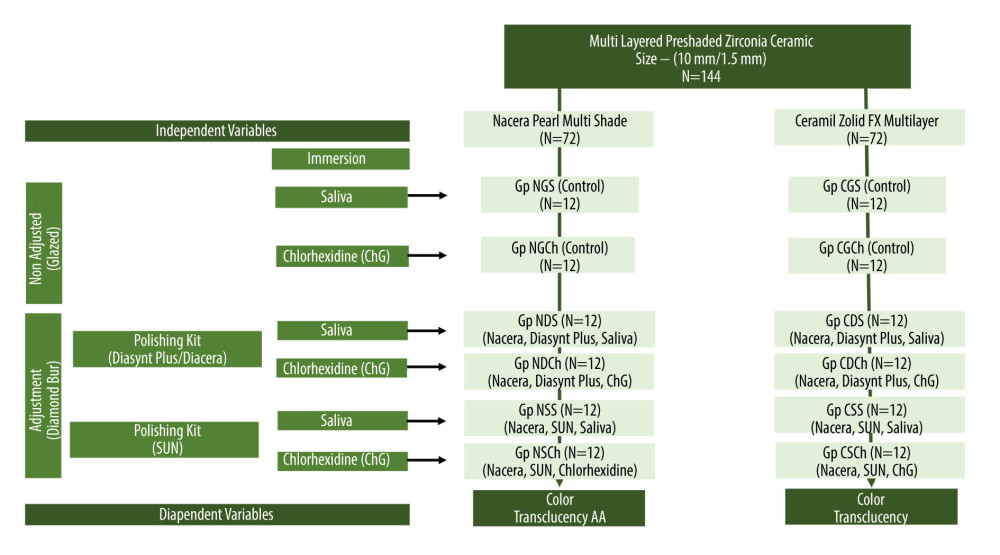 Figure 1. Flowchart showing study sampling and group distribution. [Basis of Grouping – First alphabet represents type of ceramic (N – Nacera Pearl Multi-Shade, C – Ceramill Zolid FX Multilayer), Second alphabet represents surface treatment/adjustment (G – glazed, D – with Diasynt Plus, Diacera, S – with SUN) for finishing/polishing, third alphabet represents the medium used (S – saliva and Ch – chlorhexidine)]. [Example Gp NDCh represents Nacera ceramic adjusted with Diasynt Plus and immersed in chlorhexidine]. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), windows 11 Pro, Microsoft corporation).
Figure 1. Flowchart showing study sampling and group distribution. [Basis of Grouping – First alphabet represents type of ceramic (N – Nacera Pearl Multi-Shade, C – Ceramill Zolid FX Multilayer), Second alphabet represents surface treatment/adjustment (G – glazed, D – with Diasynt Plus, Diacera, S – with SUN) for finishing/polishing, third alphabet represents the medium used (S – saliva and Ch – chlorhexidine)]. [Example Gp NDCh represents Nacera ceramic adjusted with Diasynt Plus and immersed in chlorhexidine]. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), windows 11 Pro, Microsoft corporation). Figure 2. (A) Exemplary disk-shaped specimen preparation from a block of concerned monolithic zirconia ceramic prior to sintering. (B) Exemplary surface adjustment of the adjustment kit on the surface of the specimen prior to polishing with the respective adjustment kit. (C) Finished and polished specimens of a particular individual group before measurement of color and translucency parameter. Photograph taken using a digital single-lens reflex (DSLR) camera (Canon EOS 700D) with 100 mm macro lens) with/without ring flash.
Figure 2. (A) Exemplary disk-shaped specimen preparation from a block of concerned monolithic zirconia ceramic prior to sintering. (B) Exemplary surface adjustment of the adjustment kit on the surface of the specimen prior to polishing with the respective adjustment kit. (C) Finished and polished specimens of a particular individual group before measurement of color and translucency parameter. Photograph taken using a digital single-lens reflex (DSLR) camera (Canon EOS 700D) with 100 mm macro lens) with/without ring flash. Tables
 Table 1. List of materials, instrumentation, and manufacturer.
Table 1. List of materials, instrumentation, and manufacturer. Table 2. One-way ANOVA test results for comparison of color, contrast ratio and translucency between studied ceramics (Nacera Pearl Multi-Shade/Ceramill Zolid FX multilayer) after surface alteration (finishing/polishing) with 2 different commercial kits and immersion in media (saliva/chlorhexidine).
Table 2. One-way ANOVA test results for comparison of color, contrast ratio and translucency between studied ceramics (Nacera Pearl Multi-Shade/Ceramill Zolid FX multilayer) after surface alteration (finishing/polishing) with 2 different commercial kits and immersion in media (saliva/chlorhexidine). Table 3. Post hoc (Tukey least significant difference) test results for intra (multiple) group comparison of mean values in color, contrast ratio and translucency for nacera pearl multi-shade ceramic after finishing/polishing and immersion (saliva/chlorhexidine).
Table 3. Post hoc (Tukey least significant difference) test results for intra (multiple) group comparison of mean values in color, contrast ratio and translucency for nacera pearl multi-shade ceramic after finishing/polishing and immersion (saliva/chlorhexidine). Table 4. Post hoc (Tukey least significant difference) test results for intra group comparison of mean values in color, contrast ratio and translucency for Ceramill Zolid FX multilayer ceramic after finishing/polishing and immersion (saliva/chlorhexidine).
Table 4. Post hoc (Tukey least significant difference) test results for intra group comparison of mean values in color, contrast ratio and translucency for Ceramill Zolid FX multilayer ceramic after finishing/polishing and immersion (saliva/chlorhexidine). Table 5. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Nacera Ceramic type.
Table 5. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Nacera Ceramic type. Table 6. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Ceramill Ceramic type.
Table 6. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Ceramill Ceramic type. Table 1. List of materials, instrumentation, and manufacturer.
Table 1. List of materials, instrumentation, and manufacturer. Table 2. One-way ANOVA test results for comparison of color, contrast ratio and translucency between studied ceramics (Nacera Pearl Multi-Shade/Ceramill Zolid FX multilayer) after surface alteration (finishing/polishing) with 2 different commercial kits and immersion in media (saliva/chlorhexidine).
Table 2. One-way ANOVA test results for comparison of color, contrast ratio and translucency between studied ceramics (Nacera Pearl Multi-Shade/Ceramill Zolid FX multilayer) after surface alteration (finishing/polishing) with 2 different commercial kits and immersion in media (saliva/chlorhexidine). Table 3. Post hoc (Tukey least significant difference) test results for intra (multiple) group comparison of mean values in color, contrast ratio and translucency for nacera pearl multi-shade ceramic after finishing/polishing and immersion (saliva/chlorhexidine).
Table 3. Post hoc (Tukey least significant difference) test results for intra (multiple) group comparison of mean values in color, contrast ratio and translucency for nacera pearl multi-shade ceramic after finishing/polishing and immersion (saliva/chlorhexidine). Table 4. Post hoc (Tukey least significant difference) test results for intra group comparison of mean values in color, contrast ratio and translucency for Ceramill Zolid FX multilayer ceramic after finishing/polishing and immersion (saliva/chlorhexidine).
Table 4. Post hoc (Tukey least significant difference) test results for intra group comparison of mean values in color, contrast ratio and translucency for Ceramill Zolid FX multilayer ceramic after finishing/polishing and immersion (saliva/chlorhexidine). Table 5. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Nacera Ceramic type.
Table 5. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Nacera Ceramic type. Table 6. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Ceramill Ceramic type.
Table 6. Least significant difference (LSD) test results for individual subgroup comparison with other subgroups for Ceramill Ceramic type. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








