02 July 2020: Clinical Research
Nuclear Factor κB/MicroRNA-155 Upregulates the Expression Pattern of Cytokines in Regulating the Relapse of Chronic Sinusitis with Nasal Polyps and the Underlying Mechanism of Glucocorticoid
Jianbin Du1BCE, Haijun Lv2BC, Xin Dou3BC, Zhongsheng Cao1ADEF*DOI: 10.12659/MSM.923618
Med Sci Monit 2020; 26:e923618
Abstract
BACKGROUND: The aim of this study was to explore the upregulated nuclear factor κB (NF-κB)/microRNA-155 (miR-155) in regulating inflammatory responses and relapse of chronic rhinosinusitis (CRS) with nasal polyps (NP), which underlies the molecular mechanism of glucocorticoid treatment.
MATERIAL AND METHODS: The study recruited 25 patients with eosinophilic (Eos) CRSwNP, 25 patients with Non-Eos CRSwNP, 25 patients with CRS without NP (CRSsNP) and 30 patients with nasal septum deviation (control group). The expression of NF-κB/miR-155 and inflammatory cytokines was detected in epithelial tissue specimens. Additionally, a mouse model of Eos CRSwNP was established, and the mice were treated by NF-κB inhibitor, miR-155 antagomir, or dexamethasone (DEX) to explore the role of NF-kB/miR-155 and the anti-inflammatory effects of glucocorticoid treatment.
RESULTS: Results showed that the expression level of NF-κB/miR-155 was significantly elevated in the Eos CRSwNP group, accompanied by the upregulation of cytokines: tumor necrosis factor (TNF)-α, interleukin (IL)-1, IL-4, IL-5 (P<0.05) compared with the control group, the CRSsNP group or the Non-Eos CRSwNP group. The upregulation of NF-κB/miR-155 increased inflammatory mediator cyclooxygenase2 (COX2) while decreasing anti-inflammatory mediator Src homology-2 domain-containing inositol 5-phosphatase 1 (SOCS1), which resulted in the aberrant expression pattern of cytokines in the mice model. DEX treatment inhibited the expression of cytokines and decreased the relapse rate of Eos CRSwNP via inhibiting NF-κB/miR-155 (P<0.05).
CONCLUSIONS: The upregulation of NF-κB/miR-155 was crucial in mediating the aberrant expression of inflammatory cytokines in Eos CRSwNP. This molecular mechanism is a concern with the high relapse rate of Eos CRSwNP. However, glucocorticoid treatment inhibited the relapse of CRSwNP via downregulation of NF-κB/miR-155.
Keywords: Glucocorticoids, Nasal Polyps, Chronic Disease, Cytokines, Nasal Mucosa, RNA, Messenger, Recurrence, Rhinitis, Sinusitis, transcriptional activation
Background
Chronic rhinosinusitis (CRS) with nasal polyps (NP) (CRSwNP) is one of the most common diseases in otolaryngology clinical practice due to its increasing incidence and high relapse rate [1]. CRSwNP is characterized by heterogeneous chronic inflammation of nasal and sinus mucosa. According to clinical investigations, approximate 20.6% of patients with CRSwNP receive secondary surgical treatment within 5 years after primary nasal endoscopic surgery due to relapse [2]. In a 12-year follow-up study, the relapse rate of CRSwNP even reached 78.9% [3]. The high relapse rate of CRSwNP severely affects patient quality of life and leads to global health concerns. However, until now, the molecular mechanism of relapse has not been well clarified.
Based on the degree of eosinophils infiltration, CRSwNP can be divided into 2 categories: eosinophilic CRSwNP (Eos CRSwNP) and non-eosinophilic CRSwNP (Non-Eos CRSwNP). Clinical epidemiological investigations indicate that compared with Non-Eos CRSwNP, Eos CRSwNP has more severe symptoms and higher probability of relapse in patients [4]. In past studies, several influential factors have been highlighted associated with the relapse of CRSwNP, including degree of eosinophils infiltration, asthma [5], and other factors [6]. Recent studies have considered the crucial effects of the expression pattern of cytokines in CRSwNP [7–9]. Huriyati et al. [10] and Yu et al. [11] have indicated that the expression level of inflammatory cytokines is closely associated with the relapse of nasal polyp. Additionally, postoperative application of glucocorticoid has been reported to significantly reduce the relapse of CRSwNP in patients, which may be due to its inhibitory effects on inflammatory cytokines [12]. These findings indicate that the expression phenotype of inflammatory cytokines is crucial in regulating the relapse process of CRSwNP. However, the underlying molecular mechanism of relapse and glucocorticoid’s protective effects still remains unclear.
Nuclear factor kappa B (NF-κB) is the notable inflammatory regulator in regulating cellular inflammation responses and the expression of cytokines [13]. Ramis et al. [14] showed that NF-κB was activated in both CRS and NP tissues. Additionally, IκB-α, the inhibitory proteins of NF-kB signal, has also been reported to be downregulated in NP tissues [15]. Thus, in CRSwNP, the NF-κB signal is possibly activated and regulates the expression pattern of cytokines. However, this hypothesis still needs further evidence.
Previous studies have supported a positive correlation between NF-κB activation and the upregulation of downstream microRNA (miR)-155 [16]. MiR-155 belongs to miRNAs family; miRNAs are composed of 18-24 bp and are highly conserved among species [17]. In recent years, the association between miRNAs and the expression of cytokines has attracted huge attention from researchers [18,19], especially for miR-155. O’Connell et al. [20] reported that knockout of miR-155 completely prevented the initiation of experimental autoimmune encephalomyelitis in mice. MiR-155 is also reported to regulate the expression of numerous inflammatory cytokines, including tumor necrosis factor (TNF)-α [21], interleukin (IL)-1 [22], IL-4 [23], etc. Additionally, compared with chronic rhinosinusitis without nasal polyps (CRSsNP), the expression of miR-155 is found significantly increased in CRSwNP [24]. Taken together, the activation of NF-κB may upregulate miR-155 in CRSwNP, resulting in the expression alterations of downstream inflammatory cytokines. However, reports addressing this hypothesis are still scarce.
In this study, we first examined the expression of NF-κB/miR-155 and inflammatory cytokines in the epithelial tissue of patients with nasal septum deviation (control), Eos CRSwNP, Non-Eos CRSwNP, and CRSsNP. Then we detected whether after dexamethasone (DEX) treatment for 12 weeks, the relapse rate of CRSwNP was inhibited and whether its underlying mechanism involved the NF-κB/miR-155 signaling pathway and the expression pattern of cytokines. In a mouse model of CRSwNP, pyrrolidine dithiocarbonate (PDTC) and miR-155 antagomir were used to downregulate the expression of NF-κB and miR-155, respectively. This model aimed to explore the role of NF-κB/miR-155 signaling pathway in regulating inflammatory responses and the anti-inflammatory effects of glucocorticoid treatment. Our results indicated that the upregulation of NF-κB/miR-155 was crucial in Eos CRSwNP. Upregulated NF-κB/miR-155 promoted the expression pattern of cytokines TNF-α, IL-1, IL-4, IL-5, and induced inflammatory responses via regulating cyclooxygenase2 (COX2) and SOCS1 proteins, which are known targets of miR-155. This molecular mechanism accounted for the high relapse rate of Eos CRSwNP. Additionally, glucocorticoid treatment inhibited the relapse of CRSwNP via downregulation of NF-κB/miR-155 signaling pathway. This is the first study to clarify the role of NF-κB/miR-155 signaling pathway in regulating inflammatory responses and the relapse of Eos CRSwNP. It also may provide experimental instructions for clinical practice.
Material and Methods
PATIENTS:
In total of 50 CRSwNP patients, admitted to the Second Affiliated Hospital of Suzhou University from June 2016 to December 2017 were recruited in this study. The diagnosis of CRSwNP was performed by nasal endoscopy and sinus computed tomography (CT), according to European Position Paper on Rhinosinusitis and Nasal Polyp Group [25]. Inclusion criteria were as follows: patients did not receive oral intake of glucocorticoid at least 3 months before surgery; patients did not use aerosol corticosteroids within 1 month before surgery; patients received antibiotic treatment for 3 to 5 days before surgery. Exclusion criteria were as follows: patients with posterior nostril polyps, cystic fibrosis, fungal sinusitis, primary cilia, or gastroesophageal reflux; patients were lost during follow-up; incomplete data of patient.
The pathological biopsy sections of nasal mucosal were collected and stained with hematoxylin and eosin (H&E). Ten random high microscopic fields (400×) were taken and the number of eosinophilic granulocytes were counted. If the number of eosinophilic granulocytes was more than 10% of the total infiltrating cells, patients were assigned to the Eos CRSwNP group. Otherwise, patients were included in the Non-Eos CRSwNP group [26]. Finally, 25 patients were diagnosed as Eos CRSwNP and 25 patients were diagnosed as Non-Eos CRSwNP. Additionally, 25 patients with CRSsNP and 30 patients with nasal septum deviation (without other nasal inflammatory diseases) were also recruited as control study participants. All included patients received surgical treatment. The nasal polyp tissue and ethmoid sinus mucosa in Eos CRSwNP and Non-Eos CRSwNP group were collected. The ethmoid sinus mucosa in CRSsNP group was collected. The inferior turbinate tissue and epithelial tissue of patients with nasal septum deviation were collected as control group. The clinical characteristics of recruited patients were listed in Table 1.
Atopic status was examined by the Phadiatop test. The diagnosis of asthma and aspirin intolerance was performed by physicians. Smoking history was diagnosed based on the criteria: >1 cigarette per day and lasted over 1 year. After patients were discharged from the hospital, the follow-up work lasted for 1 year by author LH, who was unaware of patient’s allocation.
ETHICAL APPROVAL:
This study was approved by the Ethics Committee of the Second Affiliated Hospital of Suzhou University. All patients signed the informed consents and agreed with the contents of experiment.
MOUSE MODEL:
The animal experiment was approved by the Ethics Committee of Animal Experiments of Suzhou University. The construction of CRSwNP model in mice was performed according to Kim et al. study [27]. In brief, the experimental mice were sensitized systemically with 25 μg of ovalbumin (OVA, Sigma), dissolved in 300 μL phosphate buffered saline (PBS) with the presence of 2 mg aluminum hydroxide gel at day 0 and day 5. One week later, mice were challenged with intranasally instillation of 3% OVA, dissolved in 40 μL PBS daily for 1 week. Then, the instillation was maintained at the same dose 3 times per week for 4 weeks. After that, 3% OVA, dissolved in 40 μL PBS was injected intranasally with intraperitoneal or intranasal injections of PDTC (Sigma), miR-155 antagomir or DEX (Sigma) for 8 weeks. During this period, the extra 10 ng of Staphylococcus aureus enterotoxin B (SEB), dissolved in 20 μL PBS was used intranasally following the instillation of OVA once a week. For control mice, only PBS was used intraperitoneally or intranasally, instead of drugs. One day after last nasal challenge in experimental group, rats were sacrificed and the diagnosis of CRSwNP was performed by pathological examination.
For drug treatment in mice, PDTC powder (Sigma) was dissolved in 0.9% sodium chloride solution and injected intraperitoneally at the concentration of 10 mg/100 g body weight. 50 mg miR-155 antagomir (Ribobio), dissolved in 50 μL sterilized saline was given to mice intranasally. DEX powder (Sigma) was dissolved in PBS and injected intraperitoneally at the concentration of 50 mg/kg body weight. Drug treatment for different group was performed within the same day when OVA was used intranasally.
GLUCOCORTICOID TREATMENT IN PATIENTS:
In total, 25 patients with Eos CRSwNP, based on whether patients agreed to receive glucocorticoid treatment for 12 weeks after surgery, 9 patients were divided into Eos CRSwNP-control group while 16 patients were defined as Eos CRSwNP-DEX group. For glucocorticoid treatment, the nasal cavity of patients was washed with 0.9% sodium chloride solution to clean the nasal cavity. Ephedrine hydrochloride and nitrofurazone nasal drops were mixed with DEX injection liquid (5 mg). Then patients received 3 drops on each side of the nasal cavity, 3 times per day, for a total of 12 weeks. For control treatment, ephedrine hydrochloride and nitrofurazone nasal drops without DEX were used at the same dose. Three months after patients received drug treatment, the epithelial tissues of Eos CRSwNP group were re-collected for following experiments.
:
The expression of miR-155 in collected tissues was performed by
QUANTITATIVE REAL-TIME POLYMERASE CHAIN REACTION (QRT-PCR):
Total RNA of collected rhinal tissues was extracted by TRIzol kit (Invitrogen). In brief, RNA was layered by chloroform, precipitated by isopropanol, and reversely transcribed by Oligo-dT. The optical density (OD) 260/280 value of RNA products was strictly controlled between 1.9 and 2.0. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed using the SYBR® Premix Ex Taq™ PCR kit (Takara).The amplification of cDNA was conducted in a thermal cycler S100 (Bio-rad) under following procedures: 95°C for 2 minutes, then 95°C for 30 seconds, 55°C for 30 seconds, 72°C for 30 seconds (40 cycles) and final incubation at 72°C for 5 minutes. The relative expression level of miRNA was calculated using the 2−ΔΔCt method. The expression level of miRNA-155 and mRNA was normalized according to the expression of U6, β-actin, respectively. The sequences of specific primers were listed in Table 2.
WESTERN BLOT:
For western blot analysis, total proteins of tissues were extracted by radioimmunoprecipitation assay (RIPA) (Beyotime). Then, bicinchoninic acid (BCA) method was performed to normalize protein concentrations among groups. After protein samples were mixed with 6×loading buffer (Beyotime), protein samples were layered by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to methyl alcohol-incubated polyvinylidene fluoride (PVDF) membrane. For immune reactions, proteins were incubated with 5% bovine serum albumin (BSA) solution for 1 hour at 37°C. Primary antibodies were diluted at a ratio of 1: 1000 and react with proteins at 4°C overnight. After washing by tris-buffered saline plus Tween (TBST) 10 minutes for 3 times, proteins were reacted with secondary antibodies: anti-mouse horseradish peroxidase (HRP) (Boster, 1: 1000) and anti-rabbit HRP (Boster, 1: 1000) for 2 hours at 37°C. Finally, the chemiluminescence was used to expose the protein bands. The blind observer photographed the results and performed analysis.
STATISTICAL ANALYSIS:
The qRT-PCR and western blot analysis were repeated for at least 3 times. The statistical analysis was performed using SPSS 19.0 software. The measurement data was depicted as mean±standard deviation (SD). The count data was depicted as frequency and percentage. Differences between 2 groups were analyzed
Results
UPREGULATION OF NF-κB/MIR-155 ASSOCIATED WITH THE INCREASED EXPRESSION OF INFLAMMATORY CYTOKINES IN EOS CRSWNP:
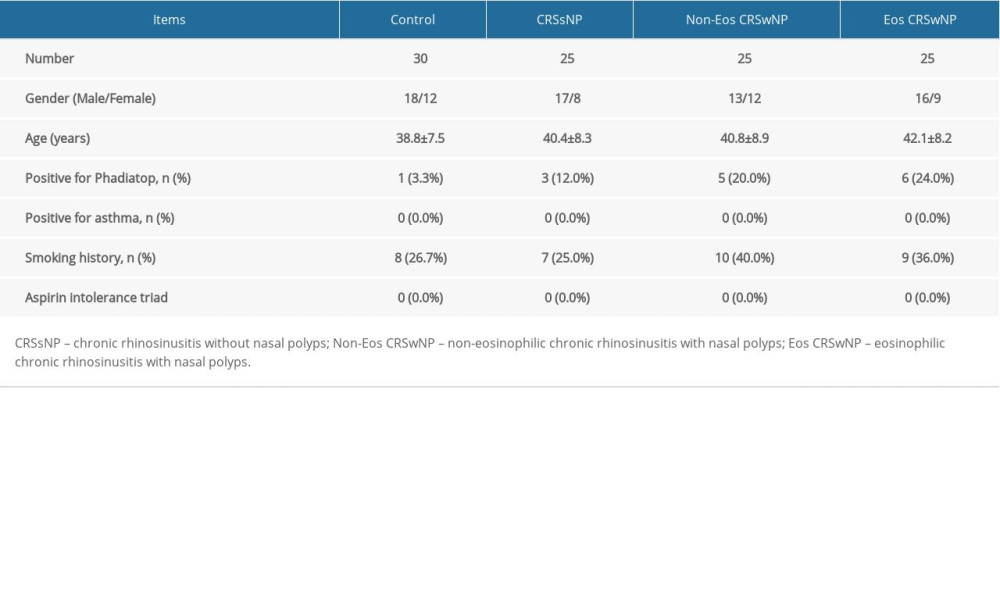
Firstly, the expression alterations of NF-κB/miR-155 signaling pathway among the control group, the Eos CRSwNP group, the Non-Eos CRSwNP, and the CRSsNP group were explored by western blot analysis and in situ hybridization. As shown in Figure 1, the expression level of NF-κB key proteins p-p65 and p-IKK were significantly increased in the Eos CRSwNP group compared with that in the control group (Figure 1A–1C, P<0.05). Despite the expression of p-p65, we found that the expression of p-IKK was also increased in the Non-Eos CRSwNP group and the CRSsNP group, however, it was still significantly less than that in the Eos CRSwNP group. On the contrary, the expression of NF-κB inhibitory protein IκB-α was significantly decreased in the Eos CRSwNP group compared with the control group, the Non-Eos CRSwNP group and the CRSsNP group (Figure 1A, 1D, P<0.05). Additionally, results showed that the number of miR-155 positive cells were significantly increased in the epithelial tissues of patients in the Eos CRSwNP group, the Non-Eos CRSwNP group and the CRSsNP group (Figure 1E, 1F, P<0.05) compared with that in the control group. The highest number of miR-155 positive cells in the epithelial tissues was recorded in the Eos CRSwNP group. These results indicated that the expression of NF-κB and miR-155 were both elevated in patients with Eos CRSwNP. The increased expression of NF-κB corresponded with the upregulation of miR-155.
We performed qRT-PCR analysis to explore the expression pattern of cytokines under the upregulation of NF-κB/miR-155 signaling pathway in patients with Eos CRSwNP. As shown in Figure 2, the messenger RNA (mRNA) expression level of the 4 inflammatory cytokines, tumor necrosis factor (TNF)-α, interleukin (IL)-1, IL-4, and IL-5, were all significantly increased in the Eos CRSwNP group compared with the control group, the Non-Eos CRSwNP group and the CRSsNP group (Figure 2A–2D, all P<0.05). These findings indicated that the upregulation of NF-κB/miR-155 might promote the expression of inflammatory cytokines in the epithelial tissues of patients with Eos CRSwNP.
DEX INHIBITED THE RELAPSE OF EOS CRSWNP VIA INHIBITING NF-κB/MIR-155:
Furthermore, we divided patients with Eos CRSwNP into the Eos CRSwNP-control group and the Eos CRSwNP-DEX group based on whether they received post-surgery DEX treatment for 12 weeks. Among 25 patients, 9 patients were assigned to Eos CRSwNP-control group while 16 patients were assigned to the Eos CRSwNP-DEX group. As shown in Figure 3, the primary expression of p-p65, p-IKK, and IκB-α was similar between the Eos CRSwNP-control group and the Eos CRSwNP-DEX group (Figure 3A–3D, P>0.05). However, after DEX treatment, the expression of p-p65 and p-IKK was significantly decreased while the expression of IκB-α was increased in the Eos CRSwNP-DEX group compared with the Eos CRSwNP-control group (Figure 3A–3D, P<0.05). The downregulation of NF-κB induced by DEX treatment was accompanied by decreased expression of miR-155 in the Eos CRSwNP-DEX group (Figure 3E, P<0.05). Additionally, after DEX treatment, the mRNA expression of the inflammatory cytokines TNF-α, IL-1, IL-4, and IL-5 were all significantly decreased in patients in the Eos CRSwNP-DEX group compared with patients in the Eos CRSwNP-control group (Figure 4A–4D, all P<0.05). During the 1-year follow-up, 3 cases in the Eos CRSwNP-control group relapsed while only 1 case in the Eos CRSwNP-DEX group relapsed. The relapse rate was also decreased in the Eos CRSwNP-DEX group compared with the Eos CRSwNP-control group (6.3% versus 33.3%). Taken together, these results suggest that post-surgery glucocorticoid treatment effectively inhibited the relapse of CRSwNP, and its underlying mechanism concerned with the downregulation of NF-κB/miR-155 and the expression pattern of cytokines.
MICE MODEL VERIFIED THE REGULATORY EFFECTS OF NF-κB/MIR-155 ON THE EXPRESSION PATTERN OF INFLAMMATORY CYTOKINES IN EOS CRSWNP:
To explore the role of NF-κB/miR-155 signaling pathway in regulating the inflammatory responses in Eos CRSwNP, the mice model of Eos CRSwNP was induced by OVA and SEB. As shown in Figure 5, the expression of p-p65 and p-IKK were significantly increased while the expression of IκB-α was decreased in the Eos CRSwNP group mice. However, application of PDTC or DEX both counterbalanced this alteration (Figure 5A–5D, P<0.05). Additionally, miR-155 was also upregulated in the Eos CRSwNP mice. Treatment of PDTC, miR-155 antagomir, or DEX also effectively inhibited the upregulation of miR-155 in the mice model of Eos CRSwNP (Figure 5E, P<0.05). This suggests that NF-κB/miR-155 was activated in Eos CRSwNP mice and miR-155 activation was dependent on NF-κB signals.
We also explored the expression alterations of miR-155 downstream target proteins COX2, the crucial inflammatory protein, as well as SOCS1, the crucial anti-inflammatory protein. Results showed that in the mice model of Eos CRSwNP, upon the upregulation of miR-155, the COX2 protein was significantly increased while the SOCS1 protein was significantly decreased (Figure 5F–5H, P<0.05) compared with the control group. However, treatment of PDTC, miR-155 antagomir, or DEX also reversed the aberrant expression pattern of COX2 and SOCS1. Additionally, the expression of cytokines was also explored by qRT-PCR analysis. Results revealed that compared with the control group, the Eos CRSwNP group had significantly elevated expressions of the cytokines TNF-α, IL-1, IL-4, and IL-5 in mice (Figure 6A–6D, all P<0.05). However, treatment of PDTC, miR-155 antagomir, or DEX abolished the upregulation of cytokines in the mice model of Eos CRSwNP. Taken together, these results indicated that the upregulation of NF-κB/miR-155 induced inflammatory responses and elevated expression pattern of cytokines in Eos CRSwNP. The protective effects of glucocorticoid treatment were derived from the inhibition of NF-κB/miR-155 signaling pathway.
Discussion
CRSwNP is one of the common diseases in otolaryngology and the incidence of CRSwNP remains approximately 4% in the general populations [28]. It severely affects the quality of patient’s health and brings a heavy financial burden to patients, due to its high relapse rate [1]. Even after proper drug treatment or surgery, it is still common for CRSwNP to relapse. According to a 12-year follow-up study, the relapse rate in patients with CRSwNP even reached 78.9% after endoscopic sinus surgery [3]. The high relapse rate is associated with the unclear pathogenesis of CRSwNP and ineffective treatments.
Clinical studies used to highlight the degree of eosinophils infiltration as the independent risk factor for the relapse of CRSwNP [29]. However, more and more studies have revealed that the expression pattern of cytokines is also associated with the relapse of nasal polyp [10,30]. Otto et al. [30] pointed that the expression pattern of cytokines in CRSwNP was Th2-related. In our study, the expression of the cytokines TNF-α, IL-1, IL-4, and IL-5 were dominant in CRSwNP, which was consistent with the Otto et al. study. TNF-α and IL-1 have been shown to be crucial pro-inflammatory cytokines and upregulation of TNF-α and IL-1 induces chronic inflammation, promotes the expression of adhesion molecules, and accelerates the recruitment of eosinophils [31]. On the contrary, prevention of TNF-α and IL-1 expression attenuates airway hyper-responsiveness and inflammation [31,32]. IL-4 and IL-5 are Th2-related cytokines and are crucial mediators in immunity response. The expression of IL-4 and IL-5 protects the survival of eosinophil and promotes eosinophils infiltration in CRSwNP [32]. Thus, the inhibition of IL-4 and IL-5 is also regarded as a potential treatment approach for CRSwNP [33]. In our study, results showed that compared with control group or the CRSsNP group, the expression of the cytokines TNF-α, IL-1, IL-4, and IL-5 were significantly increased in the CRSwNP group, especially in the Eos CRSwNP group. During a 1-year follow-up, patients who did not receive the DEX treatment for 12 weeks continued to have high expression levels of cytokines and the relapse rate was relatively high, which was consistent with other published clinical investigations. However, glucocorticoid treatment significantly decreased the expression level of cytokines in patients with Eos CRSwNP and glucocorticoid treatment led to low relapse rate. Our results also provide evidence that the expression level of inflammatory cytokines was crucial in the relapse of CRSwNP.
Until now, the role of NF-κB/miR-155 signaling pathway in regulating the inflammation responses of CRSwNP has not been well clarified. Ramis et al. [14] showed that NF-κB is activated in both CRS and NP tissues. The upregulation of NF-κB is also reported to intensively enhanced the expression of miR-155 [34]. The activation of NF-κB/miR-155 signaling pathway regulates the expression pattern of cytokines and finally regulates the inflammatory responses [16]. In our study, the expression of NF-κB active proteins was significantly increased while NF-κB inhibitory protein was significantly decreased in patients with Eos CRSwNP. The number of miR-155 positive cells in the epithelial tissues in the Eos CRSwNP group was also significantly more than the other 3 groups in our study. In the mice model of Eos CRSwNP, the upregulation of NF-κB/miR-155 was also detected by western blot and qRT-PCR analysis. The upregulation of NF-κB/miR-155 induced inflammatory responses via increase of COX2 and decrease of SOCS1. COX2 is also called the “gene of inflammatory responses”. The upregulation of COX2 has been reported in most inflammatory diseases and regulates the generation of prostaglandins. Qiu et al. [35] reported that miR-155 can bind COX2 and maintain mRNA stability. Additionally, SOCS1 is known as the anti-inflammatory mediator. SOCS1 negatively regulates the expression of inflammatory cytokines and promotes anti-inflammatory responses [36]. However, SOCS1 is inhibited by miR-155 through inducing the inflammatory mediator COX2 and inhibiting the anti-inflammatory mediator SOCS1. In our study, the upregulation of NF-κB/miR-155 promoted inflammatory responses in the mice model of Eos CRSwNP.
NF-κB/miR-155 also induced the expression of the cytokines TNF-α, IL-1, IL-4, and IL-5 in the mice model of Eos CRSwNP. However, treatment of PDTC, miR-155 antagomir, or DEX in mice all significantly reversed the aberrant expression pattern of cytokines. This indicates that glucocorticoid treatment received anti-inflammatory effects via inhibiting NF-κB/miR-155-induced inflammatory responses and aberrant expression pattern of cytokines. Our results also support the findings of Zheng et al. [37] that highlighted that downregulation of NF-κB/miR-155 by glucocorticoid treatment is a novel anti-inflammation mechanism. Despite the finding that glucocorticoid treatment had excellent treatment effects in our study, the effects of glucocorticoid treatment are still limited in glucocorticoid-resistant patients. Our study results provide a new view for the treatment of this type of patient, suggesting that direct inhibition of miR-155 may function as the potential treatment approach. Inhibition of miR-155 by miR-155 antagomir in our study also prevented inflammatory responses and significantly decreased the expression pattern of cytokines in a mice model of Eos CRSwNP. Thus, miR-155 may also function as the treatment target for patients with Eos CRSwNP.
Conclusions
In this study, we found the expression of NF-κB/miR-155 was increased in the epithelial tissues of patients with Eos CRSwNP, compared with patients with Non-Eos CRSwNP, CRSsNP or patients without inflammatory diseases. The upregulation of NF-κB/miR-155 promoted inflammatory response via inducing the inflammatory mediator COX2 and the inhibiting anti-inflammatory mediator SOCS1. This resulted in the aberrant expression pattern of several cytokines, TNF-α, IL-1, IL-4, and IL-5, in Eos CRSwNP patients. This molecular mechanism is associated with the high relapse rate of Eos CRSwNP. However, glucocorticoid treatment was found to inhibit the relapse of CRSwNP via inhibiting the upregulation of NF-κB/miR-155 signaling pathway. Our findings also highlight miR-155 as a potential therapeutic target for patients with Eos CRSwNP.
Figures
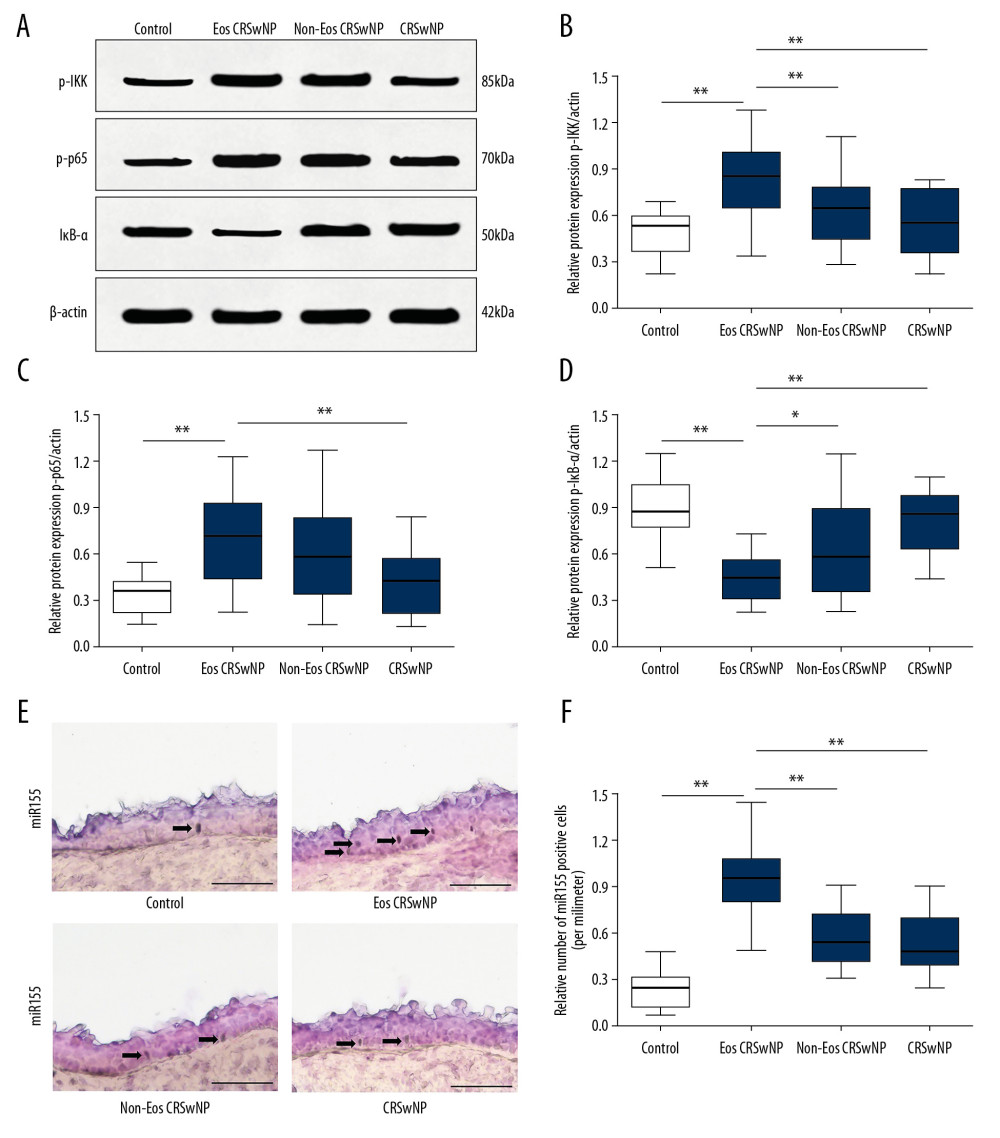 Figure 1. Expression of NF-κB/miR-155 in patients. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in Eos CRSwNP, Non-Eos CRSwNP, CRSsNP, and control group. (B–D) Statistical analysis displayed compared with Non-Eos CRSwNP, CRSsNP, and control group, the expression of p-IKK and p-p65 were significantly elevated while the expression of IκB-α was decreased in Eos CRSwNP group. (E) In situ hybridization of miR-155 in patients. Scale bar=100 μm (F) Statistical analysis displayed the number of miR-155 positive cells were significantly increased in Eos CRSwNP group. * P<0.05; ** P<0.01. NF-κB – nuclear factor κB; CRSsNP – chronic rhinosinusitis without nasal polyps; Non-Eos CRSwNP – non-eosinophilic chronic rhinosinusitis with nasal polyps; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps.
Figure 1. Expression of NF-κB/miR-155 in patients. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in Eos CRSwNP, Non-Eos CRSwNP, CRSsNP, and control group. (B–D) Statistical analysis displayed compared with Non-Eos CRSwNP, CRSsNP, and control group, the expression of p-IKK and p-p65 were significantly elevated while the expression of IκB-α was decreased in Eos CRSwNP group. (E) In situ hybridization of miR-155 in patients. Scale bar=100 μm (F) Statistical analysis displayed the number of miR-155 positive cells were significantly increased in Eos CRSwNP group. * P<0.05; ** P<0.01. NF-κB – nuclear factor κB; CRSsNP – chronic rhinosinusitis without nasal polyps; Non-Eos CRSwNP – non-eosinophilic chronic rhinosinusitis with nasal polyps; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps. 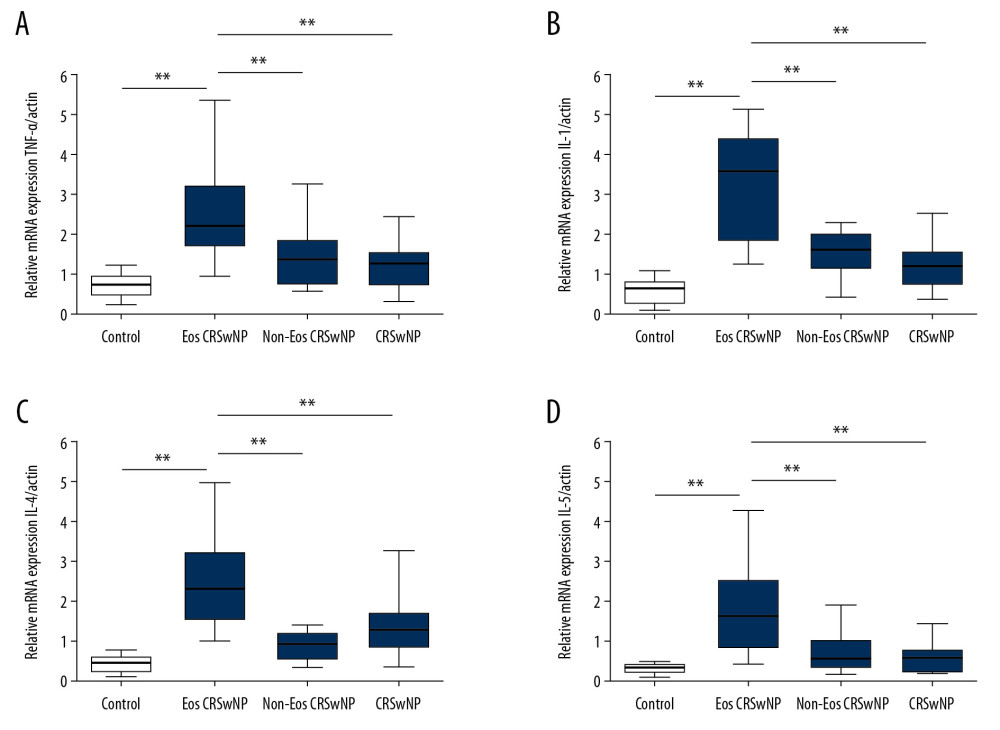 Figure 2. Expression of TNF-α, IL-1, IL-4, and IL-5 in patients. (A–D) qRT-PCR results displayed the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly increased in Eos CRSwNP group. ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; aRT-PCR – quantitative real-time polymerase chain reaction; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps.
Figure 2. Expression of TNF-α, IL-1, IL-4, and IL-5 in patients. (A–D) qRT-PCR results displayed the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly increased in Eos CRSwNP group. ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; aRT-PCR – quantitative real-time polymerase chain reaction; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps.  Figure 3. Expression of NF-κB/miR-155 in Eos CRSwNP patients with or without DEX treatment. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in Eos CRSwNP-control and Eos CRSwNP-DEX group. (B–D) Statistical analysis displayed after DEX treatment, the expression of p-IKK and p-p65 were significantly decreased while the expression of IκB-α was significantly increased in Eos CRSwNP patients. (E) After DEX treatment, the expression of miR-155 was also decreased in Eos CRSwNP patients. NF-κB – nuclear factor κB; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps; DEX – dexamethasone.
Figure 3. Expression of NF-κB/miR-155 in Eos CRSwNP patients with or without DEX treatment. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in Eos CRSwNP-control and Eos CRSwNP-DEX group. (B–D) Statistical analysis displayed after DEX treatment, the expression of p-IKK and p-p65 were significantly decreased while the expression of IκB-α was significantly increased in Eos CRSwNP patients. (E) After DEX treatment, the expression of miR-155 was also decreased in Eos CRSwNP patients. NF-κB – nuclear factor κB; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps; DEX – dexamethasone. 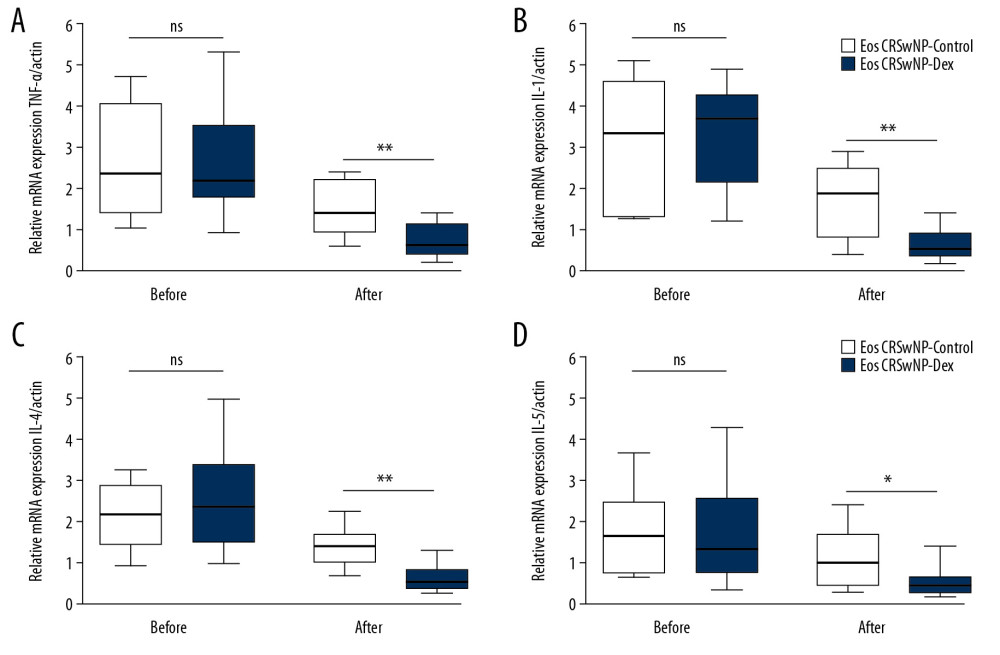 Figure 4. Expression of TNF-α, IL-1, IL-4, and IL-5 in Eos CRSwNP patients with or without DEX treatment. (A–D) qRT-PCR results displayed after DEX treatment, the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly increased in Eos CRSwNP patients. * P<0.05; ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; qRT-PCR – quantitative real-time polymerase chain reaction; DEX – dexamethasone; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps.
Figure 4. Expression of TNF-α, IL-1, IL-4, and IL-5 in Eos CRSwNP patients with or without DEX treatment. (A–D) qRT-PCR results displayed after DEX treatment, the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly increased in Eos CRSwNP patients. * P<0.05; ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; qRT-PCR – quantitative real-time polymerase chain reaction; DEX – dexamethasone; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps. 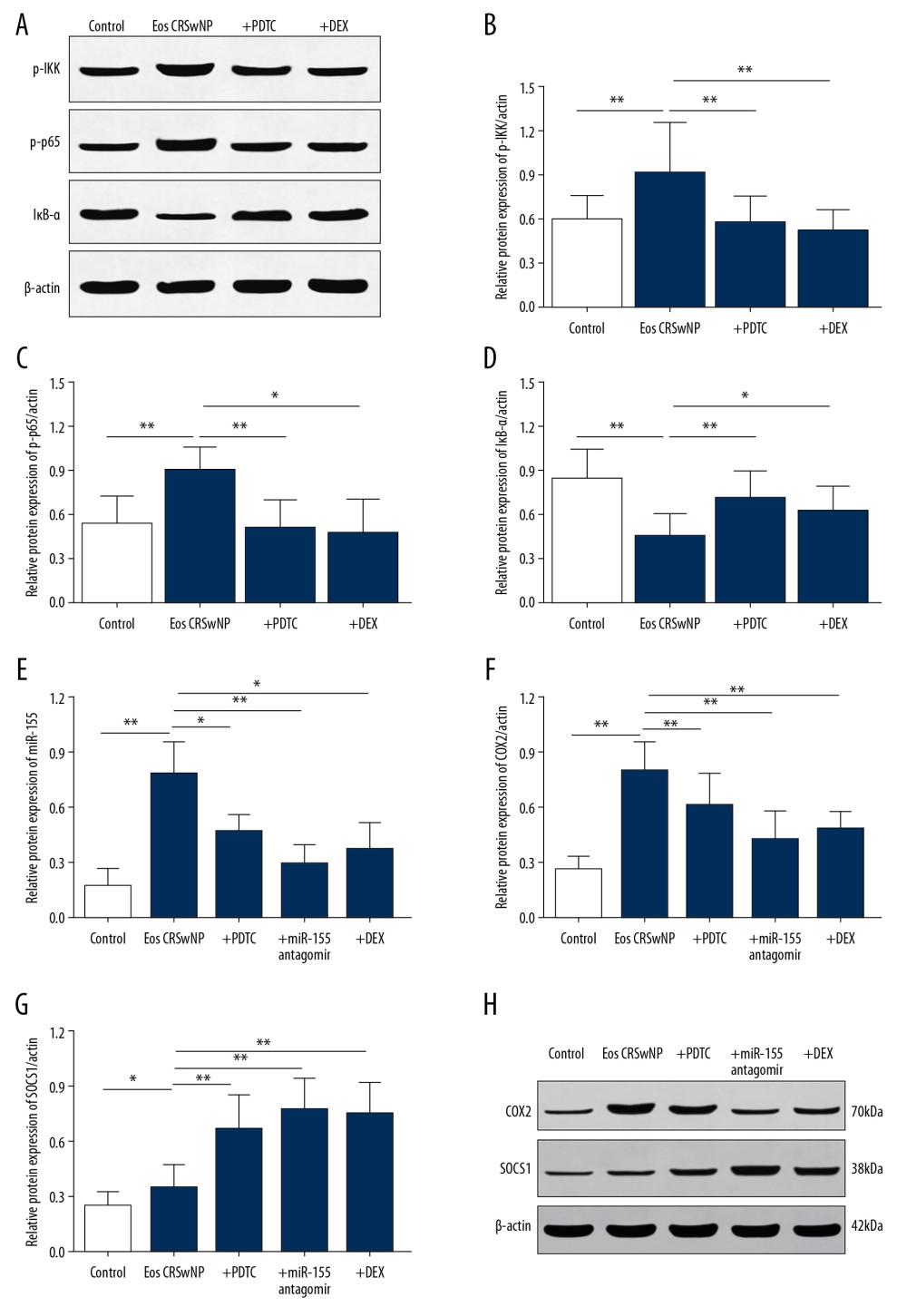 Figure 5. Expression of NF-κB/miR-155 in mice model of Eos CRSwNP. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in control, Eos CRSwNP, PDTC, and DEX groups. (B–D) Statistical analysis displayed the expression of p-IKK and p-p65 were significantly increased while the expression of IκB-α was decreased in the Eos CRSwNP group. However, the application of PDTC or DEX reversed the expression alterations, which was similar with the control group. (E) The expression of miR-155 was significantly increased in the Eos CRSwNP group. The application of PDTC, miR-155 antagomir, or DEX abolished the elevation of miR-155 in the Eos CRSwNP group. (F, G) Statistical analysis showed that after application of PDTC, miR-155 antagomir, or DEX, the expression of COX2 was significantly decreased while the expression of SOCS1 was significantly increased in the Eos CRSwNP group. (H) Western blot analysis of COX2 and SOCS1 in groups. * P<0.05; ** P<0.01. NF-κB – nuclear factor κB; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps; DEX – dexamethasone; PDTC – pyrrolidine dithiocarbonate.
Figure 5. Expression of NF-κB/miR-155 in mice model of Eos CRSwNP. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in control, Eos CRSwNP, PDTC, and DEX groups. (B–D) Statistical analysis displayed the expression of p-IKK and p-p65 were significantly increased while the expression of IκB-α was decreased in the Eos CRSwNP group. However, the application of PDTC or DEX reversed the expression alterations, which was similar with the control group. (E) The expression of miR-155 was significantly increased in the Eos CRSwNP group. The application of PDTC, miR-155 antagomir, or DEX abolished the elevation of miR-155 in the Eos CRSwNP group. (F, G) Statistical analysis showed that after application of PDTC, miR-155 antagomir, or DEX, the expression of COX2 was significantly decreased while the expression of SOCS1 was significantly increased in the Eos CRSwNP group. (H) Western blot analysis of COX2 and SOCS1 in groups. * P<0.05; ** P<0.01. NF-κB – nuclear factor κB; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps; DEX – dexamethasone; PDTC – pyrrolidine dithiocarbonate. 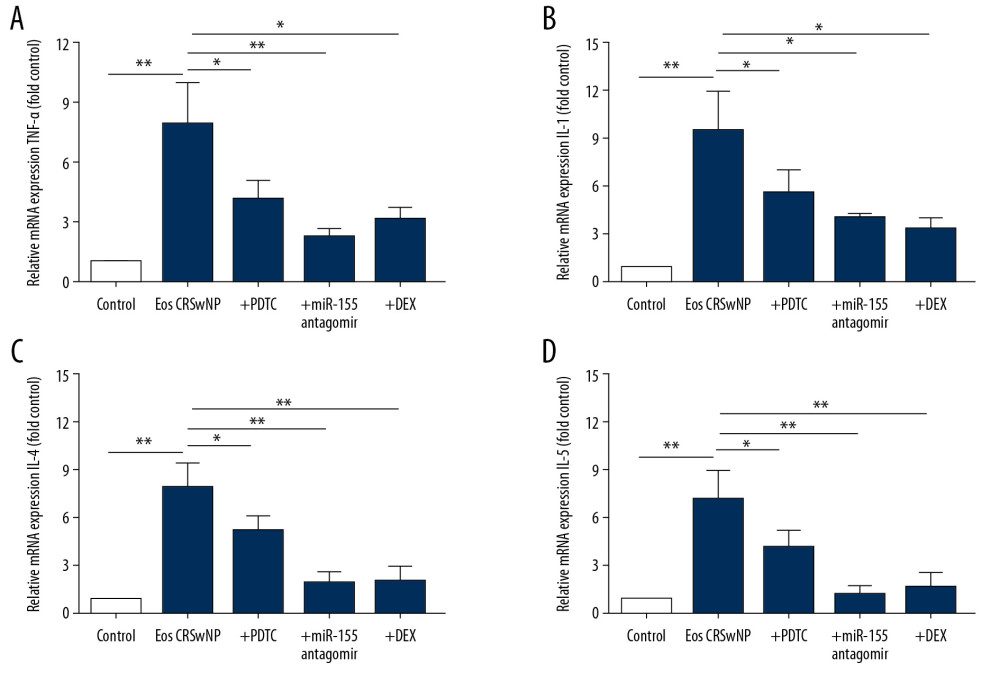 Figure 6. Expression of TNF-α, IL-1, IL-4, and IL-5 in mice model of Eos CRSwNP. (A–D) qRT-PCR results displayed after application of PDTC, miR-155 antagomir, or DEX, the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly decreased in Eos CRSwNP group. * P<0.05; ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; qRT-PCR – quantitative real-time polymerase chain reaction; PDTC – pyrrolidine dithiocarbonate; DEX – dexamethasone.
Figure 6. Expression of TNF-α, IL-1, IL-4, and IL-5 in mice model of Eos CRSwNP. (A–D) qRT-PCR results displayed after application of PDTC, miR-155 antagomir, or DEX, the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly decreased in Eos CRSwNP group. * P<0.05; ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; qRT-PCR – quantitative real-time polymerase chain reaction; PDTC – pyrrolidine dithiocarbonate; DEX – dexamethasone. References
1. Marioni G, Zanotti C, Brescia G, Chronic rhinosinusitis with nasal polyps in the elderly: Assessing current evidence: Allergy Asthma Proc, 2018; 39(1); 9-13
2. Hopkins C, Slack R, Lund V, Long-term outcomes from the English national comparative audit of surgery for nasal polyposis and chronic rhinosinusitis: Laryngoscope, 2009; 119(12); 2459-65
3. Calus L, Van Bruaene N, Bosteels C, Twelve-year follow-up study after endoscopic sinus surgery in patients with chronic rhinosinusitis with nasal polyposis: Clin Transl Allergy, 2019; 9; 30
4. Matsuwaki Y, Ookushi T, Asaka D, Chronic rhinosinusitis: Risk factors for the recurrence of chronic rhinosinusitis based on 5-year follow-up after endoscopic sinus surgery: Int Arch Allergy Immunol, 2008; 146(Suppl 1); 77-81
5. Staikuniene J, Vaitkus S, Japertiene LM, Association of chronic rhinosinusitis with nasal polyps and asthma: Clinical and radiological features, allergy, and inflammation markers: Medicina (Kaunas), 2008; 44(4); 257-65
6. Yacoub M, Trimarchi M, Cremona G, Are atopy and eosinophilic bronchial inflammation associated with relapsing forms of chronic rhinosinusitis with nasal polyps?: Clin Mol Allergy, 2015; 13(1); 23
7. Nunes FB, Becker CG, Becker HM, Cytokines profile in cystic fibrosis patients with nasal polyps: Rev Laryngol Otol Rhinol (Bord), 2009; 130(3); 163-67
8. Meng J, Zhou P, Liu Y, The development of nasal polyp disease involves early nasal mucosal inflammation and remodelling: PLoS One, 2013; 8(12); e82373
9. Zheng M, Wang M, Li YExpression of immunological and inflammatory biomarkers in chronic rhinosinusitis with nasal polyps and its predictive value for recurrence: Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi, 2019; 54(3); 174-80 [in Chinese]
10. Huriyati E, Darwin E, Yanwirasti Y, Association of inflammation mediator in mucosal and tissue of chronic rhinosinusitis with recurrent nasal polyp: Open Access Maced J Med Sci, 2019; 7(10); 1635-40
11. Yu RL, Zhu DD, Dong ZEffects of glucocorticoid on tissue remodeling of nasal mucosa of chronic rhinosinusitis with nasal polyposis after endoscopic surgery: Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi, 2006; 41(10); 773-76 [in Chinese]
12. Benson M, Pathophysiological effects of glucocorticoids on nasal polyps: An update: Curr Opin Allergy Clin Immunol, 2005; 5(1); 31-35
13. Hayden MS, Ghosh S, Signaling to nf-kappab: Genes Dev, 2004; 18(18); 2195-224
14. Ramis I, Bioque G, Lorente J, Constitutive nuclear factor-kappab activity in human upper airway tissues and nasal epithelial cells: Eur Respir J, 2000; 15(3); 582-89
15. Chen JH, Yang W, Tao ZZExpressions of nf-κbp65, iκbα in human nasal polyps: Chinese Journal of Otorhinolaryngology-Skull Base Surgery, 2006; 12; 96-100 [in Chinese]
16. Xiaodong M, Buscaglia LEB, Barker JR, MicroRNAas in NF-kappab signaling: J Mol Cell Biol, 2011; 3(3); 159-66
17. Carthew RW, Sontheimer EJ, Origins and mechanisms of miRNAs and siRNAs: Cell, 2009; 136(4); 642-55
18. Sonkoly E, Pivarcsi A, MicroRNAs in inflammation: Int Rev Immunol, 2009; 28(6); 535-61
19. Esmerina T, Jean JM, Amelia C, Modulation of mir-155 and mir-125b levels following lipopolysaccharide/TNF-alpha stimulation and their possible roles in regulating the response to endotoxin shock: J Immunol, 2007; 179(8); 5082-89
20. O’Connell RM, Kahn D, Gibson WSJ, Microrna-155 promotes autoimmune inflammation by enhancing inflammatory t cell development: Immunity, 2010; 33(4); 607-19
21. Xiaochuan L, Feng T, Fei W, Rheumatoid arthritis-associated microRNA-155 targets SOCS1 and upregulates TNFα and IL-1β in PBMCs: Int J Mol Sci, 2013; 14(12); 23910-21
22. Ceppi M, Pereira PM, Dunand-Sauthier I, MicroRNA-155 modulates the interleukin-1 signaling pathway in activated human monocyte-derived dendritic cells: Proc Natl Acad Sci USA, 2009; 106(8); 2735-40
23. Pena-Philippides JC, Caballero-Garrido E, Lordkipanidze T: J Neuroinflamm, 2016; 13(1); 287
24. Xia G, Bao L, Gao W, Differentially expressed miRNA in inflammatory mucosa of chronic rhinosinusitis: J Nanosci Nanotechnol, 2015; 15(3); 2132-39
25. Wytske F, Valerie L, Joaquim M, Ep3os 2007: European position paper on rhinosinusitis and nasal polyps 2007. A summary for otorhinolaryngologists: Rhinology, 2007; 45(2); 97-101
26. Lou H, Meng Y, Piao Y, Predictive significance of tissue eosinophilia for nasal polyp recurrence in the Chinese population: Am J Rhinol Allergy, 2015; 29(5); 350-56
27. Kim DW, Khalmuratova R, Hur DG: Am J Rhinol Allergy, 2011; 25(6); e255-61
28. Amali A, Bidar Z, Rahavi-Ezabadi S, Polypoid change of middle turbinate is associated to an increased risk of polyp recurrence after surgery in patients with chronic rhinosinusitis with nasal polyps: Eur Arch Otorhinolaryngol, 2018; 275(8); 2021-25
29. Vlaminck S, Vauterin T, Hellings PW, The importance of local eosinophilia in the surgical outcome of chronic rhinosinusitis: A 3-year prospective observational study: Am J Rhinol Allergy, 2014; 28(3); 260-64
30. Otto BA, Wenzel SE, The role of cytokines in chronic rhinosinusitis with nasal polyps: Curr Opin Otolaryngol Head Neck Surg, 2008; 16(3); 270-74
31. Oyer SL, Nagel W, Mulligan JK, Differential expression of adhesion molecules by sinonasal fibroblasts among control and chronic rhinosinusitis patients: Am J Rhinol Allergy, 2013; 27(5); 381-86
32. Cherry WB, Yoon J, Bartemes KR, A novel IL-1 family cytokine, IL-33, potently activates human eosinophils: J Allergy Clin Immunol, 2008; 121(6); 1484-90
33. Philippe G, Nicholas VB, Tom C, Mepolizumab, a humanized anti-IL-5 MAB, as a treatment option for severe nasal polyposis: J Allergy Clin Immunol, 2011; 128(5); 989-95.e1-8
34. Duan Q, Mao X, Xiao Y, Super enhancers at the mir-146a and mir-155 genes contribute to self-regulation of inflammation: Biochim Biophys Acta, 2016; 1859(4); 564-71
35. Qiu L, Zhang Y, Do DC, Mir-155 modulates cockroach allergen- and oxidative stress-induced cyclooxygenase-2 in asthma: J Immunol, 2018; 201(3); 916-29
36. Davey GM, Heath WR, Starr R, SOCS1: A potent and multifaceted regulator of cytokines and cell-mediated inflammation: Tissue Antigens, 2006; 67(1); 1-9
37. Zheng Y, Xiong S, Jiang P, Glucocorticoids inhibit lipopolysaccharide-mediated inflammatory response by downregulating microRNA-155: A novel anti-inflammation mechanism: Free Radic Biol Med, 2012; 52(8); 1307-17
Figures
 Figure 1. Expression of NF-κB/miR-155 in patients. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in Eos CRSwNP, Non-Eos CRSwNP, CRSsNP, and control group. (B–D) Statistical analysis displayed compared with Non-Eos CRSwNP, CRSsNP, and control group, the expression of p-IKK and p-p65 were significantly elevated while the expression of IκB-α was decreased in Eos CRSwNP group. (E) In situ hybridization of miR-155 in patients. Scale bar=100 μm (F) Statistical analysis displayed the number of miR-155 positive cells were significantly increased in Eos CRSwNP group. * P<0.05; ** P<0.01. NF-κB – nuclear factor κB; CRSsNP – chronic rhinosinusitis without nasal polyps; Non-Eos CRSwNP – non-eosinophilic chronic rhinosinusitis with nasal polyps; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps.
Figure 1. Expression of NF-κB/miR-155 in patients. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in Eos CRSwNP, Non-Eos CRSwNP, CRSsNP, and control group. (B–D) Statistical analysis displayed compared with Non-Eos CRSwNP, CRSsNP, and control group, the expression of p-IKK and p-p65 were significantly elevated while the expression of IκB-α was decreased in Eos CRSwNP group. (E) In situ hybridization of miR-155 in patients. Scale bar=100 μm (F) Statistical analysis displayed the number of miR-155 positive cells were significantly increased in Eos CRSwNP group. * P<0.05; ** P<0.01. NF-κB – nuclear factor κB; CRSsNP – chronic rhinosinusitis without nasal polyps; Non-Eos CRSwNP – non-eosinophilic chronic rhinosinusitis with nasal polyps; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps. Figure 2. Expression of TNF-α, IL-1, IL-4, and IL-5 in patients. (A–D) qRT-PCR results displayed the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly increased in Eos CRSwNP group. ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; aRT-PCR – quantitative real-time polymerase chain reaction; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps.
Figure 2. Expression of TNF-α, IL-1, IL-4, and IL-5 in patients. (A–D) qRT-PCR results displayed the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly increased in Eos CRSwNP group. ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; aRT-PCR – quantitative real-time polymerase chain reaction; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps. Figure 3. Expression of NF-κB/miR-155 in Eos CRSwNP patients with or without DEX treatment. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in Eos CRSwNP-control and Eos CRSwNP-DEX group. (B–D) Statistical analysis displayed after DEX treatment, the expression of p-IKK and p-p65 were significantly decreased while the expression of IκB-α was significantly increased in Eos CRSwNP patients. (E) After DEX treatment, the expression of miR-155 was also decreased in Eos CRSwNP patients. NF-κB – nuclear factor κB; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps; DEX – dexamethasone.
Figure 3. Expression of NF-κB/miR-155 in Eos CRSwNP patients with or without DEX treatment. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in Eos CRSwNP-control and Eos CRSwNP-DEX group. (B–D) Statistical analysis displayed after DEX treatment, the expression of p-IKK and p-p65 were significantly decreased while the expression of IκB-α was significantly increased in Eos CRSwNP patients. (E) After DEX treatment, the expression of miR-155 was also decreased in Eos CRSwNP patients. NF-κB – nuclear factor κB; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps; DEX – dexamethasone. Figure 4. Expression of TNF-α, IL-1, IL-4, and IL-5 in Eos CRSwNP patients with or without DEX treatment. (A–D) qRT-PCR results displayed after DEX treatment, the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly increased in Eos CRSwNP patients. * P<0.05; ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; qRT-PCR – quantitative real-time polymerase chain reaction; DEX – dexamethasone; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps.
Figure 4. Expression of TNF-α, IL-1, IL-4, and IL-5 in Eos CRSwNP patients with or without DEX treatment. (A–D) qRT-PCR results displayed after DEX treatment, the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly increased in Eos CRSwNP patients. * P<0.05; ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; qRT-PCR – quantitative real-time polymerase chain reaction; DEX – dexamethasone; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps. Figure 5. Expression of NF-κB/miR-155 in mice model of Eos CRSwNP. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in control, Eos CRSwNP, PDTC, and DEX groups. (B–D) Statistical analysis displayed the expression of p-IKK and p-p65 were significantly increased while the expression of IκB-α was decreased in the Eos CRSwNP group. However, the application of PDTC or DEX reversed the expression alterations, which was similar with the control group. (E) The expression of miR-155 was significantly increased in the Eos CRSwNP group. The application of PDTC, miR-155 antagomir, or DEX abolished the elevation of miR-155 in the Eos CRSwNP group. (F, G) Statistical analysis showed that after application of PDTC, miR-155 antagomir, or DEX, the expression of COX2 was significantly decreased while the expression of SOCS1 was significantly increased in the Eos CRSwNP group. (H) Western blot analysis of COX2 and SOCS1 in groups. * P<0.05; ** P<0.01. NF-κB – nuclear factor κB; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps; DEX – dexamethasone; PDTC – pyrrolidine dithiocarbonate.
Figure 5. Expression of NF-κB/miR-155 in mice model of Eos CRSwNP. (A) Western blot analysis of p-IKK, p-p65, and IκB-α in control, Eos CRSwNP, PDTC, and DEX groups. (B–D) Statistical analysis displayed the expression of p-IKK and p-p65 were significantly increased while the expression of IκB-α was decreased in the Eos CRSwNP group. However, the application of PDTC or DEX reversed the expression alterations, which was similar with the control group. (E) The expression of miR-155 was significantly increased in the Eos CRSwNP group. The application of PDTC, miR-155 antagomir, or DEX abolished the elevation of miR-155 in the Eos CRSwNP group. (F, G) Statistical analysis showed that after application of PDTC, miR-155 antagomir, or DEX, the expression of COX2 was significantly decreased while the expression of SOCS1 was significantly increased in the Eos CRSwNP group. (H) Western blot analysis of COX2 and SOCS1 in groups. * P<0.05; ** P<0.01. NF-κB – nuclear factor κB; Eos CRSwNP – eosinophilic chronic rhinosinusitis with nasal polyps; DEX – dexamethasone; PDTC – pyrrolidine dithiocarbonate. Figure 6. Expression of TNF-α, IL-1, IL-4, and IL-5 in mice model of Eos CRSwNP. (A–D) qRT-PCR results displayed after application of PDTC, miR-155 antagomir, or DEX, the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly decreased in Eos CRSwNP group. * P<0.05; ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; qRT-PCR – quantitative real-time polymerase chain reaction; PDTC – pyrrolidine dithiocarbonate; DEX – dexamethasone.
Figure 6. Expression of TNF-α, IL-1, IL-4, and IL-5 in mice model of Eos CRSwNP. (A–D) qRT-PCR results displayed after application of PDTC, miR-155 antagomir, or DEX, the expression of TNF-α, IL-1, IL-4, and IL-5 were all significantly decreased in Eos CRSwNP group. * P<0.05; ** P<0.01. TNF – tumor necrosis factor; IL – interleukin; qRT-PCR – quantitative real-time polymerase chain reaction; PDTC – pyrrolidine dithiocarbonate; DEX – dexamethasone. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387