23 December 2020: Clinical Research
A Real-World Study of Cerebral 99mTc-TRODAT-1 Single-Photon Emission Computed Tomography (SPECT) Imaging of the Dopamine Transporter in Patients with Parkinson Disease from a Tertiary Hospital in Brazil
Michelly Arjona1CDEF, Juliana M.P. Toldo1BCDEF, Natalia Carvalho Queiroz1ABCD, Jose Luiz Pedroso2ACD, Guilherme de Carvalho Campos Neto2ACD, Orlando G.P. Barsottini2ACD, Andre C. Felicio1ABCDEF*DOI: 10.12659/MSM.925130
Med Sci Monit 2020; 26:e925130
Abstract
BACKGROUND: Studies on the routine clinical use of dopamine transporter (DAT) imaging have largely been conducted in Europe and the United States. In this real-world study, we investigated the use of cerebral 99mTc-TRODAT-1 SPECT imaging of DAT in patients with Parkinson disease (PD) at a tertiary hospital in Brazil.
MATERIAL AND METHODS: We included 119 patients with suspected PD or clinically unclear parkinsonism who underwent brain scintigraphy with 99mTc-TRODAT-1 during a 3-year period. Additionally, a brief interview was conducted with the physician who requested the scan to determine the usefulness of the method in clinical decision-making.
RESULTS: Regarding the scan requests, most were intended to evaluate or confirm dopaminergic denervation (69%), distinguish PD from essential tremor (10%), or distinguish degenerative parkinsonism from drug-induced parkinsonism (6%). Data analysis showed that scintigraphy with 99mTc-TRODAT-1 was useful in 85% of cases, changing the management of 75% of the patients who underwent a scan. The majority of physicians who requested the scan were neurologists, and 54% were self-reported movement disorder specialists. An inappropriate use of DAT imaging was seen in 5% of cases.
CONCLUSIONS: This study demonstrated that brain scintigraphy with the DAT ligand 99mTc-TRODAT-1 may influence diagnostic or therapeutic interventions, meaning that Brazilian physicians who requested the exam have taken in vivo DAT results into account at the time of clinical decision-making.
Keywords: Brain Mapping, perfusion imaging, Brain, Brazil, Dopamine Plasma Membrane Transport Proteins, Organotechnetium Compounds, Radionuclide Imaging, Tertiary Care Centers, Tomography, Emission-Computed, Single-Photon, Tropanes
Background
Parkinson disease (PD) is a slowly progressive neurodegenerative disorder with motor (bradykinesia, rigidity, rest tremor, and postural instability) and nonmotor (hyposmia, REM sleep behavior disorder, depression, constipation) symptoms [1]. PD is the second most common neurodegenerative disorder after Alzheimer disease. The overall prevalence of PD is estimated at 1% of the population above 60 years [2]. In Brazil, one of the most populous countries in the world, there is little epidemiological information on PD. One study conducted in a small Brazilian population showed a 3.3% prevalence of PD in people aged 65 or older [3].
Age is the most important unchangeable risk factor for PD [4], and the percentage of the population that is elderly has been increasing worldwide in the past several decades. For example, in Brazil, only 4.1% of the population was elderly (≥60 years) in the 1940s, and this percentage has been predicted to be 13.8% and 33.7% in 2020 and 2060, respectively [5].
Clinical diagnosis of PD can be challenging, and recent studies have shown higher rates of diagnostic error in up to 47% of suspected PD cases with less than 5 years of disease duration [6]. Additionally, an incorrect diagnosis can lead to treatment with inappropriate medications and unnecessary adverse effects [7].
As suggested by the European Federation of Neurological Society, selected subsidiary examinations such as dopamine transporter (DAT) imaging could be effective in the differential diagnosis of PD [8]. Through DAT imaging, the dopaminergic nigrostriatal system can be assessed
A systematic review of the diagnostic accuracy of DAT scintigraphy in patients with PD or clinically uncertain parkinsonism showed that sensitivity and specificity to detect nigrostriatal cell loss reached 98% to 100%, supporting the conclusion that DAT imaging is accurate in detecting nigrostriatal cell loss in patients with parkinsonism [10]. Also, another study showed that striatal DAT binding was highly correlated with postmortem substantia nigra cell counts, corroborating the validity of DAT imaging as an
Studies have assessed the role of DAT imaging in clinical practice, mainly in Europe and the United States, where this technique is widely available [12]. Indeed, DAT imaging is easier to interpret than modern magnetic resonance imaging, and it has a lower cost than positron emission techniques [13]. Two Brazilian studies using DAT imaging to differentiate PD patients from normal healthy subjects showed that this technique had high sensitivity (100%) and specificity (89%) [14,15].
DAT imaging using single-photon computed tomography is generally performed using iodine or technetium-labeled ligands, such as 123I-FP-β-CIT or 99mTc-TRODAT-1. While image acquisition using 123I-FP-β-CIT generally requires waiting 3 to 6 h after the injection of the radioligand [16], 99mTc-TRODAT-1 image acquisition can start 3 to 4 hours after this injection. In addition, technetium-based ligands are more widely available in nuclear facilities than iodine ones [17]. One study has compared the usefulness of both ligands to aid PD diagnosis and showed similar results but suggested that 123I-FP-β-CIT could be more valuable in following dopaminergic denervation longitudinally [18]. In Brazil, commercial access to DAT imaging using 99mTc-TRODAT-1 as ligand has been available since 2012. However, brain scintigraphy with 99mTc-TRODAT-1 in Brazil is limited to specific sites and is not covered by the majority of private health insurances, nor is it a covered service under the public health system. Further, there is a lack of information about how Brazilian physicians are requesting this tool in real-world clinical practice, and specifically, its role in assisting diagnoses in the context of PD or other forms of parkinsonism.
The main objective of our study was to investigate the use of cerebral 99mTc-TRODAT-1 SPECT imaging of DAT in patients with PD from a tertiary hospital in Brazil as a tool to assist in the diagnosis of PD or clinically unclear cases of parkinsonism in a series of Brazilian patients.
Material and Methods
SETTING AND ETHICS:
The study was carried out in the Nuclear Medicine Department of Hospital Israelita Albert Einstein, a private 646-bed tertiary teaching hospital in São Paulo, Brazil. Brain scintigraphy with the DAT ligand 99mTc-TRODAT-1 was first introduced in Brazil at this hospital in 2005, followed by the first study using this radioligand [19] and finally its commercial approval in 2012.
The local Research Ethics Committee approved this protocol (no. 2472-15), and the physicians who were invited to participate in the study signed an informed consent. Due to the retrospective nature of data collection, informed consent by patients was not required.
DATA COLLECTION:
We retrospectively selected patients with the clinical diagnosis of possible or probable PD according to Gelb et al. [20] and cases in which there was diagnostic doubt. The new criteria for PD suggested by the Movement Disorder Task Force [21] was not used in this study because DAT scans were mostly done before these criteria were published. The timing was confirmed when we interviewed physicians face-to-face or online. The period regarding data selection included an approximately 3-year period (January 2013 to May 2016) with 287 DAT scans. Of these, 168 (~58%) were excluded from our study (Figure 1) mainly owing to (1) nuclear physician report not found; (2) scintigraphy lacking the physician request, which prevented evaluation of the clinical indication and medical history; (3) duplicate scintigraphy (in cases in which patients were unable to complete the first examination, generally due to claustrophobia); and (4) physician not found or did not respond/agree to participate in the study. Statistical analysis was done with the remaining 119 brain scintigraphy studies.
MEDICAL INTERVIEW:
A brief structured medical interview was conducted with all physicians who requested the imaging scans included in the current study. The questionnaire contained objective questions regarding each patient’s symptoms and diagnostic criteria used, purpose of the imaging request, possible changes in management after the scintigraphy results were obtained, follow-up period, and finally, the physician’s expertise (general neurologist, neurologist self-reported as a movement disorder specialist, or others). The main objective of this questionnaire was to evaluate the clinical usefulness of the DAT imaging.
The questionnaire was also aimed at investigating the reasons why physicians considered their patients’ cases as clinically unclear parkinsonism by taking into account (1) lack of bradykinesia or tremor interfering with bradykinesia evaluation, (2) symmetric symptoms, (3) atypical tremor, (4) the presence of early cognitive impairment or early severe dysautonomia, (5) early signs of postural imbalance or ocular abnormalities, (6) the presence of cortical signs such as apraxia or agnosia and lack of a clear movement disorder, (7) sudden onset, (8) incongruent signs and symptoms with a fluctuating pattern, (9) the use of dopamine-depleting drugs, and (10) poor or inconclusive response to levodopa.
SCINTIGRAPHY WITH 99MTC-TRODAT-1 PROCEDURE:
Brain scintigraphy was performed using 99mTc-TRODAT-1 to assess DAT availability. Each subject intravenously received 925-1110 MBq (25-30 mCi) of 99mTc-TRODAT-1 (Institute of Nuclear Energy Research, Lungtan, Taiwan). The brain images were acquired 240 min after the injection using a dual-head gamma camera with ultra-high-resolution fanbeam collimators (Discovery NM/CT 670, GE Healthcare), which yielded an image resolution of approximately 2.0 mm full-width at half maximum (Figure 2). Data were acquired over a circular 360° rotation in 120 steps, 50 s per step, in a 128×128 matrix. Images were reconstructed by ordered subset expectation maximization using a Butterworth filter (cutoff frequency, 0.4 Nyquist; power factor 7) and attenuation correction by the Chang method. DaTQUANT software (GE Healthcare) [22] was used to define the caudate, putamen, striatum, and the occipital regions for the measurement of striatal 99mTc-TRODAT-1 binding. The average counts from the occipital region were used to determine the nonspecific activity. The specific striatal 99mTc-TRODAT-1 binding (which represents striatal DAT availability) was calculated as follows: (striatal ROI counts – occipital ROI counts)/(occipital ROI counts). Binding was calculated for the whole striatum, putamen, and caudate head of each hemisphere. Qualitative reports such as normal or abnormal DAT results rather than quantitative DAT binding potentials were taken into consideration at the time of statistical analysis.
Normal versus abnormal scans took into consideration a previous study of our group that assigned cutoff values in DAT imaging scans of Brazilian PD patients versus healthy control individuals [14]. Figure 2 shows illustrative normal and abnormal scans.
SUITABILITY OF REQUEST FOR SCINTIGRAPHY WITH 99MTC-TRODAT-1:
In Table 1, we present settings in which brain scintigraphy with 99mTc-TRODAT-1 was considered technically appropriate in this study (i.e., when the scan was requested to differentiate intergroup pathologies, such as degenerative parkinsonism versus non-degenerative conditions). In this study, intragroup differentiation was not considered appropriate because DAT imaging studies do not allow atypical forms of parkinsonism such as multiple system atrophy, progressive supranuclear palsy, corticobasal degeneration, and Lewy body dementia to be distinguished from idiopathic PD at the individual level [16]. However, we are aware, for example, that when an observer-independent software was used for statistical parametric mapping, reduced midbrain [123I]β-CIT uptake was found in patients with Parkinson variant of multiple system atrophy, which allowed a correct classification of 95% patients with the variant or PD, respectively [23]. We are also aware of normal DAT results in corticobasal degeneration and abnormal DAT results in some vascular parkinsonism cases [24,25].
We also considered studies requesting evaluation or confirmation of dopaminergic cell loss (dopaminergic denervation) in cases of suspected PD as appropriate indications.
STATISTICAL ANALYSIS:
For categorical variable descriptive analysis, absolute and relative frequency values were used. Comparisons between 2 measures were done using chi-square, Fisher exact, or Mann-Whitney tests. When necessary, posttests with Holm correction were used. A significance level of 5% was adopted. The analysis was conducted with the R software (version 3.4.1) and Microsoft Office Excel 2010.
Results
DEMOGRAPHIC AND CLINICAL DATA:
Scintigraphy with the DAT ligand 99mTc-TRODAT-1 was abnormal in 76 of 119 patients (~64%) and all but one of the physicians who requested the examination were neurologists (Supplementary Table 1). Additionally, 54% of DAT imaging scans were requested by neurologists who were self-reported movement disorder specialists.
Demographic and clinical data of patients and the most prevalent symptoms at the time of the scan request are summarized in Table 2. Overall, the majority of patients were men, with an average age of 67 years and a short disease duration (<5 years). Bradykinesia, tremor, and rigidity accounted for the main clinical findings at the time of the scintigraphy request. A comparison of patients’ profiles based on the request for scintigraphy with 99mTc-TRODAT-1 being made by a movement disorder specialist versus other physicians did not reveal statistically significant differences, with the exception of a longer disease duration (4.8 vs. 3.0 years; P<0.031) being associated with requests from movement disorder specialists.
USEFULNESS OF SCINTIGRAPHY WITH 99MTC-TRODAT-1 IN REAL-WORLD CLINICAL PRACTICE:
The majority of scans were requested to evaluate or confirm dopaminergic cell loss (69%), with 10% being requested to differentiate degenerative parkinsonism from essential tremor and 6% to distinguish degenerative parkinsonism from drug-induced parkinsonism. The only request considered to be inappropriate was for differentiating PD from atypical parkinsonism, which occurred in 5% of cases.
Table 3 presents the major groups of scintigraphy requests as well as their clinical usefulness. Overall, in 85% of requests, the scans were useful in assisting physicians in the management of their patients. DAT imaging was not clinically useful in 18.3% of requests to evaluate or confirm dopaminergic cell loss, 2 scans (33%) to differentiate PD versus atypical parkinsonism, and 1 scan (33%) to discriminate between degenerative parkinsonism versus other mixed causes. The scintigraphy usefulness did not have statistically significant differences between movement disorder specialists and other physicians. DAT imaging assisted in the management of 75% patients.
CHANGE IN MANAGEMENT:
Change in management occurred in 75% of cases with brain scintigraphy with 99mTc-TRODAT-1 results. In 48% of cases, the treating physician started a new drug; in 12% of cases, medications were increased; and in 11% of cases, there was a switch or a medication withdrawal. Referral of the patient to another specialist occurred in 4% of cases, and in 19% of cases, another neuroimaging test was requested. Approximately 12% of diagnoses were reviewed in this retrospective analysis after DAT imaging results. The likelihood of starting a new drug (50%) or increasing current medication dosage (13%) was higher when scintigraphy with 99mTc-TRODAT-1 had abnormal results. In addition, when DAT imaging results were considered normal, there was a higher likelihood for interrupting medication (33%), requesting another neuroimaging examination (40%), or changing the diagnosis (40%).
Discussion
In this study, we demonstrated that brain scintigraphy with 99mTc-TRODAT-1, a DAT ligand, was considered useful in 85% of cases to assist decision-making in real-world clinical practice. Moreover, the majority of patients were >65 years of age, but with short disease duration (<5 years), and after DAT imaging, medical management (from diagnosis to medication withdrawal) was reported to have been changed in 75% of cases. Inappropriate use of the scan was seen in only 5% of cases. Notably, the largest number of physicians requesting this imaging technique were self-reported movement disorder specialists, which may suggest that even in large emerging countries such as Brazil, this technique has poor penetration among general neurologists, but mostly non-neurologists. In addition, the findings suggest that caution is needed if this method becomes more widely available without adequate educational information for physicians who are less familiar with the technique.
According to the initial tracer license in Brazil, 99mTc-TRODAT-1 may be used to assist in the diagnosis presynaptic dopaminergic transporters [26]; therefore, in our study, the vast majority of scans were performed in line with the license indication. In addition, even though we found that 5% of scan indications were inappropriate, all were in line with the tracer license. This finding differs from other series that indicated a larger number of inappropriate indications and those in disagreement with the tracer license. For example, Thiriez et al. [27] found that only 18% of the DAT scans performed in their series in France were in agreement with the initial tracer license. Additionally, they found a much higher number of inappropriate scans than in our study (40%
Cost effectiveness is a key factor when a biomarker is considered to play a role in the diagnosis of a certain disorder. DAT imaging is an expensive procedure, which may limit its use [28], particularly in low-income countries and where reimbursement is scarce. An alternative would be to add a very small amount of cost to health insurance with the aim to improve treatment [29]. In Brazil, the total average annual cost of PD patients attending 2 tertiary public health centers was estimated to be US$5,853.50 per person [30]. Therefore, a more widespread use of this technique in Brazil and other countries by general neurologists or family physicians would probably increase the number of inappropriate scans, thereby influencing the cost effectiveness of DAT imaging and increasing the burden of disease. However, as noted earlier, PD misdiagnosis may interfere with appropriate treatment choices, and the most frequent errors involve non-parkinsonian tremors, such as essential tremor [7]. Hence, caution as well as more knowledge about DAT imaging in aiding PD diagnosis is necessary in real-life clinical scenarios.
In our study, we found a high percentage (75%) of change in medical management after brain scintigraphy with 99mTc-TRODAT-1. This finding contrasts with the studies of Bhattacharjee et al. [31] in Ireland (n=48) and Løkkegaard et al. [32] in Denmark (n=90). These studies found that the diagnosis and management of patients changed in 23% and 28% of cases, respectively. In addition, Kupsch et al. [18] found that the clinical management plan and diagnosis changed in 49% of cases at 19 centers in Europe and the United States (n=113) [18]. However, our findings are in line with Catafau et al. [19], who found that imaging increased confidence in diagnosis, leading to changes in clinical management in 72% of patients in 15 European centers (n=118). Another study demonstrated that DAT may influence clinical management, including initiating new dopaminergic therapy, adjusting medication dose in patients with abnormal DAT, discontinuing dopaminergic medications, and discontinuing the initiation of new dopaminergic medications [7].
Interestingly, we found 2 patterns related to DAT imaging that influenced change in management. First, when brain scintigraphy with 99mTc-TRODAT-1 was abnormal, starting a new drug or increasing the current medication dosage was more likely to occur. Second, when brain scintigraphy with 99mTc-TRODAT-1 was normal, there was a trend for interrupting medication, requesting another neuroimaging examination, or changing the diagnosis. From this perspective, a previous study [33] followed 150 patients with normal DAT imaging for 2 years and showed that the vast majority of these patients had a benign movement disorder rather than degenerative parkinsonism, thus reinforcing the importance of functional imaging in making a diagnosis and understanding the prognosis.
A physician’s clinical familiarity with a particular imaging technique may influence the appropriateness of request. In our series, the main reason to order DAT imaging was to evaluate or confirm dopaminergic cell loss. During the period of this study (2013 to 2016), we did not find relevant differences in the motivation to request DAT imaging (data not shown). Indeed, this result is similar to Thiriez et al. [27] who did not find differences in the appropriateness of requests in the first 100 scans versus the last 100 scans 2.5 years apart.
There are limitations in this study that should be taken into consideration. First, its retrospective nature did not allow accurate control of clinical and imaging data collection. Second, a large number of physicians who requested a scan were not interviewed. This could have masked the skillfulness of physicians interviewed, possibly selecting physicians who were more familiar with DAT imaging. Also, merely describing the chance to change clinical management is insufficient to establish a real “clinical utility,” as this study did not have a control group of patients that were not submitted to DAT imaging. A control group could have helped prove that the scan result was the critical factor influencing clinical decision-making. Third, our results do not necessarily support the routine use of DAT imaging at all Brazilian sites where this technique is available, and inappropriate use of the scan and other outcomes may differ between sites. Finally, the lack of studies in other large emerging countries such as Brazil prevented a fair comparison between centers.
Conclusions
In conclusion, this study demonstrated that brain scintigraphy with 99mTc-TRODAT-1, a DAT ligand, may indeed affect diagnostic or therapeutic interventions, meaning that Brazilian physicians who requested the examination for patients incorporated the results into clinical decision-making. In addition, this study provides insight into the use of DAT scintigraphy in countries other than the United States and European nations. Finally, even though the change in medical management was high in our study, it would probably be lower with more widespread access to DAT scintigraphy worldwide.
Figures
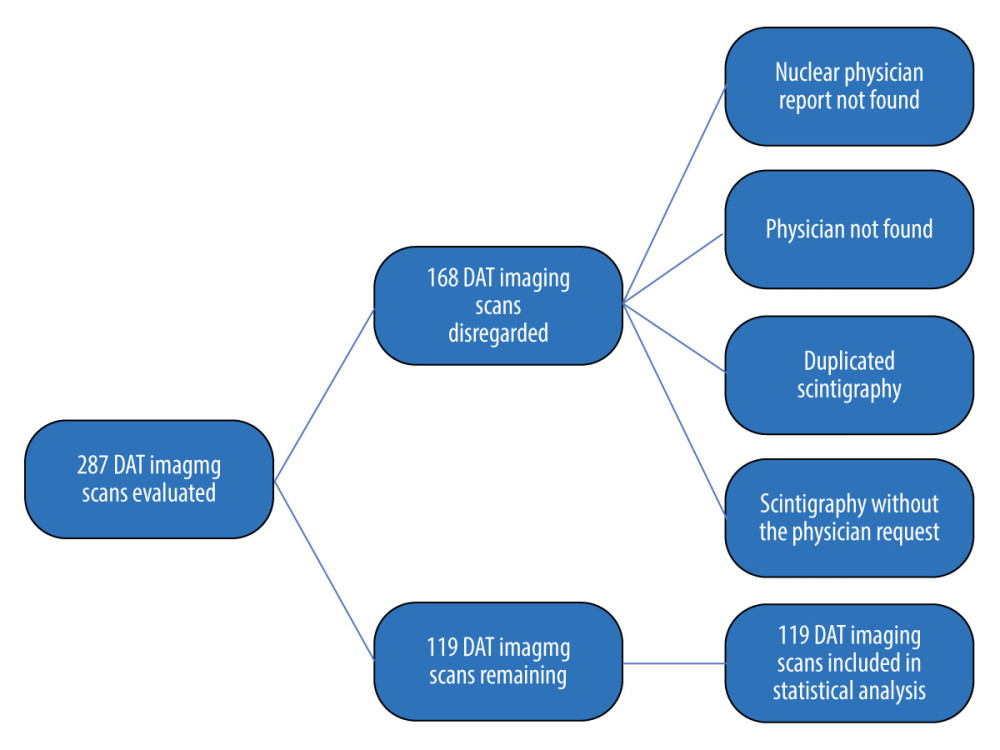 Figure 1. Flowchart of dopamine transporter (DAT) imaging scans evaluated.
Figure 1. Flowchart of dopamine transporter (DAT) imaging scans evaluated. 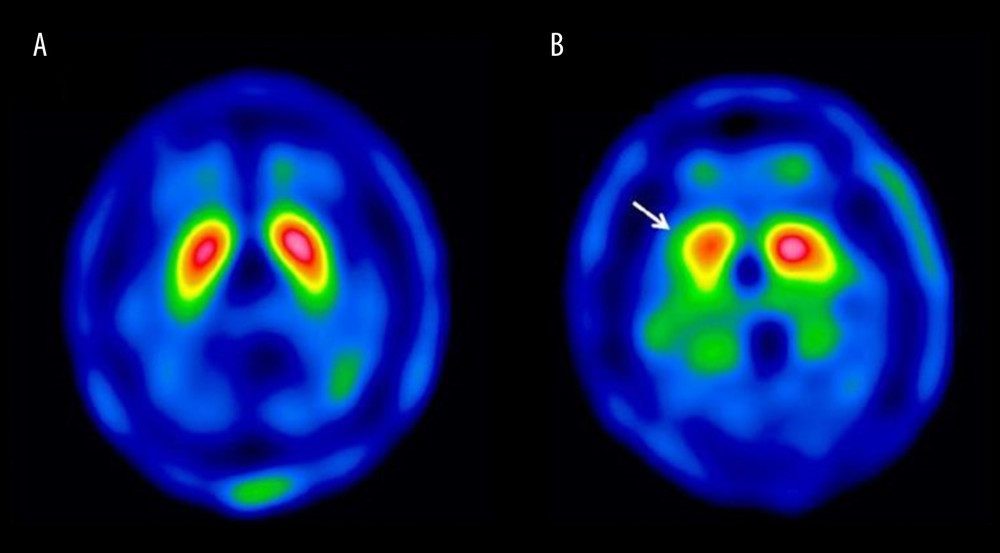 Figure 2. Illustrative dopamine transporter (DAT) imaging scans in 2 subjects from our sample who underwent a 99mTc-TRODAT-1 SPECT: (A) Normal and (B) abnormal scans are represented. Arrow (B) shows reduced DAT uptake bilaterally in the striatum, with more pronounced decrease on the right striatum.
Figure 2. Illustrative dopamine transporter (DAT) imaging scans in 2 subjects from our sample who underwent a 99mTc-TRODAT-1 SPECT: (A) Normal and (B) abnormal scans are represented. Arrow (B) shows reduced DAT uptake bilaterally in the striatum, with more pronounced decrease on the right striatum. Tables
Table 1. Suitability of brain scintigraphy with 99m Tc-TRODAT-1 requests. Indications that suggested intergroup disease (degenerative parkinsonism versus non-degenerative conditions) were considered appropriate.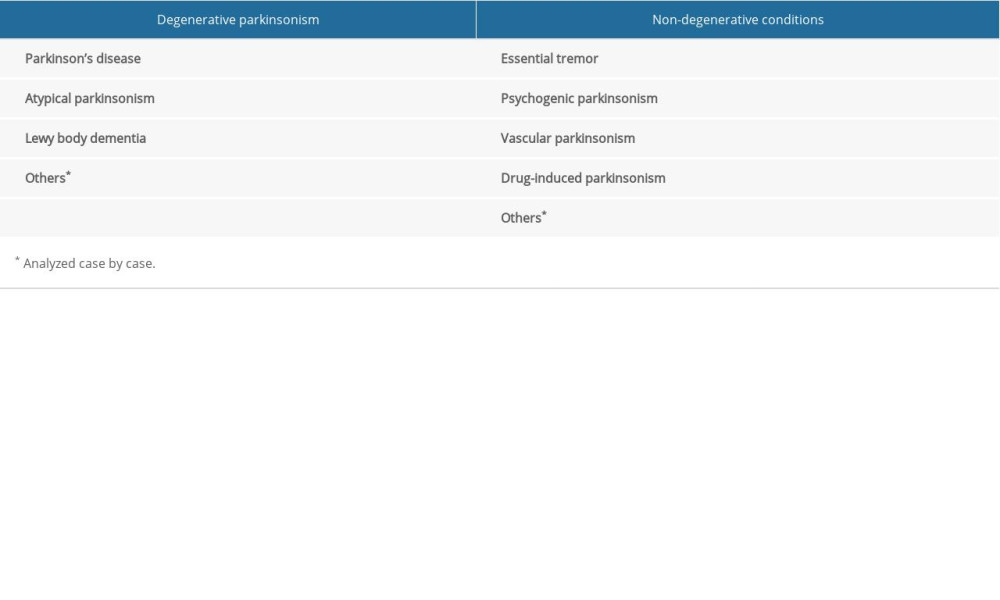 Table 2. Patient demographics and clinical data.
Table 2. Patient demographics and clinical data.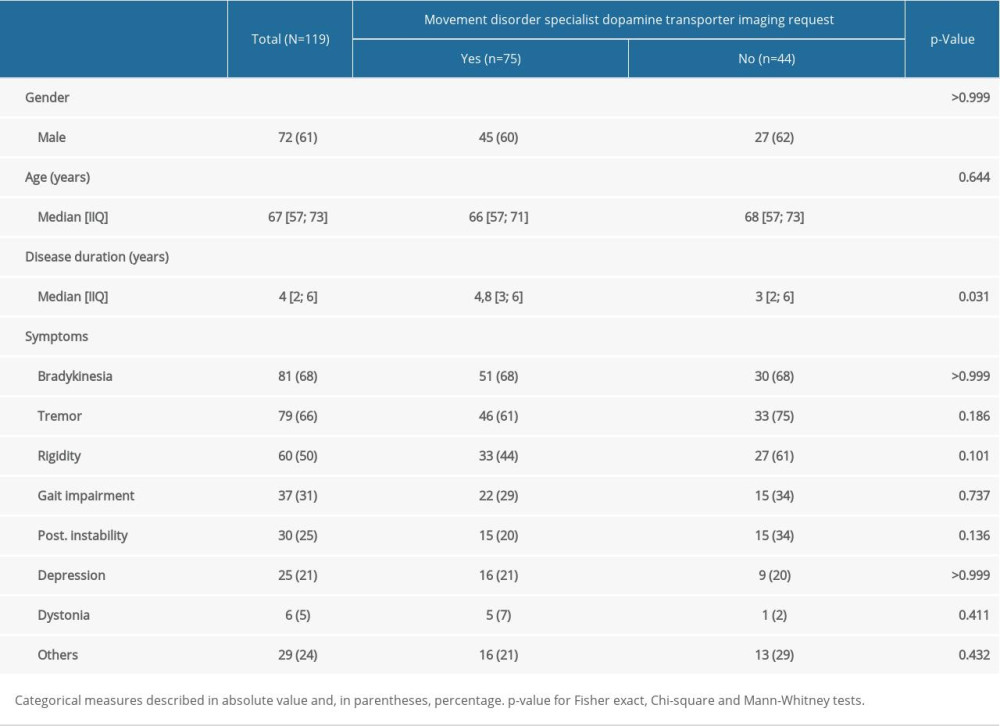 Table 3. Brain scintigraphy with 99m Tc-TRODAT-1 requests and its clinical usefulness.
Table 3. Brain scintigraphy with 99m Tc-TRODAT-1 requests and its clinical usefulness.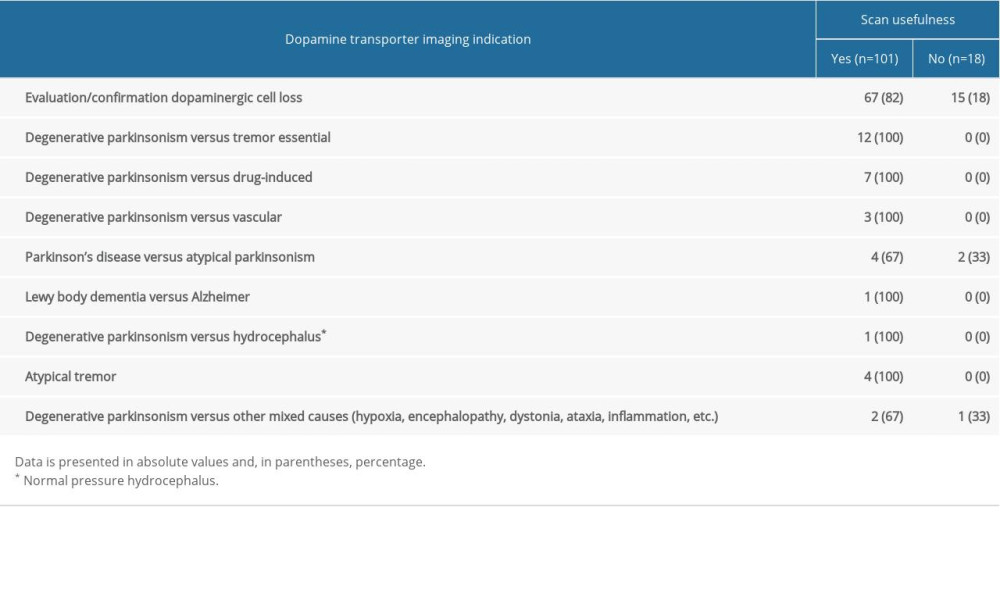 Supplementary Table 1. Scintigraphy with 99m Tc-TRODAT-1 results in patients undergoing investigation for parkinsonism.
Supplementary Table 1. Scintigraphy with 99m Tc-TRODAT-1 results in patients undergoing investigation for parkinsonism.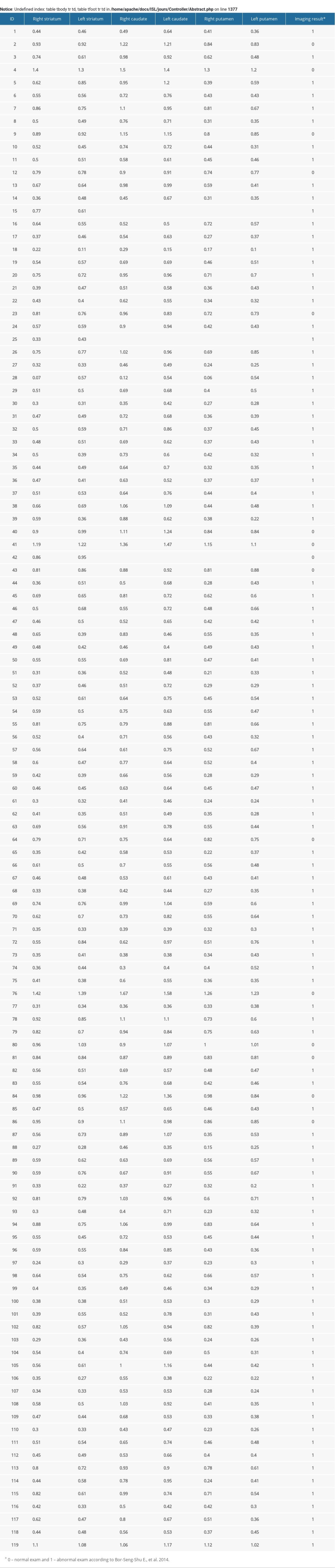
References
1. Gaig C, Tolosa E, When does Parkinson’s disease begin?: Mov Disord, 2009; 24(Suppl 2); S656-64
2. Tysnes OB, Storstein A, Epidemiology of Parkinson’s disease: J Neural Transm (Vienna), 2017; 124(8); 901-5
3. Barbosa MT, Caramelli P, Maia DP, Parkinsonism and Parkinson’s disease in the elderly: A community-based survey in Brazil (the Bambui study): Mov Disord, 2006; 21(6); 800-8
4. Reeve A, Simcox E, Turnbull D, Ageing and Parkinson’s disease: Why is advancing age the biggest risk factor?: Ageing Res Rev, 2014; 14; 19-30
5. Instituto Brasileiro de Geografia e Estatística [internet]: Mudança geográfica no Brasil no início do século XXI, 2015 [in Portuguese]https://biblioteca.ibge.gov.br/index.php/biblioteca-catalogo?view=detalhes&id=293322
6. Adler CH, Beach TG, Hentz JG: Neurology, 2014; 83(5); 406-12
7. Mirpour S, Turkbey EB, Marashdeh W, Impact of DAT-SPECT on management of patients suspected of parkinsonism: Clin Nucl Med, 2018; 43(10); 710-14
8. Berardelli A, Wenning GK, Antonini A, EFNS/MDS-ES/ENS [corrected] recommendations for the diagnosis of Parkinson’s disease: Eur J Neurol, 2013; 20(1); 16-34
9. Isaacson SH, Fisher S, Gupta F, Clinical utility of DaTscan imaging in the evaluation of patients with parkinsonism: A US perspective: Expert Rev Neurother, 2017; 17(3); 219-25
10. Suwijn SR, van Boheemen CJ, de Haan RJ, The diagnostic accuracy of dopamine transporter SPECT imaging to detect nigrostriatal cell loss in patients with Parkinson’s disease or clinically uncertain parkinsonism: A systematic review: EJNMMI Res, 2015; 5; 12
11. Kraemmer J, Kovacs GG, Perju-Dumbrava L, Correlation of striatal dopamine transporter imaging with post mortem substantia nigra cell counts: Mov Disord, 2014; 29(14); 1767-73
12. Løkkegaard A, Werdelin LM, Friberg L, Clinical impact of diagnostic SPET investigations with a dopamine re-uptake ligand: Eur J Nucl Med Mol Imaging, 2002; 29(12); 1623-29
13. Booth TC, Nathan M, Waldman AD, The role of functional dopamine-transporter SPECT imaging in parkinsonian syndromes, part 2: Am J Neuroradiol, 2015; 36(2); 236-44
14. Bor-Seng-Shu E, Felicio AC, Braga-Neto P, Dopamine transporter imaging using 99mTc-TRODAT-1 SPECT in Parkinson’s disease: Med Sci Monit, 2014; 20; 1413-18
15. Weng YH, Yen TC, Chen MC, Sensitivity and specificity of 99mTc-TRODAT-1 SPECT imaging in differentiating patients with idiopathic Parkinson’s disease from healthy subjects: J Nucl Med, 2004; 45(3); 393-401
16. Seibyl JP, Marek K, Sheff K, Iodine-123-beta-CIT and iodine-123-FPCIT SPECT measurement of dopamine transporters in healthy subjects and Parkinson’s patients: J Nucl Med, 1998; 39(9); 1500-8
17. Chen YK, Liu RS, Huang WS, The role of dopamine transporter imaging agent [99mTc]TRODAT-1 in hemi-parkinsonism rat brain: Nucl Med Biol, 2001; 28(8); 923-28
18. Van Laere K, De Ceuninck L, Dom R, Dopamine transporter SPECT using fast kinetic ligands: 123I-FP-beta-CIT versus 99mTc-TRODAT-1: Eur J Nucl Med Mol Imaging, 2004; 31(8); 1119-27
19. Shih MC, Amaro E, Ferraz HBNeuroimaging of the dopamine transporter in Parkinson’s disease: First study using [99mTc]-TRODAT-1 and SPECT in Brazil: Arq Neuropsiquiatr, 2006; 64(3A); 628-34 [in Portuguese]
20. Gelb DJ, Oliver E, Gilman S, Diagnostic criteria for Parkinson’s disease: Arch Neurol, 1999; 56(1); 33-39
21. Postuma RB, Berg D, Stern M, MDS clinical diagnostic criteria for Parkinson’s disease: Mov Disord, 2015; 30(12); 1591-601
22. GE HEALTHCARE [Internet]: NM Applications GE Healthcare DICOM conformance statement DIR 5503462-1EN (DOC1473333) Rev; 3 https://www.gehealthcare.com.au/-/jssmedia/45aae5aff8964414b592e384d4fc5c57.pdf
23. Cilia R, Rossi C, Frosini D, Dopamine transporter SPECT imaging in corticobasal syndrome: PLoS One, 2011; 6(5); e18301
24. Benítez-Rivero S, Marín-Oyaga VA, García-Solís D, Clinical features and123I-FP-CIT SPECT imaging in vascular parkinsonism and Parkinson’s disease: J Neurol Neurosurg Psychiatry, 2013; 84(2); 122-29
25. Gelb DJ, Oliver E, Gilman S, Diagnostic criteria for Parkinson disease: Arch Neurol, 1999; 56(1); 33-39
26. : TRODAT-1, 2018, Taiwan, Shinlin Sinseng Pharmaceutical http://gruporph.com.br/wp-content/uploads/2020/06/COD-002428-BULA-NAC-TRODAT-1.pdf
27. Thiriez C, Itti E, Fenelon G, Clinical routine use of dopamine transporter imaging in 516 consecutive patients: J Neurol, 2015; 262(4); 909-15
28. Miller DB, O’Callaghan JP, Biomarkers of Parkinson’s disease: Present and future: Metabolism, 2015; 64(3 Suppl 1); S40-46
29. Bhattacharjee S, Chalissery AJ, Barry T, Referral practice, reporting standards, and the impact of dopamine transporter scans done in a tertiary hospital: Neurol India, 2017; 65(6); 1264-70
30. Bovolenta TM, de Azevedo Silva SMC, Saba RA, Average annual cost of Parkinson’s disease in Sao Paulo, Brazil, with a focus on disease-related motor symptoms: Clin Interv Aging, 2017; 12; 2095-108
31. Plotkin M, Amthauer H, Klaffke S, Combined 123I-FP-CIT and 123I-IBZM SPECT for the diagnosis of parkinsonian syndromes: Study on 72 patients: J Neural Transm (Vienna), 2005; 112(5); 677-92
32. Catafau AM, Tolosa E, Impact of dopamine transporter SPECT using 123I-Ioflupane on diagnosis and management of patients with clinically uncertain parkinsonian syndromes: Mov Disord, 2004; 19(10); 1175-82
33. Marshall VL, Patterson J, Hadley DM, Two-year follow-up in 150 consecutive cases with normal dopamine transporter imaging: Nucl Med Commun, 2006; 27(12); 933-37
Figures
 Figure 1. Flowchart of dopamine transporter (DAT) imaging scans evaluated.
Figure 1. Flowchart of dopamine transporter (DAT) imaging scans evaluated. Figure 2. Illustrative dopamine transporter (DAT) imaging scans in 2 subjects from our sample who underwent a 99mTc-TRODAT-1 SPECT: (A) Normal and (B) abnormal scans are represented. Arrow (B) shows reduced DAT uptake bilaterally in the striatum, with more pronounced decrease on the right striatum.
Figure 2. Illustrative dopamine transporter (DAT) imaging scans in 2 subjects from our sample who underwent a 99mTc-TRODAT-1 SPECT: (A) Normal and (B) abnormal scans are represented. Arrow (B) shows reduced DAT uptake bilaterally in the striatum, with more pronounced decrease on the right striatum. Tables
 Table 1. Suitability of brain scintigraphy with 99m Tc-TRODAT-1 requests. Indications that suggested intergroup disease (degenerative parkinsonism versus non-degenerative conditions) were considered appropriate.
Table 1. Suitability of brain scintigraphy with 99m Tc-TRODAT-1 requests. Indications that suggested intergroup disease (degenerative parkinsonism versus non-degenerative conditions) were considered appropriate. Table 2. Patient demographics and clinical data.
Table 2. Patient demographics and clinical data. Table 3. Brain scintigraphy with 99m Tc-TRODAT-1 requests and its clinical usefulness.
Table 3. Brain scintigraphy with 99m Tc-TRODAT-1 requests and its clinical usefulness. Table 1. Suitability of brain scintigraphy with 99m Tc-TRODAT-1 requests. Indications that suggested intergroup disease (degenerative parkinsonism versus non-degenerative conditions) were considered appropriate.
Table 1. Suitability of brain scintigraphy with 99m Tc-TRODAT-1 requests. Indications that suggested intergroup disease (degenerative parkinsonism versus non-degenerative conditions) were considered appropriate. Table 2. Patient demographics and clinical data.
Table 2. Patient demographics and clinical data. Table 3. Brain scintigraphy with 99m Tc-TRODAT-1 requests and its clinical usefulness.
Table 3. Brain scintigraphy with 99m Tc-TRODAT-1 requests and its clinical usefulness. Supplementary Table 1. Scintigraphy with 99m Tc-TRODAT-1 results in patients undergoing investigation for parkinsonism.
Supplementary Table 1. Scintigraphy with 99m Tc-TRODAT-1 results in patients undergoing investigation for parkinsonism. In Press
05 Apr 2024 : Clinical Research
Comparative Analysis of Transoral Endoscopic Parathyroidectomy Vestibular Approach and Focused Open Surgery...Med Sci Monit In Press; DOI: 10.12659/MSM.944128
05 Mar 2024 : Clinical Research
Muscular Function Recovery from General Anesthesia in 132 Patients Undergoing Surgery with Acceleromyograph...Med Sci Monit In Press; DOI: 10.12659/MSM.942780
05 Mar 2024 : Clinical Research
Effects of Thermal Insulation on Recovery and Comfort of Patients Undergoing Holmium Laser LithotripsyMed Sci Monit In Press; DOI: 10.12659/MSM.942836
05 Mar 2024 : Clinical Research
Role of Critical Shoulder Angle in Degenerative Type Rotator Cuff Tears: A Turkish Cohort StudyMed Sci Monit In Press; DOI: 10.12659/MSM.943703
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








