18 April 2021: Animal Study
Left Ventricular Impaired Relaxation and Interstitial Myocarditis Identified in Sepsis-Associated Cardiac Dysfunction: Use of a Rodent Model
David J. Sturgess1ABCDEFG*, Shannon Morrison1BCDE, Brian Haluska2BCD, Glenda C. Gobe23BCDEG, Mark A. Jones4ACDE, Sonia Volante1BE, Bala Venkatesh2ACDEGDOI: 10.12659/MSM.929512
Med Sci Monit 2021; 27:e929512
Abstract
BACKGROUND: Sepsis is a serious clinical problem that results from the systemic response of the body to infection. Left ventricular (LV) diastolic dysfunction is increasingly appreciated as a contributor to morbidity and mortality in sepsis. Animal models may offer a method of studying diastolic dysfunction while controlling for many potential clinical confounders, such as sepsis duration, premorbid condition, and therapeutic interventions. This study sought to evaluate an endotoxemia (LPS) rodent model of sepsis, with regard to echocardiographic evidence, including tissue Doppler, of LV diastolic dysfunction and histopathology findings.
MATERIAL AND METHODS: Fourteen male Sprague-Dawley rats were randomly allocated (1: 1) to LPS or saline (control). Mean arterial blood pressure (MAP) was measured through cannulation of the carotid artery. After a 30-min stabilization, baseline assessment with echocardiography and blood collection was performed. Rats were administered 0.9% saline or LPS (10 mg/mL). Follow-up echocardiography and blood collection were performed after 2 h. Hearts were removed post-mortem and pathology studied using histology and immunohistochemistry.
RESULTS: LPS was associated with hypotension (MAP 81.86±31.67 mmHg; 124.29±20.16; p=0.02) and LV impaired relaxation (myocardial early diastolic velocity [e’] 0.06±0.02 m/s; 0.09±0.02; P=0.008). Histopathology and immunohistochemistry demonstrated evidence of interstitial myocarditis (hydropic changes and inflammation).
CONCLUSIONS: LPS was associated with both diastolic dysfunction (impaired relaxation) and interstitial myocarditis. These features may offer a link between the structural and functional changes that have previously been described separately in clinical sepsis. This may facilitate further studies focused upon the mechanism and potential benefit treatment of sepsis-associated cardiac dysfunction.
Keywords: Echocardiography, Echocardiography, Doppler, Immunohistochemistry, Sepsis, Ventricular Function, Left, Diastole, Heart Ventricles, myocarditis, Ventricular Dysfunction, Left
Background
Sepsis, defined as life-threatening organ dysfunction caused by a dysregulated host response to infection [1], is a serious clinical problem, accounting for substantial morbidity and mortality and imposing a significant burden on healthcare systems. The majority of sepsis patients die of refractory hypotension and cardiovascular collapse [2]. Left ventricular diastolic dysfunction (LVDD) is increasingly appreciated as a contributor to morbidity and mortality in severe sepsis and septic shock [3], and may represent a valid therapeutic target for interventions to improve clinical outcomes.
The characterization of diastolic function is complex, with non-invasive bedside investigation needed in the critically-ill. Tissue Doppler imaging (TDI) offers promise in this regard. It measures myocardial velocities, which are low-frequency, high-amplitude signals filtered from conventional Doppler imaging [4]. Transthoracic TDI was previously applied to a critically-ill patient cohort and demonstrated a high incidence of abnormal values, suggestive of diastolic dysfunction: 32% of this cohort were diagnosed with sepsis [4]. This relatively simple echocardiographic technique should be feasible in critically-ill patients, even in the presence of mechanical ventilation.
TDI appears to be a central component in characterizing LVDD in severe sepsis and septic shock, with abnormal LV diastolic TDI values associated with poor outcomes in critical illness, including sepsis [5]. Although TDI evidence for isolated and reversible impairment of ventricular relaxation in patients with septic shock has been confirmed, the precise mechanism/s of sepsis-associated LV diastolic dysfunction remain unclear. Most investigations represent effects of functional change, with little information available on concurrent structural changes in patients [6]. Histological studies in sepsis have revealed a range of abnormalities, such as inflammatory infiltrate, interstitial edema, apoptosis, and necrosis [2]. It is possible that reversible diastolic dysfunction results from myocardial inflammatory infiltration; however, the associations between structural and functional myocardial changes have not previously been investigated, particularly using clinically-applicable techniques such as TDI.
Clinical studies of cardiac function in sepsis are potentially confounded by factors such as variable timing from the onset of disease, illness severity, pre-existing cardiac disease, and the impact of therapy such as fluid resuscitation, mechanical ventilation, and vasoactive and inotropic drugs. Animal models offer a method of studying aspects of sepsis while controlling for many potential clinical confounders. Lipopolysaccharide (LPS)-induced endotoxemia is an accepted, clinically-relevant, experimental model of sepsis [7]. The aim of the current investigation was to evaluate this model for links between echocardiographic evidence of LVDD, including TDI, and structural change using histopathology.
Material and Methods
ENDOTOXEMIA (LPS) MODEL:
Male Sprague-Dawley rats were studied. They were fed autoclaved rat chow, allowed water
Rats (N=14) were randomly allocated (1: 1) to either endotoxemia (LPS) or control saline groups. A previously described model of hyperdynamic endotoxic shock was adapted for this experiment [8]. Anesthesia was induced and maintained via tracheostomy (14G Insyte cannula, Becton Dickinson Infusion Therapy Systems Inc., UT, USA) with isoflurane (up to 2%). Mechanical ventilation (Harvard Small Animal Ventilator Model 683, Harvard Apparatus, MA, USA) was titrated to normocarbia (arterial pCO2 4.8–5.8 kPa).
Body temperature was maintained throughout the experiment by use of reflective foil insulation and an underbody electric heating pad. Cannulae (24G Insyte cannula) were placed in the carotid artery for monitoring of mean arterial blood pressure (MAP) and femoral vein for intravenous access. Pressures were transduced using a fluid-filled system (BD DTXPlus™ DT-4812 disposable transducer kit, Franklin Lakes, NJ, USA 07417) combined with a Hewlett Packard M1277A transport monitor (Hewlett Packard, USA). All rats received 0.9% saline at 3 mL/h via the carotid line.
Following a 30-min period of stabilization, baseline assessment (echocardiography and blood collection) was performed. Rats then received a 30-min intravenous infusion (1 mL/kg) of either 0.9% saline or 10 mg/mL endotoxin (
ETHICS STATEMENT:
All care and research with the use of laboratory animals was performed with approval by The University of Queensland Animal Ethics Committee (UQ AEC protocol 675/05).
ECHOCARDIOGRAPHY:
Rats were studied in the left lateral position. Echocardiographic examinations were performed by a single, experienced echocardiographer (B.H.) using a commercially available echocardiographic system (Vivid 5 with 15MHz rodent probe, GE Healthcare). Unless stated otherwise, measured variables resulted from the mean of 5 consecutive cardiac cycles.
LV end-diastolic internal dimensions (LVIDd) and end-systolic internal dimensions (LVIDs) were measured by two-dimensional targeted M-mode tracings. Percent LV fractional shortening (FS) was calculated as FS=(LVIDd−LVIDs)/LVIDd×100.
Doppler interrogation of LV outflow tract velocity was guided by apical 5-chamber view [9]. Heart rate (HR), velocity time integral (VTI), mean velocity (Vmean), and peak velocity (Vpeak) were measured. Transmitral flow velocities were recorded by pulsed-wave Doppler with the sample volume placed at the mitral valve tips from the apical 4-chamber view. Peak early diastolic transmitral flow (E) velocity was measured.
Myocardial velocities were obtained using tissue Doppler settings, with the pulsed-wave Doppler sample volume at the septal mitral annulus in the apical 4-chamber view. Myocardial peak early (e′) diastolic velocity was measured. Due to anticipated rapid heart rates, A (and a′) wave fusion were expected, in which case E (and e′) were measured as described by Nagueh and colleagues in the presence of sinus tachycardia [10].
HISTOPATHOLOGY AND IMMUNOHISTOCHEMISTRY:
At the completion of each experiment, hearts were removed, bisected in an equatorial plane, fixed in 4% buffered formalin at 4°C, and prepared routinely for histopathology and IHC. Formalin-fixed tissue was embedded in paraffin using routine methods, and 4-μm sections were cut onto Superfrost Plus histology slides. For routine histology, numbered paraffin sections were stained with hematoxylin and eosin (HE; histopathology) and periodic acid-Schiff reagent (PAS; identification of glycogen). All HE and PAS-stained tissue sections were viewed using light microscopy and assessed for morphological characteristics, including hydropic change, apoptosis, and necrosis, in 10 microscope fields per section at ×200 magnification. For IHC, sections were dewaxed, rehydrated to buffer, and batch-stained routinely with primary antibodies ED-1 (monoclonal anti-rat antibody that labels tissue macrophages and monocytes; Serotec, Australian Laboratory Services Pty Ltd., Sydney Markets, Australia; 1: 50 dilution) and activated/cleaved caspase-3 (anti-rat polyclonal antibody used to indicate apoptosis; Cell Signaling, Genesearch Pty Ltd., Arundel, Australia; 1: 200 dilution). Stained slides were scanned with an Aperio ScanScope XT slide scanning system (Aperio Technologies, USA) at ×20 or ×40 magnification. Digital images of the sections were captured using Aperio ImageScope software (Leica Biosystems, Germany) to record the localization and extent of staining.
BLINDING:
The echocardiographer was blinded to the experimental group at the time of assessment. Coded echocardiographic and Doppler recordings were analyzed at least 1 month after the experiments by a single observer (D.S.) blinded to experimental group. Histopathology and immunohistochemistry were performed on coded samples by a pathologist (G.G.) blinded to the experimental group.
STATISTICAL ANALYSIS:
Differences between groups at the final assessment were characterized by analysis of covariance (ANCOVA) to adjust for baseline values. Mortality prior to completion of data collection resulted in exclusion from analysis due to the inability to perform echocardiography. Differences in categorical data (histopathology/immunohistochemistry) were assessed with Fisher’s exact test or Cochrane-Armitage test for trend. Analysis was performed using SAS for Windows, version 9.4. A P value ≤0.05 was regarded as significant. Unless otherwise stated, results are presented as mean ± SD. Sample size calculations were based upon the resource equation [11]. In consultation with an experienced biostatistician (MJ), randomization tables were created for the project. The study was regarded as complete when datasets were complete for all 7 animals.
Results
LPS ENDOTOXEMIA INDUCED DIASTOLIC DYSFUNCTION (IMPAIRED RELAXATION):
A major component of the pathophysiology of sepsis disease involves reduced blood pressure and progression to septic shock and tissue hypoperfusion. Reduction in blood pressure reduces the load the heart must pump against and has been proposed as a mechanism underlying normalization in ejection fraction in sepsis [12]. In the current study, at baseline, there were no significant differences in hemodynamic or echocardiographic variables between the sepsis and control groups. In contrast, with LPS endotoxemia compared with controls, the sepsis group demonstrated lower MAP (endotoxin 81.86±31.6 mmHg, control 124.2±20.1 mmHg, P=0.02), and myocardial early diastolic velocity (e′) was significantly lower in the sepsis group (0.06±0.02 m/s) compared with the control group (0.09±0.02; P = 0.008). At the final timepoint, however, no significant difference in E/e′ was observed between the 2 groups. These results are summarized in Table 1.
LPS ENDOTOXEMIA INDUCED MYOCARDITIS:
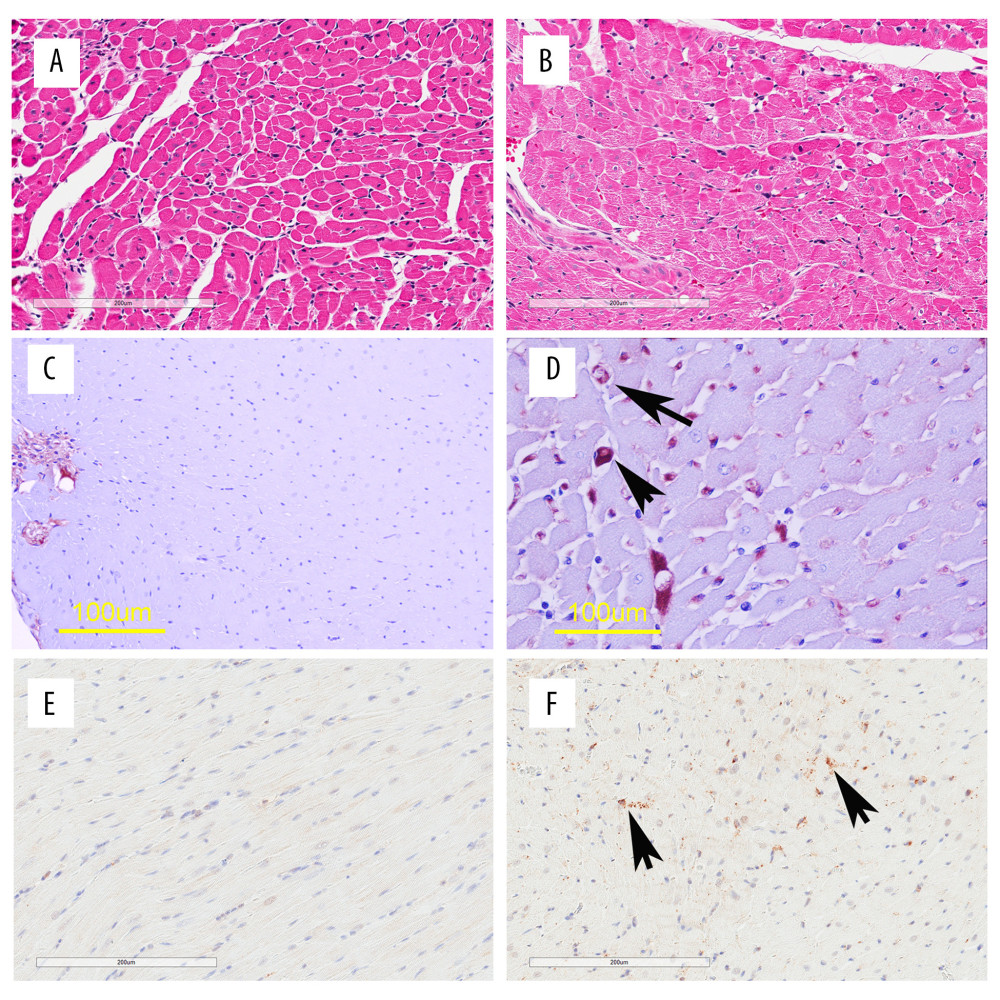
The novel component of the current study is the direct comparison of diastolic dysfunction in sepsis, and histopathology and protein biomarkers of injury in tissue sections by paired measurements from the same individuals. Results of HE and IHC staining of the ventricular myocardium are summarized in Table 2 and Figure 1. The endotoxin group demonstrated marked cardiomyocyte hydropic change/hydropic degeneration in 5 out of 7 endotoxic animals, but not present in any control animals (Fisher exact; P=0.015). Hydropic change could eventually be resolved but could also play a role in maintenance or progression of the disease. PAS staining did not indicate any differences for glycogen and mucosubstances, each sometimes indicative of interstitial myocarditis. While neutrophil counts were normal in all cases, staining for monocytes/macrophages (ED1 rat antibody) showed increased numbers in the interstitial spaces of the ventricular myocardium of the study group. Five out of 7 endotoxic animals, and no control animals, demonstrated >25 macrophages in ≥2 out of 10 high-power fields (P=0.0095; Cochrane-Armitage test for HE staining of the ventricular myocardium (Table 2) demonstrated marked hydropic changes in 5 out of 7 endotoxic animals, but was not present in any control animals, (Fisher exact; P=0.015). Neutrophil counts were normal in all cases (0–2 neutrophils per high-power field). IHC staining for monocytes/macrophages (ED1) demonstrated increased numbers in the interstitial spaces of the ventricular myocardium of the endotoxin group compared with the control group, indicative of interstitial myocarditis (Table 3). The Cochrane-Armitage test for trend was used to compare ordinal categorical data. Five out of 7 endotoxic animals, and no control animals, demonstrated >25 macrophages in at least 2 out of 10 high-power fields (P=0.0095). Activated caspase-3 staining did not demonstrate any evidence of cardiac myocyte apoptosis in any slides.
Discussion
LPS ENDOTOXEMIA INDUCED DIASTOLIC DYSFUNCTION (IMPAIRED RELAXATION):
We recently demonstrated that abnormal diastolic TDI values are common in critically-ill patients [4]. This study used previously validated echocardiographic techniques [5] and our TDI measurements are consistent with impaired LV relaxation. These results support findings in other pre-clinical in vivo experimental data, as well as echocardiographic findings in clinical sepsis [13].
LPS ENDOTOXEMIA INDUCED MYOCARDITIS:
There was a marked cardiomyocyte hydropic change seen in endotoxic animals compared to controls. These changes are typically associated with non-lethal or reversible injury of cardiomyocytes and are characterized by cell swelling, which reflects disturbance of membrane function [14]. While hydropic change can eventually be resolved, it can also play a role in maintenance or progression of the disease. In addition to these changes, there were significantly increased numbers of monocytes/macrophages in the ventricular myocardium, particularly the interstitial spaces, in the endotoxic group. This is consistent with human data that show inflammatory cell infiltrate in septic myocardium at autopsy [14]. It is also in line with previous reports of LV dysfunction associated with inflammatory processes [15], but these associations were not matched temporally in the same individual, as in the current study.
Activated caspase-3 staining did not demonstrate any evidence of cardiac myocyte apoptosis in any slides. Despite other endotoxic rat studies reporting caspase-3 activation 2 to 4 h after endotoxin administration, our findings are consistent with observations that myocardial cell death should be a rare event during sepsis [2]. Labeled cells were occasionally found in the interstitial space (Figure 1), perhaps indicative of death of immune cells with LPS treatment, as has been reported previously by Ward [16].
VALIDITY AND LIMITATIONS OF THE MODEL:
The LPS model is based upon a previously described model of hyperdynamic endotoxic shock [7]. Its adaptation has allowed evaluation of relationships between in vivo cardiac function (echocardiography incorporating diastolic tissue Doppler) and ventricular myocarditis (histopathology and immunohistochemistry). To account for many potential confounders encountered in clinical settings, our model differed significantly from clinical experience. In particular, the study period was selected to include the period of greatest hemodynamic perturbation acutely following endotoxin infusion. This contrasts with clinical sepsis, which often follows a protracted course. However, based upon observations that abnormal diastolic TDI values are relatively common in the critically-ill [4] and that these are associated with poor outcome [3], our study was able to demonstrate an association between abnormal diastolic TDI measurements and myocyte structural changes.
Conclusions
This study found an association between myocyte structural changes (inflammatory cell infiltrate and hydropic change) and abnormal diastolic TDI measurements (suggestive of in vivo ventricular diastolic dysfunction) using a direct comparison of function, histopathology, and protein biomarkers of injury by paired measurements from the same individuals. The results indicate a link between previously separate descriptions of interstitial myocarditis and diastolic dysfunction (impaired relaxation) in sepsis. The LPS model of sepsis may offer an opportunity to further elucidate the pathogenesis of and potential treatment of sepsis-associated LV diastolic dysfunction.
References
1. Verdonk F, Blet A, Mebazaa A, The new sepsis definition: Limitations and contribution to research and diagnosis of sepsis: Curr Opin Anaesthesiol, 2017; 30(2); 200-4
2. Rudiger A, Singer M, Mechanisms of sepsis-induced cardiac dysfunction: Crit Care Med, 2007; 35(6); 1599-608
3. Sturgess DJ, Marwick TH, Joyce C, A prospective comparison of tissue Doppler and cardiac biomarkers: Crit Care, 2010; 14(2); R44
4. Sturgess DJ, Marwick TH, Joyce CJJ, Tissue Doppler in critical illness: A retrospective cohort study: Crit Care, 2007; 11(5); R97
5. Lanspa MJ, Gutsche AR, Wilson EL, Application of a simplified definition of diastolic function in severe sepsis and septic shock: Crit Care, 2016; 20(1); 243
6. Rudiger A, Singer M, The heart in sepsis: From basic mechanisms to clinical management: Curr Vasc Pharmacol, 2013; 11(2); 187-95
7. Murando F, Peloso A, Cobianchi L, Experimental abdominal sepsis: Sticking to an awkward but still useful translational model: Mediators Inflamm, 2019; 2019 8971036
8. Rosser DM, Stidwill RP, Oxygen tension in the bladder epithelium rises in both high and low cardiac output endotoxemic sepsis: J Appl Physiol, 1995; 79(6); 1878-82
9. Slama M, Susic D, Varagic J, Echocardiographic measurement of cardiac output in rats: Am J Physiol Heart Circ Physiol, 2003; 284(2); H691-97
10. Nagueh SF, Appleton CP, Gillebert TC, Recommendations for the evaluation of left ventricular diastolic function by echocardiography: J Am Soc Echocardiog, 2009; 22(2); 107-33
11. Charan J, Kantharia ND, How to calculate sample size in animal studies?: J Pharmacol Pharmacother, 2013; 4(4); 303-6
12. Hoffman M, Kyriazis ID, Lucchese AM, Myocardial strain and cardiac output are preferable measurements for cardiac dysfunction and can predict mortality in septic mice: J Am Heart Assoc, 2019; 8(10); e012260
13. Bouhemad B, Nicolas-Robin A, Arbelot C, Isolated and reversible impairment of ventricular relaxation in patients with septic shock: Crit Care med, 2008; 36(3); 766-74
14. Celes MR, Torres-Duenas D, Alves-Filho JC, Reduction of gap and adherens junction proteins and intercalated disc structural remodeling in the hearts of mice submitted to severe cecal ligation and puncture sepsis: Crit Care Med, 2007; 35(9); 2176-85
15. Ikonomidis I, Tzortzis S, Lekakis J, Lowering interleukin-1 activity with anakinra improves myocardial deformation in rheumatoid arthritis: Heart, 2009; 95(18); 1502-7
16. Ward PA, Sepsis, apoptosis and complement: Biochem Pharmacol, 2008; 76(11); 1383-88
Tables
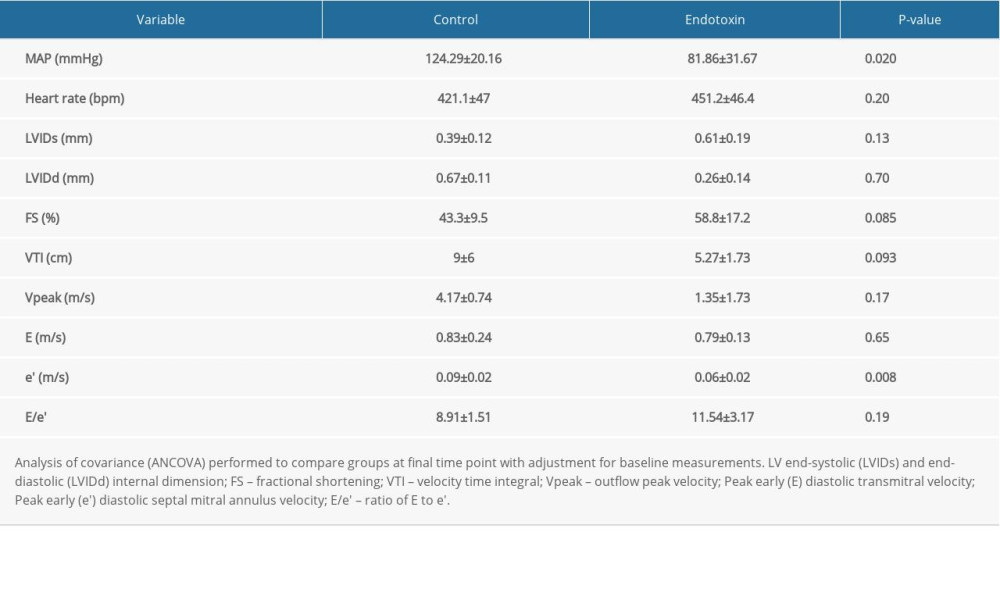 Table 1. Final assessment of function: LPS model.
Table 1. Final assessment of function: LPS model.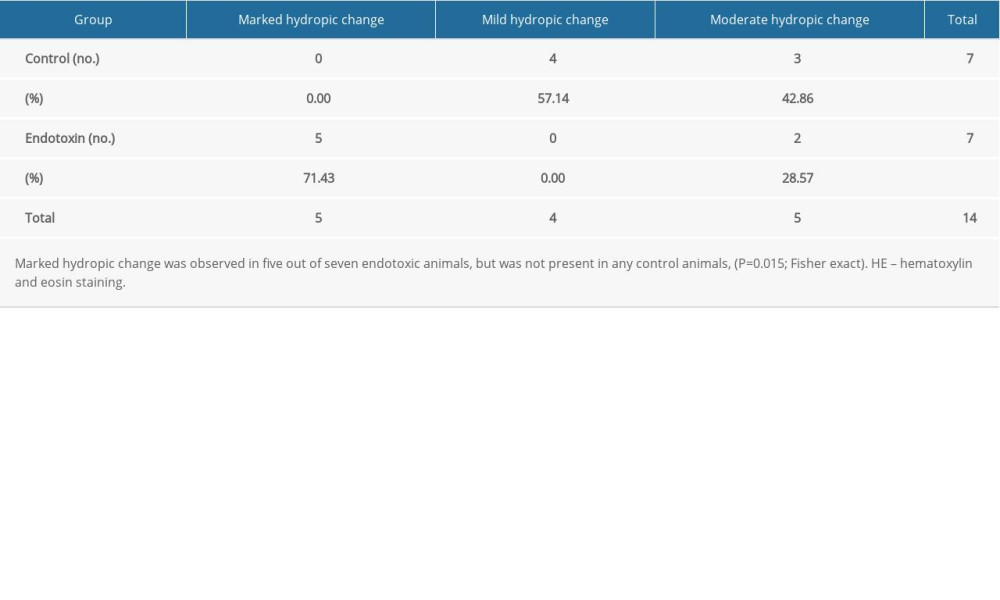 Table 2. Histopathology results for LPS model LV myocardium with HE staining.
Table 2. Histopathology results for LPS model LV myocardium with HE staining.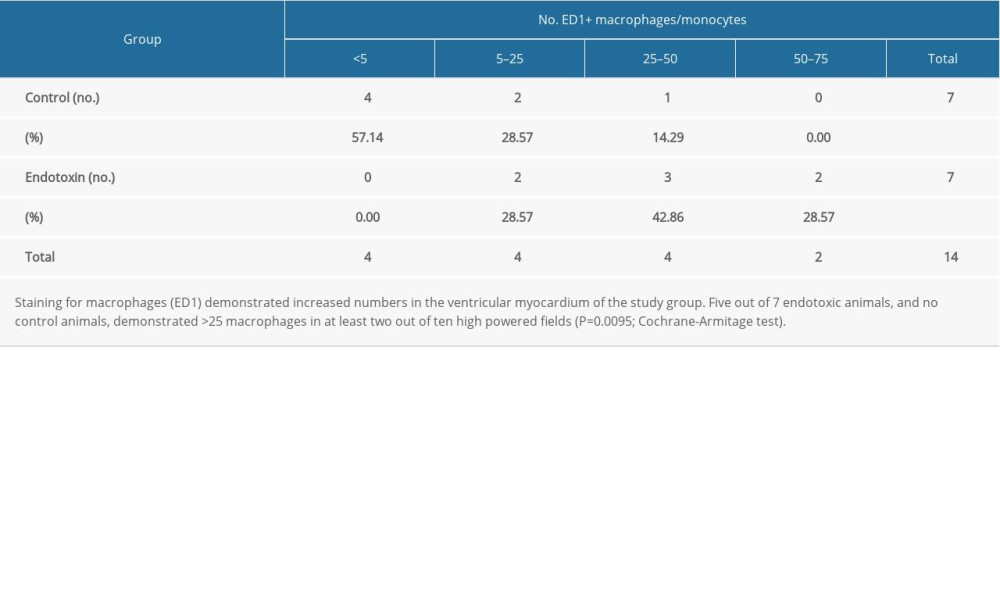 Table 3. Immunohistochemistry for monocyte/macrophage ED1 antibody in LPS model.
Table 3. Immunohistochemistry for monocyte/macrophage ED1 antibody in LPS model. Table 1. Final assessment of function: LPS model.
Table 1. Final assessment of function: LPS model. Table 2. Histopathology results for LPS model LV myocardium with HE staining.
Table 2. Histopathology results for LPS model LV myocardium with HE staining. Table 3. Immunohistochemistry for monocyte/macrophage ED1 antibody in LPS model.
Table 3. Immunohistochemistry for monocyte/macrophage ED1 antibody in LPS model. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387