06 December 2021: Animal Study
Diazepam Monotherapy or Diazepam-Ketamine Dual Therapy at Different Time Points Terminates Seizures and Reduces Mortality in a Status Epilepticus Animal Model
Ruijiao Zhou1ABCDEFG, Yanlin Wang2BCD, Xing Cao1CDEF, Zhimin Li1ABCF, Juming Yu1ACDEF*DOI: 10.12659/MSM.934043
Med Sci Monit 2021; 27:e934043
Abstract
BACKGROUND: Being refractory to drugs remains an urgent treatment problem in status epilepticus (SE). The fact that γ-aminobutyric acid A receptors (GABAARs) become internalized and inactive, N-methyl-D-aspartate receptors (NMDARs) become externalized and active during SE may explain the refractoriness to benzodiazepine. However, the real-time dynamic efficacy of antiepileptic drugs remains unclear. Therefore, we propose a hypothesis that diazepam monotherapy or diazepam-ketamine dual therapy could terminate seizures and reduce mortality in the SE model at different time points during ongoing SE.
MATERIAL AND METHODS: An SE model was established in adult Sprague-Dawley rats with lithium and pilocarpine. The GABAAR agonist diazepam was injected at 5, 10, 20, or 30 min when SE continued. In addition, diazepam and the NMDAR antagonist ketamine were injected at 10 to 60 min at 6 different time points. We measured seizure-free rates, seizure duration, degree of behavioral seizure, and mortality.
RESULTS: Diazepam monotherapy at 5 min and 10 min from the beginning of SE was able to terminate seizures and improved survival rates. Diazepam-ketamine dual therapy at 10 min, 20 min, and 30 min from the beginning of SE terminated seizures and achieved high survival rates.
CONCLUSIONS: In this parallel randomized controlled trial with a rat model, we found that diazepam monotherapy was an effective antiepileptic strategy at the early stage of SE less than 10 min after SE onset. If SE lasts more than 10 min but less than 30 min, the diazepam-ketamine dual therapy strategy may be an appropriate choice.
Keywords: Diazepam, Duration of Therapy, Ketamine, Status epilepticus, Analgesics, Animals, Anticonvulsants, Disease Models, Animal, Drug Administration Schedule, Drug Therapy, Combination, Female, Male, Rats, Rats, Sprague-Dawley, Seizures
Background
Status epilepticus (SE) is a frequently occurring neurological emergency. Its sudden, violent, self-sufficient, or repeated seizures can produce irreversible damage, including neuronal injury, respiratory and circulatory failure, and even death [1,2]. Pharmaceutical treatment as a preferred intervention can terminate SE and reduce its harm to the body as much as possible. Previous randomized controlled trials proposed a consensus about benzodiazepines as the standard first-choice drugs when treating patients with SE [3]. General anesthetics such as midazolam, propofol, and even dexmedetomidine (DMT) are commonly used to terminate SE, but these agents are associated with neuronal death, prolonged hospitalization, and poor prognosis [4]. Therefore, the simplest and most effective therapeutic strategy is generally considered the best. Benzodiazepines, as the standard first-choice drugs, are usually successful in terminating the self-sustaining or repeated seizures, but 11–57% of SE patients are resistant to benzodiazepines and develop refractory SE. Why? Studies suggested that treatment delay results in failed SE termination. For example, benzodiazepines given 1 h after the beginning of SE often fail to have an anti-seizure effect [5–7].
Different kinds of drugs have been studied in animal models for controlling SE. The NMDAR antagonist ketamine showed good anticonvulsant activity in several SE models. Even when SE lasts 1 h, ketamine could terminate SE in an electrical stimulation model [8–10]. The pilocarpine-induced SE rat model was the most used in seizure research, which mostly mimics the clinically evident SE, electrographic discharges, and short-term consequences in humans. Synergistic actions between benzodiazepine (GABAAR agonist) and ketamine (NMDAR antagonism) in this SE model were described [11,12]. One study found that ketamine effectively controlled pilocarpine-induced SE with other antiepileptic drugs when SE lasted for 30 min [13]. Niquet et al [12] found that midazolam and ketamine dual therapy instead of monotherapy could control SE successfully in animal models when the duration of SE before treatment was 40 min.
However, several issues regarding diazepam and NMDAR antagonists therapy strategy in SE treatment should be considered, and how to choose therapy strategies for controlling SE when treatment is delayed is still an unresolved issue. In the present study, we used the classical SE rat model, administered diazepam monotherapy or diazepam-ketamine dual therapy at shorter or longer time points from the beginning of SE to explore the efficacy of drugs in SE control and animal survival. This study may provide a theoretical basis for clinical applications to select the appropriate treatment strategy according to the time elapsed from the beginning of SE.
Material and Methods
ANIMALS:
We used adult female and male Sprague-Dawley rats (200–250 g) obtained from the Laboratory Animal Center of North Sichuan Medical College. Animals were provided with food and water ad libitum in a facility with constant temperature (22–24°C), humidity (40–50%), and a 12-h light/dark cycle, and housed in individual cages. The animal facilities and the experiments conformed with the Laboratory Animal Ethics Committee of North Sichuan Medical College guidelines. All drugs were administered intraperitoneally.
ELECTRODE IMPLANTATION:
The same bipolar insulated stainless-steel electrode (Plexon, Dallas, TX) was implanted into the right posterior ventral hippocampi (−5.3 mm anterior-posterior, ±4.9 mm media lateral, −5.0 depth from touchpoint) using a stereotaxic apparatus (RWD, China) under 10% pentobarbital sodium anesthesia, and another 2 electrodes were located over the left and right cortex. The electrode tips were placed in contact with the skull without penetrating the dura mater. Two fixation screws were used to ensure that the electrode adhered to the skull and was enhanced with sufficient resin, followed by standard dental acrylic cement. The procedure was based on previously described experiments [14]. Animals were given 5–7 days for recovery before starting the experiment.
SE MODEL DEVELOPMENT PROTOCOLS:
The animals were intraperitoneally (i.p.) administered 3 mmol/kg lithium chloride (Sigma, USA) dissolved in distilled water. Then, 45 mg/kg pilocarpine (Sigma, USA) was injected i.p. 16 h later to induce SE. Thirty min before pilocarpine administration, scopolamine (2 mg/kg; Sigma, USA) was administered to every model rat to avoid pilocarpine-induced cholinergic crisis. Pilocarpine was diluted with normal saline and scopolamine methyl bromide. Electroencephalographic (EEG) activity was monitored with animals moving freely for 5 h using bipolar insulated stainless-steel electrodes, and their behavior was observed and recorded. After pilocarpine injection, the rats gradually developed severe seizures; only animals that developed a seizure class up to 5 on the Racine scale [15] within 1 h were evaluated in this study.
INDUCTION OF SE AND CLASSES OF SEIZURES RECORDING:
All the model rat used for this study exhibited SE that lasted for 5 h or longer and had seizure behavior similar to that in humans; some animals had seizures that lasted 12 h or more. After a sufficient dose of pilocarpine injection, animals exhibited various classes of attacks which were reported before: wet-dog shaking, facial twitching and automatisms, chewing, staring, hind limb scratching, head bobbing, forelimb clonus, rearing, and falling with full-body convulsions, as documented in a previous study [15]. According to the Racine classification of seizures, class 2 included rats exhibiting head bobbing, class 3 included rats exhibiting forelimb clonus, class 4 included rats exhibiting rearing, and class 5 included rats exhibiting raising and falling with generalized convulsions [15]. When the epileptic seizures behavioral degree induced by pilocarpine was up to class 4–5 and continued for more than 5 min, we considered that SE was beginning [16]. During SE, most rats spent most of the time in class 3–5 seizures, with some time in class 2 seizures. Electrical attacks usually began with spikes clusters and later decreased in frequency over the first hour, then converted into rapidly repeated bursts.
SE TREATMENT:
Diazepam and ketamine were obtained from the Pharmacy of the First Affiliated Hospital of Chongqing Medical University for SE treatment in this research. Ketamine was dissolved in normal saline, and diazepam was dissolved in anhydrous alcohol so that the solution could be diluted in saline and rapidly delivered. Ketamine (water-soluble) and diazepam (liposoluble) were not mixed in a single syringe. In the diazepam-ketamine dual therapy groups, ketamine and diazepam were intraperitoneally injected sequentially without delay between injections.
Ketamine initial doses for experiments were determined according to a previous study by Borris et al [8]. Diazepam initial doses were determined according to previous research by Schneider et al [17]. Diazepam (20 mg/kg, 10 mg/kg, 5 mg/kg i.p.) monotherapy dosages or diazepam (10 mg/kg) and ketamine (90 mg/kg, 60 mg/kg, 45 mg/kg, 30 mg/kg i.p.) dual therapy dosages were tested first [13]. The dose of diazepam monotherapy was reduced from 20 mg/kg to 10 mg/kg because of mortality due to respiratory depression (3 of the 5 rats died in the 20 mg/kg group in the preliminary experiment). High mortality was experienced at ketamine 60 mg/kg combined with diazepam (3 of 5 mice died in the preliminary experiment). Therefore, we decided to use diazepam (10 mg/kg) monotherapy and diazepam (10 mg/kg) and ketamine (45mg/kg) dual therapy for subsequent experiments.
According to the parallel randomized controlled trial principle, SE model rats were randomized into groups. First, we treated each diazepam monotherapy group (1–4, n=8) at 4 different time points – 5 min, 10 min, 20 min, and 30 min – then assessed the degree of behavioral seizure and hippocampal field potential changes over time (Figure 1). Second, we treated each diazepam-ketamine dual therapy group (I–IV, n=8) at 6 different time points – 10 min, 20 min, 30 min, 40 min, 50 min, 60 min – then assessed the degree of behavioral seizure and hippocampal field potential changes over time (Figure 1). Finally, 5 SE rats in corresponding control groups at each time point were treated with 0.5 ml placebo, the same volume as diazepam.
The drug treatment efficacy was assessed by continuously monitoring eye seizure behavior and EEG activity through bipolar insulated stainless-steel electrodes for 5 h after drug injection. Experimenters who set the outcome were not the same who injected the drugs. Each of them had been trained in corresponding technology before the study started. The seizure termination event was obtained when behavioral seizures stopped and when the EEG amplitude fell below 2 times the baseline amplitude without recurrence of spikes, sharp waves, or irregular spikes in the following 5 hours. Animals reverted back to SE within 5 h after treatment were considered failures in this study. Glucose and sodium chloride were injected intraperitoneally to compensate for energy for the next 7 days. Survival rates of animals were monitored up to 7 days after SE to analyze the short-term consequences of mono- and dual therapy in SE.
Results
DIAZEPAM MONOTHERAPY TERMINATED SE AND REDUCED MORTALITY AT AN EARLY STAGE OF SE:
We used 10 mg/kg diazepam as the maximum effective dosage for seizure treatment in all subsequent experiments. We treated each group of SE animals with diazepam or an equal volume of placebo at 4 different time points – 5 min, 10 min, 20 min, and 30 min. ANOVA revealed that 10 mg/kg diazepam terminated seizures more effectively in the 5-min and 10-min groups than in the 20-min and 30-min groups in both EEG (Figure 2) and behavior (*** P<0.0001, Figure 3A). In the 5-min and 10-min groups, EEG power and amplitude quickly declined to twice the baseline. In the 20-min and 30-min groups, EEG power and amplitude had not changed obviously. Continuous behavior and seizure discharges in the 5-min and 10-min groups were distinctly shorter than in the 20-min and 30-min groups during the first hour of diazepam treatment (*** P<0.0001, Figure 3C). The degree of behavioral seizure in the 20-min and 30-min groups was controlled at grades 2 or 3 (*** P<0.0001, Figure 3B) with continuous EEG signals without obvious attenuation. We calculated the mortality in the following 7 days. A significant positive prognosis in survival was observed in cholinergic SE rats treated with diazepam at 5 min and 10 min compared to the control group and the other 2 groups. Earlier diazepam administration increased 1-week survival from 62% to 87% after SE (** P=0.0100; Figure 3D). The result was similar to that reported in a previous study [18]. In all, diazepam could terminate 5-min and 10-min SE effectively, but had less efficacy in 20-min and 30-min SE.
DIAZEPAM AND KETAMINE DUAL THERAPY TERMINATED SE AND REDUCED MORTALITY AT A LATER STAGE OF SE:
In the study, 10 mg/kg diazepam combined with 45 mg/kg ketamine as the maximum effective dosage was used in subsequent experiments. We treated each group of SE animals with dual therapy or an equal volume of placebo at 6 different time points in SE – 10 min, 20 min, 30 min, 40 min, 50 min, and 60 min. Electrographic seizure activity terminated rapidly and exhibited distinctly higher seizure-free rates in the 10-min, 20-min, and 30-min time point groups compared to the 50-min and 60-min time point groups (*** P<0.0001, Figures 4, 5A). Five animals in the 40-min time point group were well-controlled by the dual therapy, while the other 3 animals exhibited prominent seizure discharges on EEG, despite the disappearance of faint myoclonia during narcosis (Figure 4). The seizure-free rate in the 40-min time point group was also higher than in the 50-min and 60-min groups (* P=0.0221, Figure 5A). The latency to termination on EEG and behavior ranged from several minutes to half an hour (Figure 5C). Seizures were continuous during the 5-h observation period in each control group due to lack of effective treatment. Notably, the degree of behavioral seizure suddenly decreased to class 1 or disappeared entirely in the 50-min and 60-min groups (Figure 5B). The prognosis in survival obtained in post-SE rats that received dual therapy in the 30-min group was better than that in control and 60-min groups (Figure 5D). The earlier administration of diazepam combined with ketamine improved 1-week survival from 50% to 87% after SE (* P=0.0232; Figure 5D). In general, diazepam-ketamine dual therapy had better efficacy than diazepam monotherapy when SE onset was over 10 min and kept effectiveness to 30 min from the beginning of SE.
Discussion
Decreased GABAergic inhibition drives the brain state toward the ictal state at the beginning of a seizure. Improving GABAergic function controls seizures successfully. A recent randomized controlled trial recommended that benzodiazepine monotherapy was the preferred treatment strategy (Level A) for SE therapy when SE was in the early stage [19]. Our results demonstrate that a total dose of diazepam monotherapy terminated early SE successfully when administered less than 10 min from the beginning of SE. As time went on, gene expression of hippocampal tissue in SE model rats changed [20]. Thus, SE becomes progressively resistant to benzodiazepine anti-seizure drugs. Animal studies proposed a receptor-trafficking hypothesis stating that SE causes γ-aminobutyric acid A receptors (GABAARs) to be internalized and functionally inactivated [3,21,22]. This may explain why diazepam monotherapy failed when SE lasted longer. The results of the present study suggest that a sufficient dose of diazepam to control seizures has a relatively narrow therapeutic time window in cholinergic stimulation-induced SE animal models. When diazepam injection was delayed over 10 min from the beginning of SE, mild clonus in behavior was contained, and EEG spikes were monitored for the next 5 h.
The receptor-trafficking hypothesis states that internal ionotropic glutamatergic N-methyl-D-aspartate receptors (NMDARs) are transferred from the inner space to the postsynaptic membrane to be functionally activated, which likely increases excitability and promotes continued seizure activity [23]. Furthermore, ketamine, as an N-methyl-D-aspartate (NMDA) receptor noncompetitive antagonist, has been successfully used to control SE in animal models and humans in some research [24].
The present results are consistent with the hypothesis that dealing with GABAAR and NMDAR real-time trafficking would terminate prolonged SE [12]. A sufficient dose of diazepam-ketamine dual therapy had efficacy when SE onset was over 10 min and kept effectiveness to 30 min from the beginning of SE, but the effectiveness decreased rapidly when SE was over 30 min, even in combination with other anti-seizure drugs [13]; then, refractory SE developed.
In this study, the efficacy of dual therapy was consistent with the hypothesis that pharmaco-resistant involves GABAR and NMDAR trafficking, so the treatment targets were changed [25–27]. Therefore, the combination of 2 therapeutic agents, which have different mechanisms, should be considered. These results may lead to reconsideration of the traditional reliance on monotherapy as initial treatment in SE without considering other aspects. In the future, if we want to choose an effective SE treatment strategy to control self-sustained seizures, successfully avoiding behavior and EEG spikes recurrence and having a good prognosis, SE duration before therapy injection as an essential influence factor must be considered [28–30]. We still have much work to do if we want to apply results obtained from experimental animals [31] to clinical use, such as translating the ketamine dosages used in experimental animals to clinical application with pharmacologically equivalent drugs. Because of undesired psychological effects, adverse reactions, and problems with therapeutic efficacy and safety, ketamine-treated patients with prolonged SE must be studied in prospective randomized clinical trials.
Refractory SE developed when seizures lasted for 40 min or longer. Seizure activity exhausts the post-ictal state, which is intrinsically unstable, and changes to a stable refractory condition. The latter process may result from irreversible changes, such as neuronal death, receptor expression, and altered neurotransmitter release. For example, calcium entry through NMDARs activates nitric oxide synthase (NOS), which is primarily present in inflammatory cells [32]. The nitric oxide produced by NOS generally plays an important physiological role, but overproduction leads to DNA injury, lipid peroxidation, and cellular signaling change following SE. Therefore, therapeutic strategies that target the extra-synaptic δ-GABAR should be more effective in prolonged SE [33]. Studies demonstrated that extra-synaptic GABARs might not change during prolonged SE, and targeting these receptors may control refractory SE more effectively. Thus, further studies should focus on extra-synaptic refractory mechanisms in controlling SE, which have lasted for a longer time from the beginning.
Conclusions
Status epilepticus (SE) is an emergency that must be stopped as early as possible. The longer an SE episode lasts, the more significant the damage caused and the more difficult it is to stop. The present study in an animal model showed that diazepam monotherapy could terminate early SE lasting less than 10 min. Diazepam and ketamine dual therapy could end longer SE when treatment was delayed 10–30 min after SE onset. Our results provide a reference for SE treatment in humans in future research.
Figures
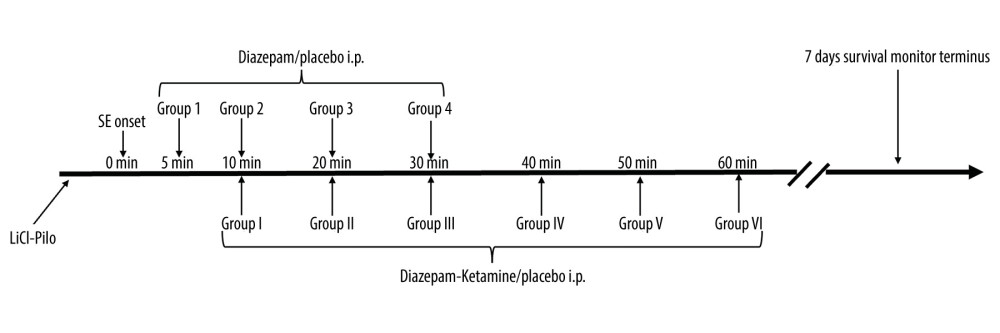 Figure 1. Experimental design. The SE model was established in adult Sprague-Dawley rats with lithium and pilocarpine (LiCl-Pilo i.p.). A few minutes later, status epilepticus began. Then, we treated each group (1–4, n=8) with diazepam (intraperitoneal injection) monotherapy at 4 different time points – 5 min, 10 min, 20 min, and 30 min – then assessed the degree of behavioral seizure and hippocampal field potential changes over time. Second, we treated each group (I–IV, n=8) with diazepam-ketamine dual therapy at 6 different time points – 10 min, 20 min, 30 min, 40 min, 50 min, 60 mi n– then assessed the degree of behavioral seizure and hippocampal field potential changes over time. At each time point in the control group, 5 SE rats were treated with a 0.5 ml placebo, the same volume as diazepam. Survival rates of animals were monitored for up to 7 days after SE to analyze the short-term consequences of mono- and dual therapy in SE.
Figure 1. Experimental design. The SE model was established in adult Sprague-Dawley rats with lithium and pilocarpine (LiCl-Pilo i.p.). A few minutes later, status epilepticus began. Then, we treated each group (1–4, n=8) with diazepam (intraperitoneal injection) monotherapy at 4 different time points – 5 min, 10 min, 20 min, and 30 min – then assessed the degree of behavioral seizure and hippocampal field potential changes over time. Second, we treated each group (I–IV, n=8) with diazepam-ketamine dual therapy at 6 different time points – 10 min, 20 min, 30 min, 40 min, 50 min, 60 mi n– then assessed the degree of behavioral seizure and hippocampal field potential changes over time. At each time point in the control group, 5 SE rats were treated with a 0.5 ml placebo, the same volume as diazepam. Survival rates of animals were monitored for up to 7 days after SE to analyze the short-term consequences of mono- and dual therapy in SE. 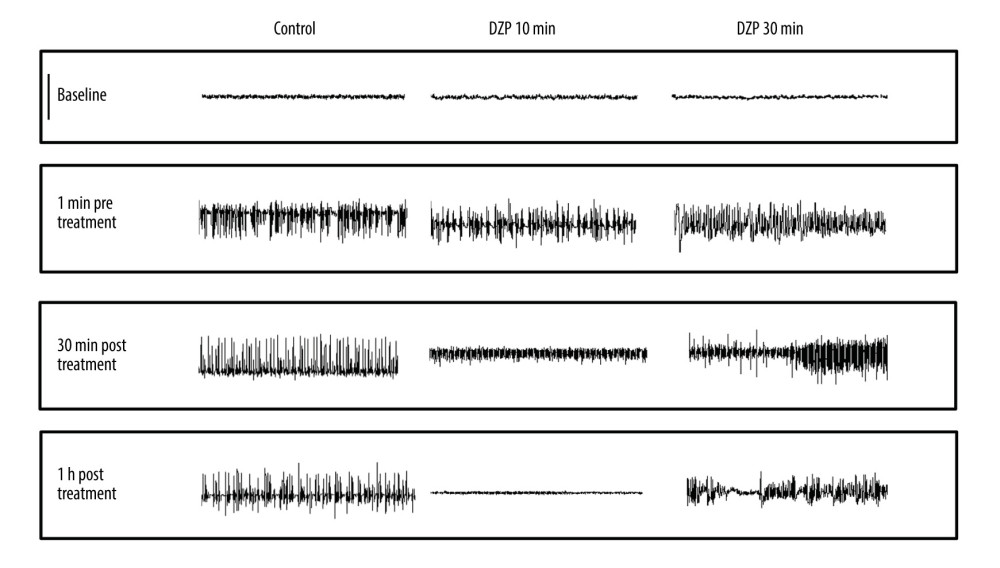 Figure 2. Representative EEG recordings from hippocampal electrodes (Omniplex Neural Data Acquisition System, Plexon, Inc.) in control, 10 min, and 30 min after SE onset with diazepam therapy. SE was induced via an injection of lithium chloride (5 mEq/kg, i.p.) and pilocarpine hydrochloride (40 mg/kg, i.p.) 16 h later. Rats in each time point group were treated with the same volume of placebo (control, n=5) or 10 mg/kg diazepam (n=8) after 5 min, 10 min, 20 min, and 30 min of SE. As illustrated in the post-treatment traces, treatments at 5 min or 10 min initially eliminated the epileptiform discharges. As a result, behavioral seizures were terminated as well. However, 10 mg/kg diazepam monotherapy, when SE lasted more than 10 min, failed to decrease EEG power to reach twice the pre-seizure baseline without bursting. Thus, each trace represents a 20-s period. Scale bar (upper left), 800 μV. EEG, electroencephalograph; SE, status epilepticus.
Figure 2. Representative EEG recordings from hippocampal electrodes (Omniplex Neural Data Acquisition System, Plexon, Inc.) in control, 10 min, and 30 min after SE onset with diazepam therapy. SE was induced via an injection of lithium chloride (5 mEq/kg, i.p.) and pilocarpine hydrochloride (40 mg/kg, i.p.) 16 h later. Rats in each time point group were treated with the same volume of placebo (control, n=5) or 10 mg/kg diazepam (n=8) after 5 min, 10 min, 20 min, and 30 min of SE. As illustrated in the post-treatment traces, treatments at 5 min or 10 min initially eliminated the epileptiform discharges. As a result, behavioral seizures were terminated as well. However, 10 mg/kg diazepam monotherapy, when SE lasted more than 10 min, failed to decrease EEG power to reach twice the pre-seizure baseline without bursting. Thus, each trace represents a 20-s period. Scale bar (upper left), 800 μV. EEG, electroencephalograph; SE, status epilepticus. 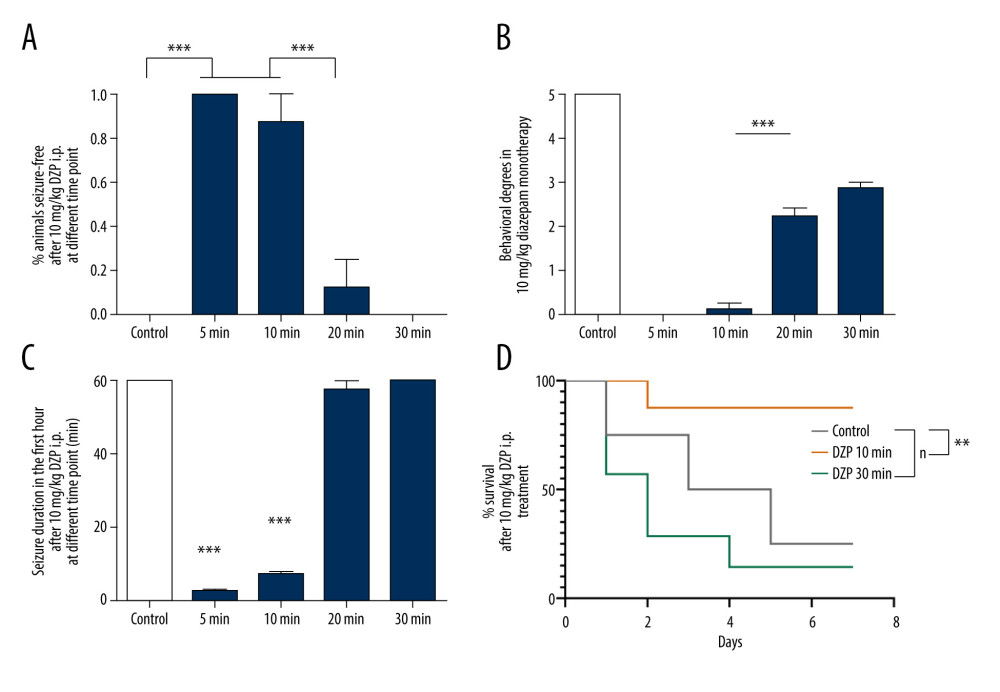 Figure 3. Comparisons of 10 mg/kg diazepam monotherapy for SE at different time points. (A) Monotherapy with 10 mg/kg diazepam when SE lasted longer than 10 min rapidly and significantly decreased the seizure-free rate. Only 1 SE rat was controlled when SE lasted for 20 min, and no rats were controlled in the 30 min group compared to the 7 controlled rats in the 10-min group and all controlled rats in the 5-min group (*** P<0.0001). ANOVA with Dunnett’s multiple comparisons. (B) The class of seizure was assessed according to the Racine scale at the end of the first hour after diazepam treatment, showing that rats in the 5-min and 10-min groups were treated successfully, but the class of seizure stayed 2–3 in the 20-min and 30-min groups with continued EEG signals (*** P<0.0001). (C) Diazepam (10 mg/kg) administration for SE in the first 10 min was effective (5 min, 10 min) and drastically reduced seizure duration (*** P<0.0001 vs 20 min, 30 min), which suggests a temporal effect of diazepam treatment. Values are means±SEM. ANOVA with Dunnett’s multiple comparisons. (D) Survival rates of animals that received placebo (n=5) or diazepam treatment at 10 min and 30 min (n=8) were monitored 7 days after therapy (** P=0.0100, log-rank test). Data are shown as the means±SEM (GraphPad Prism, Prism 8.0.2, GraphPad Software, Inc.). EEG, electroencephalograph; SE, status epilepticus.
Figure 3. Comparisons of 10 mg/kg diazepam monotherapy for SE at different time points. (A) Monotherapy with 10 mg/kg diazepam when SE lasted longer than 10 min rapidly and significantly decreased the seizure-free rate. Only 1 SE rat was controlled when SE lasted for 20 min, and no rats were controlled in the 30 min group compared to the 7 controlled rats in the 10-min group and all controlled rats in the 5-min group (*** P<0.0001). ANOVA with Dunnett’s multiple comparisons. (B) The class of seizure was assessed according to the Racine scale at the end of the first hour after diazepam treatment, showing that rats in the 5-min and 10-min groups were treated successfully, but the class of seizure stayed 2–3 in the 20-min and 30-min groups with continued EEG signals (*** P<0.0001). (C) Diazepam (10 mg/kg) administration for SE in the first 10 min was effective (5 min, 10 min) and drastically reduced seizure duration (*** P<0.0001 vs 20 min, 30 min), which suggests a temporal effect of diazepam treatment. Values are means±SEM. ANOVA with Dunnett’s multiple comparisons. (D) Survival rates of animals that received placebo (n=5) or diazepam treatment at 10 min and 30 min (n=8) were monitored 7 days after therapy (** P=0.0100, log-rank test). Data are shown as the means±SEM (GraphPad Prism, Prism 8.0.2, GraphPad Software, Inc.). EEG, electroencephalograph; SE, status epilepticus. 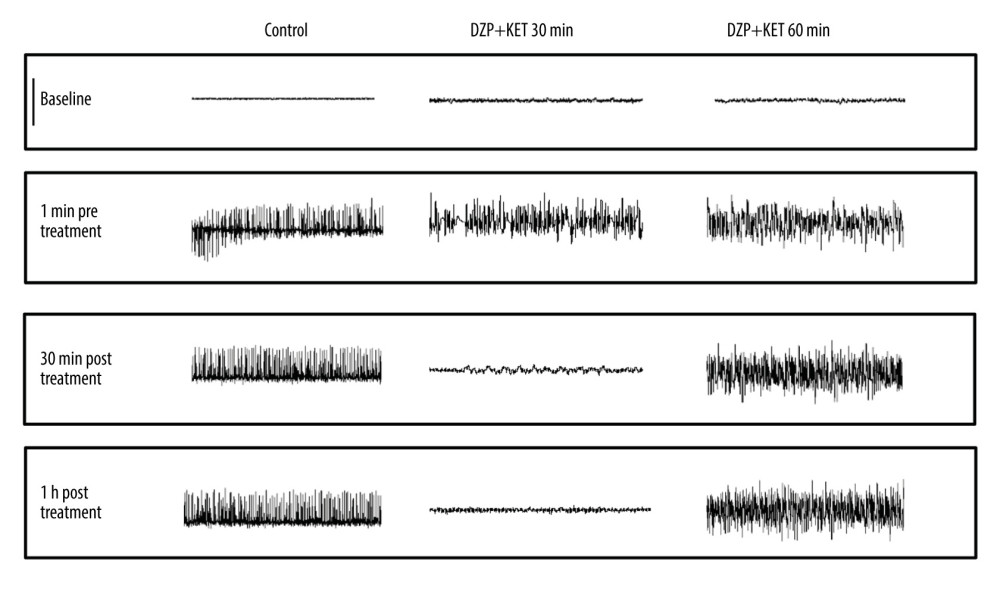 Figure 4. Representative EEG recordings from hippocampal electrodes (Omniplex Neural Data Acquisition System, Plexon, Inc.) in control, 30 min, and 60 min SE with diazepam-ketamine dual therapy. SE was induced via injection of lithium chloride (5 mEq/kg, i.p.) and pilocarpine hydrochloride (40 mg/kg, i.p.) 16 h later. The rats in each time point group were treated with the same volume of placebo (control, n=5) or 10 mg/kg diazepam combined with 45 mg/kg ketamine (n=8) at 10, 20, 30, 40, 50, or 60 min SE. As illustrated in the post-treatment traces, dual therapy at 10 min to 30 min SE initially eliminated the epileptiform discharges. Behavioral seizure activity was also terminated. The SE EEG pattern exhibited no changes when dual therapy was administered after 50 min or 60 min SE. Five animals in the 40-min group were controlled well with dual therapy. but the other 3 animals exhibited apparent seizure discharges in EEG despite the disappearance of faint myoclonia during narcosis. Each trace represents a 20-s period: scale bar (upper left), 800 μV.
Figure 4. Representative EEG recordings from hippocampal electrodes (Omniplex Neural Data Acquisition System, Plexon, Inc.) in control, 30 min, and 60 min SE with diazepam-ketamine dual therapy. SE was induced via injection of lithium chloride (5 mEq/kg, i.p.) and pilocarpine hydrochloride (40 mg/kg, i.p.) 16 h later. The rats in each time point group were treated with the same volume of placebo (control, n=5) or 10 mg/kg diazepam combined with 45 mg/kg ketamine (n=8) at 10, 20, 30, 40, 50, or 60 min SE. As illustrated in the post-treatment traces, dual therapy at 10 min to 30 min SE initially eliminated the epileptiform discharges. Behavioral seizure activity was also terminated. The SE EEG pattern exhibited no changes when dual therapy was administered after 50 min or 60 min SE. Five animals in the 40-min group were controlled well with dual therapy. but the other 3 animals exhibited apparent seizure discharges in EEG despite the disappearance of faint myoclonia during narcosis. Each trace represents a 20-s period: scale bar (upper left), 800 μV. 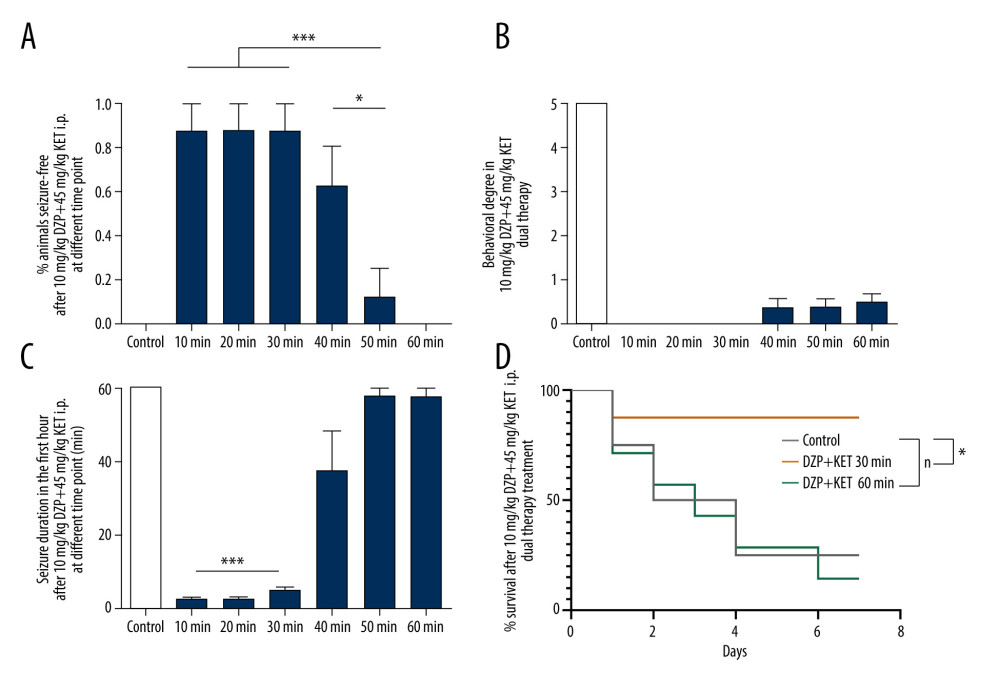 Figure 5. Comparisons of diazepam and ketamine dual therapy for SE at different time points. (A) Diazepam-ketamine dual therapy was injected when SE lasted over 40 min; the seizure-free rate decreased rapidly and significantly. SE was only controlled in 1 rat when SE was over 50 min; none of the rats were controlled in the 60-min group compared to all controlled in 10-min, 20-min, and 30-min groups (*** P<0.0001). ANOVA with Dunnett’s multiple comparisons. (B) The degree of seizures was assessed according to the Racine scale. SE in 10-min, 20-min, and 30-min groups were terminated successfully. The degree of behavioral seizure suddenly decreased to class 1 or disappeared completely in the 50-min and 60-min groups (*** P<0.0001). (C) Dual therapy was injected when SE was over 40 min, and it failed to decrease EEG power to reach twice the pre-seizure baseline, a pattern that indicates seizure termination. Dual therapy was administrated when SE lasted less than 30 min, and it drastically terminated SE. (*** P<0.0001), which suggests a temporal effect of diazepam-ketamine dual therapy. Values are means±SEM. ANOVA with Dunnett’s multiple comparisons. (D) Survival rates of animals that received placebo (n=5) or diazepam-ketamine 30 min (n=8) or diazepam-ketamine 60 min up to 7 days after SE treatment (* P=0.023, Log-rank test). Data are shown as the means±SEM (GraphPad Prism, Prism 8.0.2, GraphPad Software, Inc.). SE, status epilepticus; diazepam-ketamine, diazepam 10 mg/kg+ketamine 45 mg/kg.
Figure 5. Comparisons of diazepam and ketamine dual therapy for SE at different time points. (A) Diazepam-ketamine dual therapy was injected when SE lasted over 40 min; the seizure-free rate decreased rapidly and significantly. SE was only controlled in 1 rat when SE was over 50 min; none of the rats were controlled in the 60-min group compared to all controlled in 10-min, 20-min, and 30-min groups (*** P<0.0001). ANOVA with Dunnett’s multiple comparisons. (B) The degree of seizures was assessed according to the Racine scale. SE in 10-min, 20-min, and 30-min groups were terminated successfully. The degree of behavioral seizure suddenly decreased to class 1 or disappeared completely in the 50-min and 60-min groups (*** P<0.0001). (C) Dual therapy was injected when SE was over 40 min, and it failed to decrease EEG power to reach twice the pre-seizure baseline, a pattern that indicates seizure termination. Dual therapy was administrated when SE lasted less than 30 min, and it drastically terminated SE. (*** P<0.0001), which suggests a temporal effect of diazepam-ketamine dual therapy. Values are means±SEM. ANOVA with Dunnett’s multiple comparisons. (D) Survival rates of animals that received placebo (n=5) or diazepam-ketamine 30 min (n=8) or diazepam-ketamine 60 min up to 7 days after SE treatment (* P=0.023, Log-rank test). Data are shown as the means±SEM (GraphPad Prism, Prism 8.0.2, GraphPad Software, Inc.). SE, status epilepticus; diazepam-ketamine, diazepam 10 mg/kg+ketamine 45 mg/kg. References
1. Trinka E, Cock H, Hesdorffer D, A definition and classification of status epilepticus – Report of the ILAE Task Force on Classification of Status Epilepticus: Epilepsia, 2015; 10; 1515-23
2. Strzelczyk A, Ansorge S, Hapfelmeier J, Costs, length of stay, and mortality of super-refractory status epilepticus: A population-based study from Germany: Epilepsia, 2017; 9; 1533-41
3. Prasad K, Al-Roomi K, Krishnan PR, Sequeira R, Anticonvulsant therapy for status epilepticus: Cochrane Database Syst Rev, 2005; 4; CD003723
4. Spampanato J, Bealer SL, Smolik M, Dudek FE, Delayed adjunctive treatment of organophosphate-induced status epilepticus in rats with phenobarbital, memantine, or dexmedetomidine: J Pharmacol Exp Ther, 2020; 375; 59-68
5. Leppik IE, Derivan AT, Homan RW, Double-blind study of lorazepam and diazepam in status epilepticus: JAMA, 1983; 11; 1452-54
6. Treiman DM, Meyers PD, Walton NY, A comparison of four treatments for generalized convulsive status epilepticus. Veterans Affairs Status Epilepticus Cooperative Study Group: N Engl J Med, 1998; 12; 792-98
7. Alldredge BK, Gelb AM, Isaacs SM, A comparison of lorazepam, diazepam, and placebo for the treatment of out-of-hospital status epilepticus: N Engl J Med, 2001; 9; 631-37
8. Borris DJ, Bertram EH, Kapur J, Ketamine controls prolonged status epilepticus: Epilepsy Res, 2000; 2–3; 117-22
9. Mazarati AM, Wasterlain CG, N-methyl-D-aspartate receptor antagonists abolish the maintenance phase of self-sustaining status epilepticus in rat: Neurosci Lett, 1999; 3; 187-90
10. Shih T, McDonough JH, Koplovitz I, Anticonvulsants for soman-induced seizure activity: J Biomed Sci, 1999; 2; 86-96
11. Martin BS, Kapur J, A combination of ketamine and diazepam synergistically controls refractory status epilepticus induced by cholinergic stimulation: Epilepsia, 2008; 2; 248-55
12. Niquet J, Baldwin R, Norman K, Midazolam-ketamine dual therapy stops cholinergic status epilepticus and reduces Morris water maze deficits: Epilepsia, 2016; 9; 1406-15
13. Niquet J, Baldwin R, Suchomelova L, Treatment of experimental status epilepticus with synergistic drug combinations: Epilepsia, 2017; 4; e49-e53
14. Yang Y, Tian X, Xu D, GPR40 modulates epileptic seizure and NMDA receptor function: Sci Adv, 2018; 10; eaau2357
15. Racine RJ, Gartner JG, Burnham WM, Epileptiform activity and neural plasticity in limbic structures: Brain Res, 1972; 1; 262-68
16. Lowenstein DH, Status epilepticus: An overview of the clinical problem: Epilepsia, 1999; 40(Suppl 1); S3-8 discussion S21–22
17. Schneider HH, Stephens DN, Co-existence of kindling induced by the beta-carboline, FG 7142, and tolerance to diazepam following chronic treatment in mice: Eur J Pharmacol, 1988; 1; 35-45
18. Walton NY, Treiman DM, Response of status epilepticus induced by lithium and pilocarpine to treatment with diazepam: Exp Neurol, 1988; 2; 267-75
19. Glauser T, Shinnar S, Gloss D, Evidence-based guideline: treatment of convulsive status epilepticus in children and adults: Report of the Guideline Committee of the American Epilepsy Society: Epilepsy Curr, 2016; 11; 48-61
20. Conte G, Parras A, Alves M, High concordance between hippocampal transcriptome of the mouse intra-amygdala kainic acid model and human temporal lobe epilepsy: Epilepsia, 2020; 61; 2795-810
21. Naylor DE, Liu H, Wasterlain CG, Trafficking of GABA(A) receptors, loss of inhibition, and a mechanism for pharmacoresistance in status epilepticus: J Neurosc, 2005; 34; 7724-33
22. Goodkin HP, Joshi S, Mtchedlishvili Z, Subunit-specific trafficking of GABA(A) receptors during status epilepticus: J Neurosci, 2008; 10; 2527-38
23. Chen JW, Naylor DE, Wasterlain CG, Advances in the pathophysiology of status epilepticus: Acta Neurol Scand Suppl, 2007; 186; 7-15
24. Samanta D, Ketamine infusion for super refractory status epilepticus in alternating hemiplegia of childhood: Neuropediatrics, 2020; 51; 225-28
25. Celli R, Forna F, Targeting ionotropic glutamate receptors in the treatment of epilepsy: Curr Neuropharmacol, 2020; 18; 1
26. Naylor DE, Liu H, Niquet J, Wasterlain CG, Rapid surface accumulation of NMDA receptors increases glutamatergic excitation during status epilepticus: Neurobiol Dis, 2013; 54; 225-38
27. Niquet J, Lumley L, Baldwin R, Early polytherapy for benzodiazepine-refractory status epilepticus: Epilepsy Behav, 2019; 101; 1063-67
28. Verrotti A, Mazzocchetti C, Epilepsy: Timely treatment of refractory convulsive status epilepticus: Nat Rev Neurol, 2018; 5; 256-58
29. DeLorenzo RJ, Kirmani B, Deshpande LS, Comparisons of the mortality and clinical presentations of status epilepticus in private practice community and university hospital settings in Richmond, Virginia: Seizure, 2009; 6; 405-11
30. Loscher W, Single versus combinatorial therapies in status epilepticus: Novel data from preclinical models: Epilepsy Behav, 2015; 49; 20-25
31. Lumley L, Niquet J, Marrero-Rosado B, Treatment of acetylcholinesterase inhibitor-induced seizures with polytherapy targeting GABA and glutamate receptors: Neuropharmacology, 2021; 185; 108444
32. Forstermann U, Sessa WC, Nitric oxide synthases: regulation and function: Eur Heart J, 2012; 7; 829-837
33. Puttachary S, Sharma S, Verma S, 1400W, a highly selective inducible nitric oxide synthase inhibitor is a potential disease modifier in the rat kainate model of temporal lobe epilepsy: Neurobiol Dis, 2016; 93; 184-200
Figures
 Figure 1. Experimental design. The SE model was established in adult Sprague-Dawley rats with lithium and pilocarpine (LiCl-Pilo i.p.). A few minutes later, status epilepticus began. Then, we treated each group (1–4, n=8) with diazepam (intraperitoneal injection) monotherapy at 4 different time points – 5 min, 10 min, 20 min, and 30 min – then assessed the degree of behavioral seizure and hippocampal field potential changes over time. Second, we treated each group (I–IV, n=8) with diazepam-ketamine dual therapy at 6 different time points – 10 min, 20 min, 30 min, 40 min, 50 min, 60 mi n– then assessed the degree of behavioral seizure and hippocampal field potential changes over time. At each time point in the control group, 5 SE rats were treated with a 0.5 ml placebo, the same volume as diazepam. Survival rates of animals were monitored for up to 7 days after SE to analyze the short-term consequences of mono- and dual therapy in SE.
Figure 1. Experimental design. The SE model was established in adult Sprague-Dawley rats with lithium and pilocarpine (LiCl-Pilo i.p.). A few minutes later, status epilepticus began. Then, we treated each group (1–4, n=8) with diazepam (intraperitoneal injection) monotherapy at 4 different time points – 5 min, 10 min, 20 min, and 30 min – then assessed the degree of behavioral seizure and hippocampal field potential changes over time. Second, we treated each group (I–IV, n=8) with diazepam-ketamine dual therapy at 6 different time points – 10 min, 20 min, 30 min, 40 min, 50 min, 60 mi n– then assessed the degree of behavioral seizure and hippocampal field potential changes over time. At each time point in the control group, 5 SE rats were treated with a 0.5 ml placebo, the same volume as diazepam. Survival rates of animals were monitored for up to 7 days after SE to analyze the short-term consequences of mono- and dual therapy in SE. Figure 2. Representative EEG recordings from hippocampal electrodes (Omniplex Neural Data Acquisition System, Plexon, Inc.) in control, 10 min, and 30 min after SE onset with diazepam therapy. SE was induced via an injection of lithium chloride (5 mEq/kg, i.p.) and pilocarpine hydrochloride (40 mg/kg, i.p.) 16 h later. Rats in each time point group were treated with the same volume of placebo (control, n=5) or 10 mg/kg diazepam (n=8) after 5 min, 10 min, 20 min, and 30 min of SE. As illustrated in the post-treatment traces, treatments at 5 min or 10 min initially eliminated the epileptiform discharges. As a result, behavioral seizures were terminated as well. However, 10 mg/kg diazepam monotherapy, when SE lasted more than 10 min, failed to decrease EEG power to reach twice the pre-seizure baseline without bursting. Thus, each trace represents a 20-s period. Scale bar (upper left), 800 μV. EEG, electroencephalograph; SE, status epilepticus.
Figure 2. Representative EEG recordings from hippocampal electrodes (Omniplex Neural Data Acquisition System, Plexon, Inc.) in control, 10 min, and 30 min after SE onset with diazepam therapy. SE was induced via an injection of lithium chloride (5 mEq/kg, i.p.) and pilocarpine hydrochloride (40 mg/kg, i.p.) 16 h later. Rats in each time point group were treated with the same volume of placebo (control, n=5) or 10 mg/kg diazepam (n=8) after 5 min, 10 min, 20 min, and 30 min of SE. As illustrated in the post-treatment traces, treatments at 5 min or 10 min initially eliminated the epileptiform discharges. As a result, behavioral seizures were terminated as well. However, 10 mg/kg diazepam monotherapy, when SE lasted more than 10 min, failed to decrease EEG power to reach twice the pre-seizure baseline without bursting. Thus, each trace represents a 20-s period. Scale bar (upper left), 800 μV. EEG, electroencephalograph; SE, status epilepticus. Figure 3. Comparisons of 10 mg/kg diazepam monotherapy for SE at different time points. (A) Monotherapy with 10 mg/kg diazepam when SE lasted longer than 10 min rapidly and significantly decreased the seizure-free rate. Only 1 SE rat was controlled when SE lasted for 20 min, and no rats were controlled in the 30 min group compared to the 7 controlled rats in the 10-min group and all controlled rats in the 5-min group (*** P<0.0001). ANOVA with Dunnett’s multiple comparisons. (B) The class of seizure was assessed according to the Racine scale at the end of the first hour after diazepam treatment, showing that rats in the 5-min and 10-min groups were treated successfully, but the class of seizure stayed 2–3 in the 20-min and 30-min groups with continued EEG signals (*** P<0.0001). (C) Diazepam (10 mg/kg) administration for SE in the first 10 min was effective (5 min, 10 min) and drastically reduced seizure duration (*** P<0.0001 vs 20 min, 30 min), which suggests a temporal effect of diazepam treatment. Values are means±SEM. ANOVA with Dunnett’s multiple comparisons. (D) Survival rates of animals that received placebo (n=5) or diazepam treatment at 10 min and 30 min (n=8) were monitored 7 days after therapy (** P=0.0100, log-rank test). Data are shown as the means±SEM (GraphPad Prism, Prism 8.0.2, GraphPad Software, Inc.). EEG, electroencephalograph; SE, status epilepticus.
Figure 3. Comparisons of 10 mg/kg diazepam monotherapy for SE at different time points. (A) Monotherapy with 10 mg/kg diazepam when SE lasted longer than 10 min rapidly and significantly decreased the seizure-free rate. Only 1 SE rat was controlled when SE lasted for 20 min, and no rats were controlled in the 30 min group compared to the 7 controlled rats in the 10-min group and all controlled rats in the 5-min group (*** P<0.0001). ANOVA with Dunnett’s multiple comparisons. (B) The class of seizure was assessed according to the Racine scale at the end of the first hour after diazepam treatment, showing that rats in the 5-min and 10-min groups were treated successfully, but the class of seizure stayed 2–3 in the 20-min and 30-min groups with continued EEG signals (*** P<0.0001). (C) Diazepam (10 mg/kg) administration for SE in the first 10 min was effective (5 min, 10 min) and drastically reduced seizure duration (*** P<0.0001 vs 20 min, 30 min), which suggests a temporal effect of diazepam treatment. Values are means±SEM. ANOVA with Dunnett’s multiple comparisons. (D) Survival rates of animals that received placebo (n=5) or diazepam treatment at 10 min and 30 min (n=8) were monitored 7 days after therapy (** P=0.0100, log-rank test). Data are shown as the means±SEM (GraphPad Prism, Prism 8.0.2, GraphPad Software, Inc.). EEG, electroencephalograph; SE, status epilepticus. Figure 4. Representative EEG recordings from hippocampal electrodes (Omniplex Neural Data Acquisition System, Plexon, Inc.) in control, 30 min, and 60 min SE with diazepam-ketamine dual therapy. SE was induced via injection of lithium chloride (5 mEq/kg, i.p.) and pilocarpine hydrochloride (40 mg/kg, i.p.) 16 h later. The rats in each time point group were treated with the same volume of placebo (control, n=5) or 10 mg/kg diazepam combined with 45 mg/kg ketamine (n=8) at 10, 20, 30, 40, 50, or 60 min SE. As illustrated in the post-treatment traces, dual therapy at 10 min to 30 min SE initially eliminated the epileptiform discharges. Behavioral seizure activity was also terminated. The SE EEG pattern exhibited no changes when dual therapy was administered after 50 min or 60 min SE. Five animals in the 40-min group were controlled well with dual therapy. but the other 3 animals exhibited apparent seizure discharges in EEG despite the disappearance of faint myoclonia during narcosis. Each trace represents a 20-s period: scale bar (upper left), 800 μV.
Figure 4. Representative EEG recordings from hippocampal electrodes (Omniplex Neural Data Acquisition System, Plexon, Inc.) in control, 30 min, and 60 min SE with diazepam-ketamine dual therapy. SE was induced via injection of lithium chloride (5 mEq/kg, i.p.) and pilocarpine hydrochloride (40 mg/kg, i.p.) 16 h later. The rats in each time point group were treated with the same volume of placebo (control, n=5) or 10 mg/kg diazepam combined with 45 mg/kg ketamine (n=8) at 10, 20, 30, 40, 50, or 60 min SE. As illustrated in the post-treatment traces, dual therapy at 10 min to 30 min SE initially eliminated the epileptiform discharges. Behavioral seizure activity was also terminated. The SE EEG pattern exhibited no changes when dual therapy was administered after 50 min or 60 min SE. Five animals in the 40-min group were controlled well with dual therapy. but the other 3 animals exhibited apparent seizure discharges in EEG despite the disappearance of faint myoclonia during narcosis. Each trace represents a 20-s period: scale bar (upper left), 800 μV. Figure 5. Comparisons of diazepam and ketamine dual therapy for SE at different time points. (A) Diazepam-ketamine dual therapy was injected when SE lasted over 40 min; the seizure-free rate decreased rapidly and significantly. SE was only controlled in 1 rat when SE was over 50 min; none of the rats were controlled in the 60-min group compared to all controlled in 10-min, 20-min, and 30-min groups (*** P<0.0001). ANOVA with Dunnett’s multiple comparisons. (B) The degree of seizures was assessed according to the Racine scale. SE in 10-min, 20-min, and 30-min groups were terminated successfully. The degree of behavioral seizure suddenly decreased to class 1 or disappeared completely in the 50-min and 60-min groups (*** P<0.0001). (C) Dual therapy was injected when SE was over 40 min, and it failed to decrease EEG power to reach twice the pre-seizure baseline, a pattern that indicates seizure termination. Dual therapy was administrated when SE lasted less than 30 min, and it drastically terminated SE. (*** P<0.0001), which suggests a temporal effect of diazepam-ketamine dual therapy. Values are means±SEM. ANOVA with Dunnett’s multiple comparisons. (D) Survival rates of animals that received placebo (n=5) or diazepam-ketamine 30 min (n=8) or diazepam-ketamine 60 min up to 7 days after SE treatment (* P=0.023, Log-rank test). Data are shown as the means±SEM (GraphPad Prism, Prism 8.0.2, GraphPad Software, Inc.). SE, status epilepticus; diazepam-ketamine, diazepam 10 mg/kg+ketamine 45 mg/kg.
Figure 5. Comparisons of diazepam and ketamine dual therapy for SE at different time points. (A) Diazepam-ketamine dual therapy was injected when SE lasted over 40 min; the seizure-free rate decreased rapidly and significantly. SE was only controlled in 1 rat when SE was over 50 min; none of the rats were controlled in the 60-min group compared to all controlled in 10-min, 20-min, and 30-min groups (*** P<0.0001). ANOVA with Dunnett’s multiple comparisons. (B) The degree of seizures was assessed according to the Racine scale. SE in 10-min, 20-min, and 30-min groups were terminated successfully. The degree of behavioral seizure suddenly decreased to class 1 or disappeared completely in the 50-min and 60-min groups (*** P<0.0001). (C) Dual therapy was injected when SE was over 40 min, and it failed to decrease EEG power to reach twice the pre-seizure baseline, a pattern that indicates seizure termination. Dual therapy was administrated when SE lasted less than 30 min, and it drastically terminated SE. (*** P<0.0001), which suggests a temporal effect of diazepam-ketamine dual therapy. Values are means±SEM. ANOVA with Dunnett’s multiple comparisons. (D) Survival rates of animals that received placebo (n=5) or diazepam-ketamine 30 min (n=8) or diazepam-ketamine 60 min up to 7 days after SE treatment (* P=0.023, Log-rank test). Data are shown as the means±SEM (GraphPad Prism, Prism 8.0.2, GraphPad Software, Inc.). SE, status epilepticus; diazepam-ketamine, diazepam 10 mg/kg+ketamine 45 mg/kg. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








