08 August 2022: Clinical Research
Identification of Hepatic Dendritic Cells in Liver Biopsies in Patients with Metabolic Dysfunction-Associated Fatty Liver Diseas (MAFLD) and Obesity
Beatriz Barranco-Fragoso12ABC, Shreya C. PalDOI: 10.12659/MSM.937528
Med Sci Monit 2022; 28:e937528
Abstract
BACKGROUND: Metabolic dysfunction-associated fatty liver disease (MAFLD) is now the term used for hepatic steatosis in patients who are overweight or obese, have type 2 diabetes mellitus (T2DM), or evidence of metabolic dysregulation. The prevalence of MAFLD among morbidly obese subjects is 65-93%. Hepatic dendritic cells (hDCs) are antigen-presenting cells that induce T cell-mediated immunity. MAFLD pathogenesis involves numerous immune cell-mediated inflammatory processes, while the particular role of hDCs is yet to be well defined. This study aimed to identify hDCs in liver biopsies from 128 patients with MAFLD associated with obesity.
MATERIAL AND METHODS: In this cross-sectional study, 128 liver biopsies from 128 patients with MAFLD (diagnosed as presence of hepatic steatosis, plus T2DM, metabolic dysregulation or overweight/obesity) were collected and assessed for CD11c⁺ immunoreactivity degree (CD11c as dendritic cell biomarker), through antigen retrieval, reaction with CD11c antibodies (primary), and marking with diaminobenzidine chromogen.
RESULTS: Among the 128 patients with MAFLD, 64 (50%) had MAFLD and fibrosis and 72 (56.2%) positively expressed hDCs (CD11c⁺). Among morbidly obese patients, 49 (64.5%) positively expressed hDCs (CD11c⁺) in liver tissue; from patients with obesity grade I- grade II (GI-II), 18 (54.5%) positively expressed hDCs (CD11c⁺) in liver tissue; and from non-obese patients with MAFLD, 5 (26.3%) positively expressed hDCs (CD11c⁺) in liver tissue.
CONCLUSIONS: hDC expression increases significantly in morbidly obese patients with MAFLD compared with non-obese patients, independent of the degree of fibrosis, suggesting the role of adaptive changes within hDCs in the perpetuation of inflammatory insults in chronic liver diseases.
Keywords: CD11c Antigen, Dendritic Cells, Non-alcoholic fatty liver disease, Obesity, Morbid, Biopsy, Cross-Sectional Studies, Diabetes Mellitus, Type 2, Fatty Liver, Fibrosis, Humans, Liver Diseases, Non-alcoholic fatty liver disease, overweight
Background
DUAL ROLE OF HDCS:
Broadly speaking, DCs have migratory and adaptive features (when changing from a tolerant to a reactive state); they are also important in antigen capture, processing, and presentation. However, hDCs are derived from bone marrow and tend to remain tolerant in the healthy liver [16]. A dual role of hDCs in the development and progression of MAFLD has been described; the reported interaction between the role of hDCs in obesity, MAFLD, and steatohepatitis in experimental models and in humans can be understood as depicted in Figure 1.
It has been proposed that in homeostatic conditions, classical hDCs (Cor+, CD11c+) aim to maintain quiescence and tolerance of HSCs and KCs by secreting specific cytokines (eg, IL-10), thereby ameliorating the magnitude of TLR response to bacterial components, as well as by clearing cellular debris [11]. Thus, initially, hDCs (CD40+ expressing CD11c+ cells) promote lipid storage in hepatocytes and suppress inflammasome and T cell activation in the healthy liver, and they induce a Treg response against obesity-induced ectopic lipid storage and metabolic dysfunction, acting as a protective factor in acute conditions [17]. In contrast, during steatohepatitis, hDCs (CD11c+ MHCII+) expand and mature, thereby assuming an activated immune phenotype which involves the expression of costimulatory molecules, production of cytokines, and increased CD4 T cell activation; furthermore, CD40+ on hDCs promotes liver inflammation and leads to higher hepatic and plasmatic cholesterol levels. Therefore, adaptive changes of hDCs contribute to the promotion and perpetuation of liver fibro-inflammatory insults in advanced chronic disease [11,18].
Interestingly, the relationship between CD11c+ cells and fibrosis might be influenced by increased cytokine production and proliferative responses of hepatic stellate cells. Thus, significant dynamic changes of CD11c+ cells during fibrosis progression might impact the inflammatory environment of the liver, but effects on fibrosis progression have not been assessed [14].
HDCS IN STEATOHEPATITIS AND FIBROSIS:
A study carried out in 2013 by Henning and colleagues showed that the depletion of hDCs in steatohepatitis models leads to the promotion and maintenance of the intrahepatic fibro-inflammatory processes by “uncontrolled” innate immune cell interactions, thereby stimulating apoptosis and accelerating fibrosis instauration [12]. Additionally, during fibro-inflammatory injury recovery, the absence of hDCs delayed the resolution of damage [12,13]. Of note, HSC cytokine production and proliferative responses might be influenced by the presence of hDCs (CD11c+) [14].
As a factor on its own, inflammation promotes massive expansion and maturation of hDCs (CD11c+/MHCIIhigh/CD103−/CD11b+) by diverse stimuli, such as CD80 stimulation [18]. As human studies progress, hDCs’ role in the development and progression of MAFLD has continuously yielded intriguing results, establishing it as a current topic of interest [19].
In this study we assessed the presence hDCs using CD11c+ as a broad biomarker of hDC expression in liver tissue from patients with MAFLD and stratified its differential distribution according to patients’ body weight and presence of liver fibrosis. Therefore, this study aimed to identify hepatic dendritic cells in liver biopsy samples from 128 patients with MAFLD associated with obesity.
Material and Methods
ETHICS STATEMENT:
The study was approved by the local Ethics Committee in Medica Sur Foundation and Clinic, with the identification number 2020-EXT-449. All participants in this study signed an informed consent acknowledging the purpose of the liver biopsies taken during their surgical procedure, establishing clearly the anonymity of their personal data and the aims of the study in which they accepted to be enrolled.
PARTICIPANTS’ SELECTION:
In this observational cross-sectional study, 128 liver biopsies from adult patients with prior Non-Alcoholic Fatty Liver Disease (NAFLD) diagnosis (ie, exclusion diagnosis criteria were used to determine NAFLD in these patients) were initially compiled from the Pathology Department of the Medica Sur Clinic & Foundation (2012–2020), with the institution’s research and ethics committee approval. Medical records from the included patients were checked for clinical data registers (eg, sex, BMI, elements of metabolic syndrome). The MAFLD diagnosis was made based on histological evidence of steatosis plus either obesity, type 2 diabetes mellitus (T2DM), or metabolic dysfunction [20].
BMI, T2DM, METABOLIC DYSFUNCTION, AND FIBROSIS: OPERATIONAL DEFINITIONS:
According to the objectives of the study, participants were classified using the World Health Organization’s stratification of BMI into those with normal weight and overweight (BMI: 18–24.9 kg/m2 and 25–29.9 km/m2, respectively), obesity grade I and II (GI–II) (BMI: >30–34.9), and morbid obesity (BMI: >40 kg/m2) [21]. Non-obese MAFLD patients included those who had other MAFLD criteria exempting obesity, such as prior T2DM diagnosis or metabolic dysfunction in medical records (based on clinical and laboratory data). Metabolic dysfunction was defined as the presence of at least 2 risk factor abnormalities (hypertriglyceridemia, low high-density lipoprotein cholesterol levels, hypertension, high fasting glucose, and/or prediabetes) [20]. Liver fibrosis categorical determination (presence vs absence) was made blindly by 1 expert pathologist using trichrome Masson staining of each liver sample.
IMMUNOHISTOCHEMISTRY METHODOLOGY:
All 128 samples of liver tissue were included for immunohistochemical determination of the expression of CD11c+ cells (considered in this study as hDCs, given the fact that they are the most widely used marker for hDC identification). The samples underwent the steps that are described below in the order in which these took place.
INITIAL SAMPLE PROCESSING:
Accordingly, the 3-μm-thick formalin buffered-fixed, paraffin-embedded liver tissue slices from Tru-Cut biopsies, laparoscopic biopsies, and partial hepatectomies were deparaffinated and rehydrated.
CONTROL (IMMUNOHISTOCHEMISTRY METHOD):
Human tonsil tissue was used as control for the immunoohistochemical marking, as suggested by BioSV (provider of the antibodies) in the catalog. Other tissues suggested to be used as control were: bone marrow, spleen, colon, and hairy cell leukemia. Human tonsil tissue samples (obtained from tonsillectomies carried out in the surgical department and preserved in the pathology department in Medica Sur Hospital) were cut 3-μm-thick and placed in each of the slides where the liver biopsies were placed for staining. The same IHC process carried out in the liver biopsies was done simultaneously on the tonsil control tissue.
METHOD OF ANTIGEN RETRIEVAL:
The antigenic recovery process was carried out using pressure-boiled Declere 1: 20 (Cell Marque, Hot Springs, AR) in a Microwave Tender Cooker (2.5 Quarts, NordicWare®); subsequently, we carried out the unspecified-site blockade (3% H2O2 for 5 min). Having finished this process, the material was cooled at room temperature for 30 min and water-washed.
IMMUNOHISTOCHEMICAL MARKERS (PRIMARY ANTIBODY):
Next, after buffering with TBS (DakoCytomation, Carpinteria, CA), the samples were treated for 1 h with the antibody (Rabbit Monoclonal Anti-CD11c IgG, BSB 6445, clone: EP 157 by BioSB®), at a dilution of 1: 100. Previously, the antibody was titrated on the fabricant’s recommended tissue (tonsil tissue). Diaminobenzidine (Catalog number BSB 0005, BioSB®) was employed as chromogen. No secondary antibodies were used for this process.
IMMUNOSTAINING EVALUATION: Lastly, the slices were presented on permanent resin and protected with coverslips for microscopic analysis by pathologists. For evaluation of immune marking, light microscopy was used (Olympus Nikon E100®). As assessed by the expert pathologist, any brown membrane staining on any field within the biopsy was considered as “positive” staining, as shown in Figure 2 (both A and B would represent positive staining).
SUB-ANALYSIS METHODOLOGY: HDCS’ EXPRESSION ACROSS DISTINCT DTAGES OF MAFLD:
We carried out a sub-analysis in 35 random liver biopsies to perform this sub-analysis based on routine hematoxylin and eosin, as well as trichrome Masson stains, to determine liver steatosis, inflammation, ballooning degeneration, and fibrosis. The operational definitions for these histopathological terms were based on standardized pathology outlines and are shown in Table 1. Furthermore, all samples were blindly reviewed by 1 expert pathologist, who semi-quantitatively stratified the expression of hDCs (CD11c+) as focal (sparse number of hDCs in less than 3 fields/40× [Figure 2A]) or diffuse (high number of hDCs were observed in more than 3/fields/40× [Figure 2B]).
STATISTICAL ANALYSIS:
Baseline characteristics of participants according to BMI are presented as number and percentage for categorical variables. The uncorrected χ2 test for categorical variables was used for statistical inference. We considered significant differences as those with a 2-sided
Results
OVERALL ANALYSIS OF 128 PATIENTS, INDEPENDENT OF MAFLD STAGE:
The most relevant characteristics of the 128 included patients with MAFLD are summarized in Table 2.
The main characteristics of the gathered data include 49 (64.5%) patients who positively expressed hDCs (CD11c+) in liver tissue.
Of the 33 patients with obesity GI–II, 18 (54.5%) positively expressed hDCs (CD11c+) in liver tissue, while from the 19 non-obese patients, a total of 5 (26.3%) positively expressed hDCs (Table 3).
Importantly, from the 72 liver biopsies positively expressing hDCs (CD11c+), 49 (68.1%) were from morbidly obese individuals, 18 (25.0%) from patients with obesity GI–II, and 5 (6.9%) from non-obese individuals; out of these, 33 liver samples (45.8%) had fibrosis (Table 3). Furthermore, 20 (27.8%) were morbidly obese with fibrosis, 10 (13.9%) were patients with obesity GI–II and fibrosis, and 3 (4.2%) were non-obese with fibrosis (Table 3).
Given the gathered data, it was found that the odds ratio (OR) for hDCs (CD11c+) expression in liver tissue was 1.51 (0.65–3.49; χ2=0.957; P=0.327) for morbid obese patients compared with patients with obesity GI–II, and it was 5.08 (1.65–15.63; χ2=9.021; P=0.0027) when comparing the morbidly obese with non-obese patients, showing a significant difference in dendritic cell expression, especially among morbidly obese individuals and non-obese patients. The difference was lower when comparing patients with obesity GI–II and non-obese patients, with OR for hDCs (CD11c+) expression in liver tissue of 3.36 (0.98–11.49; χ2=3.895; P=0.048) (Table 4).
SUBGROUP PARTICIPANTS’ CHARACTERISTICS: From the 35 patients with MAFLD included in this sub-analysis, 18 (51.4%) were men and 17 (48.6%) were women; 2 (5.7%) were morbidly obese, 14 (40%) had obesity GI–II, and 19 (54.3%) were non-obese; additionally, 30 (85.7%) focally expressed hDCs (CD11c+) and 5 (14.3%) diffusely expressed hDCs (CD11c+) (Table 5).
:
The 2 morbidly obese patients (100%) in the 35-sample analysis showed inflammation and fibrosis, but only 1 (50%) had ballooning degeneration. From the 14 (40%) patients with obesity GI–II, 12 (85.7%) had inflammation, 10 (71.4%) had fibrosis, and 5 (35.7%) had ballooning degeneration. From the 19 (54.3%) non-obese patients, 13 (68.4%) had inflammation, 12 (63.2%) had fibrosis, and 7 (36.8%) had ballooning degeneration; all 5 (26.3%) patients who diffusely expressed hDCs (CD11c+) in the liver were non-obese.
DIFFUSE HDC EXPRESSION ACCORDING TO MAFLD STAGE: From the 5 patients with simple steatosis (14.3%), 1 (20%) diffusely expressed hDCs (CD11c+) on liver tissue. In addition, out of the 27 patients with inflammation (77.1%) and the 13 with ballooning degeneration (37.1%), 4 (14.8%) and 2 (15.4%) patients diffusely expressed hDCs (CD11c+) on liver tissue, respectively. Finally, among the patients with fibrosis (24, 68.6%), 3 (12.5%) of them diffusely expressed hDCs (CD11c+) on liver tissue (Table 5).
FOCAL HDC EXPRESSION ACCORDING TO MAFLD STAGE:
Out of the same number of patients with simple steatosis, inflammation, ballooning degeneration, and fibrosis, focal expression of dendritic cells was present in 4 (80%), 23 (85.2%), 11 (84.6%), 13 (37.1%), and 21 (87.5%), respectively.
Discussion
STRENGTHS AND LIMITATIONS:
An important limitation of this study is that it was not a longitudinal design and, thus, results might not represent high-strength evidence for causality. Secondly, flow cytometry was not available for this study; therefore, single-cell analysis was not included. Thirdly, even though the use of different cell surface markers (eg, lin−, HLA−DR+, CD1c+, CD163+) to characterize the differentiation of hDCs from other innate immune cells has been thoroughly described, we used only the most commonly expressed marker (CD11c) in an attempt to describe hDCs. Given the lack of information on the role of these cells on MAFLD pathogenesis, the use of this wide and sensitive cell surface marker gives us a wider perspective on the role of these cells and opens assumptions regarding involvement of other immune cells which have not been described yet, questioning whether it is an actual limitation or not. Regarding methodology and analysis, one of the limitations consisted in having a single expert pathologist evaluating the immune-stained liver biopsies. In this case, blind analysis by multiple pathologists would have increased the study’s impact.
The main strength of this study is that it is the first to assess MAFLD variation among different phenotypes (according to BMI), including patients with different grades of obesity (n=109) and lean/overweight patients (n=19). It is also the first study in humans describing the expression of hDCs (CD11c+) in the liver of patients with MAFLD, with the previously mentioned stratification according to their BMI. Importantly, showing the 3-variable relations among the expression of hDCs, BMI, and MAFLD opens up discussion regarding a potential role of the innate immune system in humans, leading to future studies analyzing specific molecular mechanisms and possible variations according to BMI.
Conclusions
In this study, hDCs (CD11c+ cells) were found to be higher in liver tissues from MAFLD patients with higher BMI, especially those with morbid obesity. However, diagnosis of fibrosis in MAFLD patients did not increase the probability of expressing hDCs (CD11c+ cells). Despite the fact that hDC expression in MAFLD patients varies depending on BMI, we showed that hDC expression (CD11c+) was also increased in liver tissue from obese MAFLD patients with fibrosis. Thus, obesity might be directly associated with the increased expression of hDCs (CD11c+) and this association remains unchanged despite the presence of fibrosis. Future research for characterization of this relationship, including additional and specific hDCs markers, morbid obesity-specific histological assessment scores, determination of the source of immune effector cells, and standardized clinical measurements, is encouraged. In conclusion, hDC expression is significantly higher in morbidly obese patients with MAFLD compared with non-obese patients, independent of the degree of fibrosis, suggesting the role of adaptive changes within hDCs in the perpetuation of inflammatory insults in chronic liver diseases.
Figures
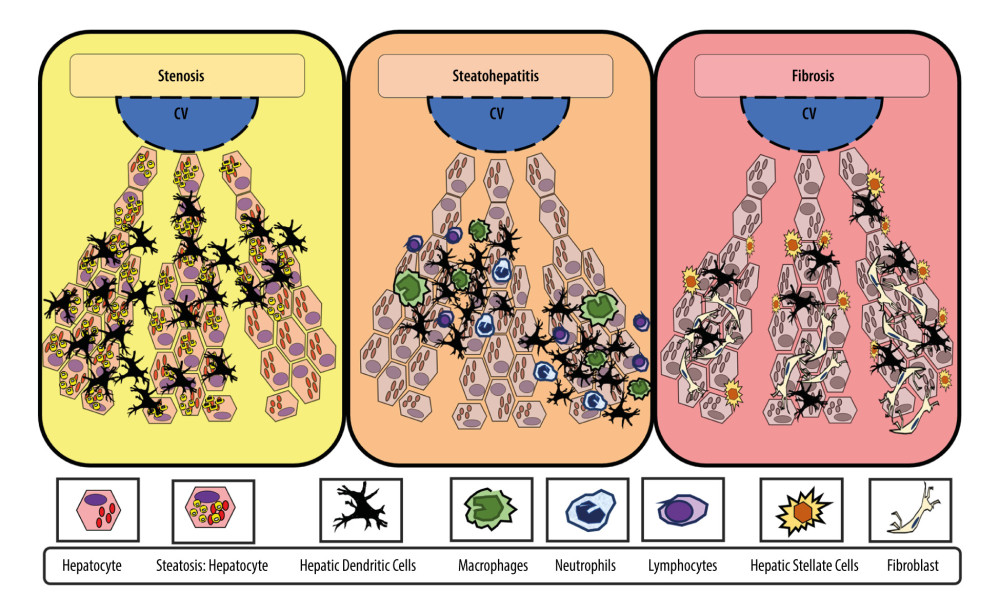 Figure 1. Adaptive changes of hepatic dendritic cells (CD11c+, hDCs) along the metabolic dysfunction-associated fatty liver disease (MAFLD) spectrum of diseasehDCs have an important role in capturing, processing, and presenting antigens. Initially, during MAFLD, when steatosis is stablished, hDCs tend to promote lipid hepatocytes storage with promotion of a tolerant and protective environment against fibro-inflammatory process. In contrast, during inflammation, the hDCs pool changes (eg, reduced lymphocytoid and plasmacytoid cells), with further expansion of matured hDCs and other innate immune cells (eg, neutrophils, macrophages) that promote and maintain fibro-inflammatory liver damage in advanced disease. In inflammation, fibrosis, and severe obesity, hDCs (CD11c+) appeared to be reduced or focally expressed, thus representing the tolerogenic and protective phenotype. Finally, during fibrosis, the hDCs (CD11c+) increased cytokine production induced a proinflammatory environment and proliferative responses of hepatic stellate cells (HSCs). Thus, focal expression of hDCs may stimulate HSCs, while diffuse expression of hDCs (CD11c+) in early stages might be characteristic of protective and tolerogenic cells (CD11c+).
Figure 1. Adaptive changes of hepatic dendritic cells (CD11c+, hDCs) along the metabolic dysfunction-associated fatty liver disease (MAFLD) spectrum of diseasehDCs have an important role in capturing, processing, and presenting antigens. Initially, during MAFLD, when steatosis is stablished, hDCs tend to promote lipid hepatocytes storage with promotion of a tolerant and protective environment against fibro-inflammatory process. In contrast, during inflammation, the hDCs pool changes (eg, reduced lymphocytoid and plasmacytoid cells), with further expansion of matured hDCs and other innate immune cells (eg, neutrophils, macrophages) that promote and maintain fibro-inflammatory liver damage in advanced disease. In inflammation, fibrosis, and severe obesity, hDCs (CD11c+) appeared to be reduced or focally expressed, thus representing the tolerogenic and protective phenotype. Finally, during fibrosis, the hDCs (CD11c+) increased cytokine production induced a proinflammatory environment and proliferative responses of hepatic stellate cells (HSCs). Thus, focal expression of hDCs may stimulate HSCs, while diffuse expression of hDCs (CD11c+) in early stages might be characteristic of protective and tolerogenic cells (CD11c+). 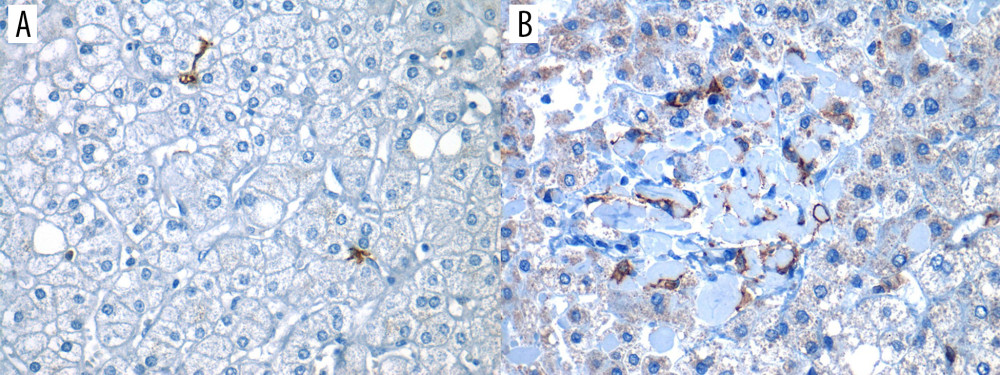 Figure 2. Positive expression of hepatic dendritic cells (CD11c+, hDCs) in liver tissue (40×)Photomicrograph showing CD11c+ immunostained cells in 2 different liver biopsies, visualized in 40× objective. (A) Focal expression: low number of hDCs (CD11c+) in less than 3 fields/40×. (B) Diffuse expression: high number of hDCs (CD11c+) are found in more than 3 fields/40×.
Figure 2. Positive expression of hepatic dendritic cells (CD11c+, hDCs) in liver tissue (40×)Photomicrograph showing CD11c+ immunostained cells in 2 different liver biopsies, visualized in 40× objective. (A) Focal expression: low number of hDCs (CD11c+) in less than 3 fields/40×. (B) Diffuse expression: high number of hDCs (CD11c+) are found in more than 3 fields/40×. 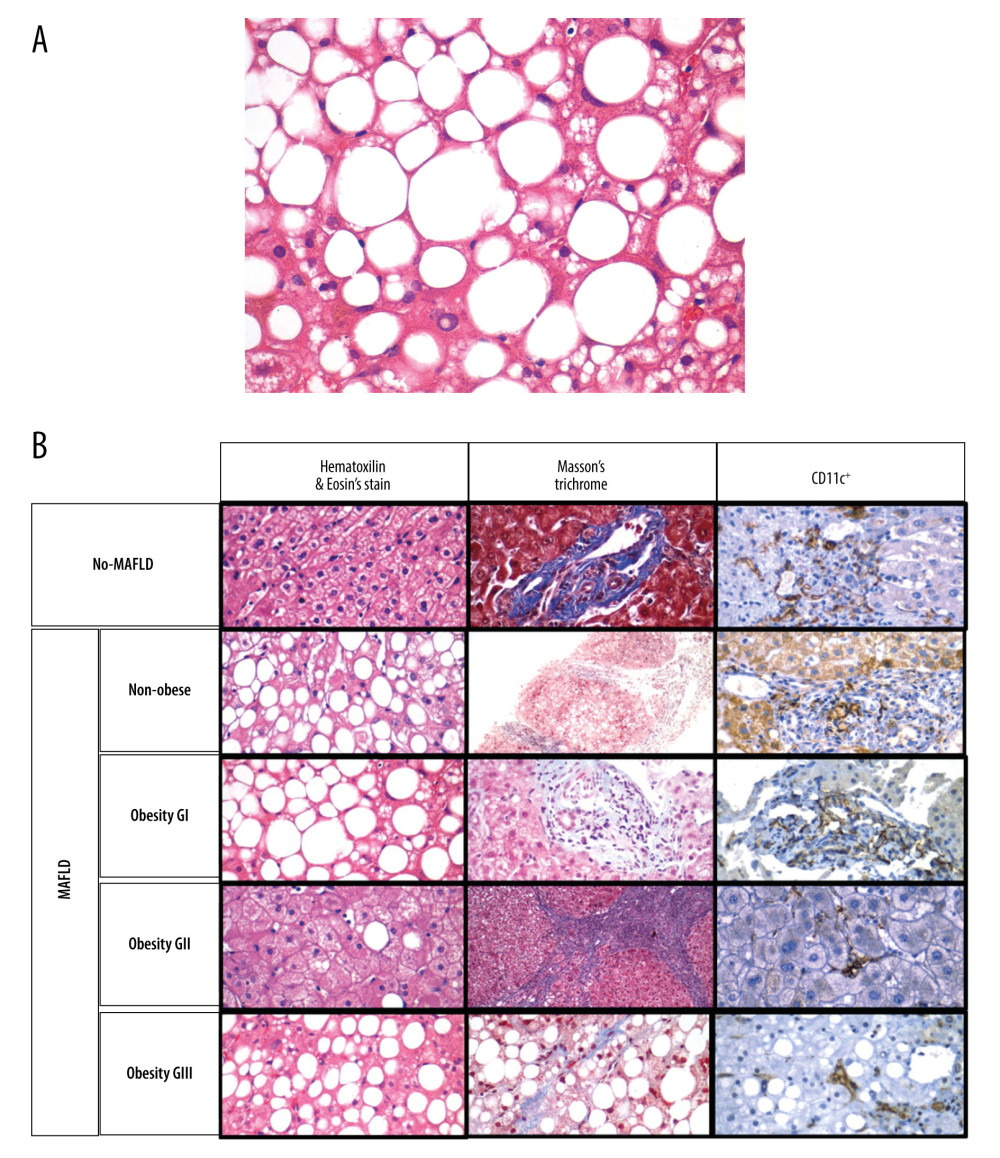 Figure 3. (A) A photomicrograph of the liver biopsy from a 54-year-old woman with metabolic dysfunction-associated fatty liver disease (MAFLD)The histology of the liver biopsy shows steatosis with round vacuoles in the liver cells where lipid has been removed during tissue processing. Note the lack of inflammation or fibrosis in this biopsy, although there may be areas of increased mononuclear cells. No necrosis is seen. Hematoxylin and eosin (H&E). Magnification ×40. (B) Comparison of liver biopsy (stained by hematoxylin and eosin, and Masson’s trichrome stain, as well as CD11c+ expression) from individuals with no metabolic dysfunction-associated fatty liver disease (MAFLD) and from patients with MAFLD in different categories according to body mass index (BMI). Based on different stains, it is possible to see the expression of dendritic cells (last row, as brown colored) varies in number and in distribution. Furthermore, the histological differences on the degree of lipid droplet saturation as well as fibrosis (with both hematoxylin and eosin, as with Masson’s stain) can be observed.
Figure 3. (A) A photomicrograph of the liver biopsy from a 54-year-old woman with metabolic dysfunction-associated fatty liver disease (MAFLD)The histology of the liver biopsy shows steatosis with round vacuoles in the liver cells where lipid has been removed during tissue processing. Note the lack of inflammation or fibrosis in this biopsy, although there may be areas of increased mononuclear cells. No necrosis is seen. Hematoxylin and eosin (H&E). Magnification ×40. (B) Comparison of liver biopsy (stained by hematoxylin and eosin, and Masson’s trichrome stain, as well as CD11c+ expression) from individuals with no metabolic dysfunction-associated fatty liver disease (MAFLD) and from patients with MAFLD in different categories according to body mass index (BMI). Based on different stains, it is possible to see the expression of dendritic cells (last row, as brown colored) varies in number and in distribution. Furthermore, the histological differences on the degree of lipid droplet saturation as well as fibrosis (with both hematoxylin and eosin, as with Masson’s stain) can be observed. 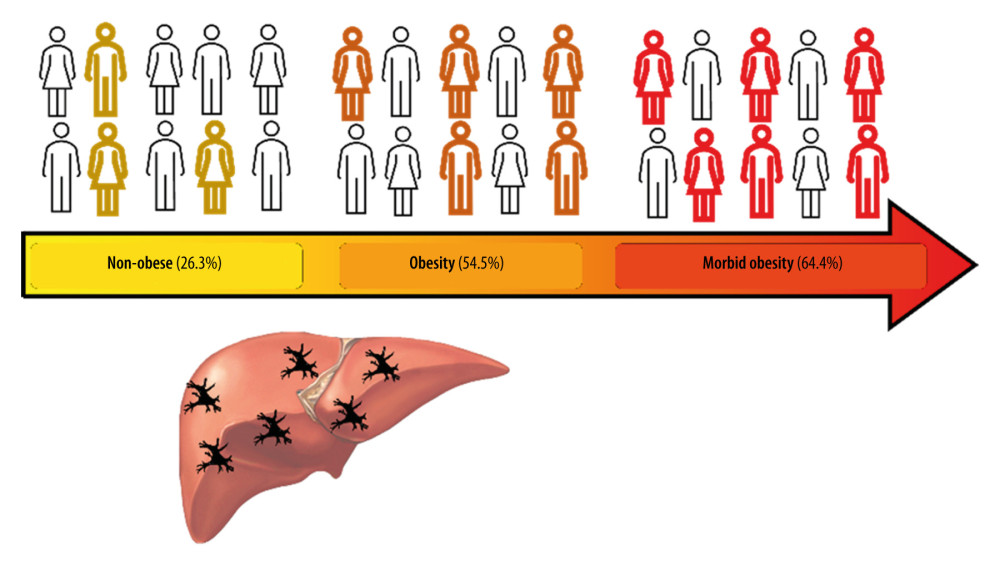 Figure 4. Increased hepatic dendritic cells (CD11c+, hDCs) expression in liver tissue according to patient’s body mass index (BMI)A graphic/schematic distribution of the liver expression of hDCs (CD11c+) according to patients’ BMI. Hematoxylin and Eosin (H), and Masson trichrome stains in liver biopsy form patients with different BMIs. Liver expression of hDCs (CD11c+) according to patients’ BMI.
Figure 4. Increased hepatic dendritic cells (CD11c+, hDCs) expression in liver tissue according to patient’s body mass index (BMI)A graphic/schematic distribution of the liver expression of hDCs (CD11c+) according to patients’ BMI. Hematoxylin and Eosin (H), and Masson trichrome stains in liver biopsy form patients with different BMIs. Liver expression of hDCs (CD11c+) according to patients’ BMI. Tables
Table 1. Operational definitions for histopathological diagnosis of liver injury:The histological diagnoses established by the expert pathologist are presented below. These are written-out here for reference to the terms used within the text. Table 2. Participant information database-main characteristics:The main characteristics of the patients enrolled in the study are presented below, with most being composed of women, morbidly obese individuals, and variable CD11c+ cell expression.
Table 2. Participant information database-main characteristics:The main characteristics of the patients enrolled in the study are presented below, with most being composed of women, morbidly obese individuals, and variable CD11c+ cell expression.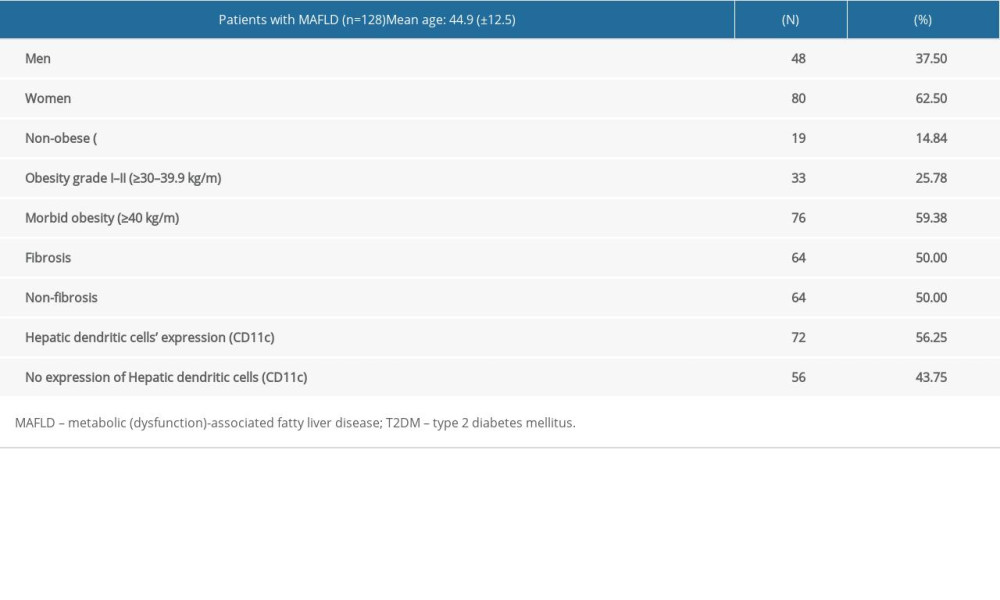 Table 3. Hepatic dendritic cells’ (CD11c+) expression in MAFLD according to patients’ BMI and either presence or absence of fibrosis:The findings of liver fibrosis among different weight groups was variable, finding that the most marked difference is dependent on the number of patients in each group (eg, non-obese patients constitute a lower percentage of the population, for which they constitute a lower percentage of patients with fibrosis, as well as without fibrosis).
Table 3. Hepatic dendritic cells’ (CD11c+) expression in MAFLD according to patients’ BMI and either presence or absence of fibrosis:The findings of liver fibrosis among different weight groups was variable, finding that the most marked difference is dependent on the number of patients in each group (eg, non-obese patients constitute a lower percentage of the population, for which they constitute a lower percentage of patients with fibrosis, as well as without fibrosis).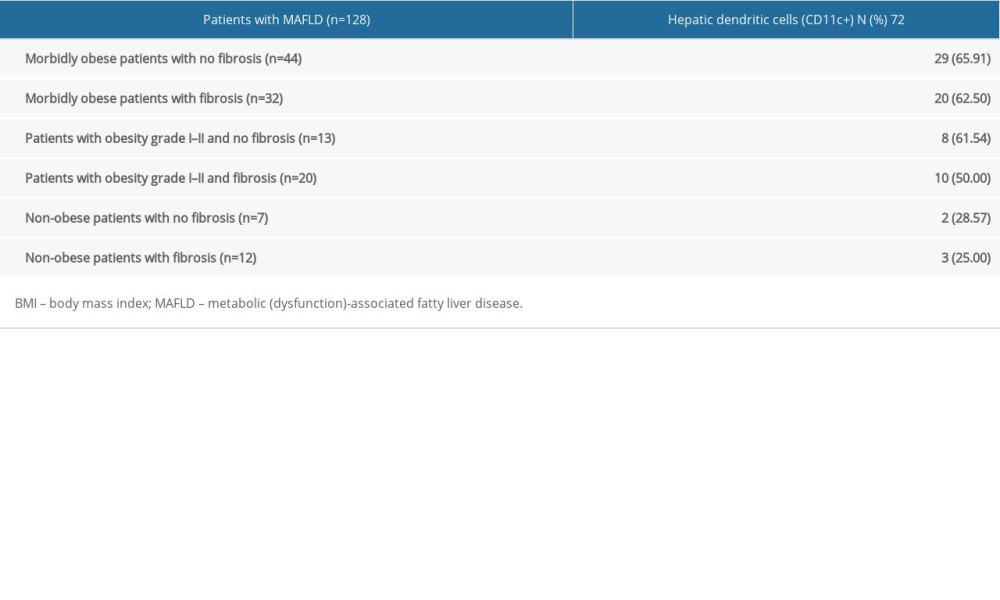 Table 4. Hepatic dendritic cells’ expression in MAFLD according to patients’ BMI and either the presence or absence of fibrosis:The data show the difference in dendritic cell expression among different weight groups; the most outstanding is the difference between CD11c+ cell expression in morbidly obese individuals compared to lower degrees in obese and non-obese individuals.
Table 4. Hepatic dendritic cells’ expression in MAFLD according to patients’ BMI and either the presence or absence of fibrosis:The data show the difference in dendritic cell expression among different weight groups; the most outstanding is the difference between CD11c+ cell expression in morbidly obese individuals compared to lower degrees in obese and non-obese individuals.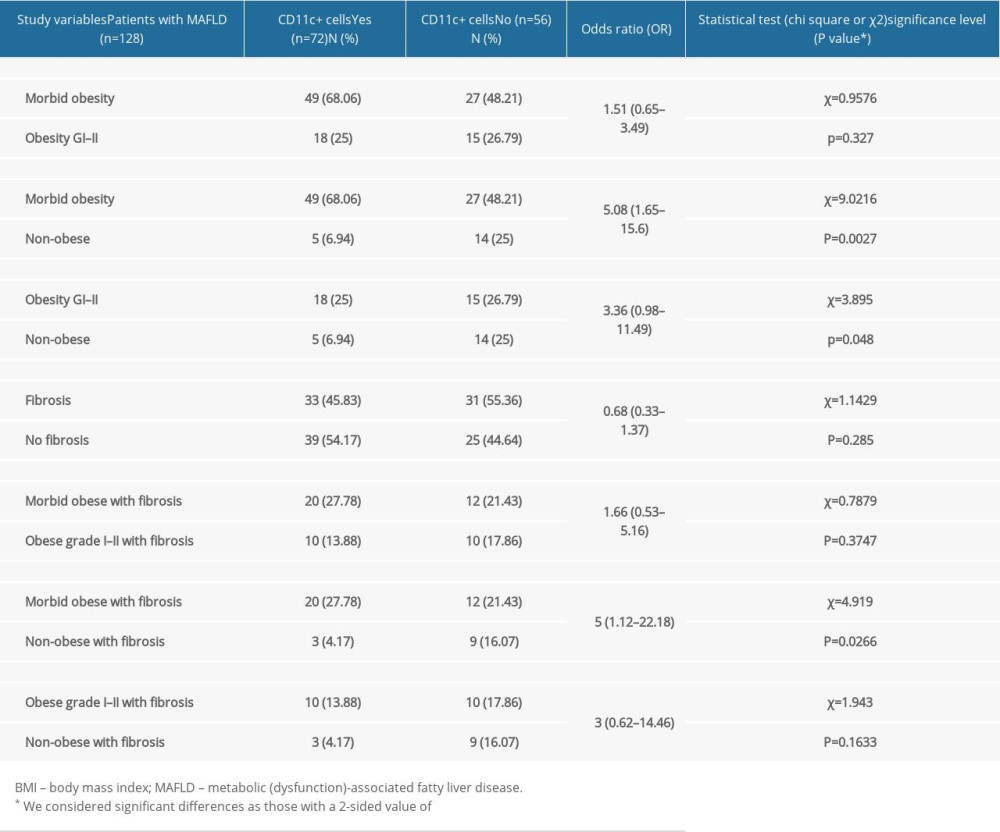 Table 5. Sub-analysis of focal and diffuse expression of hepatic dendritic cells (CD11c+) in MAFLD according to patients’ BMI and histologic stage of disease. Based on the data that the table presents, we can see how the totality of patients who present CD11c+ cell expression or any degree of liver damage also present with focal expression of these cells.
Table 5. Sub-analysis of focal and diffuse expression of hepatic dendritic cells (CD11c+) in MAFLD according to patients’ BMI and histologic stage of disease. Based on the data that the table presents, we can see how the totality of patients who present CD11c+ cell expression or any degree of liver damage also present with focal expression of these cells.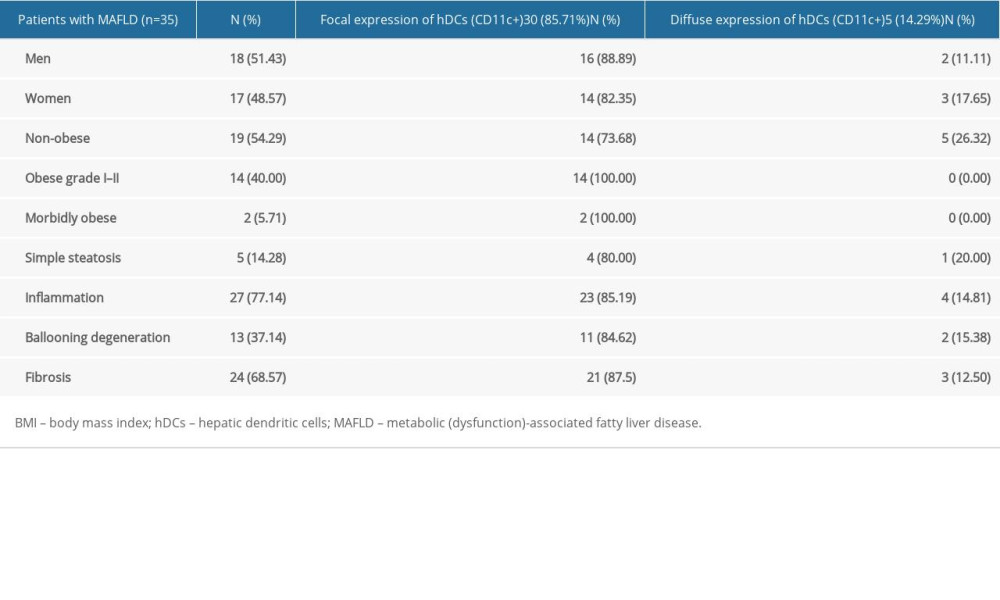
References
1. Younossi ZM, Koenig AB, Abdelatif D, Global epidemiology of nonalcoholic fatty liver disease – meta-analytic assessment of prevalence, incidence, and outcomes: Hepatology, 2016; 64(1); 73-84
2. Estes C, Razavi H, Loomba R, Younossi Z, Sanyal AJ, Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease: Hepatology, 2018; 67(1); 123-33
3. Di Cesare M, Bentham J, Stevens GA, Trends in adult body-mass index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19.2 million participants: Lancet, 2016; 387(10026); 1377-96
4. Praveenraj P, Gomes RM, Kumar S, Prevalence and predictors of non-alcoholic fatty liver disease in morbidly obese South Indian patients undergoing bariatric surgery: Obes Surg, 2015; 25(11); 2078-87
5. Ong JP, Elariny H, Collantes R, Predictors of nonalcoholic steatohepatitis and advanced fibrosis in morbidly obese patients: Obes Surg, 2005; 15(3); 310-15
6. Morita S, Neto DDS, Morita FHA, Prevalence of non-alcoholic fatty liver disease and steatohepatitis risk factors in patients undergoing bariatric surgery: Obes Surg, 2015; 25(12); 2335-43
7. Qureshi K, Abrams GA, Prevalence of biopsy-proven non-alcoholic fatty liver disease in severely obese subjects without metabolic syndrome: Clin Obes, 2016; 6(2); 117-23
8. Lonardo A, Bellentani S, Argo CK, Epidemiological modifiers of non-alcoholic fatty liver disease: Focus on high-risk groups: Dig Liver Dis, 2015; 47(12); 997-1006
9. Mendez-Sanchez N, Cruz-Ramon VC, Ramirez-Perez OL, New aspects of lipotoxicity in nonalcoholic steatohepatitis: Int J Mol Sci, 2018; 19(7); 2034
10. Nguyen MTA, Favelyukis S, Nguyen AK, A subpopulation of macrophages infiltrates hypertrophic adipose tissue and is activated by free fatty acids via toll-like receptors 2 and 4 and JNK-dependent pathways: J Biol Chem, 2007; 282(48); 35279-92
11. Méndez-Sánchez N, Córdova-Gallardo J, Barranco-Fragoso B, Eslam M, Hepatic dendritic cells in the development and progression of metabolic steatohepatitis: Front Immunol, 2021; 12(March); 1-11
12. Henning JR, Graffeo CS, Rehman A, Dendritic cells limit fibroinflammatory injury in nonalcoholic steatohepatitis in mice: Hepatology, 2013; 58(2); 589-602
13. Jiao J, Sastre D, Fiel MI, Dendritic cell regulation of carbon tetrachloride-induced murine liver fibrosis regression: Hepatology, 2012; 55(1); 244-55
14. Aloman C, Tacke F, Dendritic cells in liver fibrosis: Conductor of the inflammatory orchestra?: Hepatology, 2010; 5(1); 705-11
15. Deczkowska A, David E, Ramadori P, XCR1+ type 1 conventional dendritic cells drive liver pathology in non-alcoholic steatohepatitis: Nat Med, 2021; 27(6); 1043-54
16. Heier EC, Meier A, Julich-Haertel H, Murine CD103+ dendritic cells protect against steatosis progression towards steatohepatitis: J Hepatol, 2017; 66(6); 1241-50
17. Aarts S, Reiche M, den Toom M, Depletion of CD40 on CD11c+ cells worsens the metabolic syndrome and ameliorates hepatic inflammation during NASH: Sci Rep, 2019; 9(1); 1-11
18. Lukacs-Kornek V, Schuppan D, Dendritic cells in liver injury and fibrosis: Shortcomings and promises: J Hepatol, 2013; 59(5); 1124-26
19. Bernsmeier C, Albano E, Liver dendritic cells and NAFLD evolution: A remaining open issue: J Hepatol, 2017; 66(6); 1120-22
20. Eslam M, Newsome PN, Sarin SK, A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement: J Hepatol, 2020; 73(1); 202-9
21. Flynn MAT, McNeil DA, Maloff B, Reducing obesity and related chronic disease risk in children and youth: A synthesis of evidence with “best practice” recommendations: Obses Rev, 2006; 7(Suppl 1); 7-66
22. Nakajima S, Koh V, Kua L-F, Accumulation of CD11c + CD163 + adipose tissue macrophages through upregulation of intracellular 11β-HSD1 in human obesity: J Immunol, 2016; 197(9); 3735-45
23. Wentworth JM, Naselli G, Brown WA, Pro-inflammatory CD11c+CD206+ adipose tissue macrophages are associated with insulin resistance in human obesity: Diabetes, 2010; 59(7); 1648-56
24. Kochumon S, Al Madhoun A, Al-Rashed F, Elevated adipose tissue associated IL-2 expression in obesity correlates with metabolic inflammation and insulin resistance: Sci Rep, 2020; 10(1); 1-13
25. Engin A, Human protein kinases and obesity: Adv Exp Med Biol, 2017; 960; 111-34
26. Cho KW, Zamarron BF, Muir LA, Adipose tissue dendritic cells are independent contributors to obesity-induced inflammation and insulin resistance: J Immunol, 2016; 197(9); 3650-61
27. Fuchs A, Samovski D, Smith GI, Associations among adipose tissue immunology, inflammation, exosomes and insulin sensitivity in people with obesity and nonalcoholic fatty liver disease: Gastroenterology, 2021; 161(3); 968-81 e12
28. Stefanovic-Racic M, Yang X, Turner MS, Dendritic cells promote macrophage infiltration and comprise a substantial proportion of obesity-associated increases in CD11c+ cells in adipose tissue and liver: Diabetes, 2012; 61(9); 2330-39
Figures
 Figure 1. Adaptive changes of hepatic dendritic cells (CD11c+, hDCs) along the metabolic dysfunction-associated fatty liver disease (MAFLD) spectrum of diseasehDCs have an important role in capturing, processing, and presenting antigens. Initially, during MAFLD, when steatosis is stablished, hDCs tend to promote lipid hepatocytes storage with promotion of a tolerant and protective environment against fibro-inflammatory process. In contrast, during inflammation, the hDCs pool changes (eg, reduced lymphocytoid and plasmacytoid cells), with further expansion of matured hDCs and other innate immune cells (eg, neutrophils, macrophages) that promote and maintain fibro-inflammatory liver damage in advanced disease. In inflammation, fibrosis, and severe obesity, hDCs (CD11c+) appeared to be reduced or focally expressed, thus representing the tolerogenic and protective phenotype. Finally, during fibrosis, the hDCs (CD11c+) increased cytokine production induced a proinflammatory environment and proliferative responses of hepatic stellate cells (HSCs). Thus, focal expression of hDCs may stimulate HSCs, while diffuse expression of hDCs (CD11c+) in early stages might be characteristic of protective and tolerogenic cells (CD11c+).
Figure 1. Adaptive changes of hepatic dendritic cells (CD11c+, hDCs) along the metabolic dysfunction-associated fatty liver disease (MAFLD) spectrum of diseasehDCs have an important role in capturing, processing, and presenting antigens. Initially, during MAFLD, when steatosis is stablished, hDCs tend to promote lipid hepatocytes storage with promotion of a tolerant and protective environment against fibro-inflammatory process. In contrast, during inflammation, the hDCs pool changes (eg, reduced lymphocytoid and plasmacytoid cells), with further expansion of matured hDCs and other innate immune cells (eg, neutrophils, macrophages) that promote and maintain fibro-inflammatory liver damage in advanced disease. In inflammation, fibrosis, and severe obesity, hDCs (CD11c+) appeared to be reduced or focally expressed, thus representing the tolerogenic and protective phenotype. Finally, during fibrosis, the hDCs (CD11c+) increased cytokine production induced a proinflammatory environment and proliferative responses of hepatic stellate cells (HSCs). Thus, focal expression of hDCs may stimulate HSCs, while diffuse expression of hDCs (CD11c+) in early stages might be characteristic of protective and tolerogenic cells (CD11c+). Figure 2. Positive expression of hepatic dendritic cells (CD11c+, hDCs) in liver tissue (40×)Photomicrograph showing CD11c+ immunostained cells in 2 different liver biopsies, visualized in 40× objective. (A) Focal expression: low number of hDCs (CD11c+) in less than 3 fields/40×. (B) Diffuse expression: high number of hDCs (CD11c+) are found in more than 3 fields/40×.
Figure 2. Positive expression of hepatic dendritic cells (CD11c+, hDCs) in liver tissue (40×)Photomicrograph showing CD11c+ immunostained cells in 2 different liver biopsies, visualized in 40× objective. (A) Focal expression: low number of hDCs (CD11c+) in less than 3 fields/40×. (B) Diffuse expression: high number of hDCs (CD11c+) are found in more than 3 fields/40×. Figure 3. (A) A photomicrograph of the liver biopsy from a 54-year-old woman with metabolic dysfunction-associated fatty liver disease (MAFLD)The histology of the liver biopsy shows steatosis with round vacuoles in the liver cells where lipid has been removed during tissue processing. Note the lack of inflammation or fibrosis in this biopsy, although there may be areas of increased mononuclear cells. No necrosis is seen. Hematoxylin and eosin (H&E). Magnification ×40. (B) Comparison of liver biopsy (stained by hematoxylin and eosin, and Masson’s trichrome stain, as well as CD11c+ expression) from individuals with no metabolic dysfunction-associated fatty liver disease (MAFLD) and from patients with MAFLD in different categories according to body mass index (BMI). Based on different stains, it is possible to see the expression of dendritic cells (last row, as brown colored) varies in number and in distribution. Furthermore, the histological differences on the degree of lipid droplet saturation as well as fibrosis (with both hematoxylin and eosin, as with Masson’s stain) can be observed.
Figure 3. (A) A photomicrograph of the liver biopsy from a 54-year-old woman with metabolic dysfunction-associated fatty liver disease (MAFLD)The histology of the liver biopsy shows steatosis with round vacuoles in the liver cells where lipid has been removed during tissue processing. Note the lack of inflammation or fibrosis in this biopsy, although there may be areas of increased mononuclear cells. No necrosis is seen. Hematoxylin and eosin (H&E). Magnification ×40. (B) Comparison of liver biopsy (stained by hematoxylin and eosin, and Masson’s trichrome stain, as well as CD11c+ expression) from individuals with no metabolic dysfunction-associated fatty liver disease (MAFLD) and from patients with MAFLD in different categories according to body mass index (BMI). Based on different stains, it is possible to see the expression of dendritic cells (last row, as brown colored) varies in number and in distribution. Furthermore, the histological differences on the degree of lipid droplet saturation as well as fibrosis (with both hematoxylin and eosin, as with Masson’s stain) can be observed. Figure 4. Increased hepatic dendritic cells (CD11c+, hDCs) expression in liver tissue according to patient’s body mass index (BMI)A graphic/schematic distribution of the liver expression of hDCs (CD11c+) according to patients’ BMI. Hematoxylin and Eosin (H), and Masson trichrome stains in liver biopsy form patients with different BMIs. Liver expression of hDCs (CD11c+) according to patients’ BMI.
Figure 4. Increased hepatic dendritic cells (CD11c+, hDCs) expression in liver tissue according to patient’s body mass index (BMI)A graphic/schematic distribution of the liver expression of hDCs (CD11c+) according to patients’ BMI. Hematoxylin and Eosin (H), and Masson trichrome stains in liver biopsy form patients with different BMIs. Liver expression of hDCs (CD11c+) according to patients’ BMI. Tables
 Table 1. Operational definitions for histopathological diagnosis of liver injury:The histological diagnoses established by the expert pathologist are presented below. These are written-out here for reference to the terms used within the text.
Table 1. Operational definitions for histopathological diagnosis of liver injury:The histological diagnoses established by the expert pathologist are presented below. These are written-out here for reference to the terms used within the text. Table 2. Participant information database-main characteristics:The main characteristics of the patients enrolled in the study are presented below, with most being composed of women, morbidly obese individuals, and variable CD11c+ cell expression.
Table 2. Participant information database-main characteristics:The main characteristics of the patients enrolled in the study are presented below, with most being composed of women, morbidly obese individuals, and variable CD11c+ cell expression. Table 3. Hepatic dendritic cells’ (CD11c+) expression in MAFLD according to patients’ BMI and either presence or absence of fibrosis:The findings of liver fibrosis among different weight groups was variable, finding that the most marked difference is dependent on the number of patients in each group (eg, non-obese patients constitute a lower percentage of the population, for which they constitute a lower percentage of patients with fibrosis, as well as without fibrosis).
Table 3. Hepatic dendritic cells’ (CD11c+) expression in MAFLD according to patients’ BMI and either presence or absence of fibrosis:The findings of liver fibrosis among different weight groups was variable, finding that the most marked difference is dependent on the number of patients in each group (eg, non-obese patients constitute a lower percentage of the population, for which they constitute a lower percentage of patients with fibrosis, as well as without fibrosis). Table 4. Hepatic dendritic cells’ expression in MAFLD according to patients’ BMI and either the presence or absence of fibrosis:The data show the difference in dendritic cell expression among different weight groups; the most outstanding is the difference between CD11c+ cell expression in morbidly obese individuals compared to lower degrees in obese and non-obese individuals.
Table 4. Hepatic dendritic cells’ expression in MAFLD according to patients’ BMI and either the presence or absence of fibrosis:The data show the difference in dendritic cell expression among different weight groups; the most outstanding is the difference between CD11c+ cell expression in morbidly obese individuals compared to lower degrees in obese and non-obese individuals. Table 5. Sub-analysis of focal and diffuse expression of hepatic dendritic cells (CD11c+) in MAFLD according to patients’ BMI and histologic stage of disease. Based on the data that the table presents, we can see how the totality of patients who present CD11c+ cell expression or any degree of liver damage also present with focal expression of these cells.
Table 5. Sub-analysis of focal and diffuse expression of hepatic dendritic cells (CD11c+) in MAFLD according to patients’ BMI and histologic stage of disease. Based on the data that the table presents, we can see how the totality of patients who present CD11c+ cell expression or any degree of liver damage also present with focal expression of these cells. Table 1. Operational definitions for histopathological diagnosis of liver injury:The histological diagnoses established by the expert pathologist are presented below. These are written-out here for reference to the terms used within the text.
Table 1. Operational definitions for histopathological diagnosis of liver injury:The histological diagnoses established by the expert pathologist are presented below. These are written-out here for reference to the terms used within the text. Table 2. Participant information database-main characteristics:The main characteristics of the patients enrolled in the study are presented below, with most being composed of women, morbidly obese individuals, and variable CD11c+ cell expression.
Table 2. Participant information database-main characteristics:The main characteristics of the patients enrolled in the study are presented below, with most being composed of women, morbidly obese individuals, and variable CD11c+ cell expression. Table 3. Hepatic dendritic cells’ (CD11c+) expression in MAFLD according to patients’ BMI and either presence or absence of fibrosis:The findings of liver fibrosis among different weight groups was variable, finding that the most marked difference is dependent on the number of patients in each group (eg, non-obese patients constitute a lower percentage of the population, for which they constitute a lower percentage of patients with fibrosis, as well as without fibrosis).
Table 3. Hepatic dendritic cells’ (CD11c+) expression in MAFLD according to patients’ BMI and either presence or absence of fibrosis:The findings of liver fibrosis among different weight groups was variable, finding that the most marked difference is dependent on the number of patients in each group (eg, non-obese patients constitute a lower percentage of the population, for which they constitute a lower percentage of patients with fibrosis, as well as without fibrosis). Table 4. Hepatic dendritic cells’ expression in MAFLD according to patients’ BMI and either the presence or absence of fibrosis:The data show the difference in dendritic cell expression among different weight groups; the most outstanding is the difference between CD11c+ cell expression in morbidly obese individuals compared to lower degrees in obese and non-obese individuals.
Table 4. Hepatic dendritic cells’ expression in MAFLD according to patients’ BMI and either the presence or absence of fibrosis:The data show the difference in dendritic cell expression among different weight groups; the most outstanding is the difference between CD11c+ cell expression in morbidly obese individuals compared to lower degrees in obese and non-obese individuals. Table 5. Sub-analysis of focal and diffuse expression of hepatic dendritic cells (CD11c+) in MAFLD according to patients’ BMI and histologic stage of disease. Based on the data that the table presents, we can see how the totality of patients who present CD11c+ cell expression or any degree of liver damage also present with focal expression of these cells.
Table 5. Sub-analysis of focal and diffuse expression of hepatic dendritic cells (CD11c+) in MAFLD according to patients’ BMI and histologic stage of disease. Based on the data that the table presents, we can see how the totality of patients who present CD11c+ cell expression or any degree of liver damage also present with focal expression of these cells. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








