14 October 2022: Clinical Research
Correlation Between Trough Level of Abiraterone and Prostate-Specific Antigen (PSA) Response in Metastatic Hormone-Sensitive Prostate Cancer
Zin W. Myint12ABCDEFG*, Jill M. Kolesar23CDE, Joseph Robert McCorkle2CDE, Jianrong Wu4C, Carleton S. Ellis32CDE, Danielle E. Otto23CDE, Peng Wang12ACDEDOI: 10.12659/MSM.938091
Med Sci Monit 2022; 28:e938091
Abstract
BACKGROUND: Prostate cancer growth is primarily driven by testosterone and 5a-dihydrotestosterone. Abiraterone is an irreversible inhibitor of CYP17, and CYP17 inhibition is a required step in testosterone biosynthesis. Previous studies have shown that abiraterone trough levels are predictive of prostate-specific antigen (PSA) response in metastatic castrate-resistant prostate cancer (mCRPC). It has not been demonstrated if this association exists for patients with metastatic hormone-sensitive prostate cancer (mHSPC). In this study, we aimed to explore the correlation and association between abiraterone trough levels and PSA levels in patients with mHSPC.
MATERIAL AND METHODS: This was a single-center, prospective, observational study of patients with mHSPC being treated with abiraterone acetate (AA) 1000 mg once daily. Abiraterone trough levels (22-26 h after drug administration) were drawn at 1, 3, and 7 months after treatment initiation.
RESULTS: Thirteen patients with mHSPC were enrolled, and complete pharmacokinetic data were available for 8 patients. The mean trough levels at 1 month, 3 months, and 7 months were 34.49 ng/mL (3.36-240.46), 13.82 ng/mL (2.91-29.96), and 15.7 ng/mL (3.58-26.86), respectively. The correlation between the 1-month abiraterone trough level and 1-month PSA level was 0.29 (P=0.38), between 3-month abiraterone trough and 3-month PSA was -0.61 (P=0.08), and between 7-month abiraterone trough and 7-month PSA was -0.31 (P=0.54).
CONCLUSIONS: This study demonstrated a trend toward a negative correlation between 3-month abiraterone trough levels and PSA levels, but the correlation was not statistically significant. A study with a larger prospective sample size is needed to validate these findings.
Keywords: Abiraterone Acetate, Prostate-Specific Antigen, Androstenes, dihydrotestosterone, Humans, Male, Prospective Studies, Prostatic Neoplasms, Castration-Resistant, Steroid 17-alpha-Hydroxylase, Treatment Outcome
Background
Prostate cancer cell growth is mainly controlled by testosterone and 5α-dihydrotestosterone, which are driven from the androgen receptor-signaling pathway (80–90%) and the adrenal steroidal pathway (10–20%) [1]. Abiraterone acetate (AA) in combination with androgen-deprivation therapy demonstrated survival benefit in men with metastatic hormone-sensitive prostate cancer (mHSPC) [2] and in men with metastatic castrate-resistant prostate cancer (mCRPC) prior to [3] or after docetaxel treatment [4]. AA, a prodrug of abiraterone, is metabolized by hydroxyl-delta-5-steroid dehydrogenase, 3 beta and steroid delta-isomerase 1 (
Previous studies have evaluated the plasma trough abiraterone level as a predictive factor for prostate-specific antigen (PSA) response in mCRPC. Carton et al performed a prospective observational study of the relationship between the abiraterone plasma trough concentration and 3-month PSA response (≥50% reduction) in mCRPC [8]. They found that there was a correlation with PSA response at 3 months if the abiraterone concentration was above 8.4 ng/mL. In those patients with levels higher than 8.4 ng/mL, a prolonged progression-free survival (PFS) was observed compared with that in patients whose abiraterone concentration was below 8.4 ng/mL (PFS 12.2 months vs 7.4 months, HR: 0.55) [8]. Stein et al also determined that maintenance of abiraterone trough levels above 8.4 ng/mL was consistent with sustained testosterone suppression to <50 ng/dL and PSA ≥50% reduction through day 84 [9]. This led us to question whether the correlation of plasma abiraterone trough levels above 8.4 ng/mL and attainment of PSA 0.2 ng/mL or less after treatment remain predictive in the setting of mHSPC.
Based on this rationale, in the present study, we aimed to explore the correlation and association between steady-state trough abiraterone plasma concentrations and 3-month and 7-month PSA levels in patients with mHSPC.
Material and Methods
STUDY POPULATION:
This was a single-center, prospective, observational study in patients with newly diagnosed mHSPC who were treated with AA and recruited from July 2019 to October 2021 in the Medical Oncology Clinic at University of Kentucky, Markey Cancer Center. Abiraterone trough levels were measured at 1 month, 3 months, and 7 months after starting AA for routine therapeutic drug monitoring. Trough levels were collected between 22 and 26 h after AA was taken. Eligible patients included men with mHSPC (either de novo or with failure after definitive localized prostate therapy), pathological diagnosis of prostate cancer, PSA elevation, radiologic evidence of metastatic disease, and an Eastern Cooperative Oncology Group (ECOG) performance status score of 0, 1, or 2. Prior adjuvant androgen-deprivation therapy was allowed if progression had occurred more than 12 months after completion of therapy. Patients were excluded if they were not eligible to take AA as their first-line therapy for any reason or if they did not meet the above inclusion criteria. Patients’ baseline characteristics (age, race, body mass index, documented metastases as either low volume vs high volume, PSA values, AA dosing, adverse effects, and clinical outcomes) were collected prospectively by reviewing electronic patient records. The definition of high volume was defined by the CHAARTED criteria: the presence of visceral metastases or ≥4 bone lesions with ≥1 beyond the vertebral bodies and pelvis [10].
TREATMENT:
Participating patients were treated with combined androgen-deprivation therapy and AA/prednisone. All patients were treated with AA at 1000 mg once daily on an empty stomach. A pharmacist checked the potential drug-drug interactions on all patients before initiation of AA. Medication diaries completed by the patients were reviewed by treating physicians for adherence. All adverse events were graded using the National Cancer Institute Common Toxicity Criteria version 6.0. PSA level was monitored at baseline, 1 month, 3 months, and 7 months. Radiologic evaluation was performed at 3 to 4 months after starting AA.
PHARMACOKINETIC ANALYSIS:
Plasma samples were obtained from enrolled patients for analysis of AA at 1, 3, and 7 months after the start of treatment. AA was quantified from EDTA-plasma using methods described by Alyamani et al [11], with modifications. Briefly, 100 μL plasma samples were spiked with 50 ng/mL abiraterone-d4 (internal standard) then combined with 400 μL methyl tertiary butyl ether and vortex-mixed for 5 min. Samples were centrifuged 5 min at 15 000×g, and 250 μL of the supernatant was transferred to a 96-well polypropylene plate and then evaporated to dryness at 40°C under a stream of nitrogen gas. Dried residues were reconstituted in a 100-μL 2: 1: 1 mixture (v: v: v) of 0.1% formic acid in water: acetonitrile: methanol. Abiraterone and abiraterone-d4 were separated on a 1260 Infinity II LC system (Agilent) with an InfinityLab Poroshell 120 HPH-C18 column (Agilent), 2.1×100 mm, 2.7 μm. Mobile phase A consisted of 0.1% formic acid in water, and mobile phase B was 1: 1 (v: v) acetonitrile: methanol. The flow rate was set to 0.4 mL/min and started with 0.5-min 50% mobile phase A, 50% mobile phase B, then the mobile phase B increased to 80% over the next 1 min, was maintained for additional 1.5 min, then returned back to 50% over the next 0.5 min. Abiraterone and abiraterone-d4 were quantified using an Agilent Ultivo triple quadrupole mass spectrometer operating in positive ion mode with an electrospray ionization source. Multiple reaction monitoring was used to quantify mass to transition ions of 350.2 to 156 for abiraterone and 354.3 to 160.1 for abiraterone-d4. The abiraterone standard curve was linear from 1 to 1000 ng/mL, with r2 >0.99.
:
Genomic DNA was extracted from 750 μL of whole blood samples for each enrolled patient using QIAamp DNA blood midi kits (Qiagen) according to the manufacturer’s instructions. DNA was eluted from columns using 200 μL AE buffer and stored at 4°C. TaqMan SNP genotyping assay (Applied Biosystems), C___8695674_10, was used to genotype 20 ng of genomic DNA at the
STUDY ENDPOINTS:
We examined (1) the correlation between steady-state trough abiraterone plasma concentrations and PSA levels in patients with mHSPC and (2) the association between abiraterone trough levels and 3-month PSA levels ≤1 ng/mL and 7-month PSA levels ≤0.2 ng/mL in patients with mHSPC. The language of correlation and association can be interchangeable; however, they are technically different. Correlation refers to 2 variables that are correlated in a linear relationship; whereas, association refers to a general relationship between 2 variables [12]. In addition, we measured the germline HSD3B1 (1245 C) allele at baseline and its association with clinical outcomes. We also analyzed the correlation between HSD3B1 and abiraterone trough concentrations.
STATISTICAL ANALYSIS:
Trough plasma concentrations of abiraterone were correlated with PSA levels by the Spearman correlation coefficient. Abiraterone levels were associated with 3-month PSA levels (categorized as PSA ≤1 ng/mL vs >1 ng/mL) and 7-month PSA levels (categorized as PSA ≤0.2 ng/mL vs >0.2 ng/mL) by logistic regression. Germline
Results
PATIENT BASELINE CHARACTERISTICS:
Thirteen patients were enrolled from July 2019 to October 2021. Among them, 8 patients had complete pharmacokinetic (PK) data, 4 patients did not complete the study due to disease progression, and 1 patient refused 1 PK blood draw. Patients with mHSPC who had at least 1 abiraterone plasma trough concentration level were included in this study. Patient demographic and baseline characteristics are listed in Table 1. The median age was 64 (56–83) years; 11 patients were White and 2 were African-American. Ten patients presented with de novo mHSPC, while 3 patients received prior local therapy (2 had a prostatectomy and 1 had prostate radiation) and later developed mHSPC. Ten patients had high-volume disease and 3 had low-volume disease, as defined per CHAARTED [10]. Most patients had an ECOG performance status of 0 or 1 and only 3 patients had an ECOG performance status of 2. The median PSA at diagnosis was 132 (57–4279). The median body mass index was 30.1 (20.7–38.7). At a median of 11 months (3–14) of follow-up, 9 patients were still alive; 1 patient had a complete response, 5 had a partial response, 3 had stable disease, and 4 had disease progression and died from prostate cancer. At the cut-off in November 2021, nine patients were still being treated with AA.
PHARMACOKINETICS:
There was high inter-patient variability in abiraterone trough levels; the mean abiraterone trough levels at 1 month, 3 months, and 7 months were 34.49 ng/mL (3.36–240.46), 13.82 ng/mL (2.91–29.96), and 15.7 ng/mL (3.58–26.86), respectively (Table 2). We removed patient 5 and patient 12 from Table 2 in the analyses due to extremely high trough levels (240 and 81.8 ng/mL) to avoid sampling error. The correlation between the 1-month abiraterone trough and 1-month PSA levels was 0.29 (P=0.38), between the 3-month abiraterone trough and 3-month PSA was −0.61 (P=0.08), and between the 7-month abiraterone trough and 7-month PSA was −0.31 (P=0.54) (Table 3). We did not find any correlation between abiraterone trough and PSA levels even when stratified by body mass index (obese vs non-obese) (Table 4). There was no significant correlation between abiraterone trough levels and PSA alteration (from baseline to 1 month of abiraterone treatment) (Table 5). There was no significant association between abiraterone trough and 3-month PSA (P=0.25) and 7-month PSA (P=0.56) (Table 6). Six patients carried the germline HSD3B1 (1245 C) allele, and among those, 2 patients had stable disease, 2 had a partial response, and 2 had disease progression (Table 2). We did not observe an association between HSD3B1 genotype and abiraterone trough levels (Table 7). There was no significant association between 1245 C allele and OS status (P=0.5) (Table 8).
SAFETY AND TOLERABILITY:
Safety data were collected for all patients who received at least 1 dose (n=13). Nine of 13 patients did experience AA-related adverse events. Most of the adverse events were mild and did not require dose reduction; however, 2 patients required dose reduction due to moderate to severe fatigue. However, these 2 patients were removed from the study when the dose reduction was initiated, and thus, the reduced dose did not contribute to PK variability. The most frequently reported adverse events were fatigue (56%), hypertension (22%), and abnormal liver function tests (11%).
Discussion
STRENGTH AND LIMITATION:
The strength of this study is that it was a prospective observational study; however, it had some limitations. This was a single-center and not a multi-center study. The main limitation of the study was a small sample size, which reduced the power of the study. Also, the standardization of sampling was not always feasible. Other limitations were (1) assessment of compliance: we instructed patients to take AA 22–26 h prior to blood draw; however, we could not confirm whether all patients followed this instruction; (2) lack of record of time points of patients taking AA; and (3) only patients with metastatic disease on AA were eligible.
Conclusions
AA showed a high inter- and intra-patient variability in PK but is currently utilized as a standard fixed dose. Despite the lack of support in utilizing AA trough levels in predicting PSA changes as a prognostic or predictive marker in mHSPC in our study, we found a trend toward a negative correlation between abiraterone trough levels and PSA levels 3 months after treatment initiation. A prospective study with a larger sample size is needed to validate these findings. Additional prospective clinical trials are indicated to assess the clinical outcomes or PSA changes by increasing and decreasing AA doses based on targeted trough plasma concentrations (for example, to maintain 8.4 ng/mL) in a population with prostate cancer.
Tables
Table 1. Patient baseline characteristics.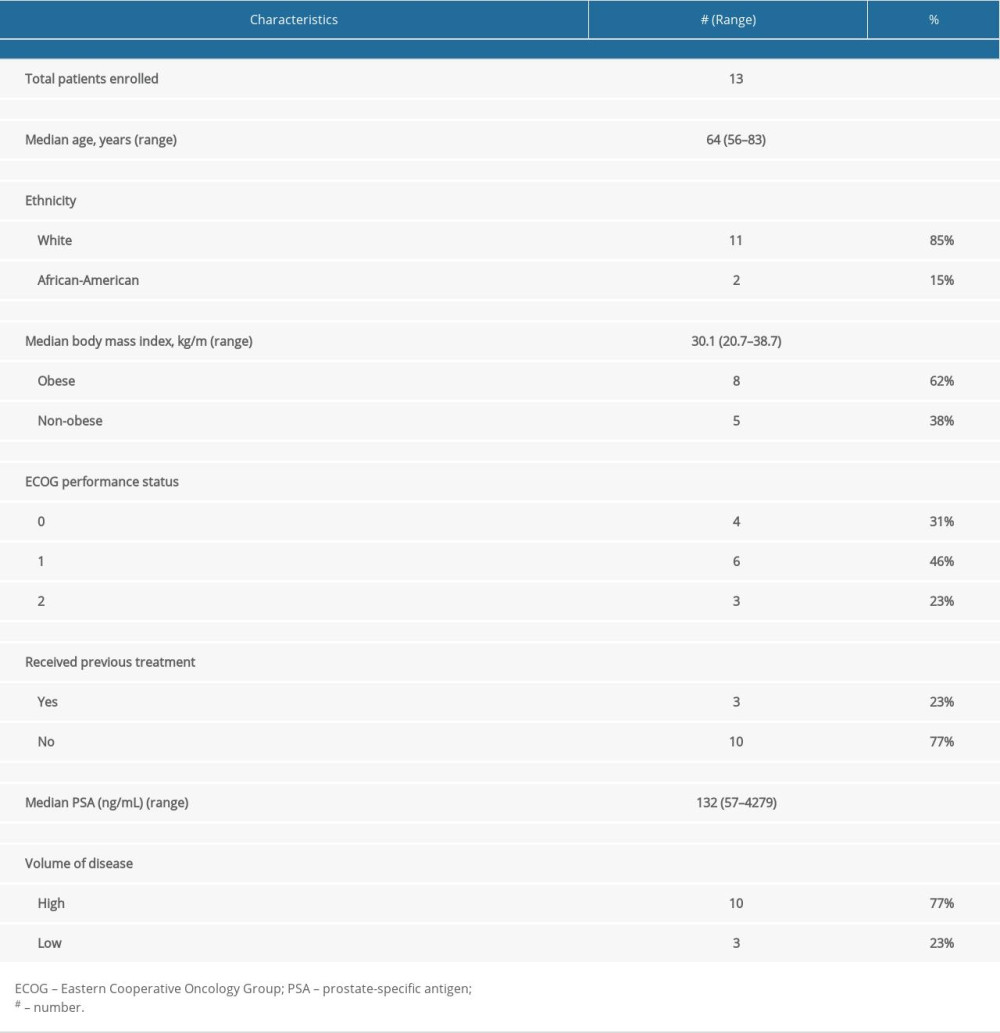 Table 2. Summary of abiraterone trough concentration levels, prostate-specific antigen levels, germline HSD3B1, and survival data of all patients.
Table 2. Summary of abiraterone trough concentration levels, prostate-specific antigen levels, germline HSD3B1, and survival data of all patients.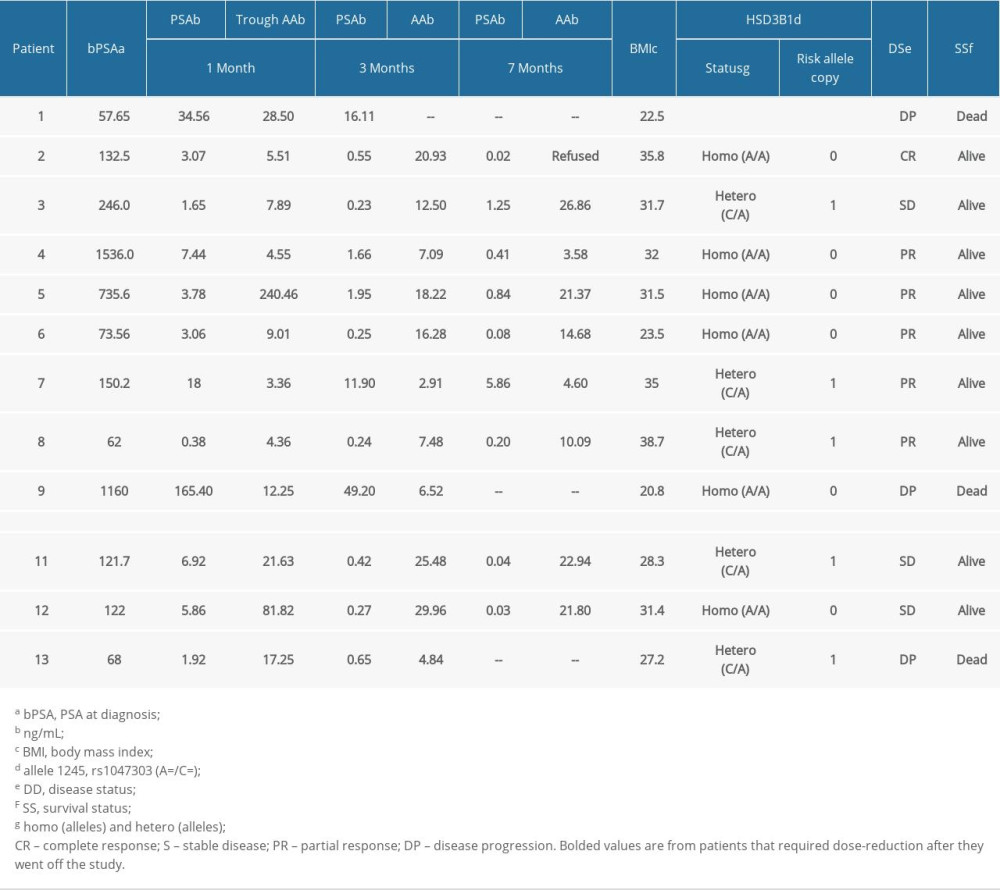 Table 3. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen levels.
Table 3. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen levels.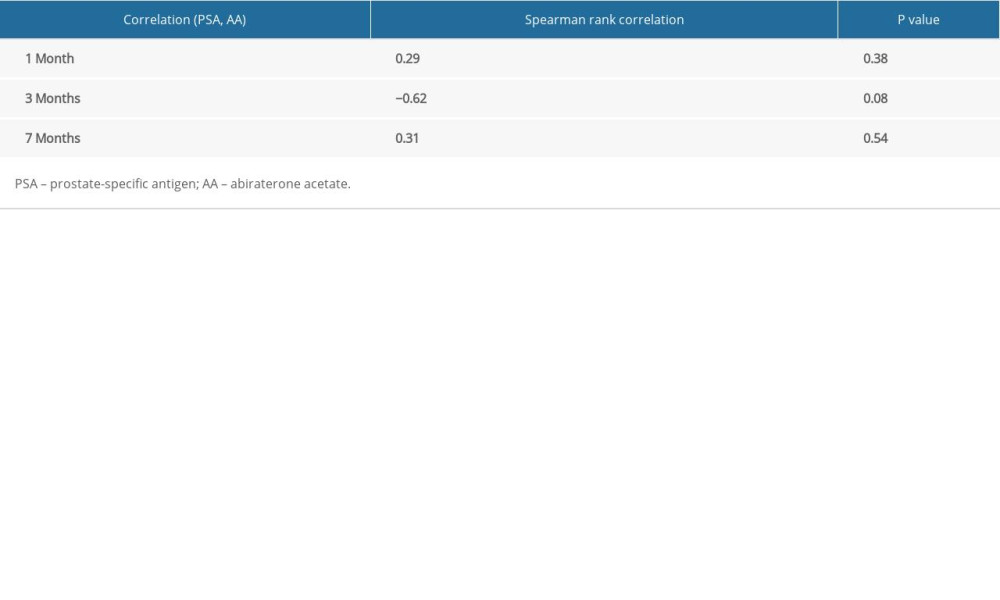 Table 4. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen levels stratified by obesity vs non-obesity.
Table 4. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen levels stratified by obesity vs non-obesity.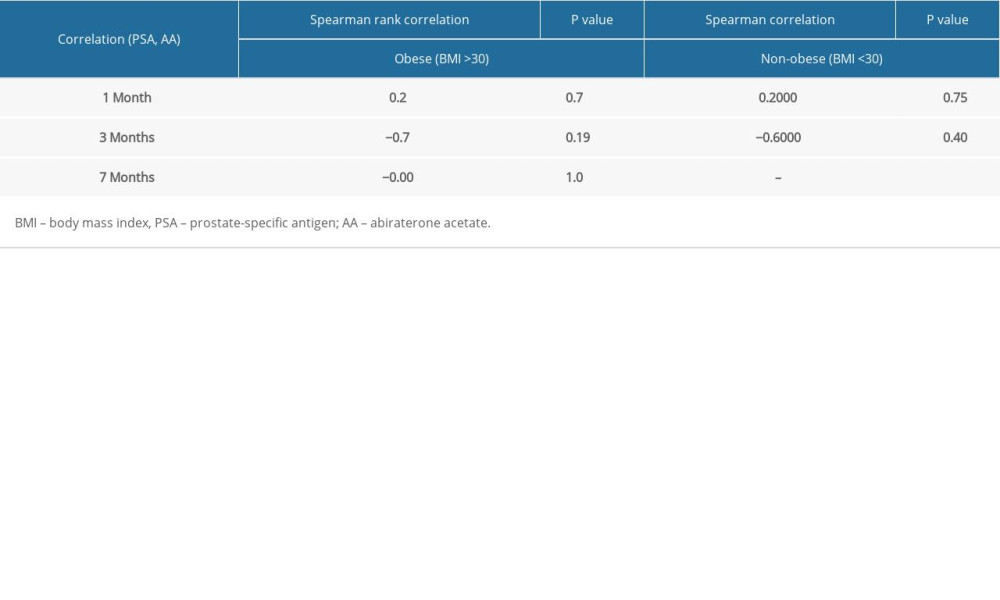 Table 5. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen alteration (change from baseline to 1 month of starting treatment).
Table 5. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen alteration (change from baseline to 1 month of starting treatment).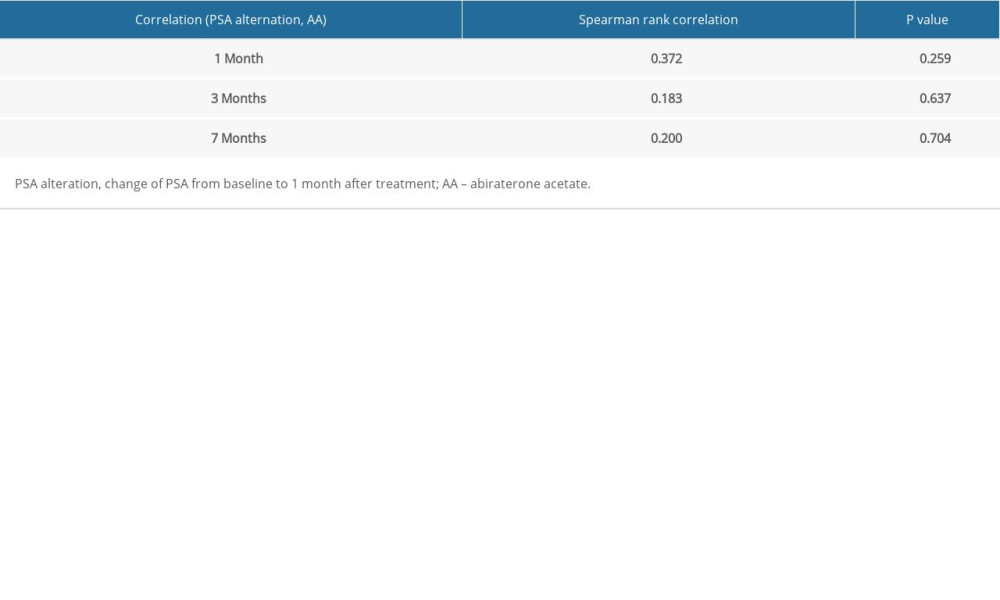 Table 6. Logistic regression between 3-month abiraterone trough level and 3-month prostate-specific antigen ≤1 ng/mL and 7-month abiraterone trough level and 7-month prostate-specific antigen ≤0.2 ng/mL.
Table 6. Logistic regression between 3-month abiraterone trough level and 3-month prostate-specific antigen ≤1 ng/mL and 7-month abiraterone trough level and 7-month prostate-specific antigen ≤0.2 ng/mL.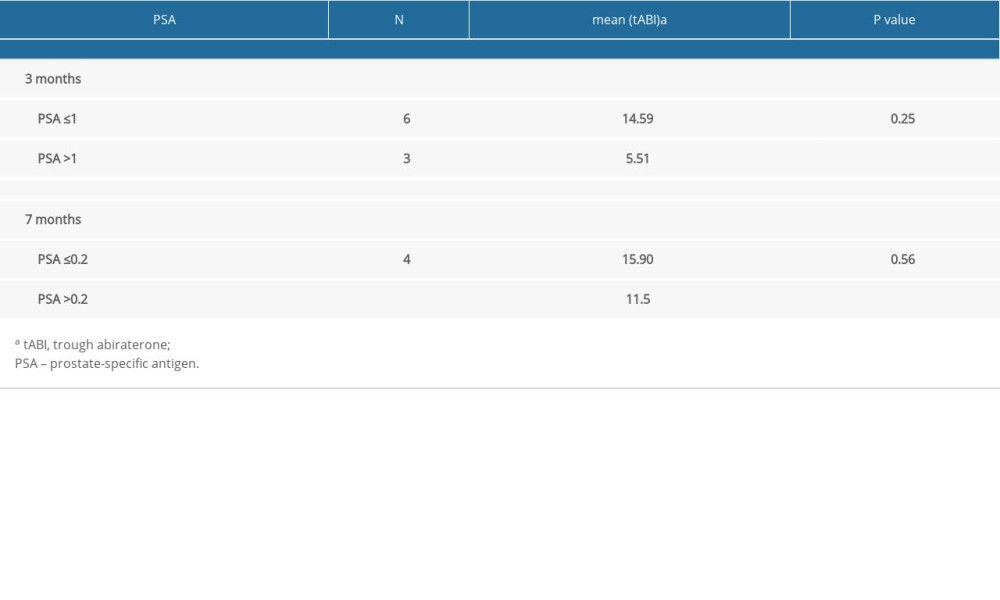 Table 7. Association between HSD3B1 genotype risk allele and 1-month, 3-month, and 7-month abiraterone trough levels.
Table 7. Association between HSD3B1 genotype risk allele and 1-month, 3-month, and 7-month abiraterone trough levels.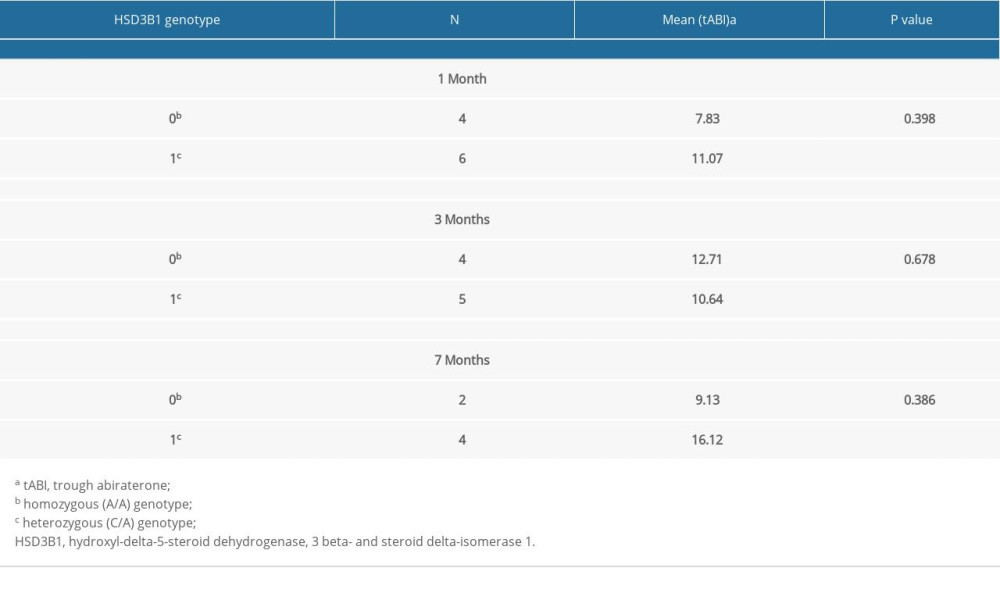 Table 8. Association between HSD3B1 genotype risk-allele and survival outcomes (overall survival and response).
Table 8. Association between HSD3B1 genotype risk-allele and survival outcomes (overall survival and response).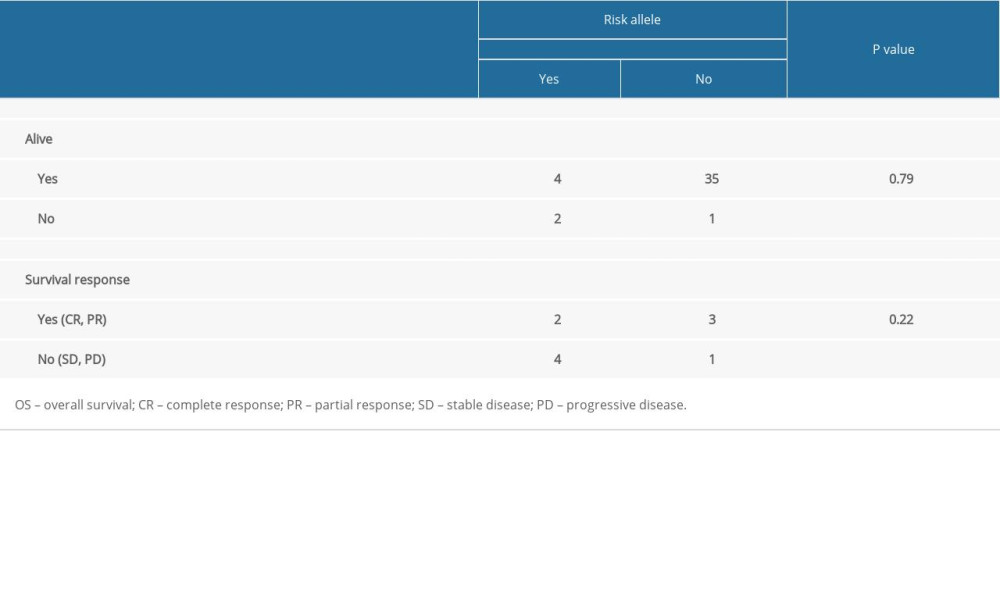
References
1. Aurilio G, Cimadamore A, Mazzucchelli R, Androgen receptor signaling pathway in prostate cancer: From genetics to clinical applications: Cells, 2020; 9(12); 2653
2. Fizazi K, Tran NP, Fein L, Abiraterone plus prednisone in metastatic, castration sensitive prostate cancer: N Engl J Med, 2017; 377; 32-360
3. Ryan CJ, Smith MR, de Bono JS, Abiraterone in metastatic prostate cancer without previous chemotherapy: N Engl J Med, 2012; 368; 138-48
4. De Bono JS, Logothetis CJ, Molina A, Abiraterone and increased survival in metastatic prostate cancer: N Engl J Med, 2011; 364; 1995-2005
5. Chang KH, Li R, Kuri B, A gain-of-function mutation in DHT synthesis in castration resistant prostate cancer: Cell, 2013; 154(5); 1074-84
6. Barrie SE, Potter AG, Goddard PM, Pharmacology of novel steroidal inhibitors of cytochrome P450(17) alpha (17 alpha-hydroxylase/C17–20 lyase): J Steroid Biochem Mol Biol, 1994; 50(5–6); 267-73
7. Hou Z, Huang S, Mei J, Inhibiting 3βHSD1 to eliminate the oncogenic effects of progesterone in prostate cancer: Cell Rep Med, 2022; 3(3); 100561
8. Carton E, Noe G, Huillard O, Relation between plasma trough concentration of abiraterone and prostate-specific antigen response in metastatic castration-resistant prostate cancer patients: Eur J Cancer, 2017; 72; 54-61
9. Stein CA, Levin R, Given R, Randomized phase 2 therapeutic equivalence study of abiraterone acetate fine particle formulation vs. originator abiraterone acetate in patients with metastatic castration resistant prostate cancer: The STAAR study: Urol Oncol, 2018; 81(36); e9-81e16
10. Sweeney CJ, Chen YH, Carducci M, Chemohormonal therapy in metastatic hormone sensitive prostate cancer: N Engl J Med, 2015; 373; 737-46
11. Alyamani M, Li Z, Upadhyay Sk, Development and validation of a novel LC-MS/MS method for simultaneous determination of abiraterone and its seven steroidal metabolites in human serum: Innovation in separation of diastereoisomers without use of a chiral column: J Steroid Biochem Mol Biol, 2017; 172; 231-39
12. Altman N, Krzywinski M, Association, correlation and causation: Nature Methods, 2015; 12; 899-900
13. Nakanishi S, Goya M, Tamaki M, Three-month early change in protate-specific antigen levels as a predictive marker for overall survival during hormonal therapy for metastatic hormone-sensitive prostate cancer: BMC Res Notes, 2021; 14(1); 227
14. Buttigliero C, Tucci M, Bertaglia V, Understanding and overcoming the mechanisms of primary and acquired resistance to abiraterone and enzalutamide in castration resistant prostate cancer: Cancer Treat Rev, 2015; 41(10); 884-92
15. Caffo O, Veccia A, Maines F, Potential value of rapid prostate-specific antigen decline in identifying primary resistance to abiraterone acetate and enzalutamide: Future Oncol, 2014; 10; 985-93
16. Antonarakis ES, Lu C, Wang H, AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer: N Engl J Med, 2014; 371(11); 1028-38
17. Boerrigter E, Benoist GE, Oort IMV, RNA biomarkers as a response measure for survival in patients with metastatic castration resistant prostate cancer: Cancers (Basel), 2021; 13(24); 6279
18. Pal SK, Patel J, He M, Identification of mechanisms of resistance to treatment with abiraterone acetate or enzalutamide in patients with castration resistant prostate cancer (CRPC): Cancer, 2018; 124(6); 1216-24
19. Agarwal N, Hahn AW, Gill DM: JAMA Oncol, 2017; 3(6); 856-57
20. Khalaf DJ, Aragon IM, Annala M: Ann Oncol, 2020; 31(9); 1186-97
21. Almassi N, Reichard C, Li J: JAMA Oncol, 2018; 4(4); 554-57
22. Hearn JW, Sweeney CJ, Almassi N: JAMA Oncol, 2020; 6(4); e196496
23. Mei Z, Yang T, Liu Y, Management of prostate cancer by targeting 3βHSD1 after enzalutamide and abiraterone treatment: Cell Rep Med, 2022; 3(5); 100608
Tables
 Table 1. Patient baseline characteristics.
Table 1. Patient baseline characteristics. Table 2. Summary of abiraterone trough concentration levels, prostate-specific antigen levels, germline HSD3B1, and survival data of all patients.
Table 2. Summary of abiraterone trough concentration levels, prostate-specific antigen levels, germline HSD3B1, and survival data of all patients. Table 3. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen levels.
Table 3. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen levels. Table 4. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen levels stratified by obesity vs non-obesity.
Table 4. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen levels stratified by obesity vs non-obesity. Table 5. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen alteration (change from baseline to 1 month of starting treatment).
Table 5. Correlation between 1-month, 3-month, and 7-month abiraterone trough levels and prostate-specific antigen alteration (change from baseline to 1 month of starting treatment). Table 6. Logistic regression between 3-month abiraterone trough level and 3-month prostate-specific antigen ≤1 ng/mL and 7-month abiraterone trough level and 7-month prostate-specific antigen ≤0.2 ng/mL.
Table 6. Logistic regression between 3-month abiraterone trough level and 3-month prostate-specific antigen ≤1 ng/mL and 7-month abiraterone trough level and 7-month prostate-specific antigen ≤0.2 ng/mL. Table 7. Association between HSD3B1 genotype risk allele and 1-month, 3-month, and 7-month abiraterone trough levels.
Table 7. Association between HSD3B1 genotype risk allele and 1-month, 3-month, and 7-month abiraterone trough levels. Table 8. Association between HSD3B1 genotype risk-allele and survival outcomes (overall survival and response).
Table 8. Association between HSD3B1 genotype risk-allele and survival outcomes (overall survival and response). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








