17 October 2023: Clinical Research
Time-Dependent Factors Influencing Cardiocerebral Vascular Events in Chronic Hemodialysis Patients: Insights from a Longitudinal Study
Haowen Zhong1CDE, Mengbi Zhang2CD, Xiaoyan Su2DG, Yuqin Qing3C, Yanhong Lai2B, Jianhua Chen2F, Linqing Chen2BF, Aiqin Cao2F, Xun Liu4AG*, Shaohong Li5AGDOI: 10.12659/MSM.941553
Med Sci Monit 2023; 29:e941553
Abstract
BACKGROUND: Cardiocerebral vascular events (CVCs) are significant complications in patients undergoing hemodialysis (HD). Given the increased morbidity and mortality associated with CVCs in this population, understanding the factors influencing CVC occurrence over time is crucial. This study aimed to investigate these time-dependent factors in HD patients.
MATERIAL AND METHODS: A total of 228 HD patients from 2 dialysis centers, with at least 3 months of treatment between 2017 and 2021, were included. Annual clinical data were collected, and patients were monitored until CVC development. Kaplan-Meier analysis and a time-dependent Cox regression model were used for data analysis.
RESULTS: The mean age of 228 patients was 55.0±15.0 years, and 64.76% were male. For 5 years of monitoring, the mean follow-up interval was 3.1±1.0 years for patients to develop CVCs. The 1-year, 3-year, and 5-year CVC-free rates were 97.47%, 81.31%, and 70.71%, respectively. Time-dependent Cox regression revealed that C-reactive protein was an independent time-dependent risk factor in HD patients and blood flow rate was an independent time-dependent protective factor. The male subgroup and non-diabetic subgroup had these same results. The following were was the independent time-dependent risk factors: white blood cell count for the female subgroup; blood flow rate for the non-elderly subgroup; and C-reactive protein for the diabetic subgroup. None were risk factors for the elderly subgroup.
CONCLUSIONS: It took an average of 3.1±1.0 years for patients with HD to develop CVCs. C-reactive protein and blood flow rate emerged as key time-dependent influencing factors for CVCs in this population.
Keywords: C-Reactive Protein, Hemodialysis Solutions, Risk Factors, protective factors, Humans, Male, Female, Middle Aged, Aged, Longitudinal Studies, Renal Dialysis, Proportional Hazards Models, Kidney Failure, Chronic
Background
With population aging and the popularization of medical technology, the incidence of end-stage renal disease has gradually increased worldwide [1,2]. Instead of peritoneal dialysis and kidney transplantation, hemodialysis (HD) remains the primary kidney replacement therapy [3]. With the development of HD techniques, the quality of life for HD patients has improved [1,4]. However, this group still is observed to have a high mortality rate, with the occurrence of cardiocerebral vascular events (CVCs) as the leading cause [5,6]. Further exploration of relevant CVC risk factors is warranted.
Given that HD patients are in a fresh situation compared with the pre-dialysis state, such as the use of anticoagulants, hemodynamic changes during HD, and blood flow diversion due to the establishment of autogenous arteriovenous fistulas, conventional risk factors of CVCs in non-HD patients, cannot be applied to HD patients. However, little is known about the underlying factors that influence CVCs in patients undergoing HD.
To address these issues, a time-dependent Cox regression model was used to explore the time-dependent influencing factors, considering the degree of change in clinical indicators over time. The goal of this study was to explore the time-dependent factors influencing the occurrence of CVCs in patients on HD, with full consideration of time variation instead of time points.
Material and Methods
STUDY POPULATION:
In this prospective study, 228 patients on HD were enrolled from Tungwah Hospital and the Third Affiliated Hospital of Sun Yat-sen University between 2017 and 2021. These patients received 2 to 3 weekly sessions of HD for at least 3 months, using bicarbonate-based dialysate and polysulfone membrane dialyzers. The criteria for sample selection were as follows: patients were at least 18 years old when HD was initiated and underwent quarterly biochemical tests; patients without a history of prior renal transplantation; patients without significant blood loss; and patients who had not undergone surgery within 1 year. The censored data were as follows: of the 56 screened patients who were excluded, 35 patients declined to participate, 10 underwent renal transplant, 8 withdrew consent, and 3 had poor compliance with dialysis. Predefined sub-studies were conducted according to the patients’ grouping based on sex, whether they had diabetes, or whether they were elderly (65 years as the cutoff).
ETHICS STATEMENT:
The protocol was approved by the institutional review boards of Tungwah Hospital and the Third Affiliated Hospital of Sun Yat-sen University (approval No. 2020DHLL010). All patients or their immediate relatives provided written informed consent.
LABORATORY TESTS AND OUTCOME EVENTS:
All demographic characteristics and clinical information of the patients were collected, along with other relevant variables that were averaged based on periodic dialysis evaluations. These variables included the average weekly dialysis time, blood flow rate (BFR), dry weight, hemoglobin, hematocrit value, white blood cell (WBC) count, and C-reactive protein (CRP), serum albumin, serum creatinine, uric acid, cholesterol, serum potassium, and parathyroid hormone levels. Venous blood samples were collected after an overnight fast during the first month of each quarter. The outcomes of CVCs included acute heart failure, acute hematencephalon, cardiac- or brain-derived death, acute myocardial infarction, acute cerebral infarction, ischemic cardiomyopathy, unstable angina pectoris, and stable angina pectoris.
When a patient experienced a cardiovascular event, we promptly activated the consultation system and collaborated with the cardiologist to establish an accurate diagnosis of the cardiovascular event. In cases of cerebrovascular events, we sought consultation from a specialist in cerebrovascular medicine. We ensured that both diagnosing physicians held the title of attending physician or higher.
STATISTICAL ANALYSIS:
Statistical analyses were performed using SPSS software (version 20.0; IBM Corp, Armonk, NY, USA). Continuous clinical variables are reported as medians and interquartile ranges or as means and standard deviations. Data are expressed as percentages. A Kaplan-Meier estimate was generated to describe the CVC-free rate. Univariate and multivariate time-dependent Cox regression analyses were used to estimate time-dependent protective or risk factors for the outcomes. A
TIME-DEPENDENT COX REGRESSION MODEL:
A Cox proportional hazard regression model was built. The Cox regression model parameters were obtained using the partial likelihood function, and the Cox proportional hazard regression model was produced. The basic form of the Cox proportional hazard regression model is as follows:
where h(t, X) is the hazard function of an individual with covariate X at time t, also known as the instantaneous mortality rate. Survival time is t, and X=(
Results
BASELINE CHARACTERISTICS AND SURVIVAL ANALYSIS OF HD PATIENTS:
We enrolled 228 patients in this study. The baseline characteristics of the study population are shown in Table 1. The study included 132 male (64.76%) and 66 female (35.24%) patients. Their mean age was 55.0±15.0 years, and the mean duration of HD was 4.0±2.5 years.
It took an average of 3.1±1.0 years for the patients to develop CVCs. The major CVCs were acute heart failure (36.14%), acute hematencephalon (19.80%), cardiac- or brain-derived death (18.32%), and acute myocardial infarction (10.40%). The 1-year, 3-year, and 5-year CVC-free rates were 97.47%, 81.31%, and 70.71%, respectively (Figure 1). In the first 3 years after HD initiation, the incidence of CVCs gradually increased, with a slower trend in the third to fifth years.
TIME-DEPENDENT COX REGRESSION REVEALED TIME-DEPENDENT INFLUENCING FACTORS FOR CVCS IN HD PATIENTS:
In clinical practice, most clinical data or indicators vary with the changes in treatment strategies. Despite its widespread application, the traditional Cox regression model cannot take advantage of such varying tendencies to accurately reflect the prognostic efficacy. A time-dependent Cox regression model was used for analysis.
A univariate time-dependent Cox regression revealed that hemoglobin (RR=0.981, 95% CI 0.969–0.992, P=0.001), serum albumin (RR=0.940, 95% CI 0.898–0.985, P=0.009), and BFR (RR=0.989, 95% CI 0.981–0.996, P=0.005) were time-dependent protective factors for CVCs in HD patients, while only BFR (RR=0.991, 95% CI 0.984–0.999, P=0.028) was the independent time-dependent protective factor. WBC count (RR=1.102, 95% CI 1.051–1.156, P=0.001) and CRP level (RR=1.008, 95% CI 1.005–1.011, P=0.001) were time-dependent risk factors for CVCs in HD patients, while only the CRP level (RR=1.009, 95% CI 1.004–1.013, P=0.001) was an independent time-dependent risk factor. The results are summarized in Table 2.
INDEPENDENT TIME-DEPENDENT INFLUENCING FACTORS FOR CVCS IN SUBGROUPS:
For the subgroup analysis, the patients were stratified into 2 subgroups based on sex, age, and diabetes status.
For sex stratification, CRP (RR=1.008, 95% CI 1.002–1.014,
For age stratification, BFR (RR=0.987, 95% CI 0.979–0.995,
For diabetes stratification, both CRP (RR=1.009, 95% CI 1.004–1.014, P=0.001) and BFR (RR=0.981, 95% CI 0.968–0.994, P=0.007) exhibited similar effects in non-diabetic patients compared with in the overall patient population; however, only CRP (RR=1.007, 95% CI 1.001–1.013, P=0.027) was identified as an independent time-dependent risk factor in diabetic patients. The results are summarized in Table 3.
Discussion
For patients who initiate HD, the early mortality rate is high [7], with up to 40% of deaths attributed to cardiovascular disease [8,9]. Among patients undergoing maintenance HD, cardiovascular incidents remain the leading cause of mortality [10]. Furthermore, cerebrovascular events occur at a significantly higher rate among HD patients than in the general population [6,11]. Despite this knowledge, the risk factors for CVCs in patients have yet to be fully elucidated. Insufficient research has merged cardiovascular and cerebrovascular incidents in patients undergoing HD.
Most clinical follow-up studies on HD patients undergoing HD have utilized the Cox regression model to analyze the prognostic factors, relying on point-in-time demographics and baseline laboratory data. However, in clinical practice, most indicators are time-varying, which limits the applicability of Cox regression models. In contrast, the time-dependent covariate Cox regression model is a superior approach that adequately accounts for the temporal trends in these indicators.
This study highlights the significance of inflammation and dialysis adequacy indicators in predicting the occurrence of CVC in HD patients. CRP, which is commonly used as a marker of inflammation, serves as a traditional cardiovascular risk factor in non-HD patients and is highly effective in HD patients [12,13]. This underscores the need for clinicians to pay close attention to CRP levels when assessing the CVC risk in this patient population. The BFR during HD, an important clinical indicator unique to HD patients, serves as a protective factor against CVCs. This highlights the critical role of adequate dialysis in reducing the incidence of complications, such as CVCs.
This study revealed that the CRP level is an independent risk factor for CVCs in HD patients, and that the effect is time-dependent, particularly in the male subgroup. As the most commonly used acute-phase protein, CRP serves not only as a non-specific inflammatory marker, but also plays a direct role in inflammation [14] and atherosclerosis [15]. CRP has been confirmed as a strong predictor and risk factor for both atherosclerosis [16] and cardiovascular disease [17], as well as an essential risk factor for acute cerebral infarction in HD patients [18]. Additionally, an elevated CRP level had been found to be unresponsive to the pharmacological effects of erythropoiesis-stimulating agents [19,20], resulting in the development of anemia. Previous studies identified CRP as a predictor of both short- and long-term mortality in HD patients [21–23].
The BFR during HD is associated with dialysis adequacy; an increasing BFR will improve the removal of uremic toxins, such as urea and β2-microglobulin [24], thereby improving patient quality of life [25]. Moreover, an elevated BFR within the range of 200 to 300 mL/min was independently linked to a reduced risk of all-cause mortality, with a more pronounced effect observed in male and non-diabetic patients [26]. BFR during HD was found to be an independent protective factor against CVCs in HD patients, and the effect was time-dependent, especially in the male, non-elderly, and non-diabetic subgroups, which is in agreement with previous studies.
This study was constrained by a limited timeframe and an inadequate sample size. In future research, a larger cohort should be observed over an extended period. Classical and representative indicators were selected for inclusion. If included, other indicators, such as blood pressure measurements before, during, and after dialysis, additional dialysis adequacy markers, and other biochemical parameters may have greater clinical significance.
Conclusions
In summary, our findings indicate that the CRP level and BFR are independent time-dependent factors influencing CVCs in HD patients. This study is a step forward in our understanding of how best to manage and care for HD patients. By identifying these key predictors and protective factors, we can improve the outcomes and enhance the quality of life of patients undergoing HD.
Tables
Table 1. Baseline characteristics and survival analysis of hemodialysis (HD) patients.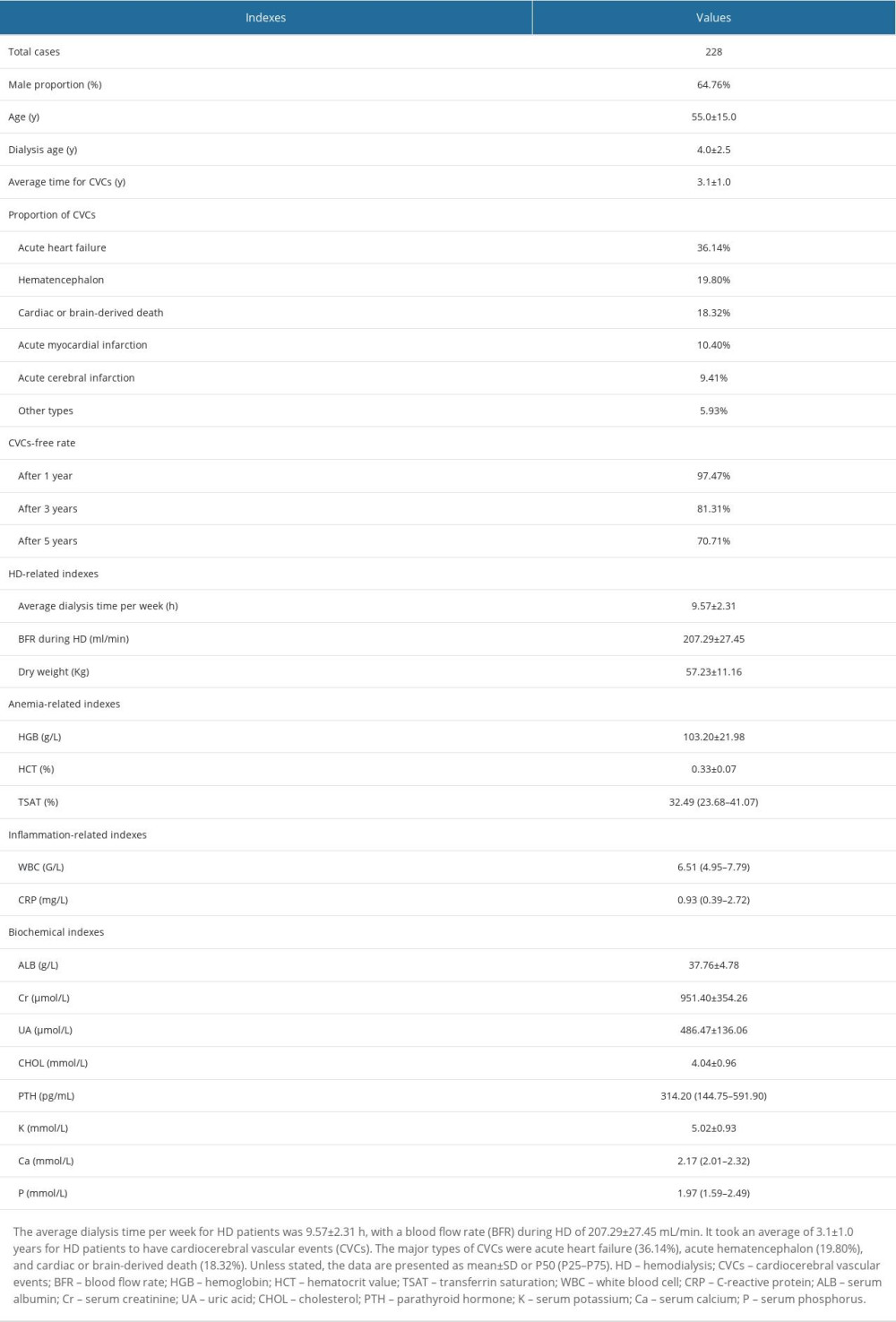 Table 2. Time-dependent Cox regression revealed the time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) patients.
Table 2. Time-dependent Cox regression revealed the time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) patients.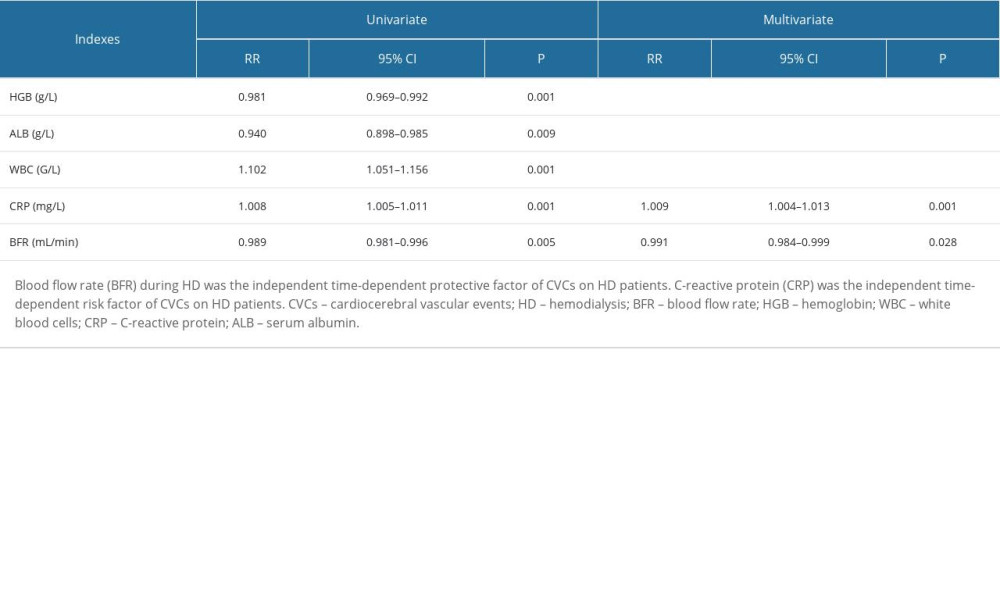 Table 3. Independent time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) subgroups.
Table 3. Independent time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) subgroups.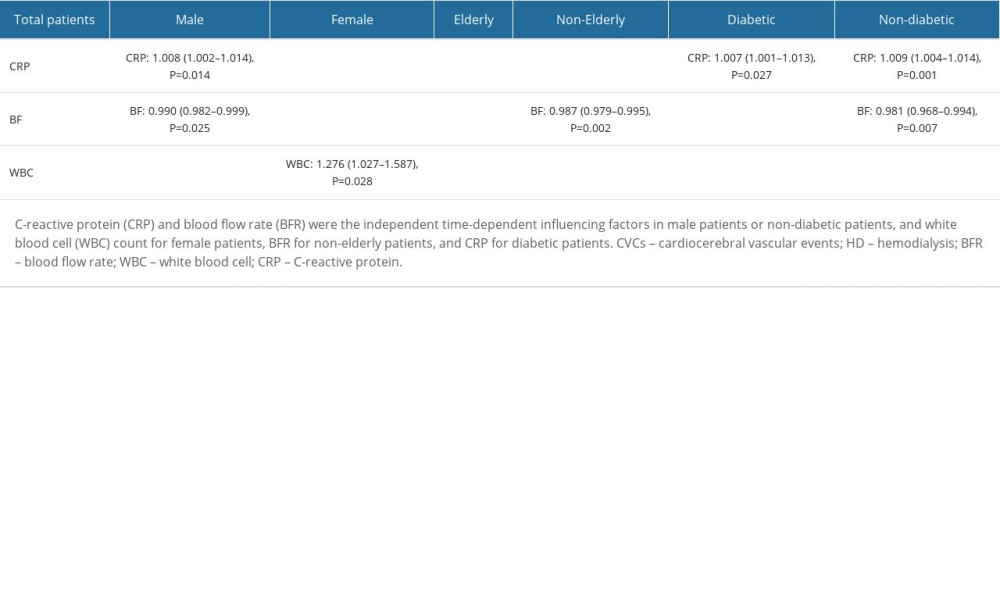
References
1. Foster BJ, Survival improvements for Europeans with ESKD: Kidney Int, 2020; 98(4); 834-36
2. McCullough KP, Morgenstern H, Saran R, Projecting ESRD incidence and prevalence in the United States through 2030: J Am Soc Nephrol, 2019; 30(1); 127-35
3. Bello AK, Levin A, Tonelli M, Assessment of global kidney health care status: JAMA, 2017; 317(18); 1864-81
4. Boenink R, Stel VS, Waldum-Grevbo BE, Data from the ERA-EDTA Registry were examined for trends in excess mortality in European adults on kidney replacement therapy: Kidney Int, 2020; 98(4); 999-1008
5. Drüeke TB, Floege J, Cardiovascular complications of chronic kidney disease: Pioneering studies: Kidney Int, 2020; 98(3); 522-26
6. Belmar L, de Francisco AL, Bueno L, Strokes in patients on haemodialysis: Incidence, onset time and associated factors: Nefrologia, 2014; 34(3); 347-52
7. Eckardt KU, Gillespie IA, Kronenberg FARO Steering Committee, High cardiovascular event rates occur within the first weeks of starting hemodialysis: Kidney Int, 2015; 88(5); 1117-25
8. Hole BD, Casula A, Caskey FJ, Quality assuring early dialysis care: Evaluating rates of death and recovery within 90 days of first dialysis using the UK Renal Registry: Clin Kidney J, 2021; 15(8); 1612-21
9. de Jager DJ, Grootendorst DC, Jager KJ, Cardiovascular and noncardiovascular mortality among patients starting dialysis: JAMA, 2009; 302(16); 1782-89
10. Hoppe K, Schwermer K, Olewicz-Gawlik A, Dialysis vintage and cardiovascular injury as factors influencing long-term survival in peritoneal dialysis and hemodialysis: Adv Clin Exp Med, 2017; 26(2); 251-58
11. Iseki K, Fukiyama KOkawa Dialysis Study (OKIDS) Group, Clinical demographics and long-term prognosis after stroke in patients on chronic haemodialysis. The Okinawa Dialysis Study (OKIDS) Group: Nephrol Dial Transplant, 2000; 15(11); 1808-13
12. Manolis AS, Manolis AA, High C-reactive protein/low serum albumin: A hidden villain in cardiovascular disease: Angiology, 2022; 73(9); 797-99
13. Zeller J, Bogner B, McFadyen JD, Transitional changes in the structure of C-reactive protein create highly pro-inflammatory molecules: Therapeutic implications for cardiovascular diseases: Pharmacol Ther, 2022; 235; 108165
14. Thiele JR, Habersberger J, Braig D, Dissociation of pentameric to monomeric C-reactive protein localizes and aggravates inflammation: In vivo proof of a powerful proinflammatory mechanism and a new anti-inflammatory strategy: Circulation, 2014; 130(1); 35-50
15. Sun H, Koike T, Ichikawa T, C-reactive protein in atherosclerotic lesions: Its origin and pathophysiological significance: Am J Pathol, 2005; 167(4); 1139-48
16. deFilippi C, Wasserman S, Rosanio S, Cardiac troponin T and C-reactive protein for predicting prognosis, coronary atherosclerosis, and cardiomyopathy in patients undergoing long-term hemodialysis: JAMA, 2003; 290(3); 353-59
17. Forghani MS, Jadidoleslami MS, Naleini SN, Rajabnia M, Measurement of the serum levels of serum troponins I and T, albumin and C-Reactive protein in chronic hemodialysis patients and their relationship with left ventricular hypertrophy and heart failure: Diabetes Metab Syndr, 2019; 13(1); 522-25
18. Anan F, Shimomura T, Kaku T, High-sensitivity C-reactive protein level is a significant risk factor for silent cerebral infarction in patients on hemodialysis: Metabolism, 2008; 57(1); 66-70
19. Yılmaz I, Ozkok A, Kostek O, C-reactive protein but not hepcidin, NGAL and transferrin determines the ESA resistance in hemodialysis patients: Ren Fail, 2016; 38(1); 89-95
20. Kimachi M, Fukuma S, Yamazaki S, Minor elevation in C-reactive protein levels predicts incidence of erythropoiesis-stimulating agent hyporesponsiveness among hemodialysis patients: Nephron, 2015; 131(2); 123-30 Erratum in: Nephron. 2016;132(4):342
21. Bazeley J, Bieber B, Li Y, Morgenstern H, C-reactive protein and prediction of 1-year mortality in prevalent hemodialysis patients: Clin J Am Soc Nephrol, 2011; 6(10); 2452-61
22. Takahashi H, Ishii H, Aoyama T, Association of cardiac valvular calcifications and C-reactive protein with cardiovascular mortality in incident hemodialysis patients: A Japanese cohort study: Am J Kidney Dis, 2013; 61(2); 254-61
23. Amemiya N, Ogawa T, Otsuka K, Comparison of serum albumin, serum C-reactive protein, and pulse wave velocity as predictors of the 4-year mortality of chronic hemodialysis patients: J Atheroscler Thromb, 2011; 18(12); 1071-79
24. Leclerc M, Bechade C, Henri P, Does the blood pump flow rate have an impact on the dialysis dose during low dialysate flow rate hemodialysis?: Blood Purif, 2018; 46(4); 279-85
25. Aliasgharpour M, Zabolypour S, Asadinoghabi A, The effect of increasing blood flow rate on severity of uremic pruritus in hemodialysis patients: A single clinical trial: J Natl Med Assoc, 2018; 110(3); 270-75
26. Zhao X, Niu Q, Gan L, Blood flow rate: An independent risk factor of mortality in Chinese hemodialysis patients: Semin Dial, 2022; 35(3); 251-57
Tables
 Table 1. Baseline characteristics and survival analysis of hemodialysis (HD) patients.
Table 1. Baseline characteristics and survival analysis of hemodialysis (HD) patients. Table 2. Time-dependent Cox regression revealed the time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) patients.
Table 2. Time-dependent Cox regression revealed the time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) patients. Table 3. Independent time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) subgroups.
Table 3. Independent time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) subgroups. Table 1. Baseline characteristics and survival analysis of hemodialysis (HD) patients.
Table 1. Baseline characteristics and survival analysis of hemodialysis (HD) patients. Table 2. Time-dependent Cox regression revealed the time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) patients.
Table 2. Time-dependent Cox regression revealed the time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) patients. Table 3. Independent time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) subgroups.
Table 3. Independent time-dependent influencing factors of cardiocerebral vascular events (CVCs) on hemodialysis (HD) subgroups. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








