07 June 2024: Clinical Research
Risk Factors for Postpartum Hemorrhage in Severe Pre-Eclampsia: A Retrospective Single-Centre Study of 1953 Cases
Yingzi Pan1ABCDE, Yuchuan Wang1ABCDE, Jiayan Miao1ABCDEF, Xiaohong Ji1BCEF, Chengqian Wu1ABC, Yixiao Wang1ABCDEF*, Hongjuan Ding1ABCDEFGDOI: 10.12659/MSM.943772
Med Sci Monit 2024; 30:e943772
Abstract
BACKGROUND: Severe pre-eclampsia (sPE) and postpartum hemorrhage (PPH) in pregnancy have serious impact on maternal and fetal health and life. Co-occurrence of sPE and PPH often leads to poor pregnancy outcomes. We explored risk factors associated with PPH in women with sPE.
MATERIAL AND METHODS: This retrospective study included 1953 women with sPE who delivered at the Women’s Hospital of Nanjing Medical University between April 2015 and April 2023. Risk factors for developing PPH in sPE were analyzed, and subgroups were analyzed by delivery mode (cesarean and vaginal).
RESULTS: A total of 197 women with PPH and 1756 women without PPH were included. Binary logistic regression results showed twin pregnancy (P<0.001), placenta accreta spectrum disorders (P=0.045), and placenta previa (P<0.001) were independent risk factors for PPH in women with sPE. Subgroup analysis showed risk factors for PPH in cesarean delivery group were the same as in the total population, but vaginal delivery did not reduce risk of PPH. Spinal anesthesia reduced risk of PPH relative to general anesthesia (P=0.034). Vaginal delivery group had no independent risk factors for PPH; however, magnesium sulfate (P=0.041) reduced PPH incidence.
CONCLUSIONS: Women with twin pregnancy, placenta accreta spectrum disorders, placenta previa, and assisted reproduction with sPE should be alerted to the risk of PPH, and spinal anesthesia should be preferred in cesarean delivery. Magnesium sulfate should be used aggressively in women with sPE; however, the relationship between magnesium sulfate and PPH risk needs further investigation.
Keywords: Postpartum Hemorrhage, Pre-Eclampsia, Risk Factors, retrospective study, Humans, Female, Pregnancy, Retrospective Studies, adult, Cesarean Section, China, Placenta Previa, Delivery, Obstetric, Pregnancy, Twin, Placenta Accreta, Pregnancy Outcome, Logistic Models, Incidence
Introduction
Pre-eclampsia (PE) is a pregnancy-induced hypertensive disorder that occurs after 20 weeks of gestation and is characterized by new-onset hypertension, proteinuria, and/or multi-organ dysfunction [1]. Severe PE (sPE) is a severe stage of PE with specific manifestations suggestive of the severity of the condition, with a prevalence of approximately 1% [2]. sPE can cause serious complications in pregnant women and fetuses, including cardiovascular and cerebrovascular disease, disseminated intravascular coagulation, placental abruption, eclampsia, and postpartum hemorrhage (PPH) [3–5]. Of these, there is substantial evidence that sPE can cause an increased risk of PPH [6,7].
PPH is one of the most common complications in obstetrics, accounting for 3% to 5% of all deliveries [8]. The Society of Obstetricians and Gynecologists of Canada (SOGC) annual meeting defines it as bleeding of more than 1000 mL within 24 h after cesarean delivery and more than 500 mL within 24 h after vaginal delivery [9]. PPH is the leading cause of maternal mortality, contributing to 27.1% of global maternal deaths [10,11]. PPH is associated with serious complications, such as disseminated intravascular coagulation, acute renal failure, respiratory distress syndrome, and Sheehan syndrome [12]. However, 54% to 93% of maternal deaths due to obstetric hemorrhage are preventable [13].
Clinically, the study of predictive indicators and risk factors of PPH has received an increasing amount of attention, with the aim of adopting interventions for women at high risk of PPH and reducing the morbidity and mortality of PPH. The combination of sPE with PPH will lead to further deterioration of adverse maternal and child outcomes; therefore, early prediction of hemorrhage risk and proper prevention and treatment planning are of positive significance for patients with sPE combined with PPH. Because PPH has a high prevalence in the group of pregnant women with sPE and little research has been done on the predictors of PPH in patients with sPE, analyzing the risk factors for PPH in women with sPE is of value in guiding the clinical treatment of women with sPE. Based on the above, we collected data on pregnant women with sPE who delivered at our hospital during the past 8 years to explore the risk factors for PPH in pregnant women with sPE.
Material and Methods
PATIENTS AND STUDY DESIGN:
This study was a retrospective case-control study that included women with sPE who delivered from April 2015 to April 2023 at the Women’s Hospital of Nanjing Medical University. Diagnosis, management, and treatment of sPE were based on the American College of Obstetricians and Gynecologists (ACOG) guidelines [14]. When elevated blood pressure was detected, labetalol and/or nifedipine tablets were first given orally and maternal blood pressure was continuously checked. If the blood pressure could not be controlled or was already very severe at the time of presentation, intravenous labetalol or phentolamine was given, and magnesium sulphate application was necessary to prevent the progression of sPE to eclampsia. In cases of less than 34 weeks of gestation, we tried to promote fetal lung maturation through the use of corticosteroids whenever possible; however, immediate termination of pregnancy was considered in cases of persistent uncontrolled hypertension, eclampsia, pulmonary edema, placental abruption, disseminated intravascular coagulation, fetal distress, or intrauterine fetal death. Vaginal delivery was preferred if labor could be accomplished within reason; however, if the attending physician determined that the cervix was not ripe enough for vaginal delivery, a cesarean delivery was considered, as soon as possible.
All cesarean deliveries in women with sPE were performed by 2 chief physicians, and vaginal assisted deliveries were performed with the participation of at least 1 chief physician and 1 experienced midwife.
PPH was diagnosed according to the SOGC guidelines, namely, bleeding of more than 1000 mL within 24 h after cesarean delivery and more than 500 mL within 24 h after vaginal delivery [9]. Blood loss was systematically measured using graduated collection bags or collection containers that are routinely used in recruitment centers and systematically reported in the medical record. If the mother was bleeding in the maternity ward, blood loss was quantified by weighing the sanitary pads.
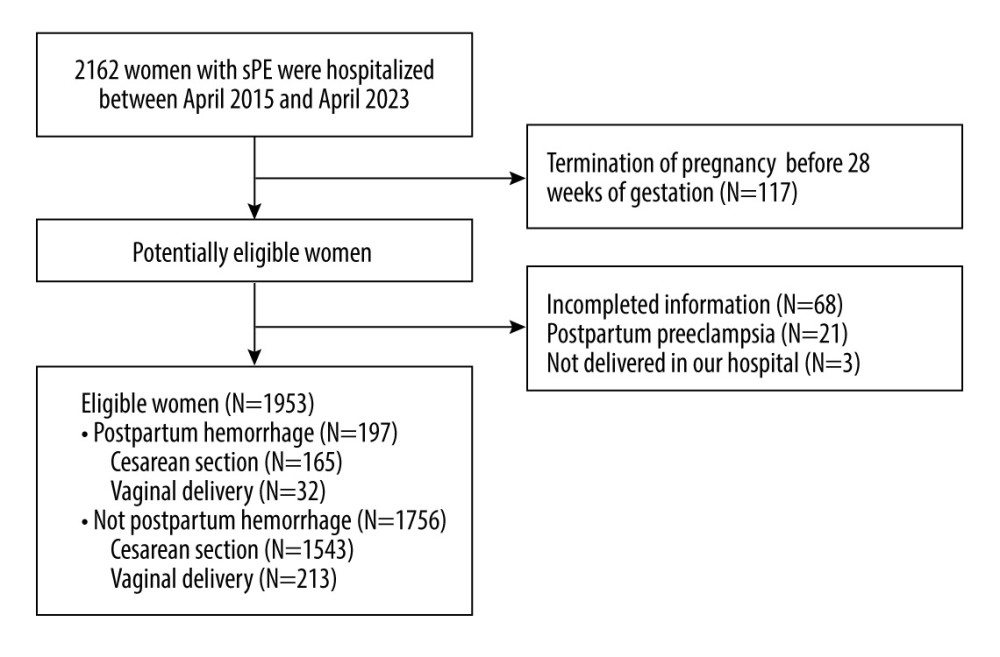
The inclusion criterion was diagnosis of sPE. The exclusion criteria were (1) termination of pregnancy at less than 28 weeks of gestation for any reason; (2) incomplete recording of clinical information; (3) not delivered in our hospital; and (4) postpartum PE. Exclusion of termination of pregnancy before 28 weeks of gestation was due to the fact that a miscarriage within 28 weeks of gestation is considered a miscarriage and not a successful delivery [15].
A total of 1953 women with sPE were finally included in this study (Figure 1). Among them, 197 had PPH (PPH group) and 1756 did not have PPH (non-PPH group).
ETHICS APPROVAL AND CONSENT TO PARTICIPATE:
The study followed the Declaration of Helsinki and was conducted in accordance with relevant local guidelines and regulations. The study was approved by the Medical Ethics Committee of the Affiliated Maternity Hospital of Nanjing Medical University, and informed consent was waived (No. 2022KY-081).
SUBGROUP ANALYSIS:
Considering the different definitions of PPH for vaginal delivery and cesarean delivery, we performed subgroup analyses of the different modes of delivery. All women were divided into a cesarean delivery group and vaginal delivery group according to the mode of delivery, and the risk factors for PPH were analyzed separately in both groups.
STATISTICAL ANALYSIS:
Statistical software SPSS 26.0 was used for data analysis and processing. In this study, continuous variables that conformed to a normal distribution were analyzed using the
Results
RISK FACTORS FOR WOMEN WITH AND WITHOUT PPH:
Women in the PPH group had much higher postpartum hemorrhage than did women in the non-PPH group (P<0.001). Comparison of baseline information between women in the PPH and non-PPH groups revealed that women in the PPH group were older (P=0.018), had higher systolic (P=0.030) and diastolic blood pressure (P=0.012), and had higher proportions of primipara (P=0.042), assisted reproduction (P<0.001), and twin pregnancy (P<0.001). However, the proportion of patients ever having hypertensive disorders of pregnancy, including gestational hypertension and PE, was lower (P=0.020; Table 1).
Among the clinical characteristics, women in the PPH group had higher rates of combined premature rupture of membranes (P<0.001), thrombocytopenia (P=0.046), placenta accreta spectrum disorders (P<0.001), and placenta previa (P<0.001) than did women in the non-PPH group. The rate of blood transfusion (P<0.001) and reoperation to stop bleeding (P<0.001) were higher, and hospitalization was significantly longer (P=0.002). Fetal birth weight (P=0.001) was significantly higher in the non-PPH group, while the proportion of fetal growth restriction was significantly lower (P=0.004). In terms of treatment modalities, the proportion of magnesium sulfate use was significantly lower in women in the PPH group (P<0.001). With regard to the type of anesthesia, a higher percentage of women in the PPH group received general anesthesia (P=0.010). In addition, women in the PPH group had a lower rate of lower extremity edema (P=0.032), amniotic fluid insufficiency (P=0.007), and spinal anesthesia (P=0.006; Table 2).
Risk factors or protective factors for PPH as described above were included in binary logistic regression equations, and the gestational week of delivery was added to correct for confounders. The analysis showed that twin pregnancy (OR 4.386, 95% CI 2.740–7.023, P<0.001), placenta accreta spectrum disorders (OR 3.079, 95% CI 1.039–9.119, P=0.042), and placenta previa (OR 6.736, 95% CI 2.798–16.214, P<0.001) were independent risk factors for PPH in women with sPE. Also, natural pregnancy (OR 0.610, 95% CI 0.385–0.966, P=0.035) was an independent protective factor for reducing combined PPH in women with sPE (Table 3).
RISK FACTORS FOR PPH AFTER CESAREAN DELIVERY:
An analysis of women in the PPH and non-PPH groups after cesarean delivery revealed that the PPH group had older maternal age (P=0.007), higher systolic (P=0.006) and diastolic blood pressure (P=0.017), and greater fetal birth weight (P=0.009). The women in the PPH group had a higher rate of previous miscarriages (P=0.013), assisted reproduction (P<0.001), twin pregnancy (P<0.001), premature rupture of membranes (P=0.049), placenta accreta spectrum disorders (P<0.001) and placenta previa (P<0.001; Tables 4, 5). However, a lower percentage of women in the PPH group had a history of hypertensive disorder complicating pregnancy (P=0.012), lower extremity edema (P=0.022), fetal growth restriction (P=0.008), and insufficient amniotic fluid (P=0.031). In addition, women in the PPH group had higher rates of blood transfusion (P<0.001) and reoperation to stop bleeding (P=0.001) and longer hospital stays (P=0.001) than those in the non-PPH group. In addition, more pregnant women in the PPH group used general anesthesia (P=0.006), but fewer used magnesium sulfate (P=0.007).
Week of gestation at delivery and having been a primiparous woman were also included in the logistic regression to adjust for confounders. Binary logistic regression showed that twin pregnancy (OR 5.058, 95% CI 3.089–8.283, P<0.001), placenta accreta spectrum disorders (OR 3.353, 95% CI 1.104–10.180, P=0.033), and placenta previa (OR 7.056. 95%CI 2.918–17.061, P<0.001) were independent risk factors for PPH (Table 6). In addition, cesarean delivery under spinal anesthesia reduced the incidence of PPH after sPE (OR 0.434, 95%CI 0.200–0.940, P=0.034).
RISK FACTORS FOR PPH AFTER VAGINAL DELIVERY:
An analysis of the characteristics of women in the PPH and non-PPH groups who delivered vaginally revealed that women in the PPH group had a greater gestational week of delivery (P=0.045), greater fetal birth weight (P=0.007), higher rate of fetal distress (P=0.008) and forceps use (P<0.001), and a longer hospital stay (P=0.010; Tables 4, 5). However, the use of magnesium sulfate (P=0.016) and labor analgesia (P<0.001) was significantly lower in women in the PPH group than in women in the non-PPH group. In a binary logistic regression, no risk factors for PPH were identified; however, magnesium sulfate use (OR 0.418, 95% CI 0.181–0.967, P=0.041) prevented PPH following vaginal delivery (Table 6).
Discussion
Further progression of PE to sPE poses a serious threat to maternal and infant health and safety. In sPE, small vessel spasm leads to endothelial injury, local ischemia, and insufficient perfusion of systemic organs, resulting in weak uterine contractions, which leads to PPH [16]. PPH is one of the most important causes of maternal death, with approximately 150 000 maternal deaths occurring globally each year [17]. Therefore, this paper discusses the general information and clinical features of PPH in order to identify potential risk factors and provide a basis for subsequent clinical work to improve the prognosis of mothers and infants.
Overall, twin pregnancy, placental abruption spectrum disorders, and placenta previa are independent risk factors for PPH in combination with sPE, and all of these factors may be closely related to the use of widely applied assisted reproductive technologies. Also, this study found that vaginal delivery was a protective factor in reducing PPH; in other words, assisted reproduction increased the risk of PPH. In recent years, with the development of assisted reproductive technology and the use of ovulation-promoting drugs, the incidence of twin pregnancy has increased yearly. Twin pregnancy is susceptible to PPH due to uterine overdistension and myofibrillar hyperextension, which affects myofibrillar retraction, leading to weak uterine contractions in the postpartum period, which affects complete placental abruption and closure of blood sinuses at the abruption surface of the placenta [18,19]. In addition, twin fetuses have a large placental area, richer blood flow, more bleeding from small uterine arteries, and vein dissections during placental abruption, and are prone to complications of hypertensive disorders of pregnancy (especially sPE), amniotic fluid, placenta previa, and intrahepatic cholestasis in pregnancy, which increase the incidence of PPH in twin pregnancy. In addition, the incidence of placenta previa and placenta accreta spectrum disorders were higher in the sPE-combined PPH group than in the non-PPH group, which is consistent with the results of a previous study [20]. Placental abnormalities, including placental abruption spectrum disorders and placenta previa, can lead to difficulties in placental abruption during labor, which affects uterine contraction, and the opening of a large number of blood sinuses after placental abruption can increase the risk of PPH.
For cesarean delivery in the present study, spinal anesthesia reduced the incidence of PPH, which is consistent with existing results [21]. This may be related to the fact that anesthesiologists may prefer to use general anesthesia for patients with placenta previa or placental anomalies [22] or that patients with expected PPH risk factors may be more likely to receive general anesthesia. Women undergoing general anesthesia are 3 times more likely to develop severe PPH than women undergoing single spinal anesthesia [23]. Since spinal and general anesthesia usually use equal doses of anesthetic drugs, it is questionable whether drug effects can explain this finding.
Interestingly, the present study showed that the use of magnesium sulfate reduced the risk of PPH in those who had vaginal delivery. In this study, the use of magnesium sulfate was also lower in the PPH group among cesarean deliveries. We cannot explain this association at this time; however, healthcare providers should be reminded that magnesium sulfate is not an antihypertensive drug. Magnesium sulfate has also been widely used as a neuroprotective agent in recent years, as it has been proposed that inhibition of NMDA receptors in the brain prevents cellular damage that may result from hypoxic stress activation [24]. In addition to this, magnesium sulfate reduces acetylcholine in the neuromuscular junction which interferes with actin-myosin, blocks NMDA receptors, and inhibits selective catecholamine. Such effects will result in myometrial relaxation, decreased uterine contractility, arterial vasodilation, and decreased systolic blood pressure. On the other hand, considering this pathophysiological pathway, one may hypothesize that magnesium sulfate may also interfere with post-partum uterine resuscitation, and therefore, its use may increase the risk of uterine asystole and PPH. Observational studies have shown a significant effect of magnesium sulfate on the risk of postpartum uterine contraction weakness and hemorrhage; however, evidence from randomized trials does not support this [25]. Given the limitations of our study, we believe that future studies could help to elucidate the actual effects of magnesium sulfate on uterine blood vessels (with the exception of uterine arteries), as well as the potential portion of the difference in expected blood loss between vaginal and cesarean delivery cases. In addition, special consideration should be given to specific cases with coagulation disorders, as magnesium sulfate appears to shorten normal bleeding time.
Our study is the first to assess the risk factors for PPH in women with sPE. Nonetheless, this study has some limitations and unexplained results. It was a single-center retrospective study that could not adequately account for and correct for multiple confounding factors. In addition, the low prevalence of certain pregnancy complications in the vaginal delivery group precludes a convincing statistical analysis. A larger sample size from multiple centers is needed to address these issues.
Conclusions
In conclusion, this study provides a preliminary investigation and analysis of the risk factors for the occurrence of sPE combined with PPH. In clinical practice, identifying the risk factors of women with sPE in different modes of delivery can assist in the development of precise management measures for women with sPE, adequate preoperative preparation, and adequate blood preparation in order to reduce the incidence of sPE combined with PPH, improve maternal outcomes, and optimize perinatal outcomes.
Tables
Table 1. Basic characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage.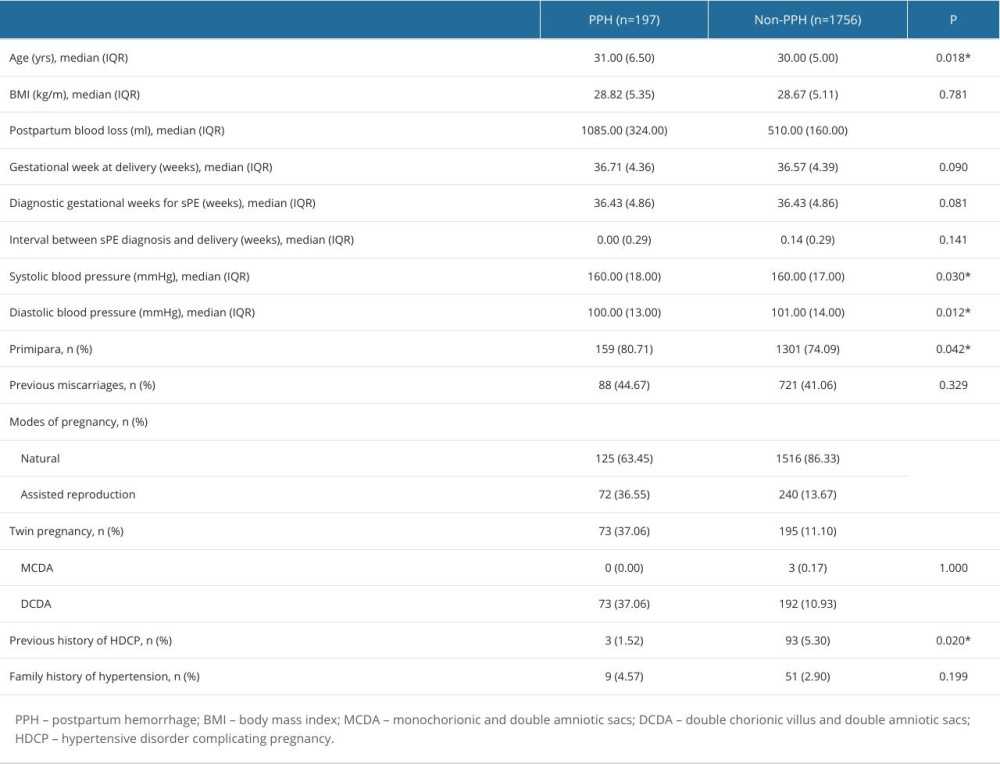 Table 2. Clinical characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage.
Table 2. Clinical characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage.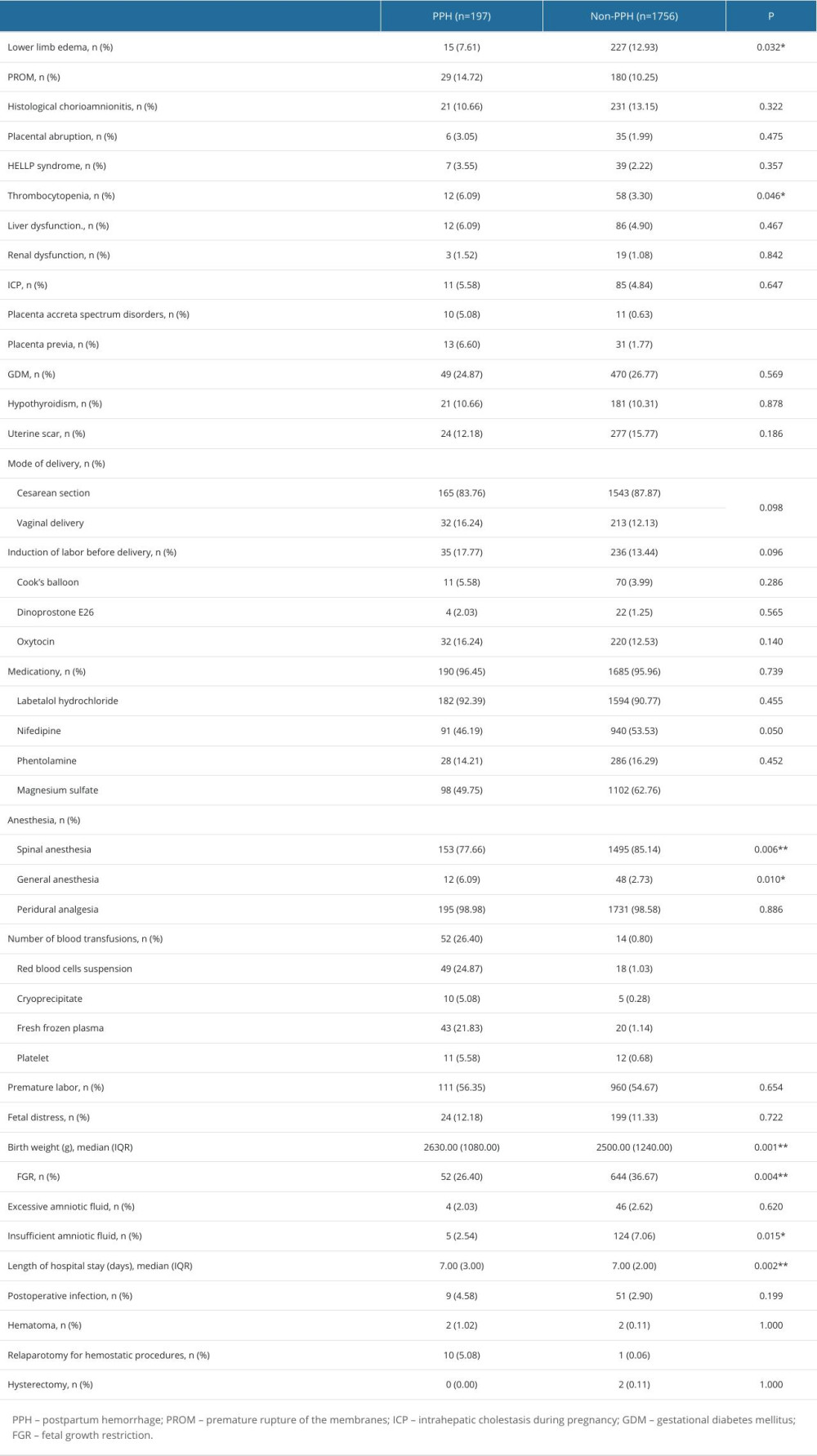 Table 3. Binary logistic regression of risk factors for severe pre-eclampsia with postpartum hemorrhage.
Table 3. Binary logistic regression of risk factors for severe pre-eclampsia with postpartum hemorrhage. Table 4. Basic characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups.
Table 4. Basic characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups. Table 5. Clinical characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups.
Table 5. Clinical characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups. Table 6. Binary logistic regression of risk factors for postpartum hemorrhage in cesarean delivery and vaginal delivery groups.
Table 6. Binary logistic regression of risk factors for postpartum hemorrhage in cesarean delivery and vaginal delivery groups.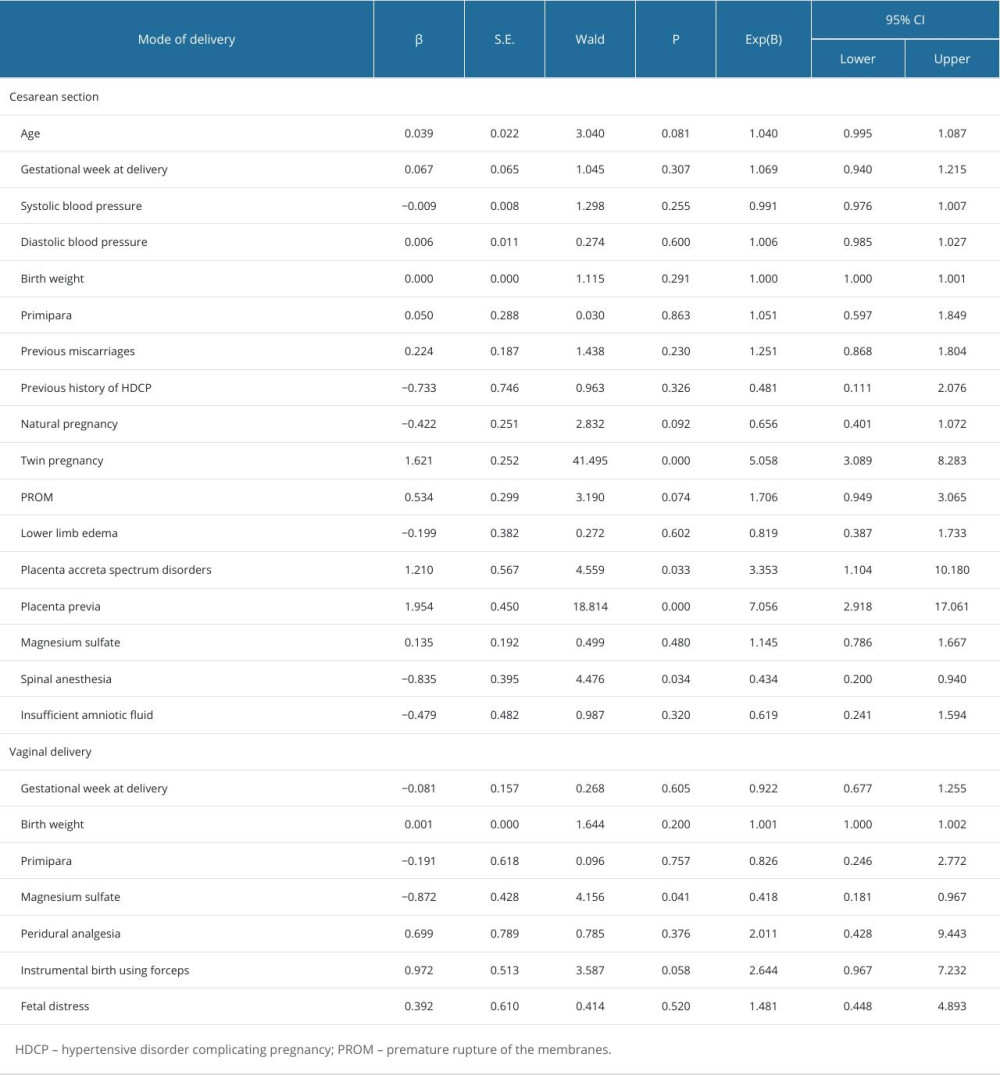
References
1. Chappell LC, Cluver CA, Kingdom J, Tong S, Pre-eclampsia: Lancet, 2021; 398(10297); 341-54
2. Zhang J, Meikle S, Trumble A, Severe maternal morbidity associated with hypertensive disorders in pregnancy in the United States: Hypertens Pregnancy, 2003; 22(2); 203-12
3. Magee LA, Yong PJ, Espinosa V, Expectant management of severe preeclampsia remote from term: A structured systematic review: Hypertens Pregnancy, 2009; 28(3); 312-47
4. van Wassenaer AG, Westera J, van Schie PE, Outcome at 4.5 years of children born after expectant management of early-onset hypertensive disorders of pregnancy: Am J Obstet Gynecol, 2011; 204(6); 510e1-9
5. Kongwattanakul K, Saksiriwuttho P, Chaiyarach S, Thepsuthammarat K, Incidence, characteristics, maternal complications, and perinatal outcomes associated with preeclampsia with severe features and HELLP syndrome: Int J Womens Health, 2018; 10; 371-77
6. Zewdu D, Tantu T, Incidence and predictors of severe postpartum hemorrhage after cesarean delivery in South Central Ethiopia: A retrospective cohort study: Sci Rep, 2023; 13(1); 3635
7. Nyfløt LT, Sandven I, Stray-Pedersen B, Risk factors for severe postpartum hemorrhage: A case-control study: BMC Pregnancy Childbirth, 2017; 17(1); 17
8. Giouleka S, Tsakiridis I, Kalogiannidis I, Postpartum hemorrhage: A comprehensive review of guidelines: Obstet Gynecol Surv, 2022; 77(11); 665-82
9. Leduc D, Senikas V, Lalonde AB, No. 235-active management of the third stage of labour: Prevention and treatment of postpartum hemorrhage: J Obstet Gynaecol Can, 2018; 40(12); e841-e55
10. Alliance for Maternal and Newborn Health Improvement (AMANHI) Mortality Study Group, Population-based rates, timing, and causes of maternal deaths, stillbirths, and neonatal deaths in south Asia and sub-Saharan Africa: A multi-country prospective cohort study: Lancet Glob Health, 2018; 6(12); e1297-e308
11. Say L, Chou D, Gemmill A, Global causes of maternal death: A WHO systematic analysis: Lancet Glob Health, 2014; 2(6); e323-33
12. Bateman BT, Berman MF, Riley LE, Leffert LR, The epidemiology of postpartum hemorrhage in a large, nationwide sample of deliveries: Anesth Analg, 2010; 110(5); 1368-73
13. , Quantitative Blood Loss in Obstetric Hemorrhage: ACOG Committee Opinion Summary, Number 794: Obstet Gynecol, 2019; 134(6); 1368-69
14. , Gestational Hypertension and Preeclampsia: ACOG Practice Bulletin, Number 222: Obstet Gynecol, 2020; 135(6); e237-e60
15. Wang H, Gao H, Chi H, Effect of levothyroxine on miscarriage among women with normal thyroid function and thyroid autoimmunity undergoing in vitro fertilization and embryo transfer: A randomized clinical trial: JAMA, 2017; 318(22); 2190-98
16. Holleboom CA, van Eyck J, Koenen SV, Carbetocin in comparison with oxytocin in several dosing regimens for the prevention of uterine atony after elective caesarean section in the Netherlands: Arch Gynecol Obstet, 2013; 287(6); 1111-17
17. Khan KS, Wojdyla D, Say L, WHO analysis of causes of maternal death: A systematic review: Lancet, 2006; 367(9516); 1066-74
18. Blitz MJ, Yukhayev A, Pachtman SL, Twin pregnancy and risk of postpartum hemorrhage: J Matern Fetal Neonatal Med, 2020; 33(22); 3740-45
19. Cao X, Luo Y, Zhou S, Twin growth discordance and risk of postpartum hemorrhage: A retrospective cohort study: Front Med (Lausanne), 2022; 9; 876411
20. Mehrabadi A, Assisted reproductive technologies: An additional risk factor for severe postpartum haemorrhage: BJOG, 2017; 124(8); 1205
21. Butwick AJ, Ramachandran B, Hegde P, Risk factors for severe postpartum hemorrhage after cesarean delivery: Case-control studies: Anesth Analg, 2017; 125(2); 523-32
22. Snegovskikh D, Clebone A, Norwitz E, Anesthetic management of patients with placenta accreta and resuscitation strategies for associated massive hemorrhage: Curr Opin Anaesthesiol, 2011; 24(3); 274-81
23. Butwick AJ, Ramachandran B, Hegde P, Risk factors for severe postpartum hemorrhage after cesarean delivery: Case-control studies: Anesth Analg, 2017; 125(2); 523-32
24. James MF, Magnesium in obstetrics: Best Pract Res Clin Obstet Gynaecol, 2010; 24(3); 327-37
25. Pergialiotis V, Bellos I, Constantinou T, Magnesium sulfate and risk of postpartum uterine atony and hemorrhage: A meta-analysis: Eur J Obstet Gynecol Reprod Biol, 2021; 256; 158-64
Tables
 Table 1. Basic characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage.
Table 1. Basic characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage. Table 2. Clinical characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage.
Table 2. Clinical characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage. Table 3. Binary logistic regression of risk factors for severe pre-eclampsia with postpartum hemorrhage.
Table 3. Binary logistic regression of risk factors for severe pre-eclampsia with postpartum hemorrhage. Table 4. Basic characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups.
Table 4. Basic characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups. Table 5. Clinical characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups.
Table 5. Clinical characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups. Table 6. Binary logistic regression of risk factors for postpartum hemorrhage in cesarean delivery and vaginal delivery groups.
Table 6. Binary logistic regression of risk factors for postpartum hemorrhage in cesarean delivery and vaginal delivery groups. Table 1. Basic characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage.
Table 1. Basic characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage. Table 2. Clinical characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage.
Table 2. Clinical characteristics between severe pre-eclampsia with postpartum hemorrhage and without postpartum hemorrhage. Table 3. Binary logistic regression of risk factors for severe pre-eclampsia with postpartum hemorrhage.
Table 3. Binary logistic regression of risk factors for severe pre-eclampsia with postpartum hemorrhage. Table 4. Basic characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups.
Table 4. Basic characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups. Table 5. Clinical characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups.
Table 5. Clinical characteristics of women with and without postpartum hemorrhage in the cesarean delivery and vaginal delivery groups. Table 6. Binary logistic regression of risk factors for postpartum hemorrhage in cesarean delivery and vaginal delivery groups.
Table 6. Binary logistic regression of risk factors for postpartum hemorrhage in cesarean delivery and vaginal delivery groups. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387