27 June 2020: Animal Study
Pyrroloquinoline Quinone Inhibits Oxidative Stress in Rats with Diabetic Nephropathy
Min Zhang12ACD, Jiangzhao Zhang3BE, Yan Xiong2CF, Jiaqing Peng2BF, Xiaoyan Wu1AEG*DOI: 10.12659/MSM.924372
Med Sci Monit 2020; 26:e924372
Abstract
BACKGROUND: Diabetic nephropathy (DN) is one of the chronic microvascular complications of diabetes. This study focused on the protective effects of pyrroloquinoline quinone (PQQ) on oxidative stress (OS) in DN.
MATERIAL AND METHODS: Thirty Sprague Dawley rats were randomly selected for this study; 10 rats were randomly selected as the control group. The other 20 rats were established for the DN model. After establishment of the successful model, the DN model rats were randomly divided into a DN group and a PQQ group. The PQQ group was fed with a PQQ diet. Blood urea nitrogen (BUN), serum creatinine (SCr), and blood glucose levels were measured in each group, and OS-related protein expression and AMPK pathway were detected by western blot and quantitative real-time polymerase chain reaction (qRT-PCR). At the same time, we constructed a DN model by culturing NRK-52E cells with high glucose to detect the molecular mechanisms.
RESULTS: The kidney function of the DN group was significantly decreased, SCr and BUN levels were significantly increased, and the renal structure under the microscope was disordered, and interstitial edema was obvious. The expression of SOD1, SOD2, GPX1, and GPX3 were significantly decreased, and the level of reactive oxygen species (ROS) was significantly increased. PQQ treatment can effectively alleviate renal function, improve structural damage, and inhibit OS. In vivo, PQQ can effectively inhibit high glucose-induced OS damage and activate the AMPK/FOXO3a signaling pathway.
CONCLUSIONS: PQQ improves renal structural damage and functional damage, and protects kidney cells in DN by inhibiting OS, which may be related to activating the AMPK/FOXO3a pathway.
Keywords: PQQ Cofactor, adenylate kinase, Blood Glucose, Blood Urea Nitrogen, Creatinine, Diabetes Mellitus, Experimental, Forkhead Box Protein O3, Free Radical Scavengers, Kidney, Random Allocation, Real-Time Polymerase Chain Reaction, superoxide dismutase-1
Background
Diabetic nephropathy (DN) is one of the chronic microvascular complications of diabetes. In China, 30% to 50% of diabetic patients have kidney disease [1]. Early DN is characterized by microalbuminuria and progressive progression to large amounts of albuminuria, as well as elevated levels of serum creatinine (SCr) and blood urea nitrogen (BUN) levels, which eventually can progress to renal failure. Dialysis or kidney transplantation is required in patients with renal failure [2]. Clinically, effective interventions can prevent or delay the progression of DN, so it is essential to research the pathogenesis of DN. Researches have shown that the number of podocytes and tubular epithelial cells is reduced, and structural or functional integrity disruption is as-sociated with proteinuria production [3]. Therefore, studying tubular epithelial cells injury caused by oxidative stress (OS) may provide new ideas for the intervention of DN.
PQQ was first identified as a novel cofactor for ethanol and glucose dehydrogenase in methylotrophic bacteria and is currently considered to be an important vegetative growth factor [4]. Researchers have found that PQQ has a chemical molecular formula of C14H6N2O8 and a relative molecular mass of 330.21. It is widely distributed in plants, bacteria, animals, foods and many biological fluids. PQQ has water solubility and thermal stability, and it exists in the form of oxidation and reduction [5]. Anti-OS of PQQ has become a research hotspot in recent years. PQQ has been proven to have neuroprotective, cardiovascular, and anti-tumor effects as antioxidants [6]. Research has shown that PQQ, as a scavenger of reactive oxygen species (ROS) in OS, can continuously neutralize with ROS
AMPK (adenosine 5′-monophosphate-activated protein kinase) coordinates the survival and function of cells in various organs, including the kidneys. AMPK is a heterotrimer composed of a catalytic subunit a and 2 regulatory subunits: b and Y. It is a widely expressed and a highly conserved serine/threonine protein kinase [8]. FOXO3a (forkhead box protein O3a) plays an important role in regulating OS, cell differentiation, proliferation, metabolism, apoptosis, and repairing damaged DNA and prolonging the lifespan of the body [9]. The activity of FOXO3a transcriptional regulator is mainly regulated by post-translational modification, with phosphorylation/dephosphorylation being most common. FOXO3a can bind to the promoters of various genes, thereby activating the superoxide dismutase (SOD) gene against OS [10], thereby regulating cell protection from OS damage. This research focuses on the inhibition of OS damage in DN by PQQ via the AMPK/FOXO3a pathway.
Material and Methods
REAGENT:
Reagents used included Dulbecco’s Modified Eagle’s Medium (DMEM; Life Technology, Wuhan, China), fetal bovine serum (FBS) (Life Technology, Wuhan, China), dimethylammonium (DMSO, Hualianke Biotechnology Wuhan, China), 0.1% trypsin (Huagao Pharmaceutical, Chengdu, China), and PQQ (Panball Biotechnology, Beijing, China) which was dissolved in physiological saline and diluted with DMEM before use.
EXPERIMENTAL ANIMALS:
In this study, Sprague Dawley rats (Wuhan University Experimental Animal Center, Wuhan, China), 6 to 8 weeks old and weighing 270±30 g, were raised by the SPF laboratory animal center of Wuhan University. The average temperature in the animal center is 20±2°C, relative humidity 50% to 70%, day and night cycle. Feed pellets and keep the cage clean. The animal experiment strictly follows the regulations of animal experiment management of Wuhan University and was reviewed by the Animal Experiment Ethics Committee.
ANIMAL MODEL:
DN model rats were established. All rats were fasted overnight for 16 hours, then one-time intraperitoneal injection of 60 mg/kg streptozocin (STZ, Qcbic, Shanghai, China) was given; normal rats were injected with citrate buffer. The injection method and the injection volume were consistent with the DN group of rats. After 3 days, the tail vein blood of the modeled rats was collected, and the blood glucose level was detected. The blood glucose >16.7 mmol/L was the successful standard for modeling the diabetes model. After successful modeling of diabetes model, the rats were fed for another 8 weeks. The urine volume of diabetic rats was collected for 24 hours, and the 24-hour urine protein level was detected. The 24-hour urine volume was 150% before modeling; 24-hour urine protein >30 mg was regarded as the DN model successfully established. After establishing the successful model, the DN rats were randomly divided into a DN group (10 rats) and a PQQ group (10 rats). The rats in the PQQ group were fed with PQQ for 4 weeks, and the control group and the DN group were fed with normal diet.
TISSUE PREPARATION:
The rats in each group were sacrificed immediately after anesthesia, and bilateral kidney tissues were taken. A mixture of formaldehyde that could preserve enzyme activity and tissue antigenicity (2% paraformaldehyde, 75 mmol/L lysine, 10 mmol/L sodium periodate, par-aldehyde, lysine, sodium periodate, periodate-lysine-paraformaldehyde (PLP), Wuhan University, Wuhan, China) were fixed in fixed solution, routinely dehydrated, embedded in paraffin, and sectioned for subsequent staining.
CELL CULTURE AND PROCESSING:
NRK-52E cells (Cell Culture Center, Shanghai, China) were cultured in DMEM medium containing 10% FBS, 100 U/mL penicillin, 100 U/mL streptomycin, incubator at 37°C, 5% CO2, 100% humidity, and entered logarithmic growth phase at 24 hours. Inoculated after about 72 hours, the cells were overgrown, and the adherent cells were passaged by direct pipetting with 0.1% trypsin. The cells were divided into control group, high glucose group (glucose: 30 mmol×L−1), high glucose+PQQ group (PQQ: 50 μmol×L−1).
CELL COUNTING KIT-8 (CCK-8) ASSAY:
The optimal concentration of PQQ and the optimal treatment time were determined using the Cell Counting Kit-8 (CCK-8) (Construction, Nanjing, China). NRK-52E cells were seeded in 96-well plates, and the cells were cultured with different concentrations of PQQ for 0.5 hours, 1 hour, 2 hours, and 3 hours. Finally, the absorbance at 450 nm was measured using a microplate reader.
HEMATOXYLIN AND EOSIN (H&E) STAINING:
Paraffin sections were routinely dewaxed and hydrated, stained with hematoxylin (Sigma, St. Louis, MO, USA), 1% hydrochloric acid alcohol differentiated, rinsed with water, and returned to blue, then counterstained with eosin (Sigma, St. Louis, MO, USA) and then the slide rinsed with running water, and finally seal the slide with a neutral gel.
IMMUNOFLUORESCENCE:
The kidney tissues were fixed, dehydrated, and embedded in paraffin. After dewaxing, the endogenous peroxidase was extinguished with 3% H2O2, washed with phosphate-buffered saline (PBS), and then blocked with 10% goat serum. The tissue was stained with 8-OHdG (Abcam, Cambridge, MA, USA, Rabbit, 1: 1000), which reflects the DNA damage index of renal tissue. The next day was observed under a fluorescence microscope.
BIOCHEMICAL INDICATORS:
Appropriate amount of serum, tissue and cell supernatant were extracted, and SOD, lactate dehydrogenase (LDH) and catalase (CAT) activities were detected after the working fluid was configured according to the kit instructions (Jiancheng, Nanjing, China).
FLOW CYTOMETRY TO DETECT ROS LEVELS:
The kidney tissues of the three groups of rats were taken, Total ROS levels were measured by flow cytometry (Becton Dickinson, Heidelberg, Germany) at 37°C for 20 minutes using DCFH-DA (10 μM Kaiji, Nanjing, China).
WESTERN BLOT:
The radioimmunoprecipitation assay (RIPA) protein lysate (Beyotime, Shanghai, China) on the ice lysed the cells and tissues, vortexed once every 5 minutes, lysed for 20 minutes, centrifuged at 4°C, 15 000 rpm for 30 minutes, and the supernatant was aspirated to obtain total protein. After bicinchoninic acid (BCA) method (Beyotime, Shanghai, China), the protein was loaded, and 10% sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) gel electrophoresis was carried out for 2.5 hours. After the membrane transfer, the NC membrane was washed with tris-buffered saline (TBS) slightly, 5% skim milk powder was blocked at room temperature for 1 hour, and the primary antibodies (SOD1, Abcam, Cambridge, MA, USA, rabbit, 1: 3000; SOD2, Abcam, Cambridge, MA, USA, rabbit, 1: 3000; GPX1, Abcam, Cambridge, MA, USA, mouse, 1: 2000; GPX3, Abcam, Cambridge, MA, USA, mouse, 1: 2000; collagen I, Abcam, Cambridge, MA, USA, rabbit, 1: 2000; AMPK, Abcam, Cambridge, MA, USA, rabbit, 1: 5000; p-AMPK, Abcam, Cambridge, MA, USA, rabbit, 1: 5000; FOXO3a, Abcam, Cambridge, MA, USA, rabbit, 1: 500; GAPDH, Proteintech, Rosemont, IL, USA, 1: 5000) were incubated at 4°C overnight, tris buffered saline-tween (TBST) was washed 10 minutes for 3 times, the secondary antibody (goat anti-rabbit IgG antibody, Yifei Xue Biotechnology, Nanjing, China, 1: 3000) was incubated for 1 hour at room temperature, washed for 10 minutes at TBST, repeated 3 times, exposed and developed; gray value analysis was performed using BIO-RAD Image Lab software (Bio-Rad, Hercules, CA, USA).
EXTRACT RNA AND QUANTITATIVE REAL-TIME POLYMERASE CHAIN REACTION (QRT-PCR):
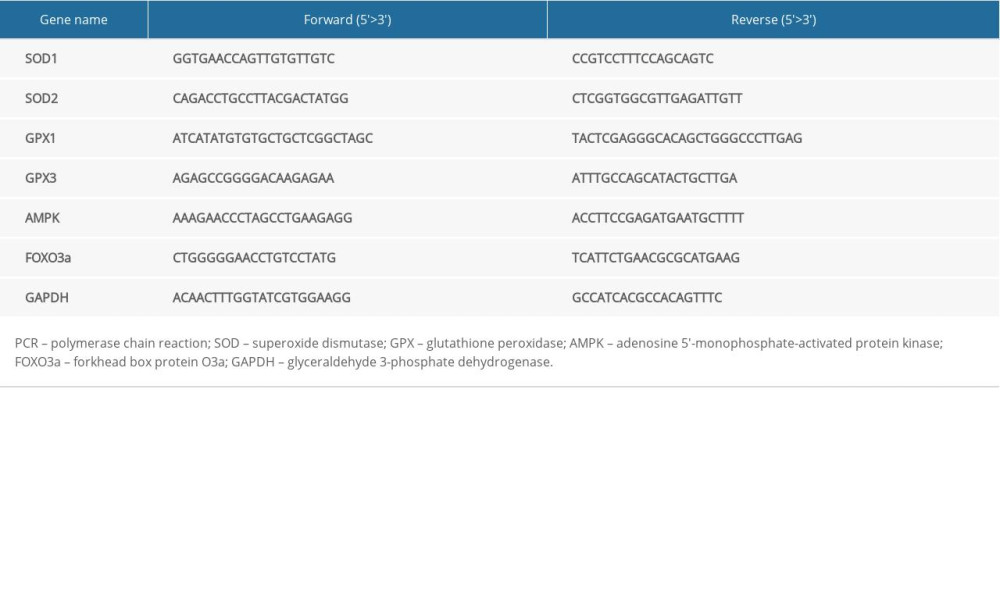
The cells were collected, total RNA was extracted by TRIzol reagent (Thermo Fisher Scientific, Waltham, MA, USA), and cDNA was reverse transcribed. The polymerase chain reaction (PCR) amplification conditions were denaturation at 95°C for 5 minutes, 95°C for 5 seconds, 60°C for 35 seconds, 72°C for 20 seconds, for 40 cycles, and extension at 72°C for 5 minutes. The amount of target gene expression was calculated by the 2−ΔΔCt method. Primers were designed using Primer 6.0 software. Primers used are shown in Table 1.
ENZYME-LINKED IMMUNOSORBENT ASSAY (ELISA):
The kidney tissues and NRK-52E cells were taken from each treatment, and the contents of malondialdehyde (MDA) and glutathione peroxidase (GSH-Px) were detected with the ELISA kit (Elabscience, Wuhan, China) according to the manufacturer’s instructions.
STATISTICAL ANALYSIS:
The tissue staining images were analyzed by Image Pro Plus (Silver Springs, MD, USA), image processing and analysis software. Percentage of total collagen positive area and percentage of 8-OHdG positive cells using Statistical Product and Service Solutions (SPSS) 11.0 software (SPSS Inc., Chicago, IL, USA) and Image analysis software. Comparison between multiple groups was done using one-way ANOVA test followed by post hoc test (least significant difference). The difference was statistically significant with
Result
:
Using serum, we tested BUN, SCr and blood glucose content. The results suggest that renal function impairment in DN group: BUN, SCr and blood glucose were significantly increased, while PQQ significantly improved renal function (Figure 1A–1C). At the same time, we detected the expression of collagen I by western blot, and found that the expression of collagen I was significantly increased in the DN group, and the expression of collagen I in the PQQ group was significantly decreased (Figure 1D). H&E staining results showed that in DN group the glomeruli were significantly hypertrophied and the interstitial edema was severe, while PQQ treatment significantly improved renal structural changes. T-col staining also confirmed that renal interstitial fibrosis was evident in the DN group (Figure 1E), but PQQ treatment can effectively relieve renal fibrosis.
:
In vivo, the expression of GPX1 (glutathione peroxidase 1) and GPX3 protein in DN group was significantly decreased, while PQQ treatment significantly inhibited the decrease of GPX1 and GPX3 (Figure 2A). The qRT-PCR results showed that GPX1 mRNA and GPX3 mRNA levels were the same as before (Figure 2B, 2C). In addition, we measured plasma SOD, LDH and MDA levels. It was found that PQQ can significantly promote the increase of SOD content and inhibit the increase of plasma LDH and MDA levels (Figure 2D–2F). The results of flow cytometry also confirmed that the ROS level in the PQQ group was significantly lower than that in the DN group (Figure 2G). In addition, we detected the expression of 8-OHdG by immunofluorescence, and found that the total amount of fluorescence in the DN group was significantly higher than that in the control group, and PQQ treatment was effective in reducing the total amount of fluorescence (Figure 2H).
:
First, we measured the optimal drug concentration and time of PQQ for NRK-52E cells by CCK-8 assay. It was found that when the PQQ concentration was 50 μmol×L−1 and the treatment time was 2 hours, the NRK-52E cell proliferation rate was the highest (Figure 3A). Secondly, the expression of SOD1 and SOD2 protein was detected by western blot, and the results also confirmed that PQQ treatment can effectively alleviate the oxidative damage of NRK-52E cells induced by high glucose (Figure 3B, 3C). Similar results were obtained with the qRT-PCR (Figure 3D, 3E). And we found that the content of CAT and GSH-Px in the high glucose group could be reduced, while the PQQ treatment could significantly promote the expression of CAT and GSH-Px (Figure 3F, 3G). Therefore, we speculate that PQQ can alleviate OS caused by high glucose
:
The results of western blot showed that the expression of AMPK in high glucose group and PQQ group was not significantly different from that in the control group, while high glucose treatment of NRK-52E cells could inhibit the expression of p-AMPK, and the expression of p-AMPK was significantly increased after PQQ treatment. Then, we examined the downstream FOXO3a, and PQQ treatment can effectively promote FOXO3a expression (Figure 4A). The p-AMPK/AMPK results confirmed that PQQ treatment significantly inhibited the high glucose-induced ratio reduction (Figure 4B). The qRT-PCR results also confirmed that high glucose can inhibit FOXO3a mRNA, and PQQ treatment can effectively alleviate this phenomenon, but there was no significant difference in AMPK mRNA among the three groups (Figure 4C, 4D). These results suggest that PQQ activates the AMPK/FOXO3a pathway.
Discussion
As early as 2001, Giacco and Brownlee proposed that OS is a common pathogenesis of diabetes and its complications [11]. Under high glucose stimulation, the cells produce a large amount of advanced glycation end products (AGES), which accumulate inside the cells. During the production of AGES, mitochondria also release a large amount of ROS. Excess ROS breaks the balance between the body’s oxidation system and the antioxidant system, and damages cells [12]. OS can directly lead to podocyte apoptosis, or by rearranging actin, increasing endothelin synthesis, altering microvascular permeability and ultimately leading to podocyte injury [13]. Our results also confirmed that in DN rats, the kidney tissues ROS levels were significantly elevated, and PQQ treatment was effective in inhibiting ROS elevation.
The damage of free radicals to cells is one of the causes of promoting kidney disease. Oxidative free radicals (OFR) have strong oxidizing ability, which can cause peroxidation of unsaturated lipids in biofilms to form lipid peroxides. The final product of MDA is strong. The cross-linking agent forms a poorly soluble substance with a protein, a nucleic acid or a lipid, hardens the biofilm, causes a decrease in permeability, affects the exchange of cellular substances, and then causes it to rupture and die. OFR can attack specific receptor proteins on biofilms, causing them to undergo oxidative degeneration, resulting in reduced numbers and reduced sensitivity [14]. Our results also confirmed that MDA and LDH levels were significantly increased in DN rats, while PQQ was effective in clearing ROS, thereby reducing lipid oxidation and reducing MDA and LDH levels. The results of 8-OHdG fluorescence confirmed that the expression of 8-OHdG was significantly increased after high glucose treatment, and PQQ treatment can effectively alleviate this phenomenon.
SOD is an important metalloenzyme for the removal of superoxide anion radicals (O2−) in living organisms. SOD is the number one killer of OFR in the body, and has anti-inflammatory, anti-oxidation, and anti-apoptosis effects [15]. It can be divided into 2 groups from the structure: CuZu-SOD is the first group, and Mn-SOD and Fe-SOD are the second group. Our results have been confirmed both
Researches have reported that AMPK/FOXO3a has a regulatory effect on OS [16]. AMPK activation can reduce the expression of adhesion molecules, reduce inflammatory cell migration and adhesion, play an anti-inflammatory effect [17]. FOXO3a binds to the promoters of various genes, thereby activating the Mn-SOD gene against OS, thereby protecting cells from OS. Mn-SOD is a tetrameric protein with a relative molecular mass of 88 600. It is an important antioxidant enzyme that is encoded by the SOD2 gene and exists in the mitochondria of cells to maintain normal function and survival [18]. OFR produced by mitochondria during metabolism and cytokines such as TNF-a, IL-1b, and IFN-g have significant effects on the expression of Mn-SOD [19]. Our results also found that PQQ treatment can promote AMPK phosphorylation, thereby activating FOXO3a, and FOXO3a can bind to its downstream promoter, thereby activating the increase of SOD gene expression, thereby protecting NRK-52E cells from OS damage and resisting OS. However, this study has not studied the inhibition of high glucose-induced inflammatory response by PQQ through the AMPK pathway, and the induction of Mn-SOD by related inflammatory factors.
Conclusions
PQQ improvs renal structural damage and functional damage, and protecting kidney cells in DN by inhibiting OS, which may be related to activating the AMPK/FOXO3a pathway. This provides great value for the treatment of DN.
Figures
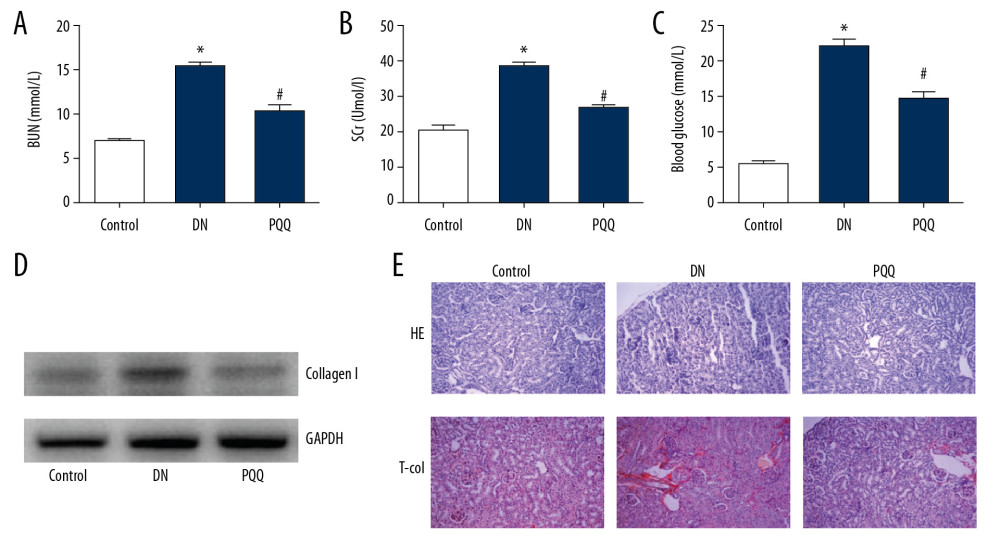 Figure 1. PQQ inhibits the structural and functional changes induced by STZ in DN. (A) Serum BUN. (B) Serum SCr. (C) Blood glucose. (D) Western blot detects collagen I expression. (E) H&E and T-col staining (magnification: 200×). * Compared with the control group, P<0.05; # Compared with the DN group, P<0.05. PQQ – pyrroloquinoline quinone; STZ – streptozocin; DN – diabetic nephropathy; BUN – blood urea nitrogen; SCr – serum creatinine; H&E – hematoxylin and eosin.
Figure 1. PQQ inhibits the structural and functional changes induced by STZ in DN. (A) Serum BUN. (B) Serum SCr. (C) Blood glucose. (D) Western blot detects collagen I expression. (E) H&E and T-col staining (magnification: 200×). * Compared with the control group, P<0.05; # Compared with the DN group, P<0.05. PQQ – pyrroloquinoline quinone; STZ – streptozocin; DN – diabetic nephropathy; BUN – blood urea nitrogen; SCr – serum creatinine; H&E – hematoxylin and eosin. 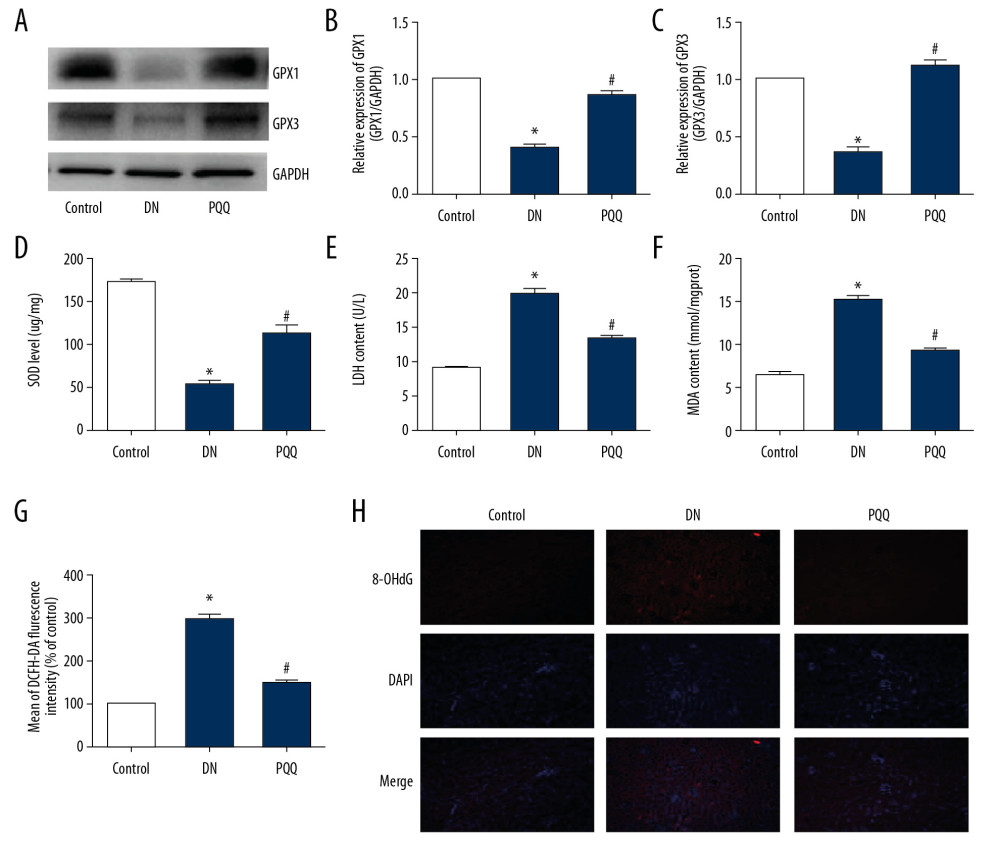 Figure 2. PQQ inhibits OS induced by STZ in DN. (A) Western blot detects GPX1 and GPX3 expression. (B) GPX1 mRNA. (C) GPX3 mRNA. (D) SOD content. (E) LDH content. (F) MDA content. (G) Flow cytometry was used to detect ROS levels. (H) Immunofluorescence detection of 8-OHdG expression (magnification: 200×). * Compared with the control group, P<0.05; # Compared with the DN group, P<0.05. PQQ – pyrroloquinoline quinone; OS – oxidative stress; STZ – streptozocin; DN – diabetic nephropathy; GPX – glutathione peroxidase; mRNA – messenger RNA; SOD – superoxide dismutase; LDH – lactate dehydrogenase; MDA – malondialdehyde; ROS – reactive oxygen species.
Figure 2. PQQ inhibits OS induced by STZ in DN. (A) Western blot detects GPX1 and GPX3 expression. (B) GPX1 mRNA. (C) GPX3 mRNA. (D) SOD content. (E) LDH content. (F) MDA content. (G) Flow cytometry was used to detect ROS levels. (H) Immunofluorescence detection of 8-OHdG expression (magnification: 200×). * Compared with the control group, P<0.05; # Compared with the DN group, P<0.05. PQQ – pyrroloquinoline quinone; OS – oxidative stress; STZ – streptozocin; DN – diabetic nephropathy; GPX – glutathione peroxidase; mRNA – messenger RNA; SOD – superoxide dismutase; LDH – lactate dehydrogenase; MDA – malondialdehyde; ROS – reactive oxygen species. 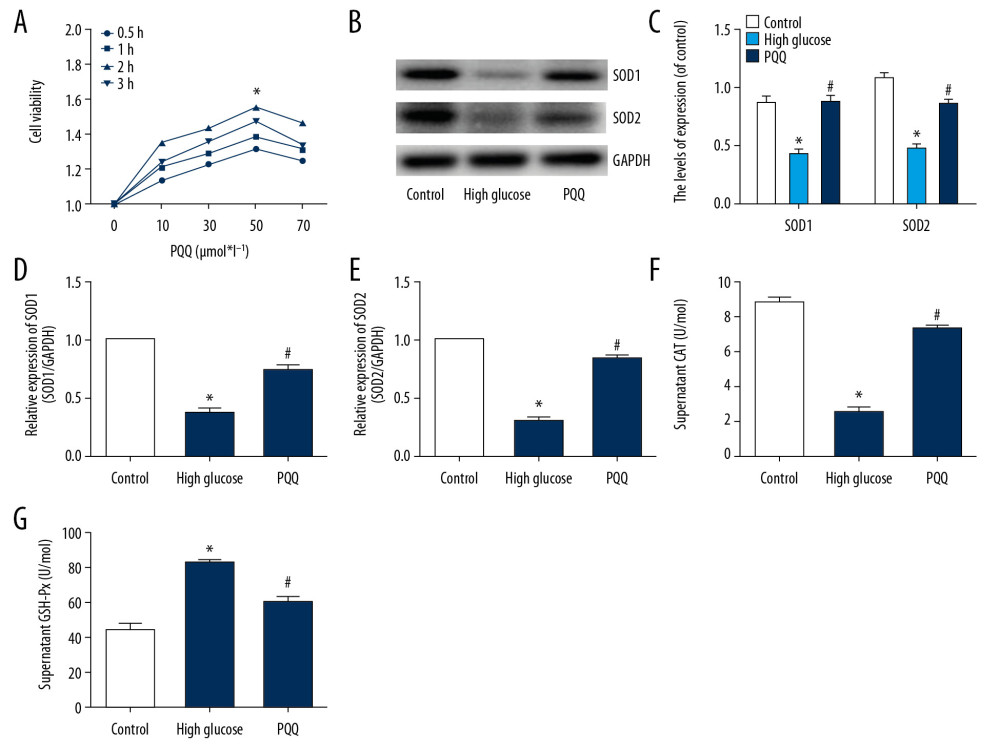 Figure 3. PQQ inhibits high glucose-induced OS damage in NRK-52E cells. (A) CCK-8 method for detecting optimal culture concentration and time of PQQ. (B) Western blot detects SOD1 and SOD2 expression. (C) Protein analysis. (D) SOD1 mRNA. (E) SOD2 mRNA. (F) CAT activity. (G) GSH-Px content. * Compared with the control group, P<0.05; # Compared with the high glucose group, P<0.05. PQQ – pyrroloquinoline quinone; OS – oxidative stress; CCK-8 – Cell Counting Kit-8; SOD – superoxide dismutase; mRNA – messenger RNA; CAT – catalase; GSH-Px – glutathione peroxidase.
Figure 3. PQQ inhibits high glucose-induced OS damage in NRK-52E cells. (A) CCK-8 method for detecting optimal culture concentration and time of PQQ. (B) Western blot detects SOD1 and SOD2 expression. (C) Protein analysis. (D) SOD1 mRNA. (E) SOD2 mRNA. (F) CAT activity. (G) GSH-Px content. * Compared with the control group, P<0.05; # Compared with the high glucose group, P<0.05. PQQ – pyrroloquinoline quinone; OS – oxidative stress; CCK-8 – Cell Counting Kit-8; SOD – superoxide dismutase; mRNA – messenger RNA; CAT – catalase; GSH-Px – glutathione peroxidase. 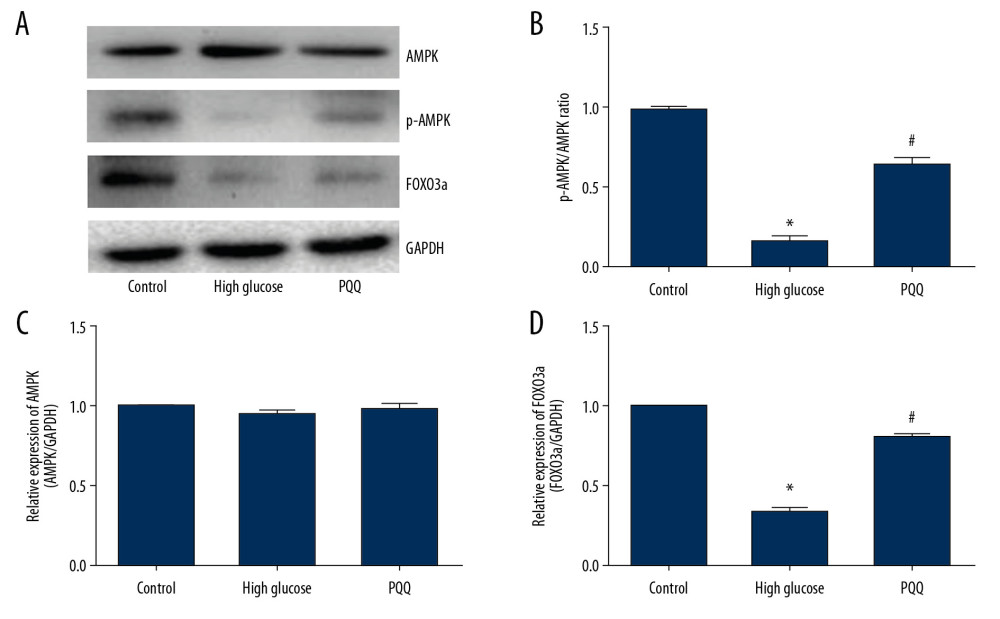 Figure 4. PQQ activates AMPK/FOXO3a pathway. (A) Western blot detects AMPK, p-AMPK, and FOXO3a expression. (B) p-AMPK/AMPK ratio. (C) AMPK mRNA. (D) FOXO3a mRNA. * Compared with the control group, P<0.05, # Compared with the high glucose group, P<0.05. PQQ – pyrroloquinoline quinone; AMPK – adenosine 5′-monophosphate-activated protein kinase; FOXO3a – forkhead box protein O3a; mRNA – messenger RNA.
Figure 4. PQQ activates AMPK/FOXO3a pathway. (A) Western blot detects AMPK, p-AMPK, and FOXO3a expression. (B) p-AMPK/AMPK ratio. (C) AMPK mRNA. (D) FOXO3a mRNA. * Compared with the control group, P<0.05, # Compared with the high glucose group, P<0.05. PQQ – pyrroloquinoline quinone; AMPK – adenosine 5′-monophosphate-activated protein kinase; FOXO3a – forkhead box protein O3a; mRNA – messenger RNA. References
1. Liu WJ, Huang WF, Ye L, The activity and role of autophagy in the pathogenesis of diabetic nephropathy: Eur Rev Med Pharmacol Sci, 2018; 22; 3182-89
2. Alicic RZ, Rooney MT, Tuttle KR, Diabetic kidney disease: Challenges, progress, and possibilities: Clin J Am Soc Nephrol, 2017; 12; 2032-45
3. Bose M, Almas S, Prabhakar S, Wnt signaling and podocyte dysfunction in diabetic nephropathy: J Investig Med, 2017; 65; 1093-101
4. Akagawa M, Nakano M, Ikemoto K, Recent progress in studies on the health benefits of pyrroloquinoline quinone: Biosci Biotechnol Biochem, 2016; 80; 13-22
5. Hartmann C, Klinman JP, Pyrroloquinoline quinone: A new redox cofactor in eukaryotic enzymes: Biofactors, 1988; 1; 41-49
6. Rucker R, Chowanadisai W, Nakano M, Potential physiological importance of pyrroloquinoline quinone: Altern Med Rev, 2009; 14; 268-77
7. Sasakura H, Moribe H, Nakano M: J Cell Sci, 2017; 130; 2631-43
8. Ke R, Xu Q, Li C, Mechanisms of AMPK in the maintenance of ATP balance during energy metabolism: Cell Biol Int, 2018; 42; 384-92
9. Sun W, Qiao W, Zhou B, Overexpression of Sirt1 in mesenchymal stem cells protects against bone loss in mice by FOXO3a deacetylation and oxidative stress inhibition: Metabolism, 2018; 88; 61-71
10. Asadi S, Rahimi Z, Saidijam M, Effects of resveratrol on FOXO1 and FOXO3a genes expression in adipose tissue, serum insulin, insulin resistance and serum SOD activity in type 2 diabetic rats: Int J Mol Cell Med, 2018; 7; 176-84
11. Giacco F, Brownlee M, Oxidative stress and diabetic complications: Circ Res, 2010; 107; 1058-70
12. Sahajpal NS, Goel RK, Chaubey A, Pathological perturbations in diabetic retinopathy: Hyperglycemia, AGEs, oxidative stress and inflammatory pathways: Curr Protein Pept Sci, 2019; 20; 92-110
13. Raij L, Tian R, Wong JS, Podocyte injury: The role of proteinuria, urinary plasminogen, and oxidative stress: Am J Physiol Renal Physiol, 2016; 311; F1308-17
14. Bai M, Che R, Zhang Y, Reactive oxygen species-initiated autophagy opposes aldosterone-induced podocyte injury: Am J Physiol Renal Physiol, 2016; 310; F669-78
15. Offer T, Russo A, Samuni A, The pro-oxidative activity of SOD and nitroxide SOD mimics: FASEB J, 2000; 14; 1215-23
16. Kim AD, Kang KA, Piao MJ, Cytoprotective effect of eckol against oxidative stress-induced mitochondrial dysfunction: Involvement of the FOXO3a/AMPK pathway: J Cell Biochem, 2014; 115; 1403-11
17. Sharma G, Kar S, Palit S, Das PK, 18beta-glycyrrhetinic acid induces apoptosis through modulation of Akt/FOXO3a/Bim pathway in human breast cancer MCF-7 cells: J Cell Physiol, 2012; 227; 1923-31
18. Sas K, Szabo E, Vecsei L, Mitochondria, oxidative stress and the kynurenine system, with a focus on ageing and neuroprotection: Molecules, 2018; 23 pii: E191
19. Lugrin J, Rosenblatt-Velin N, Parapanov R, Liaudet L, The role of oxidative stress during inflammatory processes: Biol Chem, 2014; 395; 203-30
Figures
 Figure 1. PQQ inhibits the structural and functional changes induced by STZ in DN. (A) Serum BUN. (B) Serum SCr. (C) Blood glucose. (D) Western blot detects collagen I expression. (E) H&E and T-col staining (magnification: 200×). * Compared with the control group, P<0.05; # Compared with the DN group, P<0.05. PQQ – pyrroloquinoline quinone; STZ – streptozocin; DN – diabetic nephropathy; BUN – blood urea nitrogen; SCr – serum creatinine; H&E – hematoxylin and eosin.
Figure 1. PQQ inhibits the structural and functional changes induced by STZ in DN. (A) Serum BUN. (B) Serum SCr. (C) Blood glucose. (D) Western blot detects collagen I expression. (E) H&E and T-col staining (magnification: 200×). * Compared with the control group, P<0.05; # Compared with the DN group, P<0.05. PQQ – pyrroloquinoline quinone; STZ – streptozocin; DN – diabetic nephropathy; BUN – blood urea nitrogen; SCr – serum creatinine; H&E – hematoxylin and eosin. Figure 2. PQQ inhibits OS induced by STZ in DN. (A) Western blot detects GPX1 and GPX3 expression. (B) GPX1 mRNA. (C) GPX3 mRNA. (D) SOD content. (E) LDH content. (F) MDA content. (G) Flow cytometry was used to detect ROS levels. (H) Immunofluorescence detection of 8-OHdG expression (magnification: 200×). * Compared with the control group, P<0.05; # Compared with the DN group, P<0.05. PQQ – pyrroloquinoline quinone; OS – oxidative stress; STZ – streptozocin; DN – diabetic nephropathy; GPX – glutathione peroxidase; mRNA – messenger RNA; SOD – superoxide dismutase; LDH – lactate dehydrogenase; MDA – malondialdehyde; ROS – reactive oxygen species.
Figure 2. PQQ inhibits OS induced by STZ in DN. (A) Western blot detects GPX1 and GPX3 expression. (B) GPX1 mRNA. (C) GPX3 mRNA. (D) SOD content. (E) LDH content. (F) MDA content. (G) Flow cytometry was used to detect ROS levels. (H) Immunofluorescence detection of 8-OHdG expression (magnification: 200×). * Compared with the control group, P<0.05; # Compared with the DN group, P<0.05. PQQ – pyrroloquinoline quinone; OS – oxidative stress; STZ – streptozocin; DN – diabetic nephropathy; GPX – glutathione peroxidase; mRNA – messenger RNA; SOD – superoxide dismutase; LDH – lactate dehydrogenase; MDA – malondialdehyde; ROS – reactive oxygen species. Figure 3. PQQ inhibits high glucose-induced OS damage in NRK-52E cells. (A) CCK-8 method for detecting optimal culture concentration and time of PQQ. (B) Western blot detects SOD1 and SOD2 expression. (C) Protein analysis. (D) SOD1 mRNA. (E) SOD2 mRNA. (F) CAT activity. (G) GSH-Px content. * Compared with the control group, P<0.05; # Compared with the high glucose group, P<0.05. PQQ – pyrroloquinoline quinone; OS – oxidative stress; CCK-8 – Cell Counting Kit-8; SOD – superoxide dismutase; mRNA – messenger RNA; CAT – catalase; GSH-Px – glutathione peroxidase.
Figure 3. PQQ inhibits high glucose-induced OS damage in NRK-52E cells. (A) CCK-8 method for detecting optimal culture concentration and time of PQQ. (B) Western blot detects SOD1 and SOD2 expression. (C) Protein analysis. (D) SOD1 mRNA. (E) SOD2 mRNA. (F) CAT activity. (G) GSH-Px content. * Compared with the control group, P<0.05; # Compared with the high glucose group, P<0.05. PQQ – pyrroloquinoline quinone; OS – oxidative stress; CCK-8 – Cell Counting Kit-8; SOD – superoxide dismutase; mRNA – messenger RNA; CAT – catalase; GSH-Px – glutathione peroxidase. Figure 4. PQQ activates AMPK/FOXO3a pathway. (A) Western blot detects AMPK, p-AMPK, and FOXO3a expression. (B) p-AMPK/AMPK ratio. (C) AMPK mRNA. (D) FOXO3a mRNA. * Compared with the control group, P<0.05, # Compared with the high glucose group, P<0.05. PQQ – pyrroloquinoline quinone; AMPK – adenosine 5′-monophosphate-activated protein kinase; FOXO3a – forkhead box protein O3a; mRNA – messenger RNA.
Figure 4. PQQ activates AMPK/FOXO3a pathway. (A) Western blot detects AMPK, p-AMPK, and FOXO3a expression. (B) p-AMPK/AMPK ratio. (C) AMPK mRNA. (D) FOXO3a mRNA. * Compared with the control group, P<0.05, # Compared with the high glucose group, P<0.05. PQQ – pyrroloquinoline quinone; AMPK – adenosine 5′-monophosphate-activated protein kinase; FOXO3a – forkhead box protein O3a; mRNA – messenger RNA. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387