04 March 2023: Clinical Research
Perfusate Neutrophil Gelatinase-Associated Lipocalin, Kidney Injury Molecular-1, Liver-Type Fatty Acid Binding Protein, and Interleukin-18 as Potential Biomarkers to Predict Delayed Graft Function and Long-Term Prognosis in Kidney Transplant Recipients: A Single-Center Retrospective Study
Zejia Sun1AEF, Zihao Gao1ACE, Xin Li1ABC, Xiang Zheng1BD, Wei Wang1AEG*, Peng Qiao1AEFDOI: 10.12659/MSM.938758
Med Sci Monit 2023; 29:e938758
Abstract
BACKGROUND: Delayed graft function (DGF) caused by ischemia-reperfusion injury is a common pathophysiological process that should be monitored by specific biomarkers in addition to serum creatinine. Thus, this single-center retrospective study aimed to investigate the association between levels of neutrophil gelatinase-associated lipocalin (NGAL), kidney injury molecular-1 (KIM-1), liver-type fatty acid binding protein (L-FABP), and interleukin-18 (IL-18) in DGF associated with acute kidney injury in kidney transplant recipients (KTRs) and estimated glomerular filtration rate (eGFR) at 3 years post-transplant.
MATERIAL AND METHODS: A total of 102 KTRs [14(13.7%) of DGF and 88(86.3%) of NON-DGF] were enrolled. DGF was defined as “dialysis is needed within 1 week after kidney transplantation”. NGAL, KIM-1, L-FABP, and IL-18 were obtained from perfusate samples of donation-after-cardiac-death (DCD) kidneys, and measured by ELISA.
RESULTS: Compared to the NON-DGF group, KTRs in the DGF group had a statistically significant increase in levels of NGAL (P<0.001) and KIM-1 (P<0.001). Multiple logistic regression analyses showed that NGAL (OR=1.204, 95% CI 1.057-1.372, P=0.005) and KIM-1 (OR=1.248, CI=1.065-1.463, P=0.006) could be regarded as independent risk factors. The accuracy of NGAL and KIM-1 was 83.3% and 82.1%, respectively, calculated using the area under the receiver operating characteristic curve. Furthermore, the eGFR at 3 years post-transplant had a moderate negative correlation with NGAL (r=-0.208, P=0.036) and KIM-1 (r=-0.260, P=0.008).
CONCLUSIONS: Our results support those from previous studies showing that perfusate levels of NGAL and KIM-1 are associated with DGF in KTRs and also with reduced eGFR at 3 years post-transplant.
Keywords: Biomarkers, Delayed Graft Function, Kidney Transplantation, Predictive Value of Tests, Prognosis, Humans, Interleukin-18, Lipocalin-2, Fatty Acid-Binding Proteins, Kidney, Liver
Background
Kidney transplantation is the best choice for treatment of end-stage kidney disease (ESKD). For patients with ESKD, this operation of can improve quality of life and help rebuild patient confidence and return to society [1], and the cost of long-term treatment is significantly lower than that of dialysis [2].
With the development of surgical techniques and immunotherapy, the success rate of surgery is significantly increased. Unfortunately, the incidence of delayed graft function (DGF) and long-term graft loss caused by ischemia-reperfusion injury (IRI) are still increasing [3]. One of the reasons for this is the expansion of the donor pool through increased donation-after-cardiac-death (DCD) in the last few decades [4]. Due to insufficient organ perfusion, infection, use of vasoactive drugs, chronic diseases, and other factors, transplanted kidneys often experience severe IRI, which is the main cause of DGF [5].
Multiple studies have shown that DGF can significantly reduce the long-term graft survival rate and will also lead to a higher rejection rate, prolonged hospitalization, and increased costs. Thus, predicting DGF at an early stage is of great importance. The perfect definition of DGF is still controversial. In general, most transplantation centers define DGF as follows: renal function has not recovered within 1 week and requires dialysis, or the average creatinine reduction rate within 3 days post-operative is less than 10% [6, 7]. However, this definition has some limitations. Serum creatinine may be influenced by factors such as the hemodynamics present or the presence of a urinary tract obstruction. Moreover, traditional diagnostic criteria cannot provide an early warning or diagnosis for DGF [8].
The most common cause of DGF is acute tubular necrosis (ATN) caused by IRI, but the diagnosis relies on renal biopsy, which has some risks, and many patients are unwilling to take the risk [9]. For a long time, transplantation doctors have been committed to exploring non-invasive diagnostic methods for the early detection of DGF. At present, kidney injury biomarkers are mainly collected from serum, urine, perfusate, and tissue [10]. Among them, serum and urine samples are greatly affected by the body’s general condition, and tissue samples need to be obtained by invasive puncture, so perfusate has a great advantage. According to previous studies, neutrophil gelatinase-associated lipocalin (NGAL), kidney injury molecular-1 (KIM-1), liver-type fatty acid binding protein (L-FABP), and interleukin-18 (IL-18) may be early biomarkers of acute kidney injury (AKI) [11]. NGAL is a 25-KD protein that is mainly expressed in the liver and kidney endothelial cells. The reactivity increases when there is infection, poisoning, or hypoxia. NGAL is filtered in the glomerulus and reabsorbed in the proximal tubule [12]. KIM-1 is a phosphatidylserine receptor that can recognize apoptotic cells and guide degradation. At the same time, KIM-1 can also convert proximal tubular endothelial cells into macrophages, which may be related to the immune response in kidney injury. IL-18 is an inflammation-mediator molecule that is mainly found in monocytes, fibroblasts, and proximal tubular epithelial cells. It can mediate ischemic renal tubular damage and participate in inflammation through Toll-like receptor 4 [13]. L-FABP is expressed in both the proximal and distal renal tubules and can be used to predict acute kidney injury [14].
NGAL, KIM-1, L-FABP, and IL-18 also have predictive value for the prognosis of kidney transplantation. A recent multi-center prospective study showed that urinary NGAL and IL-18 are early predictors of both the need for dialysis within the first week of kidney transplantation and 3-month recovery of graft function [15]. Cappuccilli et al [16] compared the predictive performance of NGAL levels in plasma, urine, and perfusate on DGF, and found that each one has limitations and benefits, which may be related to different cut-off levels, designs, and patient sample sizes. In addition, another prospective study evaluated the effect of urinary KIM-1, L-FABP, and NGAL on the prognosis of kidney transplantation, and found that urinary KIM-1 was a potent biomarker for the long-term prognosis of graft function [17].
Therefore, this retrospective study from a single center aimed to investigate the association between levels of NGAL, KIM-1, L-FABP, and IL-18 in delayed graft function (DGF) associated with AKI in kidney transplant recipients (KTRs) and estimated glomerular filtration rate (eGFR) at 3 years post-transplant.
Material and Methods
ETHICS STATEMENT:
Ethics approval for this study from the medical ethics committee boards of Beijing Chaoyang Hospital was received. According to the decision of the medical ethical committees, no informed consent was required. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
STUDY POPULATION:
We present a retrospective analysis of collected data. Patients who underwent their first kidney transplantation in Beijing Chaoyang Hospital from August 7, 2016 to July 30, 2018 and met the inclusion criteria were included in the study. All kidneys of DCD kidney transplantation in this study were procured from donation after citizens’ death, adhering to the guidelines of the National Program for Deceased Organ Donation in China. Data on donor characteristics, recipient characteristics, number of DGFs, and outcomes at the 3-year follow-up visit were obtained from the electronic medical records. Cohort generation is shown in Figure 1. The inclusion criteria were: (1) grafts obtained from DCD, (2) surgery performed as single-kidney transplantation for allografts with a single renal artery, and (3) recipients older than 16 years during the study period. The exclusion criteria were: (1) recipients who did not have at least 1 biomarker measured, and (2) recipients who lacked medical records for 3 years after surgery due to death or loss to follow-up. Patients were followed with blood test and urine analysis every 3 weeks for a period of 3 months, every 2 weeks thereafter until 9 months, and then monthly. DGF was defined as “dialysis is needed within 1 week after kidney transplantation,” and the diagnosis of DGF was assessed by 2 independent transplant surgeons. The eGFR was calculated using the abbreviated Modification of Diet in Renal Disease (MDRD) equation. The formula used for eGFR was as follows:
Serum creatinine reduction ratio (CRR) was defined as “the average percentage of creatinine reduction within 3 days after kidney transplantation as compared to the previous day.” The formula used for CRR was as follows:
PERFUSION PROCEDURE:
Perfusate samples were obtained from strictly controlled static perfusion solution. Histidine-tryptophan-ketoglutarate (HTK) solution was used as the perfusion preservation solution for kidney transplantation. At a targeted pressure of 100 cmH2O and targeted temperature of 4°C, HTK solution to which heparin was added (concentration: 20%) was perfused from the renal artery into the donor kidney. The total kidney perfusion volume was 250 ml, and the end of the perfusion was marked by the donor kidney turning pale and uniform, with clear perfusion solution flowing out of the renal vein [18,19].
Perfusate samples were collected at 2 time points: a 10 ml sample (defined as “base”) within minutes of starting perfusion and a second 10 ml sample (defined as “post”) just before the end of the perfusion. The timing of sample collections was recorded by the operator. Fresh perfusate samples were immediately centrifuged at 5000×g to remove insoluble elements. Aliquots of the supernatant were transported on ice and stored at −80°C without the addition of protease inhibitors until biomarker measurement [20,21].
BIOMARKER MEASUREMENT:
Serum creatinine levels were measured by standard enzymatic methods with an automated analyzer. Enzyme-linked immunosorbent assays (ELISA) were performed in duplicate using the NGAL ELISA kit (Abcam, Cambridge, MA, USA), the human TIM-1/KIM-1/HAVCR Quantikine ELISA kit (R&D System, Minneapolis, MN), the L-FABP human ELISA kit (Hycult, Biotech, Uden, The Netherlands), and the human IL-18 ELISA kit (Medical and Biological Laboratories, Nagoya, Japan). To standardize the biomarker measurement of perfusate sample, “post” concentration minus “base” concentration was defined as the final biomarker concentration used in our study.
STATISTICAL ANALYSIS:
Descriptive statistics were reported as the mean±SD or median (interquartile range) for continuous variables and as the frequency (percentage) for categorical variables. The
Results
THE BASELINE OF CHARACTERISTICS OF KTRS:
The baseline data of the 102 KTRs included in the study are shown in Table 1. Fourteen patients (13.7%) developed DGF after the kidney transplantation. The average age of the participants was 43.36±12.07 years, and the proportion of men was 68.6%. The average BMI was 23.32±3.77 kg/m2. Among these ESKD patients, 92.2% had hypertension, 15.7% had diabetes, and 82 patients received dialysis before transplantation, which was 80.4% of patients. At the time of the transplantation, the average serum creatinine of patients in the DGF group and NON-DGF group were 10.05±2.80 mg/dl and 8.80±3.08 mg/dl, respectively, but these were not statistically significant. The main causes of ESKD included glomerulonephritis (49.0%), hypertension (15.7%), diabetes (11.8%), polycystic kidney disease (5.9%), and others (17.6%). It should be noted that compared with the NON-DGF group, the DGF group tended to experience a longer cold ischemia time (8.33±1.70 h vs 6.42±1.79 h, P<0.001) and more HLA mismatches (P=0.044). However, there was no significant difference between the 2 groups in terms of PRA, pretransplant dialysis, allograft weight, perfusion time, or warm ischemia time. Supplementary Table 1 provides the baseline data of the 69 kidney transplant donors included in the study.
COMPARISON OF PERFUSATE BIOMARKER LEVELS BETWEEN DGF AND NON-DGF PATIENTS:
To visually show the difference between the perfusate biomarkers, we drew a box diagram (Figure 2, Table 2); perfusate NGAL and KIM-1 were higher in the DGF group, the level of NGAL in the 2 groups of patients was [DGF: 203.67±44.27 vs NON-DGF: 147.03±40.34, P<0.001], and the difference was statistically significant. The level of KIM-1 in the 2 groups of patients was [DGF: 190.86±42.94 vs NON-DGF: 135.27±39.38, P<0.001], and the difference was statistically significant. However, for L-FABP and IL-18, there were no significant differences between the DGF group and the NON-DGF group.
NGAL AND KIM-1 AS INDEPENDENT RISK FACTORS AMONG KTRS:
As shown in Table 3, according to the univariate logistic analyses, higher NGAL (OR=1.033, CI=1.016–1.051, P<0.001) and KIM-1 (OR=1.035, CI=1.016–1.054, P<0.001) are associated with DGF in KTRs. To further validate this hypothesis, multiple logistic regression analyses were used and showed that NGAL (OR=1.204, CI=1.057–1.372, P=0.005) and KIM-1 (OR=1.248, CI=1.065–1.463, P=0.006) could be regarded as independent risk factors. NGAL increased the risk of a DGF by 20.4% for each 1 ng/ml of NGAL elevation, while KIM-1 increased the risk of a DGF by 24.8% for each 1 of ng/ml KIM-1 elevation. Nevertheless, no evidence has proven that perfusate L-FABP and IL-18 are relevant to DGF.
PREDICTION EFFICIENCY FOR DGF AMONG PERFUSATE BIOMARKERS:
As we can see in Figure 3 and Table 4, ROC curves revealed that the perfusate NGAL had a sensitivity of 85.7% and a specificity of 79.5%, and the cutoff value was set at 175.94 ng/ml (P<0.001), while KIM-1 had a sensitivity of 85.7% and specificity of 68.2%, with the cutoff value set at 158.30 ng/ml (P<0.001).
Additionally, compared with the traditional serum creatinine reduction ratio (CRR), which has sensitivity of 78.57% and specificity of 95.45%, NGAL and KIM-1 had similar potential diagnostic values for DGF. However, the poor sensitivities or specificities of L-FABP and IL-18 implied that these 2 markers might not be recommended to predict DGF in KTRs (Table 5).
CORRELATION BETWEEN DGF AND LONG-TERM GRAFT OUTCOMES:
We conducted a 3-year follow-up of KTRs. Through a Spearman correlation analysis, we found that the 3-year graft survival rate of patients with DGF was significantly reduced (P=0.011). However, the 3-year eGFR and urine albumin were not affected by a DGF in our study (Table 6).
CORRELATION BETWEEN PERFUSATE BIOMARKERS AND LONG-TERM GRAFT FUNCTION:
As shown in Tables 7 and 8, Spearman correlations and linear regressions were performed to evaluate the prognostic value of all 4 perfusate biomarkers. NGAL and KIM-1 were positively correlated with long-term graft function in KTRs. Spearman correlation analyses demonstrated that NGAL decreased the 3-year eGFR by 0.208 ml/min/1.73 m2 for each 1 ng/ml of NGAL elevation (P=0.036) and that KIM-1 decreased the 3-year eGFR by 0.260 ml/min/1.73 m2 for each 1 ng/ml of KIM-1 elevation (P=0.008). In our study, L-FABP and IL-18 were negatively correlated with the long-term graft function, such as the 3-year eGFR, graft survival, and urine albumin.
Discussion
The most common typical pathological manifestation of DGF is ATN [22]. We tested 4 of the most promising biomarkers within ATN. NGAL is a marker of distal renal tubular injury, while KIM-1 is produced by proximal tubule cells. When various ischemia, hypoxia, infection, and nephrotoxic drugs destroy the renal tubular structure, the expression of NGAL and KIM-1 is significantly upregulated. At present, some medical centers use NGAL, L-FABP, KIM-1, and IL-18 for the clinical monitoring of acute kidney injury (AKI), but the research mostly focused on donor or recipient urine samples [23,24]. However, although urine specimens are easy to obtain, these biomarkers are easily affected by prerenal and postrenal factors, so they have certain limitations. For example, the patient’s urine volume can directly affect the detection level of the biomarkers, and any vasoactive drugs that are used before transplant surgery can affect the hemodynamics and cause prerenal and renal dysfunction [25]. Donors who have urinary tract obstructions may have inaccurate biomarker levels that are detected [26].
On the other hand, L-FABP and IL-18 provided little value in predicting DGF. This is similar to the conclusion drawn by Ntrinias [24,27], but some researchers believe that IL-18/L-FABP can predict DGF [7,14]. These contradictory conclusions may require large-sample multi-center prospective studies or meta-analyses for further evaluation. Our research found that DGF significantly reduces the 3-year graft survival rate. This result is the same as the general research conclusion. Molecular biology studies have shown that IRI in the early stage of kidney transplantation can cause chronic inflammatory cell infiltration, release of inflammatory cytokines, and microthrombosis, which lead to long-term graft dysfunction [28,29]. In our study, we also found that the expression of KIM-1 is related to 3-year eGFR. KIM-1 is responsible for the clearance of debris from damaged renal tubules. KIM-1 elevation often indicates that the structure of renal tubules is severely damaged [30]. Early kidney allograft injury leads to interstitial fibrosis, tubular atrophy, and glomerulosclerosis [31,32]. In addition, DGF is correlated with higher incidence of rejection. Studies have shown that it may be related to inflammatory cell aggregation and activation of the complement system during IRI [33].
In recent years, many biomarkers for predicting DGF have been evaluated [22,34]. Roest et al recommend testing miRNAs such as miR-505-3p in graft preservation fluid to assess DGF. Moser et al proposed that perfusate LDH, NGAL, and MMP-2 are good biomarkers of DGF. Moreover, biomarkers in the recipient’s urine that predict DGF and long-term prognosis after kidney transplantation include TRL-4, corin, amylase, IL-18, KIM-1, L-FABP, CC motif chemokine ligand 2 (CCL2), NGAL, cystatin C, KIM-1, T-cell immunoglobulin, mucin domain-containing protein 3 (TIM3), alpha-1 antitrypsin (A1AT), alpha-2 antiplasmin (A2AP), and serum amyloid A (SAA) [17,35–37]. We believe that the ideal DGF biomarker that should be used either before KTx in the donor or immediately after it in the recipient, and should have the following characteristics: non-invasive, easy to measure, short turn-around time, accurate, and cost-effective [12,38]. In summary, we selectively tested 4 biomarkers in the perfusate to evaluate DGF and the long-term graft prognosis.
Currently, only approximately 25% of ESKD patients can receive a kidney transplantation, and nearly 5% of ESKD patients die each year while waiting for a transplantation [24]. Therefore, an increasing number of expanded criteria donors are widely used to solve the problem of organ shortage, so DGF caused by IRI has become a major problem in the field of kidney transplantation [39]. An increasing number of studies have demonstrated that DGF is a critical risk factor for acute rejection, graft loss, 30-day readmission, and poor long-term graft function [27,39,40]. The current diagnostic criteria for DGF are still controversial [16] and have some limitations. Thus, non-invasive biomarkers for early detection of DGF are urgently needed.
Compared with the urine biomarkers of KTRs, perfusate biomarkers can provide an early warning of DGF before transplantation and can help guide doctors to make full preparations during the perioperative period. For example, using hemofiltration instead of hemodialysis can allow for the adjustment of immunosuppressive agents to prevent rejection or to delay the start of CNI [41,42].
We believe that this study provides additional evidence for perfusate biomarkers to predict DGF and long-term graft outcomes. On the one hand, perfusate NGAL and KIM-1 is a safe, reliable, non-invasive method for early warning of DGF and may be used to improve patient management strategies to alleviate the adverse effect of DGF. On the other hand, several studies have found a relationship between these 4 biomarkers and AKI, so they use them to define donor AKI instead of using biopsy [4,43,44]. But in our study, all 4 biomarkers were negatively correlated with 3-year graft survival, which means although the KTRs have a higher risk of DGF, transplant centers should not refuse to accept kidneys from people with AKI.
However, this study has some limitations: (1) This was a single-center retrospective study with a small sample size. (2) The included patients were all Chinese, which may have brought bias into the study due to the ethnicity and source of patients. (3) In China, most KTRs do not accept time-zero renal biopsy, so the pathological data of the donor kidney tissues that we could obtain are extremely limited. (4) In our study, only 2 samples were collected rather than multiple samples, and the most important time point at which to measure these 4 biomarkers was not standardized, which could not fully describe the kinetics of biomarker release during perfusion. (5) Several potential confounders, such as the volume of preservation fluid used before procurement and perfusion time, may have influenced the reliability of biomarker measurement. In addition, renal pathophysiology, including obstruction of the renal microcirculation caused by thrombosis, or edema can also lead to poor washout of biomarkers released from damaged cells, thus affecting the measurement of biomarker concentration. To solve these problems, we will expand the sample size in the future, conduct prospective studies with multiple centers, optimize the study design, and conduct stratified analyses to further improve the reliability of the results.
Conclusions
We found that perfusate NGAL and KIM-1 have high specificities and sensitivities. Studying the biological pathways or establishing DGF clinical risk prediction models are expected to help achieve early diagnosis of DGF and even replace the traditional definition of DGF, and also help to explore potential targets for new drugs.
In short, perfusate NGAL and KIM-1 could be potential biomarkers to predict DGF and 3-year eGFR in KTRs.
Figures
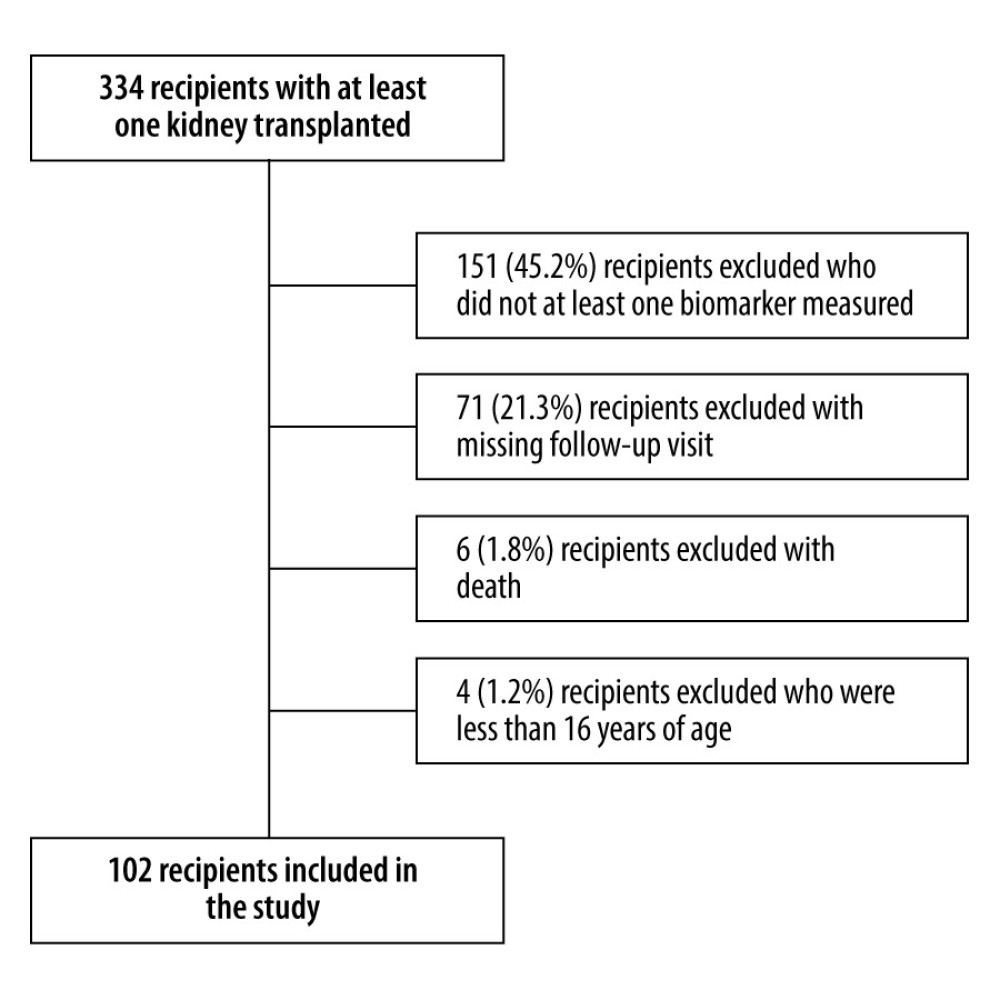 Figure 1. Enrollment of KTRs into the study cohort. KTRs: kidney transplant recipients. PowerPoint 2016, Microsoft.
Figure 1. Enrollment of KTRs into the study cohort. KTRs: kidney transplant recipients. PowerPoint 2016, Microsoft. 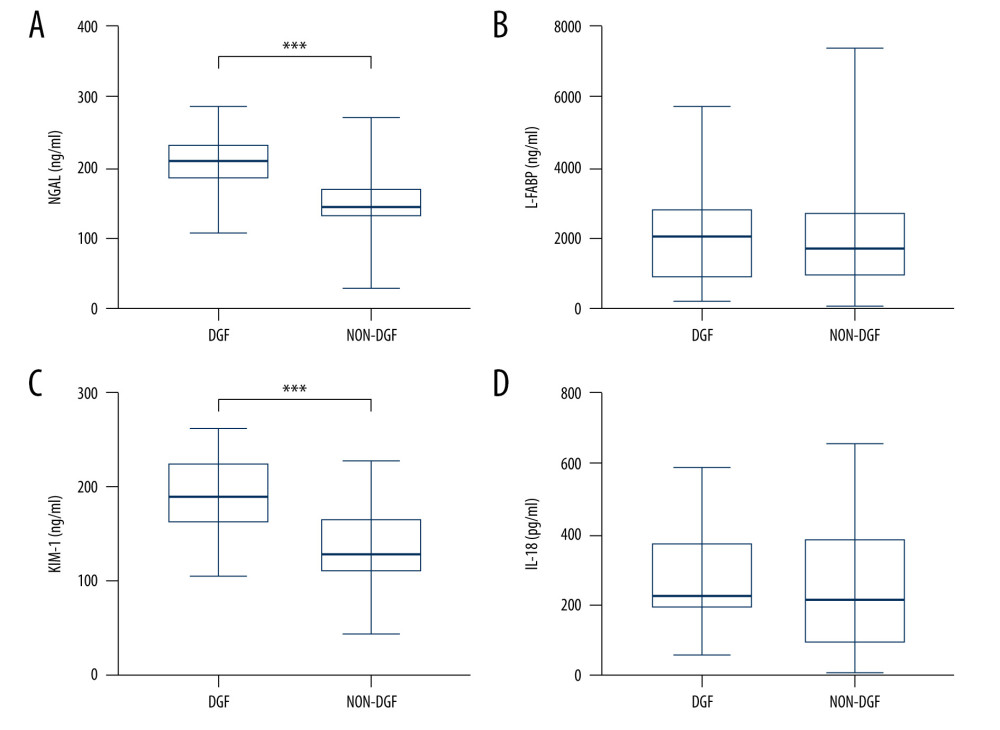 Figure 2. (A–D) Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF. DGF – delayed graft function; NGAL – neutrophil gelatinase-associated lipocalin; L-FABP – liver-type fatty acid binding protein; KIM-1 – kidney injury molecular-1; IL-18 – interleukin-18. GraphPad Prism 8, GraphPad Software, Adobe Illustrator 2022, Adobe.
Figure 2. (A–D) Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF. DGF – delayed graft function; NGAL – neutrophil gelatinase-associated lipocalin; L-FABP – liver-type fatty acid binding protein; KIM-1 – kidney injury molecular-1; IL-18 – interleukin-18. GraphPad Prism 8, GraphPad Software, Adobe Illustrator 2022, Adobe. 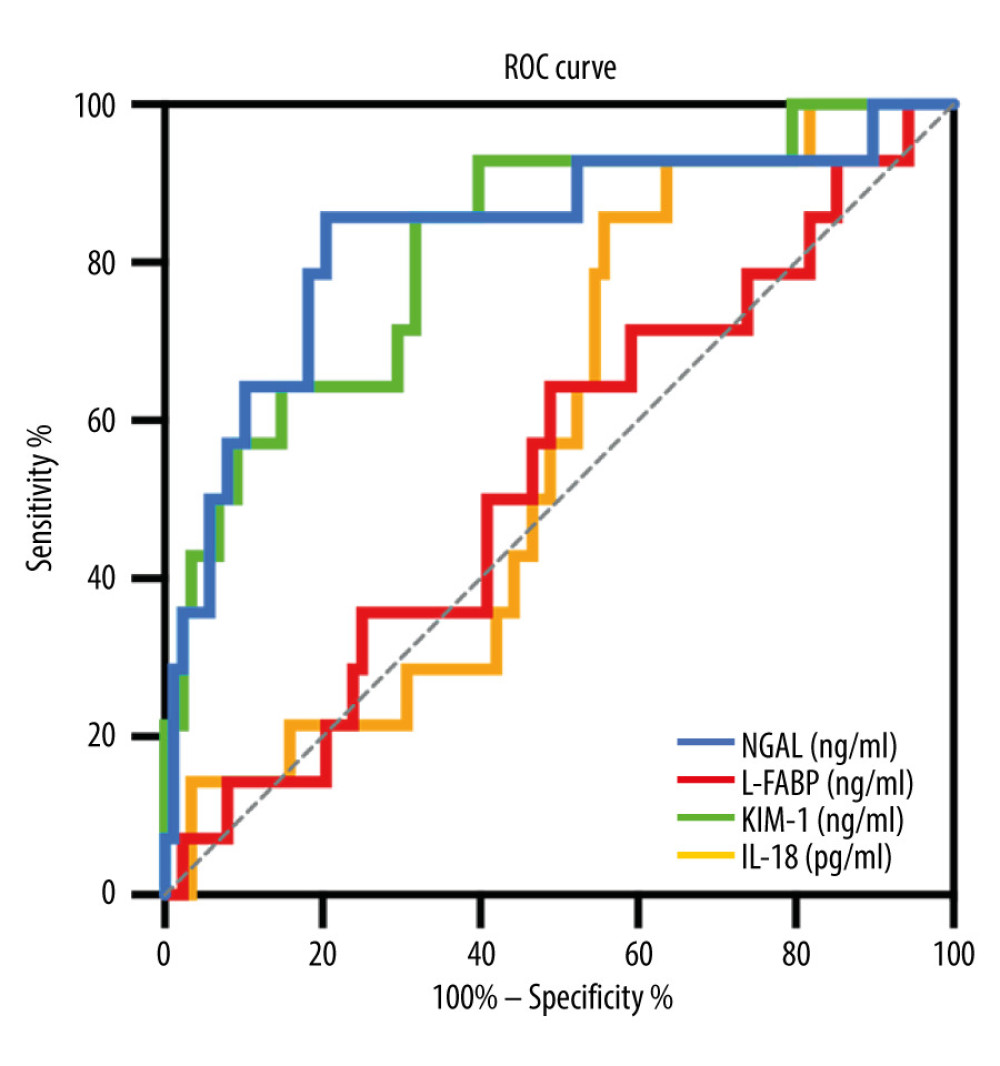 Figure 3. Receiver operating characteristic (ROC) curves for perfusate kidney injury biomarkers and DGF. DGF – delayed graft function. SPSS Statistics 22.0, IBM.
Figure 3. Receiver operating characteristic (ROC) curves for perfusate kidney injury biomarkers and DGF. DGF – delayed graft function. SPSS Statistics 22.0, IBM. Tables
Table 1. Kidney transplant recipient characteristics.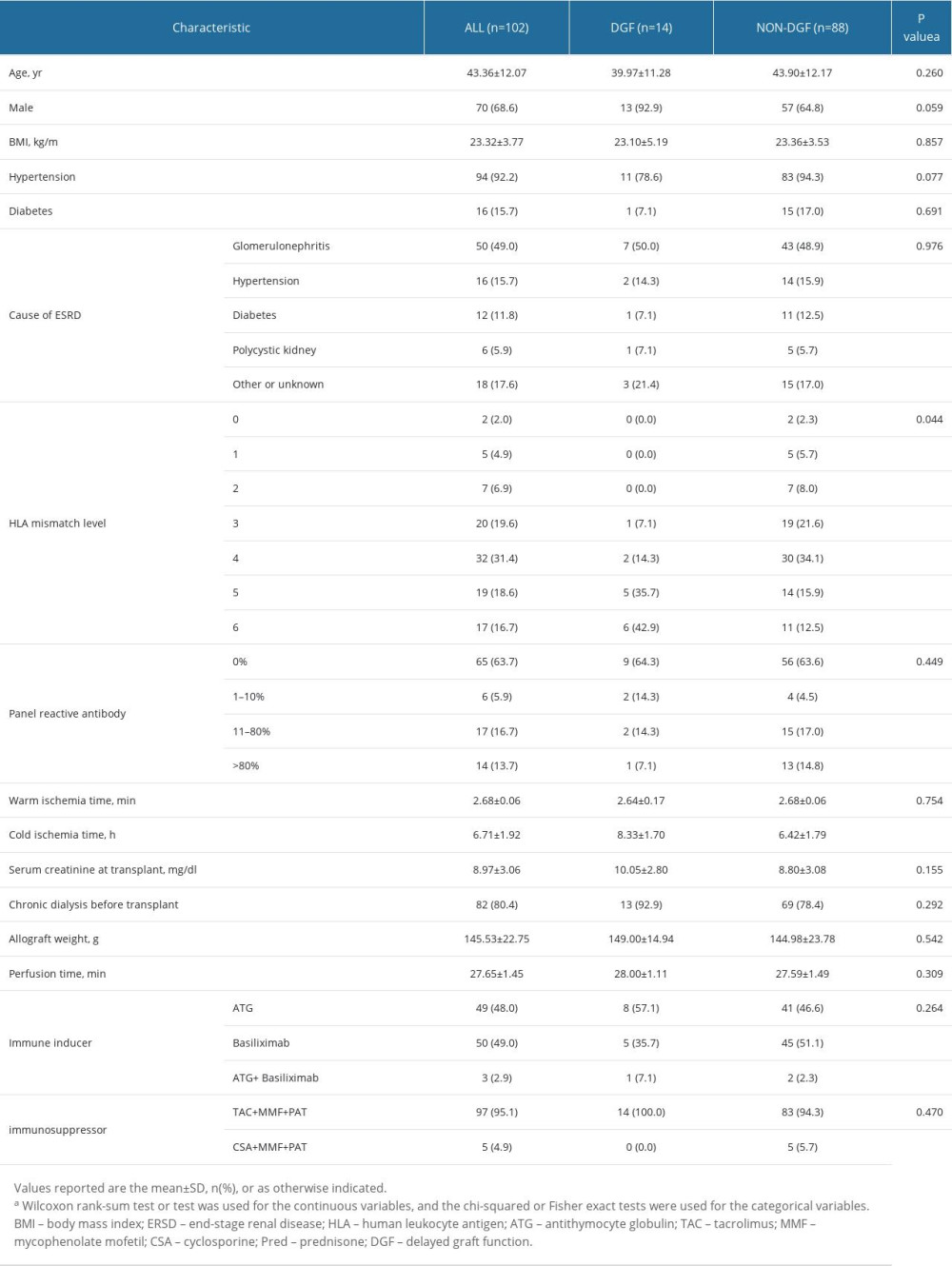 Table 2. Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF.
Table 2. Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF.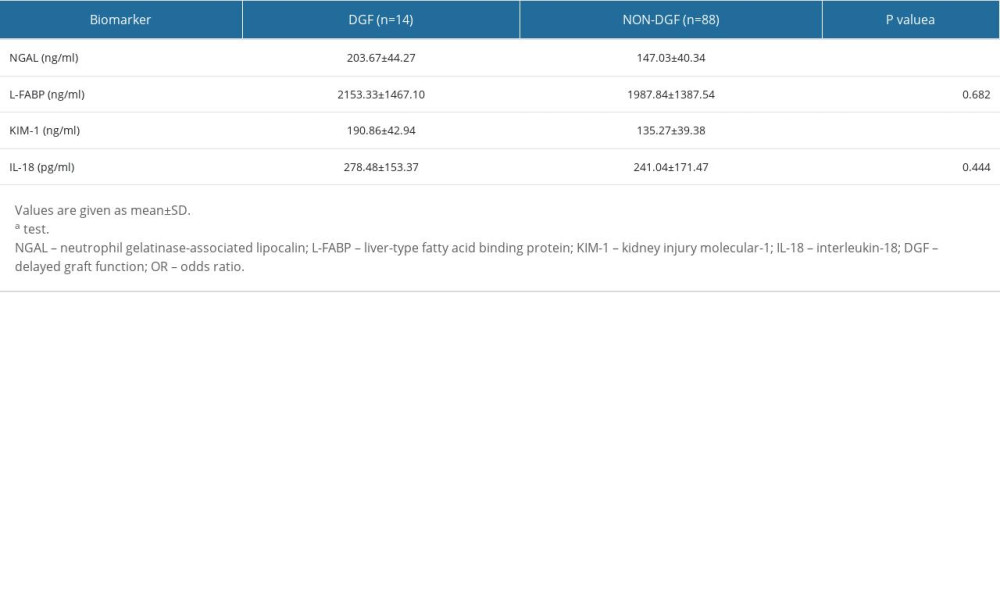 Table 3. Logistic regression of the risk of DGF according to perfusate kidney injury biomarkers (n=102).
Table 3. Logistic regression of the risk of DGF according to perfusate kidney injury biomarkers (n=102).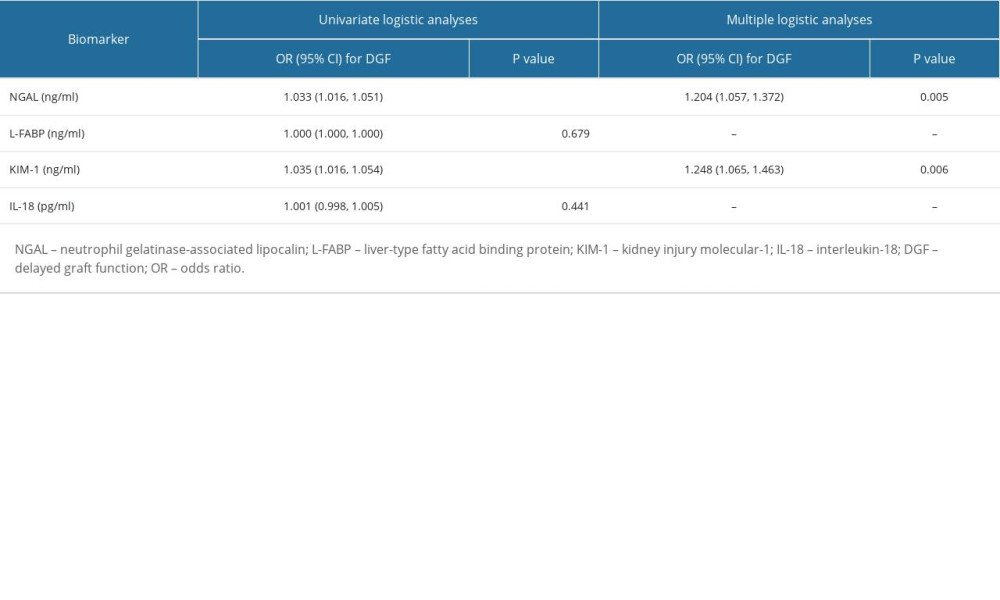 Table 4. Performance indices of perfusate kidney injury biomarkers to diagnose DGF.
Table 4. Performance indices of perfusate kidney injury biomarkers to diagnose DGF.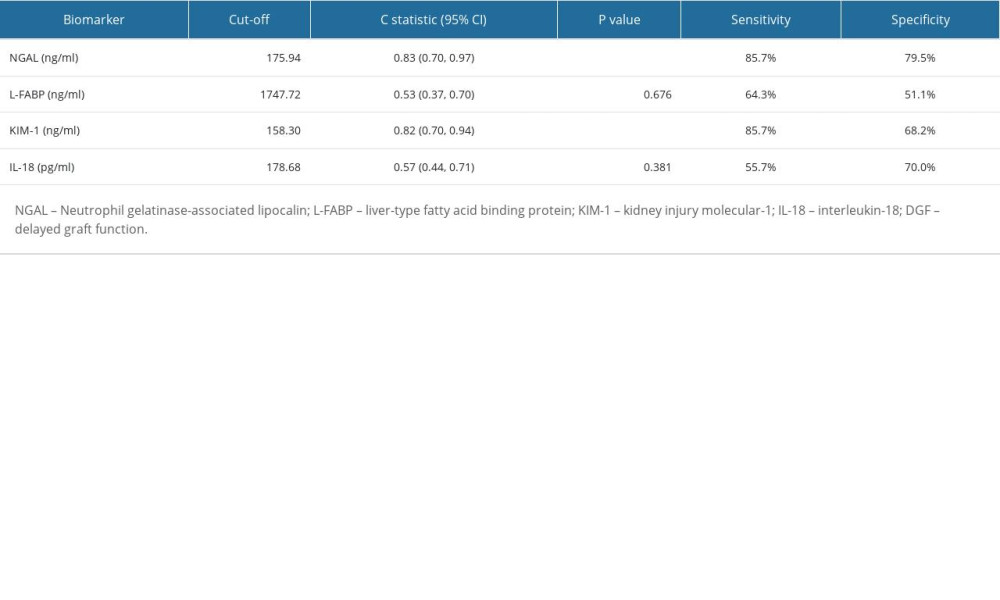 Table 5. Performance indices of perfusate NGAL, KIM-1, and serum creatinine reduction ratio to diagnose DGF.
Table 5. Performance indices of perfusate NGAL, KIM-1, and serum creatinine reduction ratio to diagnose DGF.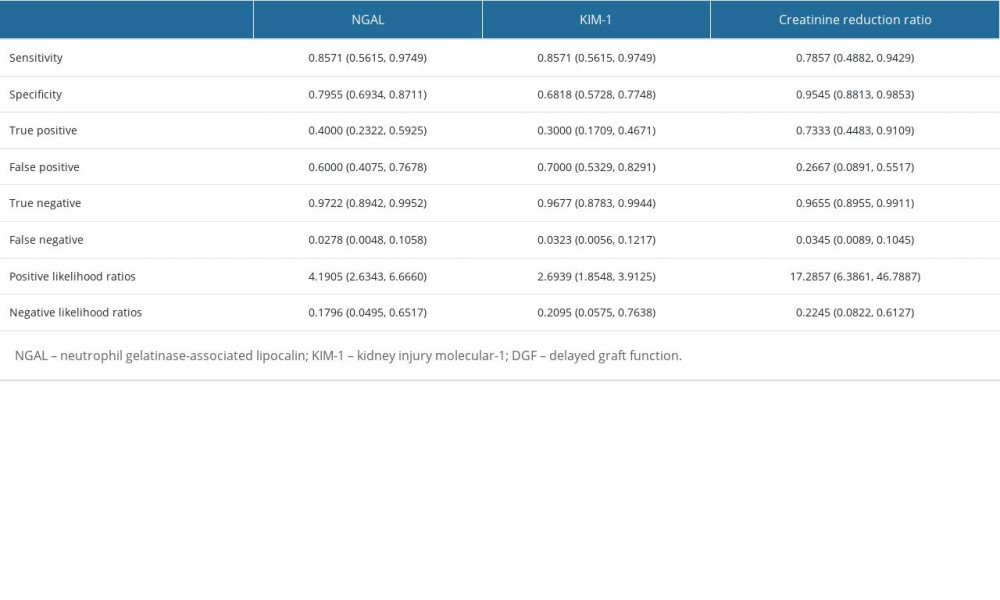 Table 6. Spearman correlations of 3-year graft survival, eGFR and urine albumin on delayed graft function.
Table 6. Spearman correlations of 3-year graft survival, eGFR and urine albumin on delayed graft function.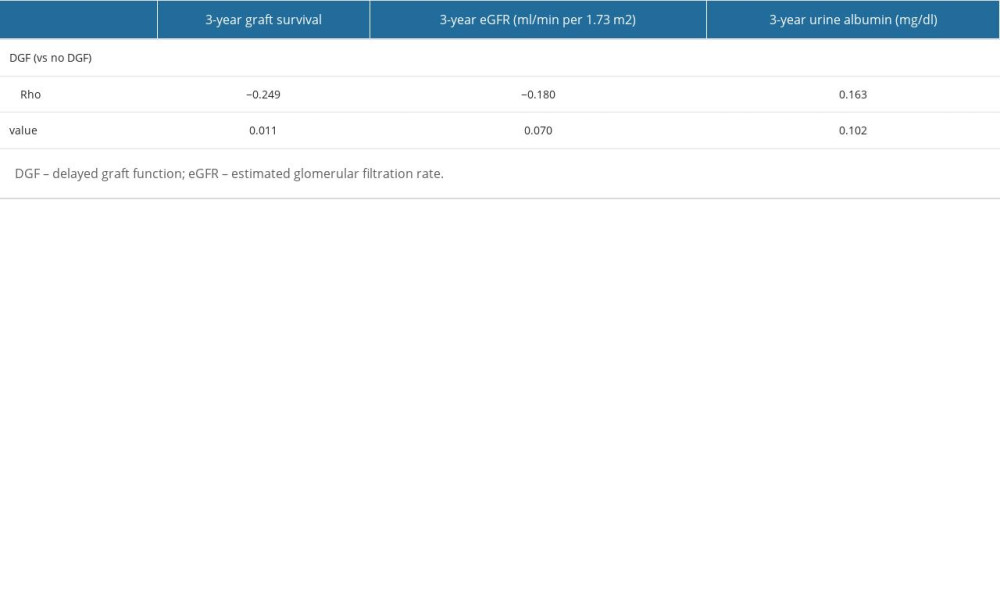 Table 7. Spearman correlations of 3-year graft survival, eGFR, urine albumin and perfusate kidney injury biomarkers in a random subcohort.
Table 7. Spearman correlations of 3-year graft survival, eGFR, urine albumin and perfusate kidney injury biomarkers in a random subcohort.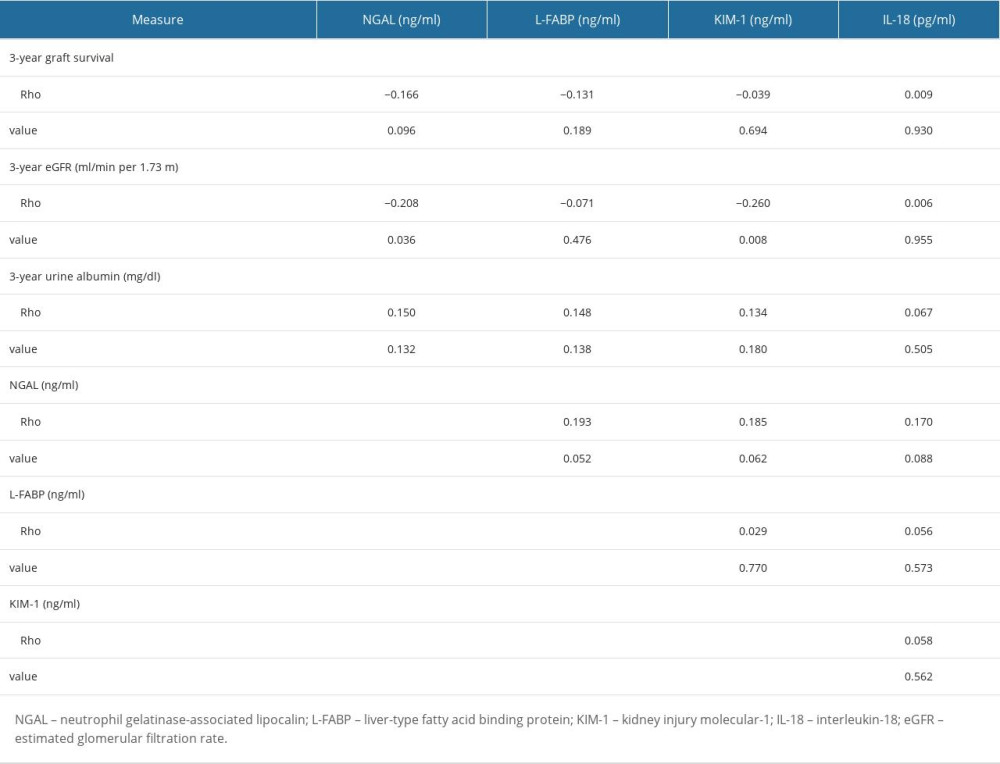 Table 8. Linear regression of 3-year eGFR on perfusate kidney injury biomarkers.
Table 8. Linear regression of 3-year eGFR on perfusate kidney injury biomarkers.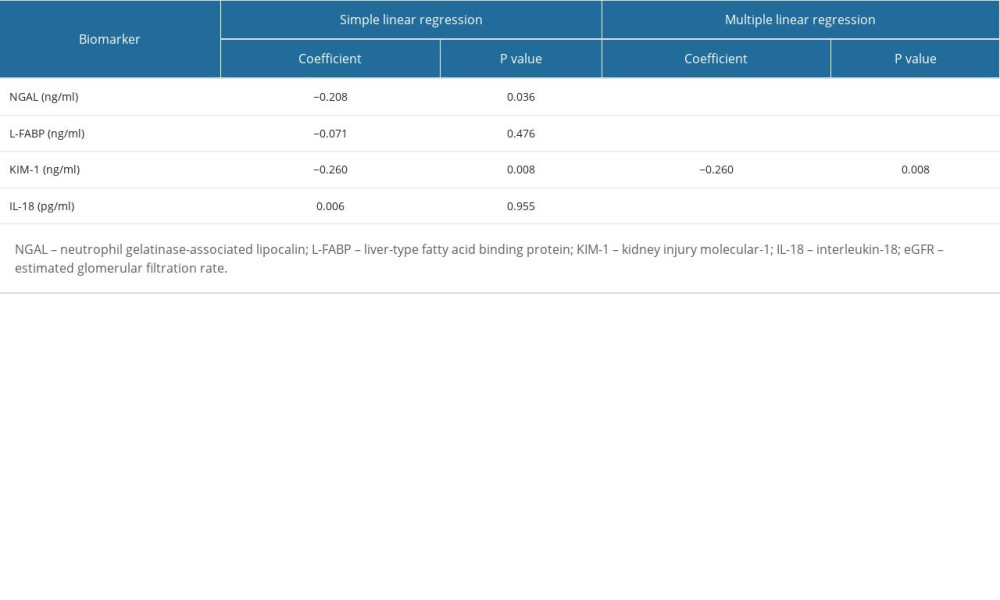 Supplementary Table 1. Kidney transplant donor characteristics, stratified by DGF.
Supplementary Table 1. Kidney transplant donor characteristics, stratified by DGF.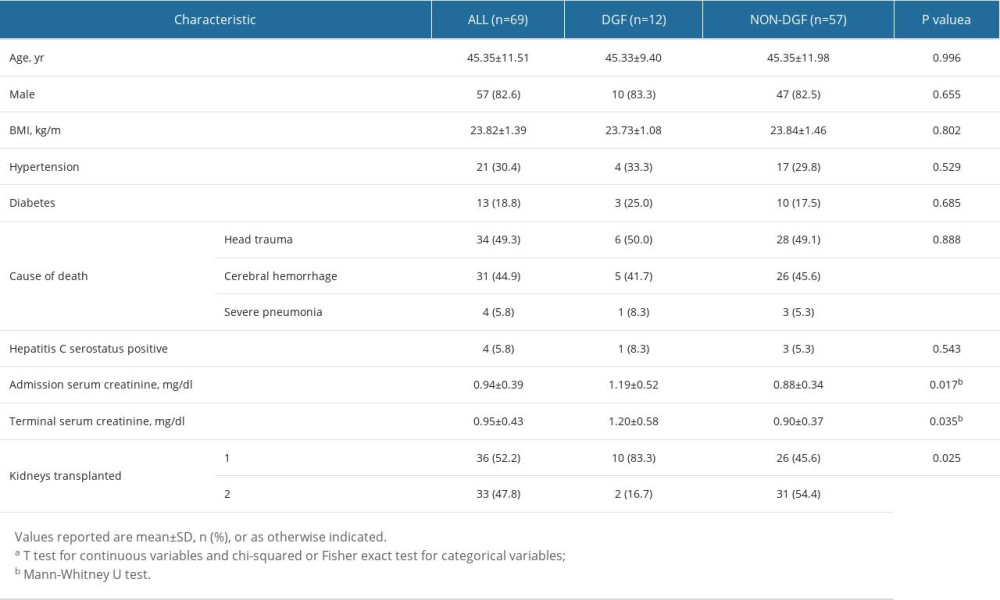
References
1. Wang Y, Jia Y, Wang C, Urinary neutrophil gelatinase-associated lipocalin rapidly decreases in the first week after kidney transplantation: J Clin Lab Anal, 2020; 34(10); e23445
2. Tejeda-Mora H, Verhoeven J, Verschoor W, Circulating endothelial cells transiently increase in peripheral blood after kidney transplantation: Sci Rep, 2021; 11(1); 8915
3. Bahl D, Haddad Z, Datoo A, Qazi YA, Delayed graft function in kidney transplantation: Curr Opin Organ Transplant, 2019; 24(1); 82-86
4. Moledina DG, Hall IE, Thiessen-Philbrook H, Performance of serum creatinine and kidney injury biomarkers for diagnosing histologic acute tubular injury: Am J Kidney Dis, 2017; 70(6); 807-16
5. Tuuminen R, Jouppila A, Salvail D, Dual antiplatelet and anticoagulant APAC prevents experimental ischemia-reperfusion-induced acute kidney injury: Clin Exp Nephrol, 2017; 21(3); 436-45
6. Seibert FS, Sitz M, Passfall J, Urinary calprotectin, NGAL, and KIM-1 in the differentiation of primarily inflammatory vs. non-inflammatory stable chronic kidney diseases: Ren Fail, 2021; 43(1); 417-24
7. Tejchman K, Nowacki A, Kotfis K, The role of endothelins, IL-18, and NGAL in kidney hypothermic machine perfusion: Biomedicines, 2021; 9(4); 417
8. Malhotra R, Katz R, Jotwani V, Urine markers of kidney tubule cell injury and kidney function decline in SPRINT trial participants with CKD: Clin J Am Soc Nephrol, 2020; 15(3); 349-58
9. Bank JR, van der Pol P, Vreeken D, Kidney injury molecule-1 staining in renal allograft biopsies 10 days after transplantation is inversely correlated with functioning proximal tubular epithelial cells: Nephrol Dial Transplant, 2017; 32(12); 2132-41
10. Bhangoo RS, Hall IE, Reese PP, Parikh CR, Deceased-donor kidney perfusate and urine biomarkers for kidney allograft outcomes: A systematic review: Nephrol Dial Transplant, 2012; 27(8); 3305-14
11. Zdziechowska M, Gluba-Brzozka A, Poliwczak AR, Serum NGAL, KIM-1, IL-18, L-FABP new biomarkers in the diagnostics of acute kidney injury (AKI) following invasive cardiology procedures: Int Urol Nephrol, 2020; 52(11); 2135-43
12. Aljumah AA, Tamim H, Saeed M, The role of urinary neutrophil gelatinase-associated lipocalin in predicting acute kidney dysfunction in patients with liver cirrhosis: J Clin Med Res, 2018; 10(5); 419-28
13. Mojtahedzadeh M, Etezadi F, Motaharinia J, Predictive values of urinary interleukin 18 and neutrophil gelatinase-associated lipocalin for delayed graft function diagnosis in kidney transplantation: Iran J Pathol, 2016; 11(4); 391-98
14. Yepes-Calderón M, Sotomayor CG, Pena M, Urinary liver-type fatty acid-binding protein is independently associated with graft failure in outpatient kidney transplant recipients: Am J Transplant, 2021; 21(4); 1535-44
15. Hall IE, Yarlagadda SG, Coca SG, IL-18 and urinary NGAL predict dialysis and graft recovery after kidney transplantation: J Am Soc Nephrol, 2010; 21(1); 189-97
16. Cappuccilli M, Capelli I, Comai G, Evaluation of the current status and future insights: Artif Organs, 2018; 42(1); 8-14
17. Zhu M, Chen Z, Wei Y, The predictive value of urinary kidney injury molecular-1 for long-term graft function in kidney transplant patients: a prospective study: Ann Transl Med Feb, 2021; 9(3); 244
18. Hosgood SA, Brown RJ, Nicholson ML, Advances in kidney preservation techniques and their application in clinical practice: Transplantation, 2021; 105(11); e202-e14
19. Radajewska A, Krzywonos-Zawadzka A, Bil-Lula I, Recent methods of kidney storage and therapeutic possibilities of transplant kidney: Biomedicines, 2022; 10(5); 1013
20. Moers C, Varnav OC, van Heurn E, The value of machine perfusion perfusate biomarkers for predicting kidney transplant outcome: Transplantation, 2010; 90(9); 966-73
21. Parikh CR, Hall IE, Bhangoo RS, Associations of perfusate biomarkers and pump parameters with delayed graft function and deceased donor kidney allograft function: Am J Transplant, 2016; 16(5); 1526-39
22. Rasmussen DGK, Nielsen PM, Kasab-Oglo Ö Y, A non-invasive biomarker of type III collagen degradation reflects ischaemia reperfusion injury in rats: Nephrol Dial Transplant, 2019; 34(8); 1301-9
23. Fuhrman DY, Kellum JA, Joyce EL, The use of urinary biomarkers to predict acute kidney injury in children after liver transplant: Pediatr Transplant, 2020; 24(1); e13608
24. Ntrinias T, Papasotiriou M, Balta L, Biomarkers in progressive chronic kidney disease. Still a long way to go: Pril (Makedon Akad Nauk Umet Odd Med Nauki), 2019; 40(3); 27-39
25. Cabello R, Fontecha-Barriuso M, Martin-Sanchez D, Urinary cyclophilin A as marker of tubular cell death and kidney injury: Biomedicines, 2021; 9(2); 217
26. Rao VS, Ahmad T, Brisco-Bacik MA, Renal effects of intensive volume removal in heart failure patients with preexisting worsening renal function: Circ Heart Fail, 2019; 12(6); e005552
27. Koyawala N, Reese PP, Hall IE, Urine injury biomarkers are not associated with kidney transplant failure: Transplantation, 2020; 104(6); 1272-79
28. Benoit SW, Dixon BP, Goldstein SL, A novel strategy for identifying early acute kidney injury in pediatric hematopoietic stem cell transplantation: Bone Marrow Transplant, 2019; 54(9); 1453-61
29. Lee SA, Noel S, Kurzhagen JT, CD4(+) T cell-derived NGAL modifies the outcome of ischemic acute kidney injury: J Immunol, 2020; 204(3); 586-95
30. Ichimura T, Asseldonk EJ, Humphreys BD, Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells: J Clin Invest, 2008; 118(5); 1657-68
31. Feldreich T, Nowak C, Fall T, Circulating proteins as predictors of cardiovascular mortality in end-stage renal disease: J Nephrol, 2019; 32(1); 111-19
32. Keshavarz Shahbaz S, Pourrezagholi F, Nafar M, Dynamic variation of kidney injury molecule-1 mRNA and protein expression in blood and urine of renal transplant recipients: A cohort study: Clin Exp Nephrol, 2019; 23(10); 1235-49
33. Virzì GM, Breglia A, Brocca A, Levels of proinflammatory cytokines, oxidative stress, and tissue damage markers in patients with acute heart failure with and without cardiorenal syndrome type 1: Cardiorenal Med, 2018; 8(4); 321-31
34. Lewandowska L, Małyszko J, Joanna Matuszkiewicz-Rowińska J, Urinary and serum biomarkers for prediction of acute kidney injury in patients undergoing liver transplantation: Ann Transplant, 2019; 24; 291-97
35. Breglia A, Godi I, Virzì GM, Subclinical contrast-induced acute kidney injury in patients undergoing cerebral computed tomography: Cardiorenal Med, 2020; 10(2); 125-36
36. Hall PS, Mitchell ED, Smith AF, Evidence synthesis, care pathway analysis and research prioritisation: Health Technol Assess May; 22(32); 1-274
37. Virzì GM, Breglia A, Castellani C, Lipopolysaccharide in systemic circulation induces activation of inflammatory response and oxidative stress in cardiorenal syndrome type 1: J Nephrol, 2019; 32(5); 803-10
38. Mojtahedzadeh M, Etezadi F, Pourmand G, Effect of hypertonic saline 5% on early graft function and urinary interleukin 18 and neutrophil gelatinase-associated lipocalin in deceased-donor kidney transplantation: Iran J Pharm Res, 2017; 16(4); 1583-92
39. Modarresi A, Nafar M, Sahraei Z, Role of N-acetylcysteine: Iran J Pharm Res, 2020; 19(1); 57-67
40. Levin A, Reznichenko A, Witasp A, Novel insights into the disease transcriptome of human diabetic glomeruli and tubulointerstitium: Nephrol Dial Transplant, 2020; 35(12); 2059-72
41. McMahon KR, Rod Rassekh S, Schultz KR, Design and methods of the Pan-Canadian applying biomarkers to minimize long-term effects of childhood/adolescent cancer treatment (ABLE) nephrotoxicity study: A prospective observational cohort study: Can J Kidney Health Dis, 2017; 4 2054358117690338
42. Thorsen IS, Bleskestad IH, Jonsson G, Neutrophil gelatinase-associated lipocalin, fibroblast growth factor 23, and soluble klotho in long-term kidney donors: Nephron Extra, 2016; 6(3); 31-39
43. Moser MAJ, Sawicka K, Arcand S, Insights into protection from injury: Ann Transplant, 2017; 22; 730-39
44. Perrenoud L, Kruse NT, Andrews E, Uric acid lowering and biomarkers of kidney damage in CKD Stage 3: A post hoc analysis of a randomized clinical trial: Kidney Med, 2020; 2(2); 155-61
Figures
 Figure 1. Enrollment of KTRs into the study cohort. KTRs: kidney transplant recipients. PowerPoint 2016, Microsoft.
Figure 1. Enrollment of KTRs into the study cohort. KTRs: kidney transplant recipients. PowerPoint 2016, Microsoft. Figure 2. (A–D) Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF. DGF – delayed graft function; NGAL – neutrophil gelatinase-associated lipocalin; L-FABP – liver-type fatty acid binding protein; KIM-1 – kidney injury molecular-1; IL-18 – interleukin-18. GraphPad Prism 8, GraphPad Software, Adobe Illustrator 2022, Adobe.
Figure 2. (A–D) Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF. DGF – delayed graft function; NGAL – neutrophil gelatinase-associated lipocalin; L-FABP – liver-type fatty acid binding protein; KIM-1 – kidney injury molecular-1; IL-18 – interleukin-18. GraphPad Prism 8, GraphPad Software, Adobe Illustrator 2022, Adobe. Figure 3. Receiver operating characteristic (ROC) curves for perfusate kidney injury biomarkers and DGF. DGF – delayed graft function. SPSS Statistics 22.0, IBM.
Figure 3. Receiver operating characteristic (ROC) curves for perfusate kidney injury biomarkers and DGF. DGF – delayed graft function. SPSS Statistics 22.0, IBM. Tables
 Table 1. Kidney transplant recipient characteristics.
Table 1. Kidney transplant recipient characteristics. Table 2. Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF.
Table 2. Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF. Table 3. Logistic regression of the risk of DGF according to perfusate kidney injury biomarkers (n=102).
Table 3. Logistic regression of the risk of DGF according to perfusate kidney injury biomarkers (n=102). Table 4. Performance indices of perfusate kidney injury biomarkers to diagnose DGF.
Table 4. Performance indices of perfusate kidney injury biomarkers to diagnose DGF. Table 5. Performance indices of perfusate NGAL, KIM-1, and serum creatinine reduction ratio to diagnose DGF.
Table 5. Performance indices of perfusate NGAL, KIM-1, and serum creatinine reduction ratio to diagnose DGF. Table 6. Spearman correlations of 3-year graft survival, eGFR and urine albumin on delayed graft function.
Table 6. Spearman correlations of 3-year graft survival, eGFR and urine albumin on delayed graft function. Table 7. Spearman correlations of 3-year graft survival, eGFR, urine albumin and perfusate kidney injury biomarkers in a random subcohort.
Table 7. Spearman correlations of 3-year graft survival, eGFR, urine albumin and perfusate kidney injury biomarkers in a random subcohort. Table 8. Linear regression of 3-year eGFR on perfusate kidney injury biomarkers.
Table 8. Linear regression of 3-year eGFR on perfusate kidney injury biomarkers. Table 1. Kidney transplant recipient characteristics.
Table 1. Kidney transplant recipient characteristics. Table 2. Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF.
Table 2. Comparison of perfusate kidney injury biomarker levels between patients diagnosed with DGF and NON-DGF. Table 3. Logistic regression of the risk of DGF according to perfusate kidney injury biomarkers (n=102).
Table 3. Logistic regression of the risk of DGF according to perfusate kidney injury biomarkers (n=102). Table 4. Performance indices of perfusate kidney injury biomarkers to diagnose DGF.
Table 4. Performance indices of perfusate kidney injury biomarkers to diagnose DGF. Table 5. Performance indices of perfusate NGAL, KIM-1, and serum creatinine reduction ratio to diagnose DGF.
Table 5. Performance indices of perfusate NGAL, KIM-1, and serum creatinine reduction ratio to diagnose DGF. Table 6. Spearman correlations of 3-year graft survival, eGFR and urine albumin on delayed graft function.
Table 6. Spearman correlations of 3-year graft survival, eGFR and urine albumin on delayed graft function. Table 7. Spearman correlations of 3-year graft survival, eGFR, urine albumin and perfusate kidney injury biomarkers in a random subcohort.
Table 7. Spearman correlations of 3-year graft survival, eGFR, urine albumin and perfusate kidney injury biomarkers in a random subcohort. Table 8. Linear regression of 3-year eGFR on perfusate kidney injury biomarkers.
Table 8. Linear regression of 3-year eGFR on perfusate kidney injury biomarkers. Supplementary Table 1. Kidney transplant donor characteristics, stratified by DGF.
Supplementary Table 1. Kidney transplant donor characteristics, stratified by DGF. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








