19 August 2021: Lab/In Vitro Research
Hyperglycemia Promotes Liver Metastasis of Colorectal Cancer via Upregulation of Integrin αvβ6
Ben Wang1ABEG, Shanjie Wang2B, Wenke Wang3BC, Enyu Liu1BD, Sen Guo1EF, Chuanzong Zhao1CF, Jun Niu1AD, Zongli Zhang1A*DOI: 10.12659/MSM.930921
Med Sci Monit 2021; 27:e930921
Abstract
BACKGROUND: Diabetes is related to higher risk of multiple cancers. This study aimed to explore the effect and mechanism of diabetes on liver metastasis of CRC.
MATERIAL AND METHODS: Overall and liver metastasis-free survival in diabetic and non-diabetic CRC patients were compared by Kaplan-Meier analysis. Expression of αvβ6 was detected by immunohistochemistry in clinical specimens. Effects of hyperglycemia on αvβ6 expression in colon cancer cells were assessed by western blot, real-time PCR, and flowcytometry. Effects of hyperglycemia on migration and invasion were demonstrated by Transwell assay. Expression and activity of MMP-9 and MMP-2 were determined by real-time PCR and gelatin zymography. Liver metastatic nodules were counted and b6 expression was detected by western blot in a liver metastasis mouse model.
RESULTS: CRC patients with diabetes had poorer overall and liver metastasis-free survival, and diabetes was associated with higher αvβ6 expression in CRC specimens. Hyperglycemia promoted the invasion and migration of colon cancer cells, and upregulated the expression and activity of MMP-9, which were attenuated by inhibition of αvβ6. Hyperglycemia upregulated the expression of β6 and cell surface expression of avb6, which was reduced by ERK inhibitor. The in vitro results were confirmed in vivo in the mouse model.
CONCLUSIONS: Our study demonstrated the enhancing effect of hyperglycemia on liver metastasis of CRC, and showed that αvβ6 was involved in this process, suggesting that control of glucose levels and inhibition of αvβ6 can reduce the risk of liver metastasis in diabetic CRC patients.
Keywords: Colorectal Neoplasms, Diabetes Mellitus, Integrin alphaV, Hyperglycemia, Integrins, Liver Neoplasms
Background
Colorectal cancer (CRC) and type 2 diabetes (T2DM) are among the most common diseases in the world. According to the Diabetes Atlas, Eighth Edition (2017), the number of diabetic adults was about 425 million (about 1 in 11), which will increase to 629 million by 2045 [1]. Colorectal cancer is the second leading cause of cancer-related death, the main cause of which is liver metastasis.
Several studies have shown that diabetes is related to a significant increase in the risk and mortality rates of various cancers, such as pancreatic, breast, lung, urinary system, and gastric cancer [2–6]. Type 2 diabetes and colorectal cancer have similar risk factors, including obesity, lack of exercise, a Western-style diet, smoking, visceral adiposity, and aging. Several studies have revealed that diabetes increases the risk of colorectal cancer and leads to a poor prognosis. A large-cohort study, in which a total of 985 815 subjects (including 104 343 diabetic patients) were undergoing follow-up from 1998 to 2009, showed that the incidence of colorectal cancer increased in diabetic people independently of hypertension, hyperlipidemia, and gout [7]. A study of 741 patients with colorectal cancer found that the overall and median survival rates of diabetic patients were significantly lower than in non-diabetic patients, while poorly controlled glycemic status (determined by glycosylated hemoglobin levels) independently predicted a worse outcome [8]. In an experimental animal study, colon cancer was induced by 1,2-dimethylhydrazine, and the average number of colon tumors in diabetic rats was significantly higher than that in non-diabetic rats [9]. Moreover, angiotensin activation induced by hyperglycemia could promote liver metastasis of colon cancer, and the combination of anti-angiotensin and hypoglycemic treatments synergistically inhibited liver metastasis [10,11]. A recent study showed that diabetes-complicated CRC patients were associated with advanced lymphovascular invasion and higher risk of liver metastasis, in which biglycan-rich cancer stroma may be involved [12]. These studies suggested a potential role of diabetes and hyperglycemia in liver metastasis of CRC. However, data on the impact of diabetes on liver metastasis of colorectal cancer and its underlying mechanism is still limited.
The process of liver metastasis is composed of several interrelated and complex steps, such as detachment from the primary site, invasion, migration, intravasation, extravasation, and proliferation [13]. Integrins are transmembrane glycoprotein receptors composed of α and β subunits, and can regulate cell migration, invasion, and adhesion by transmitting biological signals [14]. The β6 subunit can only covalently bind to the αv subunit to form the integrin αvβ6, which is highly expressed in epithelial malignant tumors [15]. Our previous studies demonstrated that αvβ6 plays an important role in migration, invasion, apoptosis, and metastasis of colorectal cancer [16–23].
The CXCL12/CXCR4 axis displays dual effects in diabetes-associated complications, including protective and adverse effects [24,25]. Considering the important role of αvβ6 in CXCL12/CXCR4 axis-induced liver metastasis of CRC in our previous study [26], we speculate that diabetes can promote liver metastasis of colorectal cancer through integrin αvβ6. The aim of this study was to investigate the effect of diabetes mellitus on liver metastasis of colorectal cancer and its underlying mechanism.
Material and Methods
PATIENT MANAGEMENT AND FOLLOW-UP:
Patients with stage II or III colorectal cancer were enrolled at the Department of General Surgery of Qilu Hospital from 2009 to 2012. The inclusion criteria were as follows: (1) undergoing radical resection of CRC and standard chemotherapy after operation; (2) diagnosis of CRC with stage II or III according to TNM system confirmed by at least 2 pathologists; (3) without distant metastasis at first diagnosis; (4) fasting blood glucose level ≥126 mg/dl, postprandial blood glucose level ≥200 mg/dl, HbA1c level ≥6.5%, or taking drugs for diabetes. The exclusion criteria were as follows: (1) receiving neoadjuvant chemotherapy or radiotherapy; (2) loss to follow-up within 6 months after surgery; (3) developing another primary malignant tumor during follow-up. Finally, 56 diabetic and 147 non-diabetic patients with CRC were included in this study. Postoperative follow-up, including blood tests and imaging examination, was carried out until December 2017. This study adhered to the tenants of the Declaration of Helsinki. The research protocol was approved by the Ethics Committee of Qilu hospital of Shandong University (grant number KYLL-2015-158) and we obtained signed informed consent from all participants.
TISSUE MICROARRAY (TMA) AND IMMUNOHISTOCHEMISTRY:
According to hematoxylin-eosin staining, the representative areas of colorectal cancer were obtained from the paraffin-embedded tissue blocks, and tissue microarray was constructed using Tissue Microarrayer. After deparaffinization and antigen repair in sodium citrate buffer solution at 95 for 15 min, the paraffin sections were washed with distilled water, immersed in 3% hydrogen peroxide, and blocked with 5% bovine serum albumin. The sections were incubated with primary antibody of αvβ6 (clone 442.5C4, Calbiochem, CA, USA) at 4 overnight, incubated with HRP-conjugated secondary antibody on the second day, and then stained with 3,3-diaminobenzidine (DAB) for 5 min. Normal mouse IgG served as a negative control of the primary antibody. Immunostaining of αvβ6 was scored using previously described methods [27]. The intensity score was divided into 0 (no staining), 1 (weak staining), 2 (medium staining), and 3 (strong staining). The score for percentage of positive cells was divided into 0 (0%), 1 (1–25%), 2 (26–50%), 3 (51–75%), and 4 (76–100%). The final immunohistochemistry score (IHS) was determined by the product of intensity score and percentage score. Immunostaining with an IHS score 0–3 was considered as negative, and with an IHS score >3 was considered as positive.
CELL CULTURE AND INTERFERENCE:
Human colon cancer cell lines WiDr and HT29 (ATCC, USA), and Balb/c mouse-derived colon cancer cells CT26, were cultured in DMEM containing 10% fetal bovine serum (Gibco) with 5% CO2 at 37°C. Colon cancer cells were stimulated with normal glucose (5.5 mM) and high glucose (15, 20, and 25 mM) for 24 h. Medium containing 5.5 mM glucose and 19.5 mM mannitol served as an osmotic control. To inhibit expression of αvβ6, cells were treated by transient transfection with specific siRNA (GenePharma) in Opti-MEM Medium (Invitrogen, CA, USA) using Lipofectamine 2000 (Invitrogen, CA, USA), and experiments were then performed 24 h later. To inhibit activation of ERK, cells were pretreated with 20 μmol/L U0126 (Cell Signaling Technology, MA, USA). Colon cancer cell lines HT-29, WiDr, and CT26 used in this study were routinely authenticated by short tandem repeat DNA profiling.
QUANTITATIVE REAL-TIME PCR (QRT-PCR):
Total RNA was extracted from colon cancer cells with Trizol reagent (Invitrogen, CA). First-strand cDNA was obtained using a RevertAid™ kit (Fermentas). Then, real-time PCR was carried out with SsoFast EvaGreen Supermix (Bio-Rad, CA, USA) using an IQ5 Real-Time PCR cycler (Bio-Rad). GAPDH served as an internal control. The primers for integrin β6, matrix metalloproteinase-2 (MMP-2), and metalloproteinase-9 (MMP-9) were as described in our previous studies [26,28]. Relative fold change of mRNA expression was calculated by 2−ΔΔCT method.
WESTERN BLOT:
Equal amounts of protein extracted from colon cancer cells by RIPA lysis buffer were separated on 10% SDS-PAGE. The protein was then transferred to a nitrocellulose membrane, which was blocked with 5% nonfat milk. Then, the membrane was incubated at 4°C overnight with the primary antibodies, including anti-β6 (C-19, Santa Cruz, CA, USA), anti-αv (P2W7, Santa Cruz, CA, USA), anti-ERK1/2 (Cell Signaling Technology, MA, USA), and anti-phospho-ERK1/2 (Cell Signaling Technology, MA, USA). After washing with TBST buffer, the membrane was incubated with horseradish peroxidase-conjugated secondary antibodies at room temperature for 1 h. Protein expression was visualized by chemiluminescence (Millipore, USA). β-actin was used as the loading control.
FLOW CYTOMETRY:
The colon cancer cell suspension (100 μl per group) with an intensity of 1×106 cells/ml was blocked by goat serum on ice for 30 min and washed twice with PBS buffer. Then, αvβ6 monoclonal antibody E7P6 (Chemicon, CA, USA) was added to colon cancer cells and incubated on ice for 20 min, followed by incubation with FITC-labeled goat anti-mouse IgG on ice for 20 min. The surface expression of αvβ6 was detected by flow cytometry (FACSCalibur, BD) and analyzed by FlowJo software.
MIGRATION AND INVASION ASSAY:
The membrane of Transwell chambers (Corning) was precoated with 10 μg/ml fibronectin. The colon cancer cells in logarithmic growth phase were starved in serum-free medium for 12 h and resuspended in serum-free medium to 2×105 cells/ml. We added 500 μl cell suspension into the upper chamber and 600 μl medium with 10% fetal bovine serum was added to the lower chamber. Both upper and lower chambers were supplemented with different concentrations of glucose and cultured at 37°C for 24 h. Cells on the upper surface of the basement membrane were gently wiped away, and cells on the lower surface were fixed with 4% paraformaldehyde and stained with 0.1% crystal violet for 20 min. The average number of cells in 5 randomly selected high-power fields was calculated per group. To avoid the effect of cell proliferation, the final number of migrating cells was adjusted by cell proliferation rate. The invasion assay was conducted in a similar way, but the membrane was precoated with Matrigel.
GELATIN ZYMOGRAPHY:
Cell-free supernatants were harvested from HT-29 cells pretreated with different concentrations of glucose and siRNA. Samples with equivalent amount of protein were reconstituted in loading buffer and subjected to electrophoresis in 10% SDS-PAGE containing 0.1% gelatin. After washing twice with 2.5% Triton X-100, the gels were incubated in activation buffer (15 mM CaCl2, 50 mM NaCl, 50 mM Tris-HCl, pH 7.5) at 37°C overnight, and stained with Coomassie blue solution. Then, gels were destained with 45% methanol and 10% acetic acid. The band intensities were quantified with Image J software.
MOUSE MODELS OF DIABETES AND LIVER METASTASIS:
Diabetes was induced in Balb/c mice (8 weeks old) by intraperitoneal injection of 50 mg/kg streptozotocin (STZ, Sigma, St. Louis, MO) dissolved in 0.1 ml citrate buffer (pH 4.5) for 5 consecutive days [29], while mice injected with citrate buffer served as the control. Random blood glucose measurements were made 5 days later, and mice with blood glucose at least 16 mmol/L were considered as diabetic mice. Then, mice were divided into 4 groups: control group (n=10), diabetes group (n=10), diabetes and metformin group (n=10), and diabetes and β6 shRNA group (n=10). In the diabetes and metformin group, metformin (50 mg/kg body mass/day) was administered orally in diabetic mice. In the diabetes and β6 shRNA group, β6-specific shRNA was injected via the tail vein in diabetic mice. Balb/c-derived colon cancer cells CT26 in logarithmic growth phase were harvested and washed with serum-free medium, and then resuspended in PBS with a concentration of 2×106 cells/ml. Each mouse was injected with 0.2 ml cell suspension via the spleen. Twenty days after injection, the mice were sacrificed to observe the number of intrahepatic metastases. All animal experiments strictly followed the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health and Shandong University, and was approved by the Ethics Committee of Qilu hospital of Shandong University (grant number KYLL-2015-158).
STATISTICAL ANALYSIS:
Survival curves were demonstrated using the Kaplan-Meier method, and the significance of differences was evaluated by the log-rank test. The statistical significance of other data was analyzed by one-way ANOVA, chi-square test, or
Results
EFFECTS OF DM ON LIVER METASTASIS AND OVERALL SURVIVAL IN CRC PATIENTS:
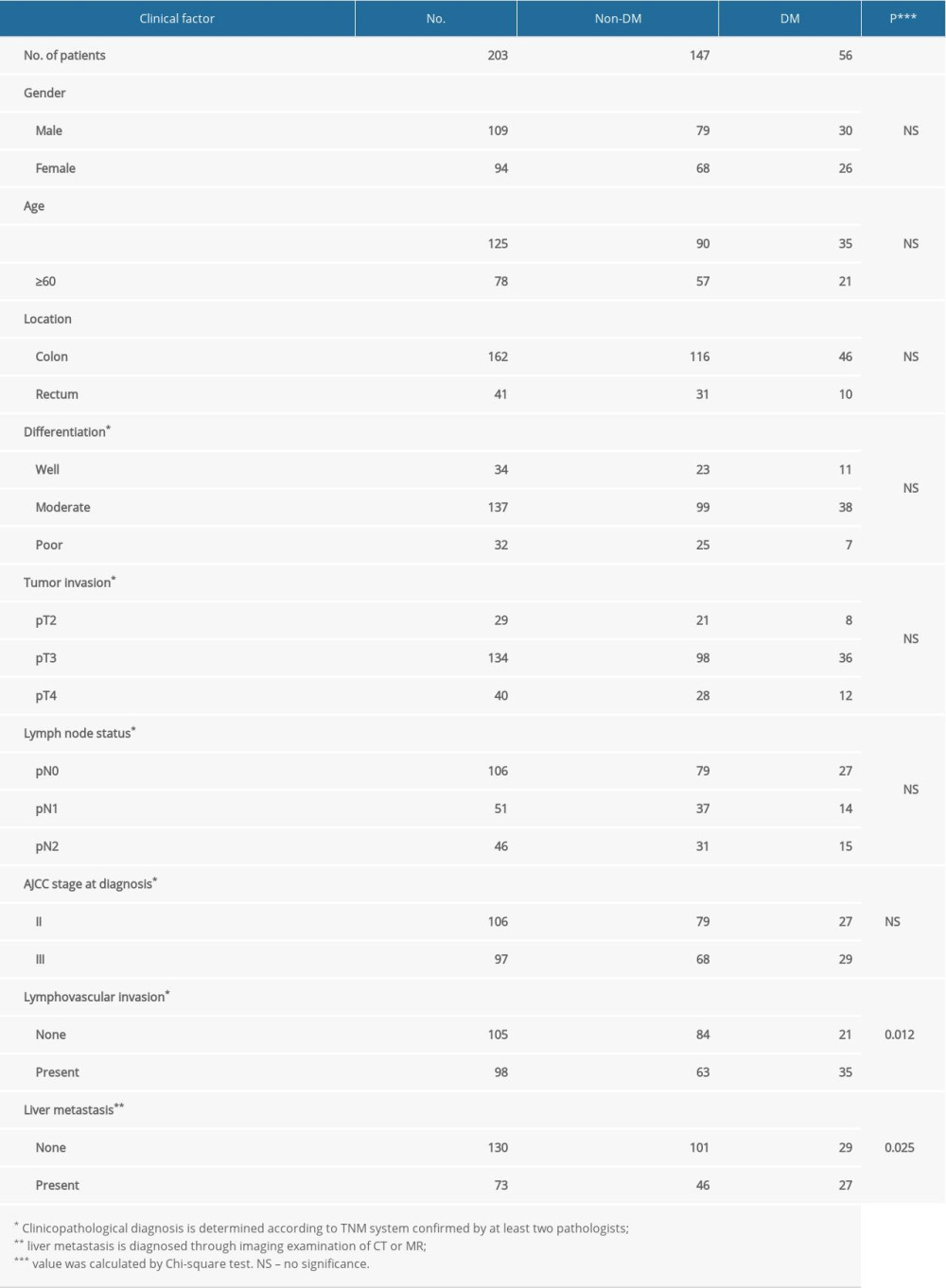
Of the 203 stage II–III CRC patients, 56 had DM and the remaining 147 patients did not. These CRC patients were separated into 2 groups: those with DM (DM group) and those without DM (non-DM group). Clinicopathological factors were compared between the 2 groups. No significant difference was found in cell differentiation, tumor invasion, lymph node invasion, or tumor stage. However, a significant difference was found in lymphovascular invasion (P<0.05) and liver metastasis (P<0.05) between the DM group and non-DM group (Table 1). The liver metastasis-free survival curve demonstrated a significant difference in time from surgery to hepatic relapse between the 2 groups (P<0.01), and patients with DM were more likely to develop liver metastasis (Figure 1A, 1B). Moreover, the overall survival curve also showed a significant difference in the overall survival time between the 2 groups (P<0.01), and suggested that prognosis was particularly unfavorable for patients with DM (Figure 1C, 1D).
EFFECTS OF DM ON EXPRESSION OF INTEGRIN αVβ6 IN CRC PATIENTS:
To explore the potential mechanism of DM in CRC progression, the expression of integrin αvβ6 was evaluated in the 203 primary CRC samples. Tissue microarray was constructed using the paraffin-embedded tissue blocks, with 203 CRC points evenly distributed in 8 sections (Figure 2C). Integrin αvβ6 was positively expressed in 34 samples (60.7%) of the DM group and in 65 samples (44.2%) of the non-DM group, and its expression was observed both in the cytoplasm and membrane of cancer cells (Figure 2A). Analysis of the immunohistochemistry scores (IHS) of αvβ6 using the Mann-Whitney test showed that the expression of αvβ6 was significantly higher in the DM group than in the non-DM group (P<0.01, Figure 2B).
HIGH GLUCOSE INDUCED UPREGULATION OF INTEGRIN αVβ6 THROUGH ERK PATHWAY:
The aforementioned data indicated a correlation between DM and integrin αvβ6 in CRC tissues. To investigate the effect of high glucose on αvβ6 expression, we examined the mRNA and protein expression of αvβ6 in medium with a physiological glucose level (5.5 mM glucose), hyperglycemic level (15, 20, and 25 mM glucose), or osmotic control (5.5 mM glucose and 19.5 mM mannitol). Real-time-PCR analysis demonstrated that high glucose induced a dose-dependent upregulation of integrin β6 at mRNA levels in HT-29 and WiDr cells (P<0.05), but had no effect on αv (Figure 3A, 3B). However, the osmotic control medium had no effect on β6 expression compared with a physiological glucose level. Similar results were shown by western blot (Figure 3C, 3D). We then explored the potential signaling pathways induced by high glucose. Western blot showed that stimulation of HT-29 or WiDr cells with high glucose increased the phosphorylation of ERK 1/2 (P<0.05, Figure 4A, 4B). Inhibition of the ERK pathway by U0126 effectively attenuated high-glucose-induced upregulation of β6 at protein and mRNA levels (P<0.05, Figure 4C–4E). The β6 subunit can only bind to αv to form the integrin αvβ6, and β6 upregulation results in increased αvβ6 on the cell surface despite unchanged αv expression [30]. Flow cytometry demonstrated that high glucose upregulated surface expression of αvβ6 in HT-29 cells, which was inhibited by U0126 (P<0.05, Figure 4F). In addition, hyperglycemia also increased mRNA expression of integrin β3 (P<0.05), but had no effect on β1 and β5 (Figure 3E).
INTEGRIN αVβ6 WAS INVOLVED IN HIGH-GLUCOSE-INDUCED CRC CELL MIGRATION:
Cell migration and invasion are essential for liver metastasis of CRC cells. Migration assay showed that high glucose significantly induced HT-29 and WiDr cells migration on fibronectin compared with normal glucose medium (P<0.05), but osmotic control had no effect. Inhibition of αvβ6 expression by siRNA attenuated high-glucose-induced migration (P<0.05, Figure 5A, 5B). These data indicated that high glucose promoted the migration of CRC cells through upregulation of αvβ6.
HIGH GLUCOSE ENHANCED INVASIVE ABILITY OF CRC CELLS THROUGH αVβ6-INDUCED UPREGULATION OF MMP-9:
The effects of high glucose on invasion of CRC cells were then examined. Invasion assay showed that high glucose significantly increased the invasion ability of WiDr and HT-29 cells compared with normal glucose medium, which was markedly attenuated by pretreatment with β6 siRNA (P<0.05, Figure 5C, 5D). Metalloproteinase-2 (MMP-2) and metalloproteinase-9 (MMP-9) played an important role in CRC cell invasion. Real-time PCR and gelatin zymography demonstrated that high glucose increased the activity and mRNA expression of MMP-9 but not MMP-2, and β6 siRNA markedly attenuated this effect (P<0.05, Figure 6). These data suggest that αvβ6-mediated MMP-9 upregulation, and activation may be involved in high-glucose-induced invasion.
EFFECT OF DIABETES ON LIVER METASTASIS AND EXPRESSION OF INTEGRIN β6 IN THE MOUSE MODEL:
To verify the effect of diabetes on liver metastasis in vivo, a liver metastasis model was conducted in diabetic and control mice using Balb/c-derived colon cancer cells CT26. The number of liver metastatic nodules in diabetic mice was significantly greater than that in the control group, which was attenuated by metformin- or β6-specific shRNA (P<0.05, Figure 7A, 7B). The expression of β6 was increased in diabetic mice, and the effect was attenuated by metformin- or β6-specific shRNA (P<0.05, Figure 7C).
Discussion
Over the last few decades, the incidence of diabetes and colorectal cancer has increased globally. Previous studies have shown a linkage between the risk of colorectal cancer and diabetes [31], but the impact of diabetes on colorectal cancer prognosis, especially liver metastasis, is not clear. In the present study, we focused on the effect and underlying mechanism of diabetes on liver metastasis of colorectal cancer. By analyzing colorectal cancer specimens, we demonstrated that diabetic patients had a shorter liver metastasis-free survival and overall survival compared with non-diabetic patients, and diabetes was associated with higher αvβ6 expression in CRC specimens. Hyperglycemia promoted the migration and invasion of CRC cells through upregulation of αvβ6 via the ERK pathway in vitro.
In our experiment, we followed up 203 stage II–III CRC patients and found that CRC patients with diabetes had shorter liver metastasis-free survival and overall survival, suggesting that diabetic patients are more prone to develop liver metastasis. These results are in good agreement with previous reports [10,11,32]. A meta-analysis by Mills et al revealed that CRC patients with diabetes have a significantly increased risk of cancer-specific and all-cause mortality, and have significantly reduced disease-free survival [33].
The process of liver metastasis is composed of several complex steps, such as detachment from the primary site, invasion, migration, intravasation, extravasation, and proliferation. In the process, migration and invasion play important roles. As a kind of adhesion receptor, integrin αvβ6 is associated with several aggressive behaviors of CRC, including cell migration, invasion, adhesion, proliferation, apoptosis, and chemoresistance [16,22,23,26,34]. Due to the poor prognosis of CRC patients with diabetes, radio-chemo-therapy after surgery is particularly important for them. Integrin αvβ6 is also involved in the radio-chemo-therapy for various cancers, especially CRC. Proton radiotherapy can inhibit the metastatic potential of HT-29 colon cancer cells by downregulating the expression of integrins, such as α5β1, α6β4, αvβ3, and αvβ6, and by modulating integrin trafficking and its downstream signaling [35]. Integrin αvβ6-targeted photodynamic therapy showed an inhibitive effect against tumor formation and metastasis [36]. Integrin αvβ6 is associated with chemoresistance of colorectal and breast cancer [37,38], and β6-targeted immunoliposomes can improve chemoresistance by mediating tumor-specific drug delivery [39]. In this study, we detected the expression of αvβ6 in CRC specimens, and found that diabetic patients displayed higher αvβ6 expression than non-diabetic patients. These results suggest integrin αvβ6 is involved in diabetes-induced liver metastasis of CRC.
The linkage between CRC and diabetes may result from hyperinsulinemia, hyperglycemia, DM therapy, and the DM microenvironment [31]. Several studies demonstrated that hyperglycemia is an independent risk factor for many kinds of cancers, although its underlying mechanism was still unclear [40,41]. Thus, we further investigated the effect of hyperglycemia on malignant behaviors of CRC cells and potential mechanisms in vitro. In our study, high glucose promoted the migration and invasion of colorectal cancer cells, and upregulated the expression and activity of MMP-9, and these enhanced effects were significantly attenuated by inhibition of avb6. Thus, it is plausible that hyperglycemia promotes CRC cells migration and invasion via integrin αvβ6. As mentioned above, αvβ6 expression was correlated with the state of diabetes, so we further explored the effect of hyperglycemia on expression of αvβ6. As expected, hyperglycemia upregulated the expression of β6 subunit at mRNA and protein levels, as well as the expression of integrin αvβ6 on the cell surface. Moreover, inhibition of the ERK signal pathway significantly reduced hyperglycemia-induced upregulation of αvβ6. Moreover, the in vitro results were verified in vivo by the liver metastatic model in diabetic mice. These results suggest that hyperglycemia promotes CRC metastasis through upregulation of αvβ6 via the ERK pathway, in addition to direct effects of hyperglycemia such as energy source and the Warburg effect.
Furthermore, some cytokines, such as CXCL12 and TGF-b1, were reported to be increased in diabetic patients and play an important role both in diabetes-related complications and liver metastasis of CRC [24–26,42–44]. Interestingly, integrin αvβ6 can activate TGF-β1 by binding to latency-associated peptide (LAP), and TGF-β1 conversely upregulates β6 expression by binding to its gene promoter through the TGF-β1 signaling mediator Smad3 [45,46], indicating a positive feedback loop between TGF-β1 and αvβ6. In addition, our previous study demonstrated that CXCL12 promotes liver metastasis of CRC by increasing αvβ6 expression [26]. These data suggest that crosstalk between αvβ6 and cytokines may play an important role in diabetes-induced liver metastasis of CRC, which needs to be further explored in future research.
Conclusions
In conclusion, this study highlights the enhancing effect of diabetes and hyperglycemia on the liver metastasis of CRC, and upregulation of integrin αvβ6 was shown to be an underlying mechanism. Control of glucose levels and inhibition of αvβ6 may be of benefit in the treatment and prevention of liver metastasis in CRC patients with diabetes.
Figures
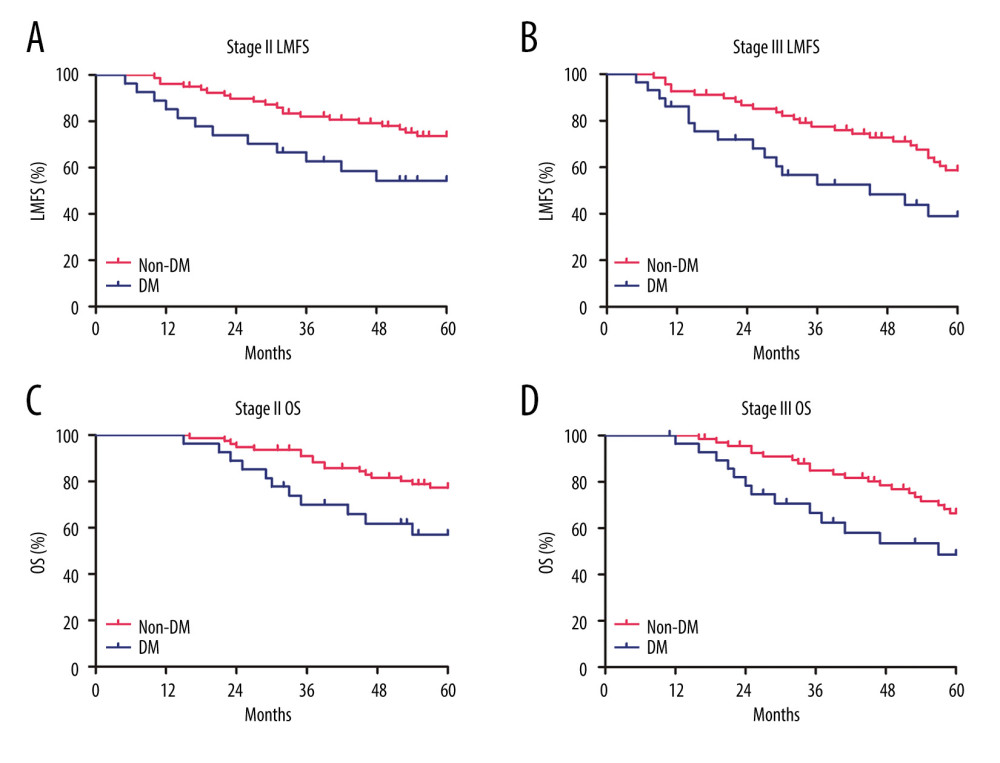 Figure 1. Kaplan-Meier analysis of liver metastasis-free survival (LMFS) and overall survival (OS) for CRC patients with diabetes mellitus (DM group) and without diabetes mellitus (non-DM group). (A) LMFS curve of stage II patients (P<0.01). (B) LMFS curve of stage III patients (P<0.01). (C) OS curve of stage II patients (P<0.01). (D) OS curve of stage III patients (P<0.01). Significance of difference was evaluated by log-rank test.
Figure 1. Kaplan-Meier analysis of liver metastasis-free survival (LMFS) and overall survival (OS) for CRC patients with diabetes mellitus (DM group) and without diabetes mellitus (non-DM group). (A) LMFS curve of stage II patients (P<0.01). (B) LMFS curve of stage III patients (P<0.01). (C) OS curve of stage II patients (P<0.01). (D) OS curve of stage III patients (P<0.01). Significance of difference was evaluated by log-rank test. 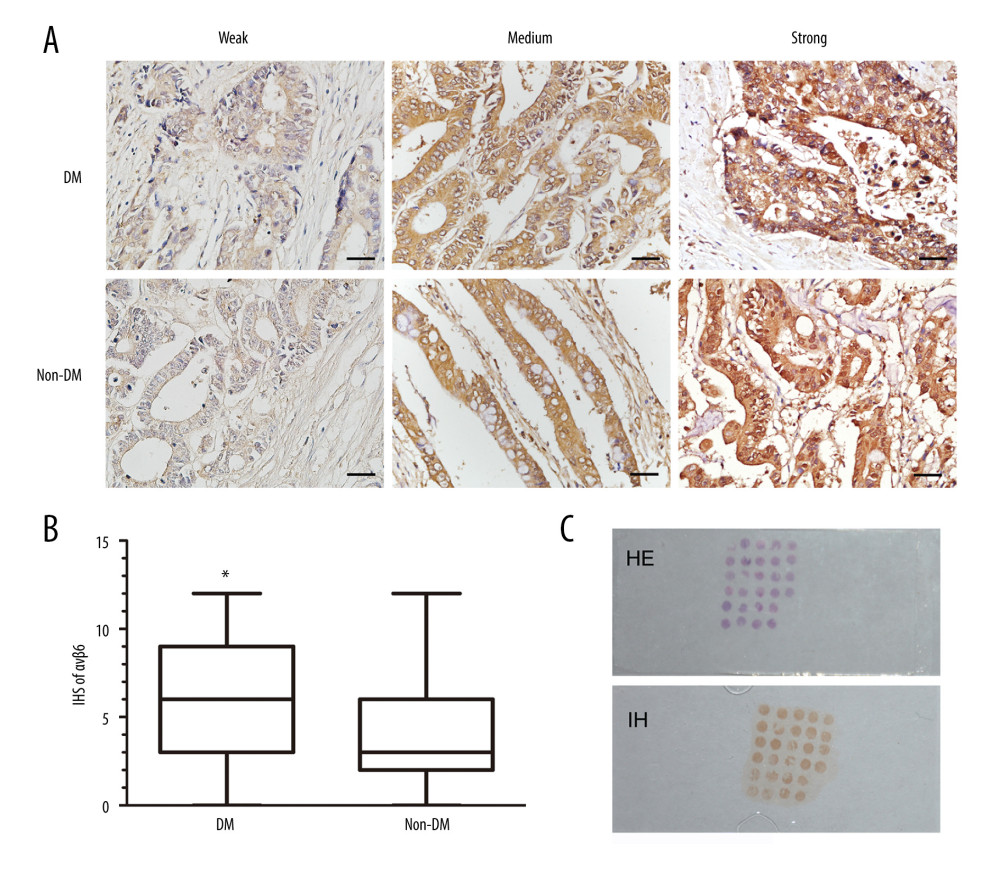 Figure 2. Immunohistochemical expression of αvβ6 in DM group and non-DM group. (A) Immunostaining of αvβ6 in representative colorectal cancer tissues of the DM group and non-DM group (×40). Bar=20 μm. (B) Box-and-whisker graph demonstrated that immunohistochemistry scores (IHS) of αvβ6 in the DM group were significantly higher than in the non-DM group. (C) Representative hematoxylin-eosin (HE) and immunohistochemical (IH) image of tissue microarray. * P<0.01 vs non-DM group.
Figure 2. Immunohistochemical expression of αvβ6 in DM group and non-DM group. (A) Immunostaining of αvβ6 in representative colorectal cancer tissues of the DM group and non-DM group (×40). Bar=20 μm. (B) Box-and-whisker graph demonstrated that immunohistochemistry scores (IHS) of αvβ6 in the DM group were significantly higher than in the non-DM group. (C) Representative hematoxylin-eosin (HE) and immunohistochemical (IH) image of tissue microarray. * P<0.01 vs non-DM group. 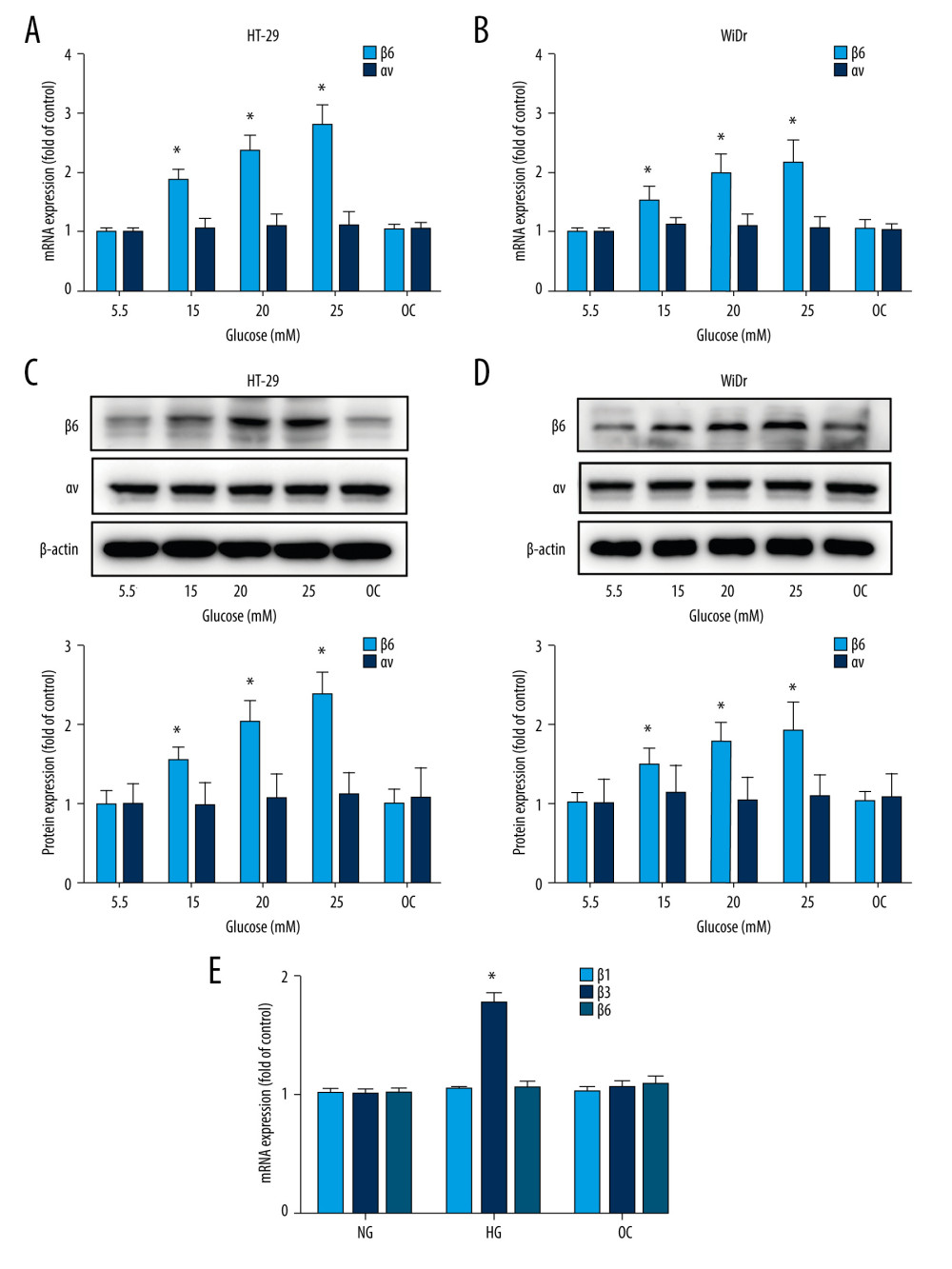 Figure 3. High glucose induced upregulation of integrin β6. HT-29 and WiDr cells were incubated with medium containing various concentrations of glucose for 24 h, and expression of integrin β6 and αv was measured by real-time PCR (A, B) and western blot (C, D). (E) Expression of integrin β1, β3, and β5 was detected by real-time PCR in HT-29 cells. Mean±SD of 3 independent experiments. * P<0.05 vs control. OC – osmotic control; NG – normal glucose; HG – high glucose.
Figure 3. High glucose induced upregulation of integrin β6. HT-29 and WiDr cells were incubated with medium containing various concentrations of glucose for 24 h, and expression of integrin β6 and αv was measured by real-time PCR (A, B) and western blot (C, D). (E) Expression of integrin β1, β3, and β5 was detected by real-time PCR in HT-29 cells. Mean±SD of 3 independent experiments. * P<0.05 vs control. OC – osmotic control; NG – normal glucose; HG – high glucose. 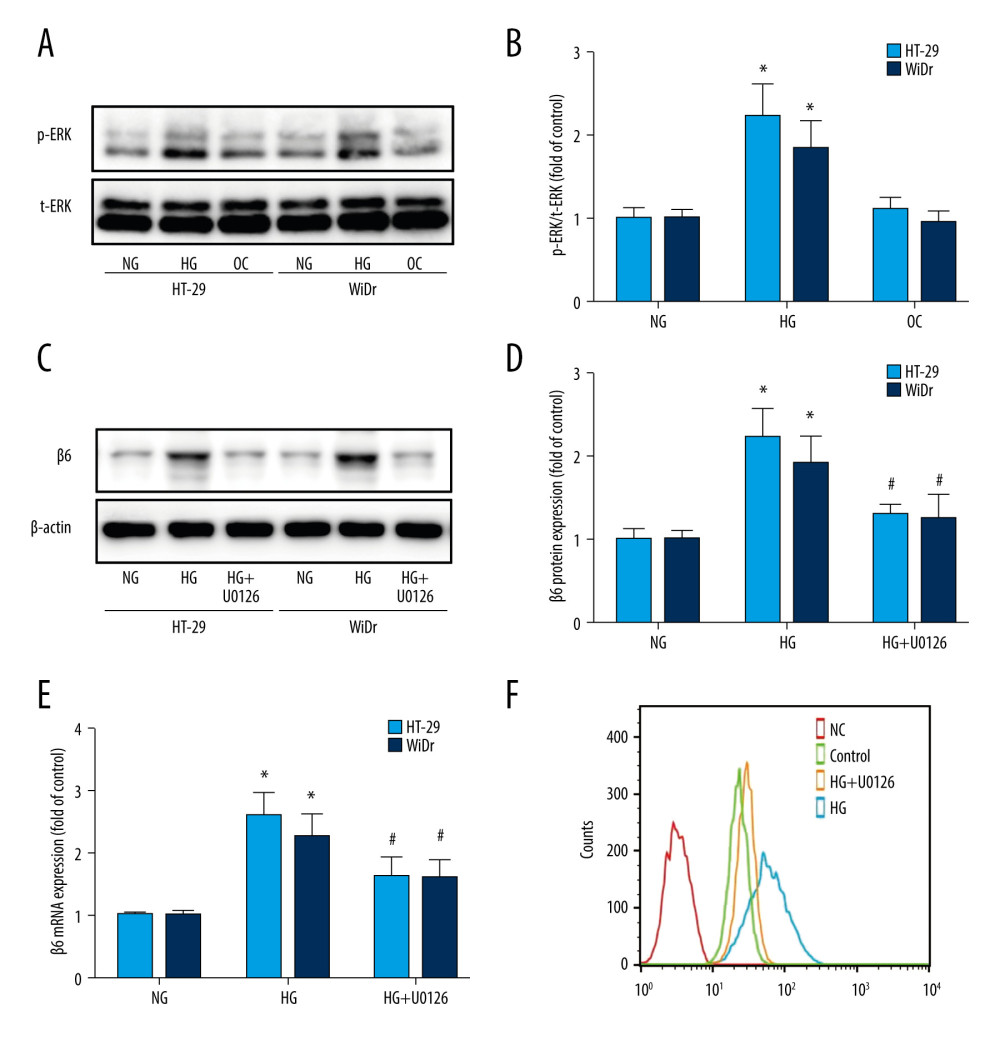 Figure 4. The ERK pathway was involved in high-glucose-induced upregulation of integrin αvβ6. (A, B) HT-29 and WiDr cells were incubated with normal glucose, high-glucose, and osmotic control medium, and expression of total-ERK (t-ERK) and phospho-ERK (p-ERK) was determined by western blot. (C–E) HT-29 and WiDr cells were pretreated with ERK-specific inhibitor U0126 for 30 min followed by stimulation with high-glucose medium for 24 h, and protein (C, D) and mRNA (E) expression of β6 subunit was detected by western blot and real-time PCR. (F) Cell surface expression of integrin αvβ6 was detected by flow cytometry in HT-29 cells. * P<0.05 vs NG. # P<0.05 vs HG. NG – normal glucose; HG – high glucose.
Figure 4. The ERK pathway was involved in high-glucose-induced upregulation of integrin αvβ6. (A, B) HT-29 and WiDr cells were incubated with normal glucose, high-glucose, and osmotic control medium, and expression of total-ERK (t-ERK) and phospho-ERK (p-ERK) was determined by western blot. (C–E) HT-29 and WiDr cells were pretreated with ERK-specific inhibitor U0126 for 30 min followed by stimulation with high-glucose medium for 24 h, and protein (C, D) and mRNA (E) expression of β6 subunit was detected by western blot and real-time PCR. (F) Cell surface expression of integrin αvβ6 was detected by flow cytometry in HT-29 cells. * P<0.05 vs NG. # P<0.05 vs HG. NG – normal glucose; HG – high glucose. 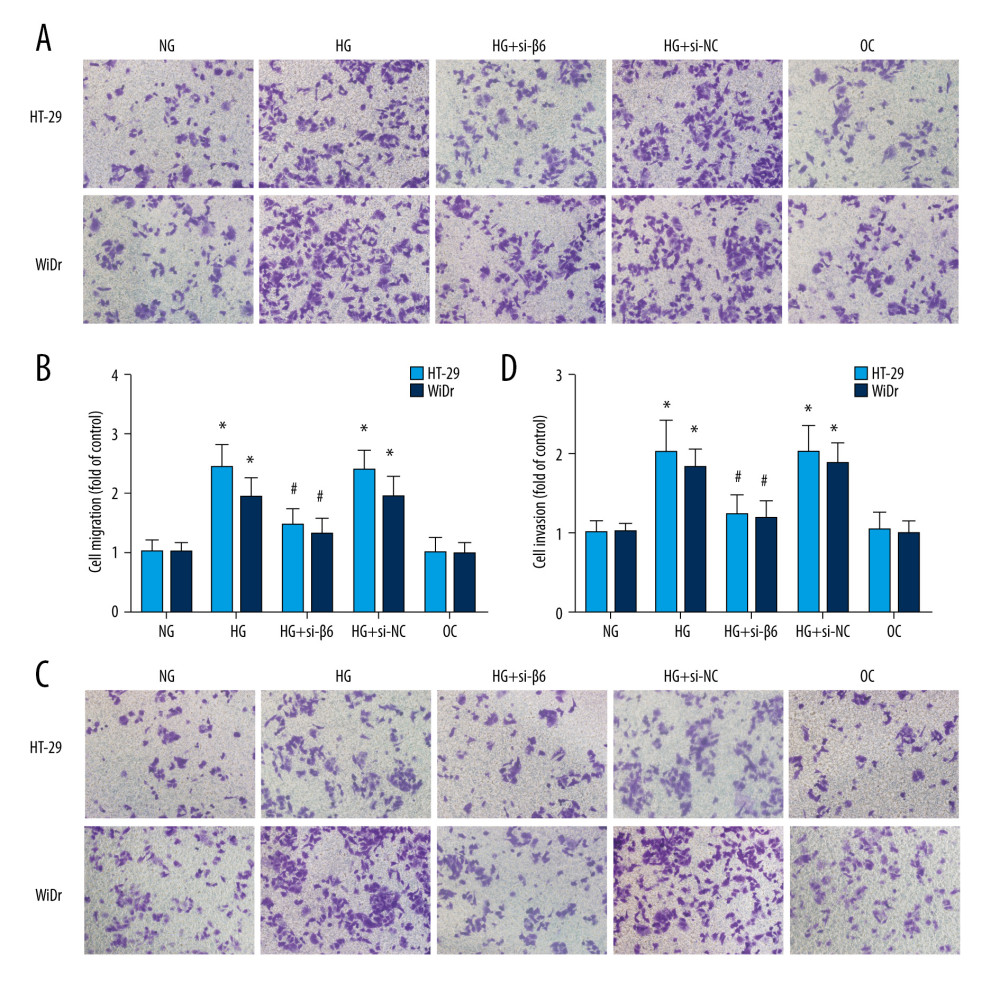 Figure 5. High glucose induced colon cancer cells migration and invasion through integrin αvβ6. HT-29 and WiDr cells were transfected with β6 siRNA (si-β6) or negative control (si-NC) for 24 h. Cell migration (A, B) and cells invasion (C, D) ability was measured with Transwell assay. * P<0.05 vs NG. # P<0.05 vs HG.
Figure 5. High glucose induced colon cancer cells migration and invasion through integrin αvβ6. HT-29 and WiDr cells were transfected with β6 siRNA (si-β6) or negative control (si-NC) for 24 h. Cell migration (A, B) and cells invasion (C, D) ability was measured with Transwell assay. * P<0.05 vs NG. # P<0.05 vs HG. 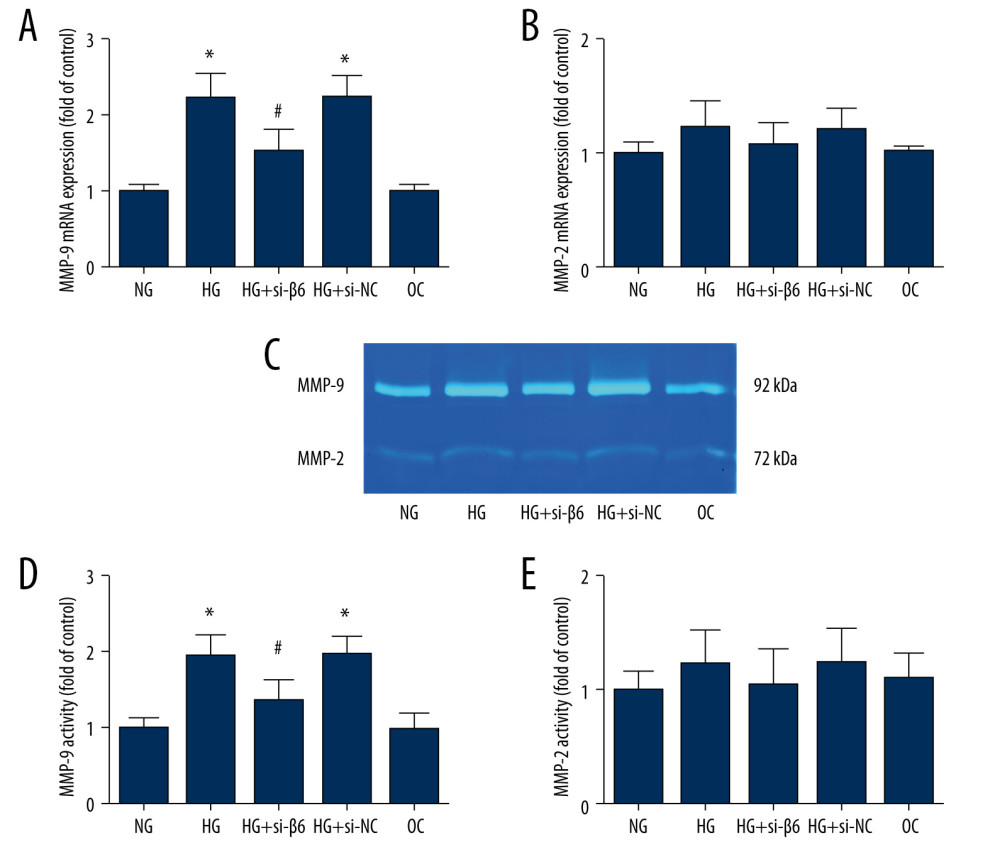 Figure 6. High glucose increased mRNA expression and activity of MMP-9. HT-29 cells were transfected with β6 siRNA or negative control for 24 h, and cells were incubated in normal glucose, high-glucose, or osmotic control medium. (A, B) mRNA expression of metalloproteinase-9 (MMP-9) and metalloproteinase-2 (MMP-2) was measured by real-time PCR. (C–E) Cell supernatant was collected, and the activity of MMP-9 and MMP-2 was determined by gelatin zymography. * P<0.05 vs NG. # P<0.05 vs HG.
Figure 6. High glucose increased mRNA expression and activity of MMP-9. HT-29 cells were transfected with β6 siRNA or negative control for 24 h, and cells were incubated in normal glucose, high-glucose, or osmotic control medium. (A, B) mRNA expression of metalloproteinase-9 (MMP-9) and metalloproteinase-2 (MMP-2) was measured by real-time PCR. (C–E) Cell supernatant was collected, and the activity of MMP-9 and MMP-2 was determined by gelatin zymography. * P<0.05 vs NG. # P<0.05 vs HG. 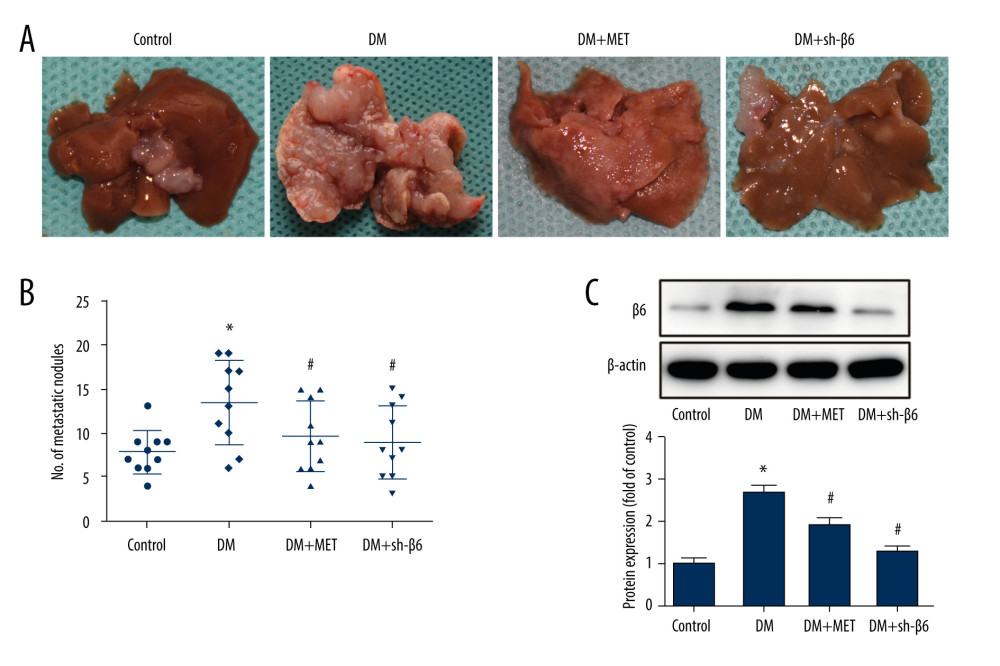 Figure 7. Diabetes promoted liver metastasis through integrin αvβ6 in the mouse model. (A) Liver metastasis was induced by intrasplenic injection of CT26 cells. (B) The number of liver metastatic nodules in each group is shown. (C) Protein expression of β6 was detected by western blot. * P<0.05 vs control. # P<0.05 vs DM. DM – diabetic mice; DM+MET – diabetic mice treated with oral administration of metformin; DM+sh-β6 – diabetic mice treated by injection of β6 shRNA.
Figure 7. Diabetes promoted liver metastasis through integrin αvβ6 in the mouse model. (A) Liver metastasis was induced by intrasplenic injection of CT26 cells. (B) The number of liver metastatic nodules in each group is shown. (C) Protein expression of β6 was detected by western blot. * P<0.05 vs control. # P<0.05 vs DM. DM – diabetic mice; DM+MET – diabetic mice treated with oral administration of metformin; DM+sh-β6 – diabetic mice treated by injection of β6 shRNA. References
1. Patel MM, Patel BM, Repurposing of sodium valproate in colon cancer associated with diabetes mellitus: Role of HDAC inhibition: Eur J Pharm Sci, 2018; 121; 188-99
2. Pothuraju R, Rachagani S, Junker WM, Pancreatic cancer associated with obesity and diabetes: an alternative approach for its targeting: J Exp Clin Cancer Res, 2018; 37(1); 319
3. Luo J, Hendryx M, Qi L, Pre-existing diabetes and lung cancer prognosis: Br J Cancer, 2016; 115(1); 76-79
4. Samuel SM, Varghese E, Varghese S, Busselberg D, Challenges and perspectives in the treatment of diabetes associated breast cancer: Cancer Treat Rev, 2018; 70; 98-111
5. Sayyid RK, Fleshner NE, Diabetes mellitus type 2: A driving force for urological complications: Trends Endocrinol Metab, 2016; 27(5); 249-61
6. Yoon JM, Son KY, Eom CS, Pre-existing diabetes mellitus increases the risk of gastric cancer: A meta-analysis: World J Gastroenterol, 2013; 19(6); 936-45
7. Lee MY, Lin KD, Hsiao PJ, Shin SJ, The association of diabetes mellitus with liver, colon, lung, and prostate cancer is independent of hypertension, hyperlipidemia, and gout in Taiwanese patients: Metabolism, 2012; 61(2); 242-49
8. Lee SJ, Kim JH, Park SJ, Optimal glycemic target level for colon cancer patients with diabetes: Diabetes Res Clin Pract, 2017; 124; 66-71
9. Terai K, Sakamoto K, Goto M, Greater development of 1,2-dimethylhydrazine-induced colon cancer in a rat model of type 2 diabetes mellitus: J Int Med Res, 2006; 34(4); 385-89
10. Shimomoto T, Ohmori H, Luo Y, Diabetes-associated angiotensin activation enhances liver metastasis of colon cancer: Clin Exp Metastasis, 2012; 29(8); 915-25
11. Luo Y, Ohmori H, Shimomoto T, Anti-angiotensin and hypoglycemic treatments suppress liver metastasis of colon cancer cells: Pathobiology, 2011; 78(5); 285-90
12. Fujiwara-Tani R, Sasaki T, Fujii K, Diabetes mellitus is associated with liver metastasis of colorectal cancer through production of biglycan-rich cancer stroma: Oncotarget, 2020; 11(31); 2982-94
13. Fidler IJ, The pathogenesis of cancer metastasis: The ‘seed and soil’ hypothesis revisited: Nat Rev Cancer, 2003; 3(6); 453-58
14. Hynes RO, Integrins: Bidirectional, allosteric signaling machines: Cell, 2002; 110(6); 673-87
15. Bandyopadhyay A, Raghavan S, Defining the role of integrin alphavbeta6 in cancer: Curr Drug Targets, 2009; 10(7); 645-52
16. Niu J, Gu X, Turton J, Meldrum C, Integrin-mediated signalling of gelatinase B secretion in colon cancer cells: Biochem Biophys Res Commun, 1998; 249(1); 287-91
17. Yang GY, Xu KS, Pan ZQ, Integrin alpha v beta 6 mediates the potential for colon cancer cells to colonize in and metastasize to the liver: Cancer Sci, 2008; 99(5); 879-87
18. Zhang ZY, Xu KS, Wang JS, Integrin alphanvbeta6 acts as a prognostic indicator in gastric carcinoma: Clin Oncol (R Coll Radiol), 2008; 20(1); 61-66
19. Zhao-Yang Z, Ke-Sen X, Qing-Si H, Signaling and regulatory mechanisms of integrin alphavbeta6 on the apoptosis of colon cancer cells: Cancer Lett, 2008; 266(2); 209-15
20. Peng C, Liu X, Liu E, Norcantharidin induces HT-29 colon cancer cell apoptosis through the alphavbeta6-extracellular signal-related kinase signaling pathway: Cancer Sci, 2009; 100(12); 2302-8
21. Niu W, Liu X, Zhang Z, Effects of alphavbeta6 gene silencing by RNA interference in PANC-1 pancreatic carcinoma cells: Anticancer Res, 2010; 30(1); 135-42
22. Wang J, Wu J, Hong J, PKC promotes the migration of colon cancer cells by regulating the internalization and recycling of integrin alphavbeta6: Cancer Lett, 2011; 311(1); 38-47
23. Liu S, Wang J, Niu W, The beta6-integrin-ERK/MAP kinase pathway contributes to chemo resistance in colon cancer: Cancer Lett, 2013; 328(2); 325-34
24. Vidakovic M, Grdovic N, Dinic S, The Importance of the CXCL12/CXCR4 axis in therapeutic approaches to diabetes mellitus attenuation: Front Immunol, 2015; 6; 403
25. Karimabad MN, Hassanshahi G, Significance of CXCL12 in type 2 diabetes mellitus and its associated complications: Inflammation, 2015; 38(2); 710-17
26. Wang B, Wang W, Niu W, SDF-1/CXCR4 axis promotes directional migration of colorectal cancer cells through upregulation of integrin alphavbeta6: Carcinogenesis, 2014; 35(2); 282-91
27. Marechal R, Demetter P, Nagy N, High expression of CXCR4 may predict poor survival in resected pancreatic adenocarcinoma: Br J Cancer, 2009; 100(9); 1444-51
28. Gao H, Peng C, Liang B, beta6 integrin induces the expression of metalloproteinase-3 and metalloproteinase-9 in colon cancer cells via ERK-ETS1 pathway: Cancer Lett, 2014; 354(2); 427-37
29. Wang WK, Wang B, Lu QH, Inhibition of high-mobility group box 1 improves myocardial fibrosis and dysfunction in diabetic cardiomyopathy: Int J Cardiol, 2014; 172(1); 202-12
30. Niu J, Dorahy DJ, Gu X, Integrin expression in colon cancer cells is regulated by the cytoplasmic domain of the beta6 integrin subunit: Int J Cancer, 2002; 99(4); 529-37
31. Gonzalez N, Prieto I, Del Puerto-Nevado L, 2017 update on the relationship between diabetes and colorectal cancer: Epidemiology, potential molecular mechanisms and therapeutic implications: Oncotarget, 2017; 8(11); 18456-85
32. Ikemura M, Nishikawa M, Kusamori K, Pivotal role of oxidative stress in tumor metastasis under diabetic conditions in mice: J Control Release, 2013; 170(2); 191-97
33. Mills KT, Bellows CF, Hoffman AE, Diabetes mellitus and colorectal cancer prognosis: A meta-analysis: Dis Colon Rectum, 2013; 56(11); 1304-19
34. Bates RC, Bellovin DI, Brown C, Transcriptional activation of integrin beta6 during the epithelial-mesenchymal transition defines a novel prognostic indicator of aggressive colon carcinoma: J Clin Invest, 2005; 115(2); 339-47
35. Ha BG, Park JE, Cho HJ, Inhibitory effects of proton beam irradiation on integrin expression and signaling pathway in human colon carcinoma HT29 cells: Int J Oncol, 2015; 46(6); 2621-28
36. Gao D, Gao L, Zhang C, A near-infrared phthalocyanine dye-labeled agent for integrin alphavbeta6-targeted theranostics of pancreatic cancer: Biomaterials, 2015; 53; 229-38
37. Zhang YH, Gao ZF, Dong GH, Suppression of alphavbeta6 downregulates P-glycoprotein and sensitizes multidrug-resistant breast cancer cells to anticancer drugs: Neoplasma, 2020; 67(2); 379-88
38. Liu S, Wang J, Niu W, The beta6-integrin-ERK/MAP kinase pathway contributes to chemo resistance in colon cancer: Cancer Lett, 2013; 328(2); 325-34
39. Liang B, Shahbaz M, Wang Y, Integrinbeta6-targeted immunoliposomes mediate tumor-specific drug delivery and enhance therapeutic efficacy in colon carcinoma: Clin Cancer Res, 2015; 21(5); 1183-95
40. Stattin P, Bjor O, Ferrari P, Prospective study of hyperglycemia and cancer risk: Diabetes Care, 2007; 30(3); 561-67
41. Park J, Sarode VR, Euhus D, Neuregulin 1-HER axis as a key mediator of hyperglycemic memory effects in breast cancer: Proc Natl Acad Sci USA, 2012; 109(51); 21058-63
42. Shi GJ, Shi GR, Zhou JY, Involvement of growth factors in diabetes mellitus and its complications: A general review: Biomed Pharmacother, 2018; 101; 510-27
43. Tan HX, Gong WZ, Zhou K, CXCR4/TGF-beta1 mediated hepatic stellate cells differentiation into carcinoma-associated fibroblasts and promoted liver metastasis of colon cancer: Cancer Biol Ther, 2020; 21(3); 258-68
44. Tsushima H, Ito N, Tamura S, Circulating transforming growth factor beta 1 as a predictor of liver metastasis after resection in colorectal cancer: Clin Cancer Res, 2001; 7(5); 1258-62
45. Bi J, Koivisto L, Dai J, Epidermal growth factor receptor signaling suppresses alphavbeta6 integrin and promotes periodontal inflammation and bone loss: J Cell Sci, 2019; 133(5); jcs263588
46. Xu R, Xu M, Fu Y, Transforming growth factor-beta1 and lysophosphatidic acid activate integrin beta6 gene promoter in Hep-3B cells: Oncol Lett, 2018; 16(1); 439-46
Figures
 Figure 1. Kaplan-Meier analysis of liver metastasis-free survival (LMFS) and overall survival (OS) for CRC patients with diabetes mellitus (DM group) and without diabetes mellitus (non-DM group). (A) LMFS curve of stage II patients (P<0.01). (B) LMFS curve of stage III patients (P<0.01). (C) OS curve of stage II patients (P<0.01). (D) OS curve of stage III patients (P<0.01). Significance of difference was evaluated by log-rank test.
Figure 1. Kaplan-Meier analysis of liver metastasis-free survival (LMFS) and overall survival (OS) for CRC patients with diabetes mellitus (DM group) and without diabetes mellitus (non-DM group). (A) LMFS curve of stage II patients (P<0.01). (B) LMFS curve of stage III patients (P<0.01). (C) OS curve of stage II patients (P<0.01). (D) OS curve of stage III patients (P<0.01). Significance of difference was evaluated by log-rank test. Figure 2. Immunohistochemical expression of αvβ6 in DM group and non-DM group. (A) Immunostaining of αvβ6 in representative colorectal cancer tissues of the DM group and non-DM group (×40). Bar=20 μm. (B) Box-and-whisker graph demonstrated that immunohistochemistry scores (IHS) of αvβ6 in the DM group were significantly higher than in the non-DM group. (C) Representative hematoxylin-eosin (HE) and immunohistochemical (IH) image of tissue microarray. * P<0.01 vs non-DM group.
Figure 2. Immunohistochemical expression of αvβ6 in DM group and non-DM group. (A) Immunostaining of αvβ6 in representative colorectal cancer tissues of the DM group and non-DM group (×40). Bar=20 μm. (B) Box-and-whisker graph demonstrated that immunohistochemistry scores (IHS) of αvβ6 in the DM group were significantly higher than in the non-DM group. (C) Representative hematoxylin-eosin (HE) and immunohistochemical (IH) image of tissue microarray. * P<0.01 vs non-DM group. Figure 3. High glucose induced upregulation of integrin β6. HT-29 and WiDr cells were incubated with medium containing various concentrations of glucose for 24 h, and expression of integrin β6 and αv was measured by real-time PCR (A, B) and western blot (C, D). (E) Expression of integrin β1, β3, and β5 was detected by real-time PCR in HT-29 cells. Mean±SD of 3 independent experiments. * P<0.05 vs control. OC – osmotic control; NG – normal glucose; HG – high glucose.
Figure 3. High glucose induced upregulation of integrin β6. HT-29 and WiDr cells were incubated with medium containing various concentrations of glucose for 24 h, and expression of integrin β6 and αv was measured by real-time PCR (A, B) and western blot (C, D). (E) Expression of integrin β1, β3, and β5 was detected by real-time PCR in HT-29 cells. Mean±SD of 3 independent experiments. * P<0.05 vs control. OC – osmotic control; NG – normal glucose; HG – high glucose. Figure 4. The ERK pathway was involved in high-glucose-induced upregulation of integrin αvβ6. (A, B) HT-29 and WiDr cells were incubated with normal glucose, high-glucose, and osmotic control medium, and expression of total-ERK (t-ERK) and phospho-ERK (p-ERK) was determined by western blot. (C–E) HT-29 and WiDr cells were pretreated with ERK-specific inhibitor U0126 for 30 min followed by stimulation with high-glucose medium for 24 h, and protein (C, D) and mRNA (E) expression of β6 subunit was detected by western blot and real-time PCR. (F) Cell surface expression of integrin αvβ6 was detected by flow cytometry in HT-29 cells. * P<0.05 vs NG. # P<0.05 vs HG. NG – normal glucose; HG – high glucose.
Figure 4. The ERK pathway was involved in high-glucose-induced upregulation of integrin αvβ6. (A, B) HT-29 and WiDr cells were incubated with normal glucose, high-glucose, and osmotic control medium, and expression of total-ERK (t-ERK) and phospho-ERK (p-ERK) was determined by western blot. (C–E) HT-29 and WiDr cells were pretreated with ERK-specific inhibitor U0126 for 30 min followed by stimulation with high-glucose medium for 24 h, and protein (C, D) and mRNA (E) expression of β6 subunit was detected by western blot and real-time PCR. (F) Cell surface expression of integrin αvβ6 was detected by flow cytometry in HT-29 cells. * P<0.05 vs NG. # P<0.05 vs HG. NG – normal glucose; HG – high glucose. Figure 5. High glucose induced colon cancer cells migration and invasion through integrin αvβ6. HT-29 and WiDr cells were transfected with β6 siRNA (si-β6) or negative control (si-NC) for 24 h. Cell migration (A, B) and cells invasion (C, D) ability was measured with Transwell assay. * P<0.05 vs NG. # P<0.05 vs HG.
Figure 5. High glucose induced colon cancer cells migration and invasion through integrin αvβ6. HT-29 and WiDr cells were transfected with β6 siRNA (si-β6) or negative control (si-NC) for 24 h. Cell migration (A, B) and cells invasion (C, D) ability was measured with Transwell assay. * P<0.05 vs NG. # P<0.05 vs HG. Figure 6. High glucose increased mRNA expression and activity of MMP-9. HT-29 cells were transfected with β6 siRNA or negative control for 24 h, and cells were incubated in normal glucose, high-glucose, or osmotic control medium. (A, B) mRNA expression of metalloproteinase-9 (MMP-9) and metalloproteinase-2 (MMP-2) was measured by real-time PCR. (C–E) Cell supernatant was collected, and the activity of MMP-9 and MMP-2 was determined by gelatin zymography. * P<0.05 vs NG. # P<0.05 vs HG.
Figure 6. High glucose increased mRNA expression and activity of MMP-9. HT-29 cells were transfected with β6 siRNA or negative control for 24 h, and cells were incubated in normal glucose, high-glucose, or osmotic control medium. (A, B) mRNA expression of metalloproteinase-9 (MMP-9) and metalloproteinase-2 (MMP-2) was measured by real-time PCR. (C–E) Cell supernatant was collected, and the activity of MMP-9 and MMP-2 was determined by gelatin zymography. * P<0.05 vs NG. # P<0.05 vs HG. Figure 7. Diabetes promoted liver metastasis through integrin αvβ6 in the mouse model. (A) Liver metastasis was induced by intrasplenic injection of CT26 cells. (B) The number of liver metastatic nodules in each group is shown. (C) Protein expression of β6 was detected by western blot. * P<0.05 vs control. # P<0.05 vs DM. DM – diabetic mice; DM+MET – diabetic mice treated with oral administration of metformin; DM+sh-β6 – diabetic mice treated by injection of β6 shRNA.
Figure 7. Diabetes promoted liver metastasis through integrin αvβ6 in the mouse model. (A) Liver metastasis was induced by intrasplenic injection of CT26 cells. (B) The number of liver metastatic nodules in each group is shown. (C) Protein expression of β6 was detected by western blot. * P<0.05 vs control. # P<0.05 vs DM. DM – diabetic mice; DM+MET – diabetic mice treated with oral administration of metformin; DM+sh-β6 – diabetic mice treated by injection of β6 shRNA. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387