10 August 2022: Clinical Research
A Retrospective Study from a Single Center to Identify Hematological Factors that Distinguish Between Patients with Colorectal Carcinoma and Colorectal Adenoma
Jun Huang12AE, Pingping Xu3BCD, Youxiang Chen4AE*DOI: 10.12659/MSM.936745
Med Sci Monit 2022; 28:e936745
Abstract
BACKGROUND: This retrospective study from a single center in China was conducted using data from medical records between 2012 and 2020, to identify hematological factors that distinguish between patients with colorectal carcinoma (CRC) and colorectal adenoma.
MATERIAL AND METHODS: In this case-control study, 856 eligible patients were randomly divided into a training set (n=600) and a testing set (n=256). Routine blood parameters, blood coagulation, and liver and kidney function parameters were collected. Univariate and multivariate Cox regression analyses were used to explore diagnostic indicators. The values of the area under the curve and calibration curves were used to evaluate the model.
RESULTS: The study included 325 colorectal adenoma and 531 CRC patients. The prediction model for diagnosing CRC using hemoglobin-to-platelet ratio, fibrinogen-albumin ratio (FAR), albumin-globulin ratio (A/G), platelet-lymphocyte ratio, carcinoembryonic antigen (CEA), and thrombin time (TT) was developed on the basis of the patients grouped into the CRC and colorectal adenoma groups. The prediction model for diagnosing CRC stage was developed using prothrombin time (PT), TT, CEA, A/G, FAR, and HPR. The prediction model for diagnosing CRC grade was developed using PT, TT, A/G, plateletcrit, FAR, and HPR. The AUCs of the 3 prediction models were [0.848, 95% CI: (0.800-0.896)], [0.806, 95% CI: (0.775-0.836)], and [0.829, 95% CI: (0.797-0.860)] in the testing set.
CONCLUSIONS: Three diagnostic prediction models for early screening of CRC, stage of CRC, and grade of CRC were established through a panel of readily available hematological parameters, which could provide auxiliary tools for early screening of CRC.
Keywords: Colorectal Neoplasms, Diagnosis, Polyposis Syndrome, Hereditary Mixed, 1, Adenoma, Albumins, Biomarkers, Tumor, Carcinoembryonic Antigen, Case-Control Studies, Humans
Background
Colorectal carcinoma (CRC) is the fourth most common cancer in women and the third most common cancer in men, and CRC is the leading cause of cancer deaths worldwide [1,2]. In 2020, there were an estimated 0.55 million new cases of CRC, and 0.28 million deaths, in China [3,4]. CRC disease progresses slowly, and many patients do not have clinical manifestations such as abdominal pain or intestinal bleeding until the cancer metastasizes, leading to delay in treatment [5]. CRC mostly results from colon or rectal epithelial polyps, of which colorectal adenoma is the most common neoplastic polyp, and is associated with an increased risk of CRC [6,7]. According to the National Comprehensive Cancer Network recommendations, early screening could prevent CRC by detecting colorectal lesions early and treating them appropriately [8]. Colonoscopy is the gold standard for screening for CRC and distinguishing between CRC and colorectal adenoma [2,9]. However, its application in early screening is limited due to its high cost and inconvenience, and the fact that it can easily lead to unnecessary treatments [10,11]. Therefore, the development of non-invasive and easily available markers to distinguish CRC from colorectal adenoma is of great significance to avoid unnecessary treatments and improve the accuracy of CRC detection [12]. Blood is invariably the most desirable analyte for cancer biomarkers due to its availability and high patient acceptance [12].
Studies have shown several hematological factors to be correlated with poor survival rates of CRC and other cancers, including platelet-to-lymphocyte (LYM) ratio (PLR) [13], monocyte (MONO)-to-LYM ratio (MLR) [14], neutrophil (NEUT)-to-LYM ratio (NLR) [15], systemic immune-inflammatory index (SII) [16], and others. Recently, the relationship between platelet-related parameters (platelets, mean platelet volume (MPV), and plateletcrit, the product of platelets and MPV) and CRC has also been reported [17,18]. Wu et al [19] stated that the MPV-to-platelets ratio could be used as an effective diagnostic index to distinguish benign from malignant CRC. In addition, carcinoembryonic antigen (CEA) is a common tumor marker that has assisted in the diagnosis of CRC [20]. A machine-learning model using biochemical and hematological markers was built to identify CRC patients [21]. Several studies have established models of diagnosing CRC based on inflammation factors, platelet-related markers, or CEA [22,23], but most studies have excluded people with inflammatory diseases. Clinically, some patients with suspected CRC have inflammatory diseases. Excluding patients with inflammatory diseases may limit the application of the prediction model to the population. The performance of the existing prediction model of CRC diagnosis is limited in its application in clinical practice, and needs to be further improved.
Therefore, this retrospective study from a single center in China was conducted using data from patient medical records between 2012 to 2020, to identify hematological factors that distinguish between patients with colorectal carcinoma and CRC.
Material and Methods
STUDY POPULATION:
This case-control study was approved by the Ethics Committee of Jiangxi Cancer Hospital (Affiliated Cancer Hospital of Nanchang University), and informed consent was obtained from all the participants. Demographic and clinical data for 980 patients were collected from the electronic medical records of Jiangxi Cancer Hospital (Affiliated Cancer Hospital of Nanchang University) from May 2012 to September 2020. The inclusion criteria for the CRC and colorectal adenoma groups were patients who had not received anti-tumor therapy and were diagnosed with CRC or colorectal adenoma by histopathology. The exclusion criteria were: (1) patient age less than 18 years, (2) patients diagnosed with distant metastasis of CRC, (3) patients with hematological disease, and (4) patients with a history of blood transfusion within 3 months. According to the inclusion and exclusion criteria, 856 CRC and colorectal adenoma patients were finally included in this study (Figure 1). Then, these patients were randomly divided into 2 groups: the training set (n=600) and the testing set (n=256), at a ratio of 7: 3.
DIAGNOSIS OF CRC AND COLORECTAL ADENOMA:
CRC and colorectal adenoma were diagnosed into distinct pathological types assigned after surgical resection or endoscopic resection. The histological types were categorized into either colorectal adenoma, on the basis of the International Classification of Diseases (ICD-0) (8210/0 and 8210/2), ICD-11 (2E92.4Y & XH7SY6)], or CRC [ICD-0 (8140/3), ICD-11 (2B90.Y & XH74S1). The diagnosis of CRC was according to the eighth edition of the American Joint Committee on Cancer/Tumor, Node, Metastases tumor staging (including pathological staging, degree of differentiation, lymph node metastasis, vascular invasion, and other values).
DATA COLLECTION:
We collected age, gender, body mass index [BMI, (kg/m2)], routine blood parameters, blood coagulation parameters, liver and kidney function parameters, and tumor biomarkers for eligible patients. The routine blood parameters and blood coagulation parameters were hemoglobin [Hb (g/L)], red blood cell count [RBC (1012/L)], white blood cell count [WBC (109/L)], NEUT (109/L), LYM (109/L), MONO (109/L), platelets (109/L), MPV (fL), plateletcrit (%), red blood cell distribution width [RDW (%)], prothrombin time [PT (s)], activated partial thromboplastin time [APTT (s)], thrombin time [TT (s)], fibrinogen [FIB (g/L)], and D-dimer (mg/L). Routine blood and blood coagulation parameters were measured by a Mindray 5800 plus automatic blood cell analyzer and Sysmex CS5001 blood coagulation analyzer, respectively. Liver and kidney function parameters included glutamic pyruvic transaminase [GPT (U/L)], glutamic oxaloacetic transaminase [GOT (U/L)], total bilirubin [TBil (μmol/L)], albumin [ALB (g/L)], globulin [GLB (g/L)], ALB/GLB (A/G), blood urea nitrogen [BUN (mmol/L)], creatinine [Cr (μmol/L)], and uric acid [UA (μmol/L)]. A Hitachi 7600 automatic biochemical analyzer was used for the determination of liver and kidney function parameters. Tumor biomarkers collected were: CEA (ng/mL), carbohydrate antigen 199 (U/mL), and carbohydrate antigen 724 (U/mL). Tumor biomarkers were measured using the Siemens ADVIA Centaur XPT. The values of NLR, PLR, MLR, FIB-to-ALB ratio (FAR), 10×ALB+0.005×LYM (PNI), SII, NEUT/(WBC-NEUT) (dNLR) and hemoglobin-to-platelets ratio (HPR) were calculated. In addition, we also collected data on whether eligible patients had diabetes or inflammatory diseases (pelvic infection, lung infection, abdominal infection, non-atrophic gastritis, chronic bronchitis, hepatitis B, or rheumatoid arthritis).
STATISTICAL ANALYSIS:
Quantitative data were described by mean±standard deviation (mean±SD) or median (interquartile range) [M (Q1, Q3)]. Categorical variable data were described by frequency and proportion [n (%)]. The statistical differences between the 2 groups of quantitative data were compared and analyzed using the Mann-Whitney U test or
All statistical tests used two-sided tests, and
Results
CHARACTERISTICS OF THE STUDY POPULATION:
A total of 856 patients [325 (37.97%) with colorectal adenoma, 531 (62.03%) with CRC] were included in the study. Among the patients with CRC, 98 (11.45%) were poorly differentiated, 419 (48.95%) were moderately differentiated, and 14 (1.64%) were well differentiated; and 68 (7.94%) had stage I disease, 249 (29.09%) had stage II disease, and 214 (25.00%) had stage III disease. There were 600 patients in the training set and 256 patients in the testing set. No statistical differences between the training and testing sets were observed (Table 1). Of all the selected patients, 511 (59.70%) were male and 345 (40.30%) were female. The average age was (56.39±12.15) years old, and the average BMI was (22.45±3.38) kg/m2. There were 38 (4.44%) participants who had diabetes, and 380 (44.39%) participants who had other inflammatory diseases. More detailed information about the demographic and clinicopathological characteristics of the selected patients is shown in Table 1. There was no statistical difference between the data before and after the missing values were interpolated (Supplementary Table 1).
COMPARISON OF CRC AND COLORECTAL ADENOMA CHARACTERISTICS IN THE TRAINING SET:
As shown in Table 2, significant differences were found between the CRC group and colorectal adenoma group in the following parameters: BMI (P=0.004), Hb (P<0.001), RBC (P<0.001), WBC (P=0.038), NEUT (P=0.004), LYM (P<0.001), platelets (P<0.001), MPV (P<0.001), plateletcrit (P<0.001), RDW (P<0.001), PT (P<0.001), APTT (P=0.007), TT (P<0.001), FIB (P<0.001), D-dimer (P<0.001), CEA (P<0.001), carbohydrate antigen 199 (P=0.023), GPT (P<0.001), GOT (P<0.001), TBil (P<0.001), ALB+GLB (P<0.001), ALB (P=0.004), GLB (P<0.001), A/G (P<0.001), BUN (P<0.001), UA (P<0.001), NLR (P<0.001), PLR (P<0.001), MLR (P=0.002), MPV/platelets (P<0.001), FAR (P<0.001), PNI (P=0.004), SII (P<0.001), dNLR (P<0.001), and HPR (P<0.001).
When comparing the characteristics of CRC stage I, II, and III, and colorectal adenoma, the variables with significant differences were basically the same as the variables with differences between the groups of CRC and colorectal adenoma, except that WBC, APTT, MPV, and plateletcrit were not significantly different (Supplementary Table 2). There were significant differences in the values of WBC and carbohydrate antigen 199 when comparing the characteristics of CRC (poorly differentiated, moderately differentiated, and well-differentiated) and colorectal adenoma (Supplementary Table 3).
DIAGNOSTIC INDICATORS FOR CRC PATIENTS:
In the patients grouped into CRC and colorectal adenoma (Table 3), the variable of HPR was associated with a decreased risk of being diagnosed with CRC [odds ratio (OR) =0.178, 95% confidence interval (CI): 0.065–0.487, (P<0.001)]. The variables of FAR [OR=1.590, 95% CI: 1.406–1.798, (P<0.001)], A/G [OR=1.410, 95% CI: 1.301–1.528, (P<0.001)], PLR [OR=1.004, 95% CI: 1.001–1.007, (P=0.010)], CEA [OR=1.064, 95% CI: 1.016–1.115, (P=0.009)], and TT [OR=1.585, 95% CI: 1.365–1.840, (P<0.001)] were associated with increased risks of being diagnosed with CRC. In the patients grouped into CRC stage I, II, and III, and colorectal adenoma, PT, TT, CEA, A/G, FAR, and HPR were diagnostic indicators (Table 4). In the patients grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma, PT, TT, A/G, plateletcrit, FAR, and HPR were the diagnostic indicators (Table 5).
THE MODEL DEVELOPED TO DIAGNOSE CRC:
The 6 diagnostic indicators (HPR, FAR, A/G, PLR, CEA, and TT) of CRC were used to jointly construct a prediction model for diagnosing CRC in the patients grouped into CRC and colorectal adenoma. A nomogram was generated based on the identified diagnostic indicators (Figure 2), which was built on HPR, FAR, A/G, PLR, CEA, and TT. As shown in Figure 3, we randomly selected an observation, the 140th patient. The value of each diagnostic indicator is indicated by a red dot in Figure 3. Each value corresponded to the score of the first row, and then the scores of the diagnostic indicators were added. The resulting total score, denoted Total Points, was 281 in the penultimate row, and the probability that this patient was diagnosed with CRC was 0.986. In the patients grouped into CRC stage I, II, and III, and those with colorectal adenoma, PT, TT, CEA, A/G, FAR, and HPR were used to construct a prediction model for diagnosing CRC. In the patients grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma, PT, TT, A/G, plateletcrit, FAR, and HPR were used to construct a prediction model for diagnosing CRC.
MODEL EVALUATION AND VERIFICATION:
The AUCs of the training set [0.864, (95% CI: 0.834–0.895)] and testing set [0.848, (95% CI: 0.800–0.896)] in patients grouped into CRC and colorectal adenoma (Table 6, Figure 4A), the AUCs of the training set [0.818, (95% CI: 0.799–0.838)] and testing set [0.806, (95% CI: 0.775–0.836)] in patients grouped into CRC stage I, II, and III, and colorectal adenoma (Table 6, Figure 4B), the AUCs of the training set [0.857, (95% CI: 0.838–0.876)] and testing set [0.829, (95% CI: 0.797–0.860)] in patients grouped into the poorly differentiated CRC, moderately and well-differentiated, CRC, and colorectal adenoma (Table 6, Figure 4C), suggest the prediction model distinguished between these groups well. The values of accuracy, sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) in the prediction model are also shown in Table 6. The calibration curves in Figure 4D–4F showed that the prediction model was well-calibrated. Moreover, the results in Table 7 of patients grouped into CRC and colorectal adenoma showed that our prediction model has higher AUC and PPV in patients with [0.879 (95% CI: 0.760–0.997) of AUC, 0.906 (95% CI: 0.805–0.999) of PPV] or without [0.835 (95% CI: 0.741–0.928) of AUC, 0.773 (95% CI: 0.649–0.897) of PPV] inflammatory diseases. The prediction models we developed showed good performance in patients divided into CRC stage I, II, or III, and in colorectal adenomas and patients divided into poorly differentiated CRC, moderately to well-differentiated CRC, and colorectal adenomas.
Discussion
In the present study, we found HPR, FAR, A/G, PLR, CEA, PT, TT, and plateletcrit to be diagnostic indicators of CRC, showing the ability to differentiate colorectal adenoma from CRC. A prediction model for diagnosing CRC using HPR, FAR, A/G, PLR, CEA, and TT was developed, and a nomogram was generated for the patients grouped into the CRC and colorectal adenoma groups. The other 2 prediction models for diagnosing CRC were developed (1) using PT, TT, CEA, A/G, FAR, and HPR in the patients grouped into CRC stages I, II, and III, and colorectal adenoma; and (2) using PT, TT, A/G, plateletcrit, FAR, and HPR in the patients grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. The prediction models showed great performance in calibration and discrimination.
In the identification of CRC and colorectal adenoma patients, Roman et al [24] created a prediction model and nomogram using Hb, FIB, ALB, and NLR to predict the risk of CRC in patients, with an AUC value of 0.744. Another study showed that the diagnostic value of plateletcrit combined with CEA in the diagnosis of CRC was 0.835 (AUC), and the sensitivity was 0.700, while the specificity was 0.830 [22]. The AUC values of the prediction models we proposed in this study using the diagnostic indicators were higher than those of prediction models that are used to diagnose CRC through hematological parameters; in addition, we also provided the reference PPV value. Clinically, the AUC may be different in the case of application of the nomogram to different cohorts, so a higher AUC may ensure the reliability of the prediction [25]. In addition, we did not exclude patients with inflammatory diseases. In our study population, patients with inflammatory diseases accounted for 17% of the total population, indicating that patients with inflammation diseases accounted for a large proportion of the CRC and colorectal adenoma population. Therefore, our CRC diagnosis prediction model used a wider population range than the prediction model that excluded patients with inflammation. A retrospective study in China stated that MPV-to-platelets ratio was used as an effective diagnostic index to distinguish benign from malignant CRC and distinguish early CRC from advanced CRC [19]. Hui et al [21] found that CEA, Hb, lipoprotein (a), and high-density lipoprotein might be powerful and noninvasive diagnostic indicators based on machine-learning approaches. Consistently, we have studied the diagnosis of CRC by hematological indicators, and there is also agreement on diagnostic indicators such as platelet-related parameters and CEA. These 2 studies considered the diagnosis and identification of early and advanced CRC. In addition to CRC stage, we also considered the grade of CRC, and established a related prediction model in participants grouped into CRC that was poorly differentiated, CRC that was moderately and well differentiated, or colorectal adenoma.
There are many diagnostic methods for CRC, including colonoscopy, colorectal barium enema, nuclear magnetic resonance, intracavitary ultrasound, blood tumor markers, fecal occult blood test (FOBT), genetic examination, and others [26]. Among these, colonoscopy is currently the most reliable method for the diagnosis of CRC, but invasiveness, poor compliance, and high cost has limited its application in large-scale screening [26,27]. Computerized tomography (CT), magnetic resonance imaging (MRI), and other imaging examinations do not easily detect small lesions, which limits the early diagnosis of CRC by these methods [28]. FOBT had the advantages of simplicity, non-invasiveness, and economy, but it is prone to false-positive results [29]. It therefore requires multiple and repeated tests to increase its detection rate. In contrast, the method of CRC detection based on inflammation markers, blood biomarkers, and CEA as combined indicators developed in the present study has the advantages of low cost, rapid detection, and acceptability [12,30].
Inflammation plays an important role in the proliferation and survival of malignant cells by changing the homeostasis of tumor tissues and participating in the construction of the tumor microenvironment [31]. Platelets have gradually become regarded as multipurpose cells. In addition to their traditional roles in hemostasis and thrombosis, platelets are also involved in the complex interaction between inflammation and cancer [18]. PLR as a typical inflammatory indicator has been previously related to CRC [32]. In a retrospective study evaluating the prognosis of 201 CRC patients treated with curative surgery, overall survival and relapse-free survival were significantly higher in CRC patients in the low-PLR group than in those in the high-PLR group [33]. Also, CRC patients have exhibited significantly higher PLR values than colorectal adenoma patients and healthy controls [34], a finding that was consistent with our results. There have been few predictive models for HPR and FAR to diagnose CRC, but HPR and FAR are related to platelets and related inflammatory factors. In our study, we found that CRC patients have lower HPR and higher FAR levels, compared with colorectal adenoma patients. A/G is a common inflammation-related indicator that has been used previously as a prognostic indicator for CRC patients [35]. The NLR was derived from NEU and LYM of the complete blood count. It has become one of the commonly used inflammation markers due to its cost effectiveness [36]. Several studies have showed NLR to be useful for the diagnosis and prognosis of breast cancer [37], cervical cancer [38,39], non-small cell lung cancer [40], CRC [15] and other malignant tumors. Although we found differences in NLR in the comparison between CRC and colorectal adenoma, no differences were found in the multivariate logistic regression.
CEA was initially considered to be CRC-specific, but later, CEA levels were also detected in other tumors, such as gastric cancer and pancreatic cancer [41]. In addition, CEA could not distinguish between benign and malignant polyps, and CEA is not currently recommended for screening tests according to the guidelines of the European Group on Tumor Markers, European Society of Medical Oncology, and American Society of Clinical Oncology [42,43]. Therefore, we used CEA as one of the combined diagnostic indicators for CRC diagnosis in our study. Similarly, the use of CEA combined with other hematological indicators for CRC diagnosis has also been reported in other studies [15,23].
However, a few limitations were present in our study. First, this study could only provide a reference for early screening of CRC; we did not conduct regular followup of the eligible patients, so we were unable to obtain prognostic status after diagnostic analysis. Second, we only studied several hematological parameters (HPR, FAR, A/G, PLR, CEA, PT, TT, and plateletcrit) for the diagnostic value of CRC and colorectal adenoma, providing a reference for the early detection of CRC. However, other indicators of potential research value, such as genes [44], were not included in the study due to the limitation of retrospective data. Third, although we have verified the prediction models used to diagnose CRC, our research lacks sufficient external data, and further support and verification are needed. Therefore, multi-center and large-sample prospective studies are still needed to verify the accuracy of the results.
Conclusions
In this study, we developed 3 prediction models for the diagnosis of CRC, stage of CRC, and grade of CRC that used several readily available hematological parameters (HPR, FAR, A/G, PLR, CEA, PT, TT, and plateletcrit) as diagnostic indicators. The prediction models were validated with good discrimination and calibration. Our research provided a reference for the development of auxiliary tools for the early screening of CRC.
Figures
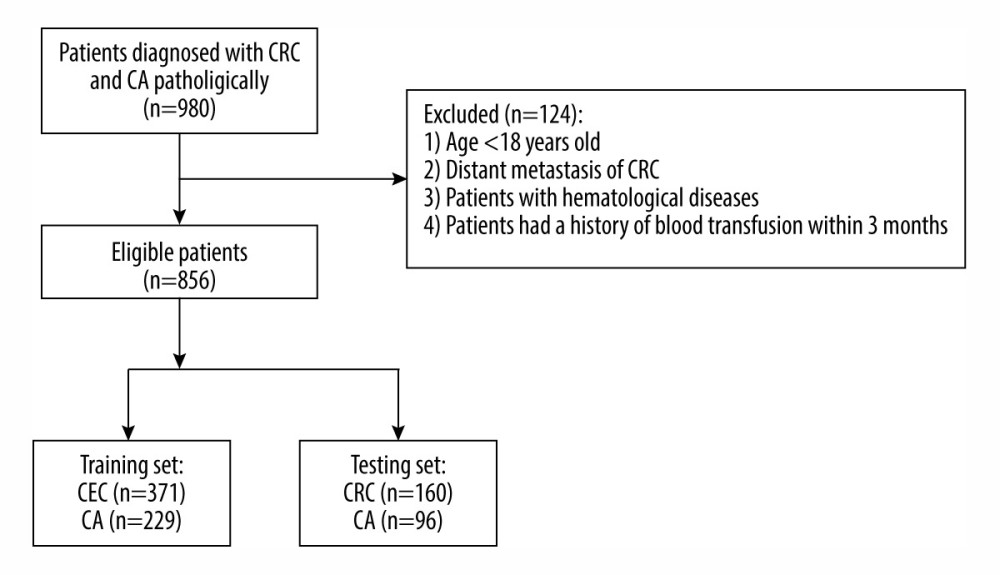 Figure 1. Flow chart of the selected patients created using Drawio v. 13.9.9 (diagrams.net). CRC – colorectal cancer; CA – colorectal adenoma; CEA – carcinoembryonic antigen.
Figure 1. Flow chart of the selected patients created using Drawio v. 13.9.9 (diagrams.net). CRC – colorectal cancer; CA – colorectal adenoma; CEA – carcinoembryonic antigen. ![Nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio.](https://jours.isi-science.com/imageXml.php?i=medscimonit-28-e936745-g002.jpg&idArt=936745&w=1000) Figure 2. Nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio.
Figure 2. Nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio. ![An example of the nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio.](https://jours.isi-science.com/imageXml.php?i=medscimonit-28-e936745-g003.jpg&idArt=936745&w=1000) Figure 3. An example of the nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio.
Figure 3. An example of the nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio. ![Receiver operating characteristic (ROC) curves for the training set and testing set in the patients when grouped into the following comparative subgroups: (A) colorectal cancer (CRC) and colorectal adenoma; (B) stage I, II, and III CRC and colorectal adenoma; (C) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. Calibration plots of the training set and testing set in the patients when grouped into the following comparative subgroups: (D) CRC and colorectal adenoma; (E) stage I, II, and III CRC and colorectal adenoma; (F) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. [Python software v. 3.8 (Python Software Foundation, DE, USA)]. AUC – the area under the curve; CI – confidence interval.](https://jours.isi-science.com/imageXml.php?i=medscimonit-28-e936745-g004.jpg&idArt=936745&w=1000) Figure 4. Receiver operating characteristic (ROC) curves for the training set and testing set in the patients when grouped into the following comparative subgroups: (A) colorectal cancer (CRC) and colorectal adenoma; (B) stage I, II, and III CRC and colorectal adenoma; (C) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. Calibration plots of the training set and testing set in the patients when grouped into the following comparative subgroups: (D) CRC and colorectal adenoma; (E) stage I, II, and III CRC and colorectal adenoma; (F) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. [Python software v. 3.8 (Python Software Foundation, DE, USA)]. AUC – the area under the curve; CI – confidence interval.
Figure 4. Receiver operating characteristic (ROC) curves for the training set and testing set in the patients when grouped into the following comparative subgroups: (A) colorectal cancer (CRC) and colorectal adenoma; (B) stage I, II, and III CRC and colorectal adenoma; (C) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. Calibration plots of the training set and testing set in the patients when grouped into the following comparative subgroups: (D) CRC and colorectal adenoma; (E) stage I, II, and III CRC and colorectal adenoma; (F) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. [Python software v. 3.8 (Python Software Foundation, DE, USA)]. AUC – the area under the curve; CI – confidence interval. Tables
Table 1. Demographic and clinicopathological characteristics of selected patients.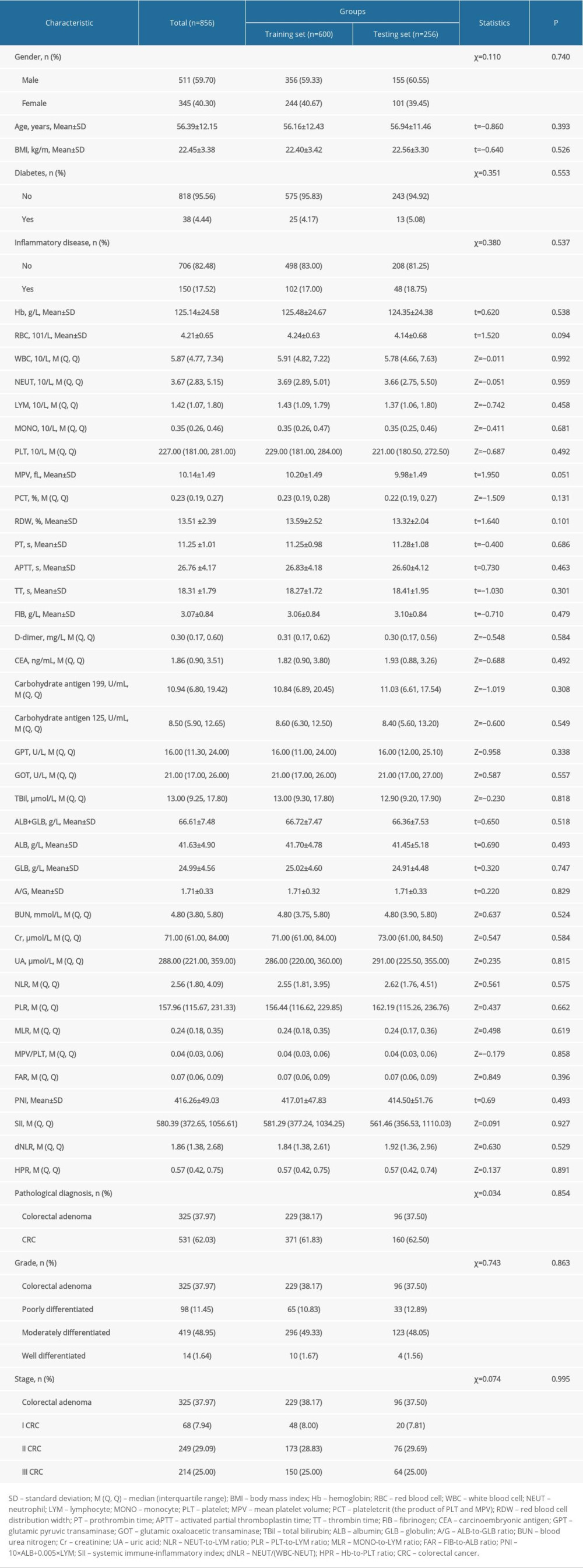 Table 2. Comparison of characteristics between colorectal adenoma and CRC in the training set.
Table 2. Comparison of characteristics between colorectal adenoma and CRC in the training set.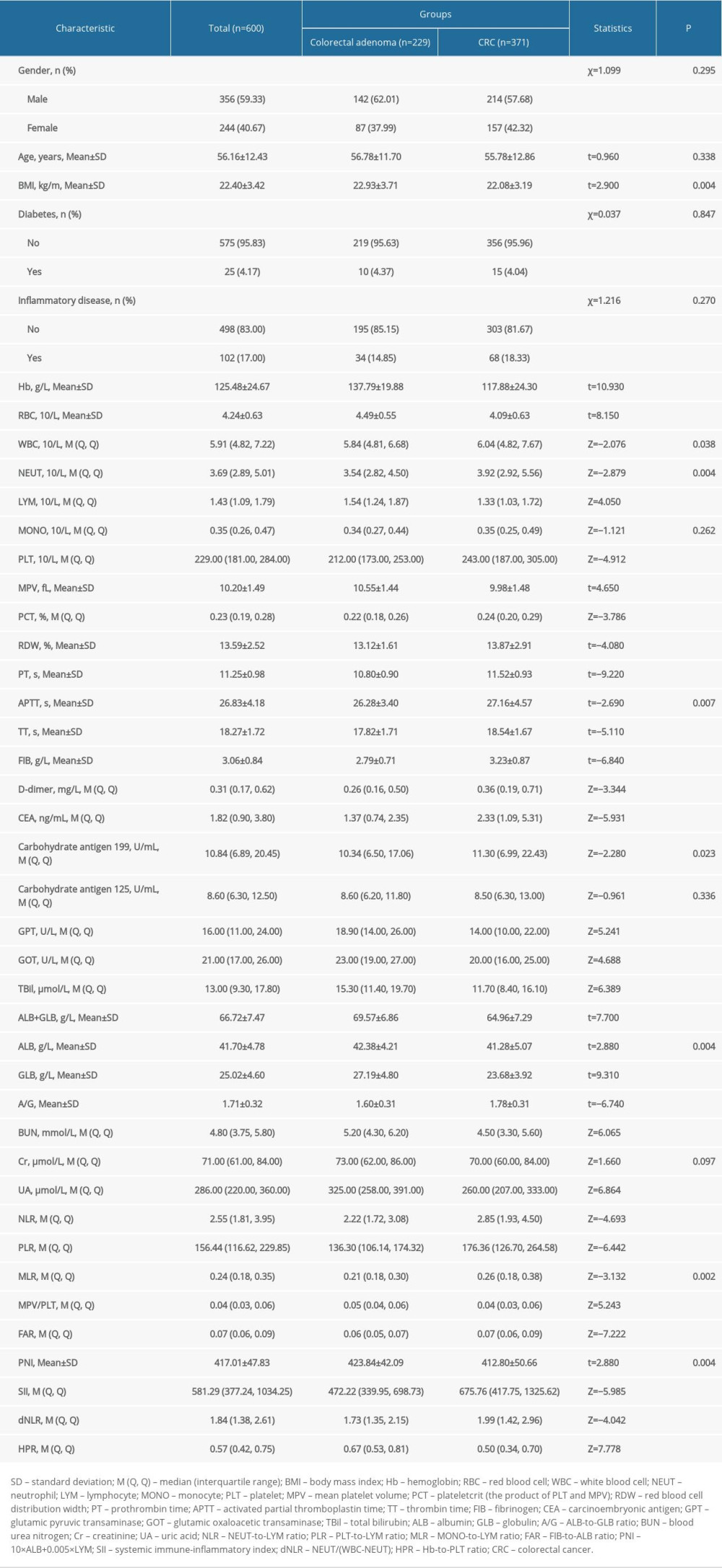 Table 3. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into CRC and colorectal adenoma.
Table 3. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into CRC and colorectal adenoma.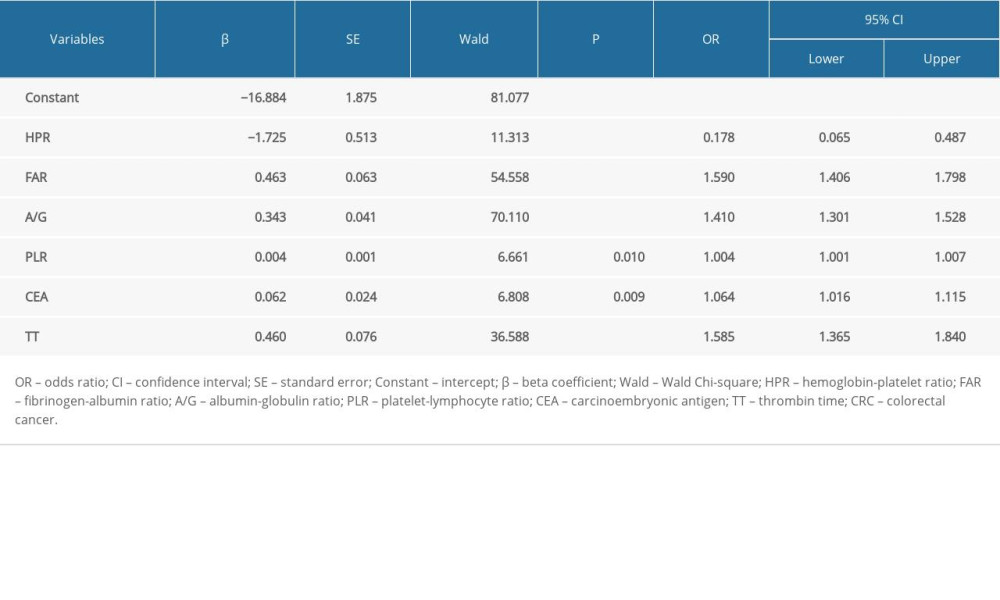 Table 4. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into stage I, II, and III CRC and colorectal adenoma.
Table 4. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into stage I, II, and III CRC and colorectal adenoma.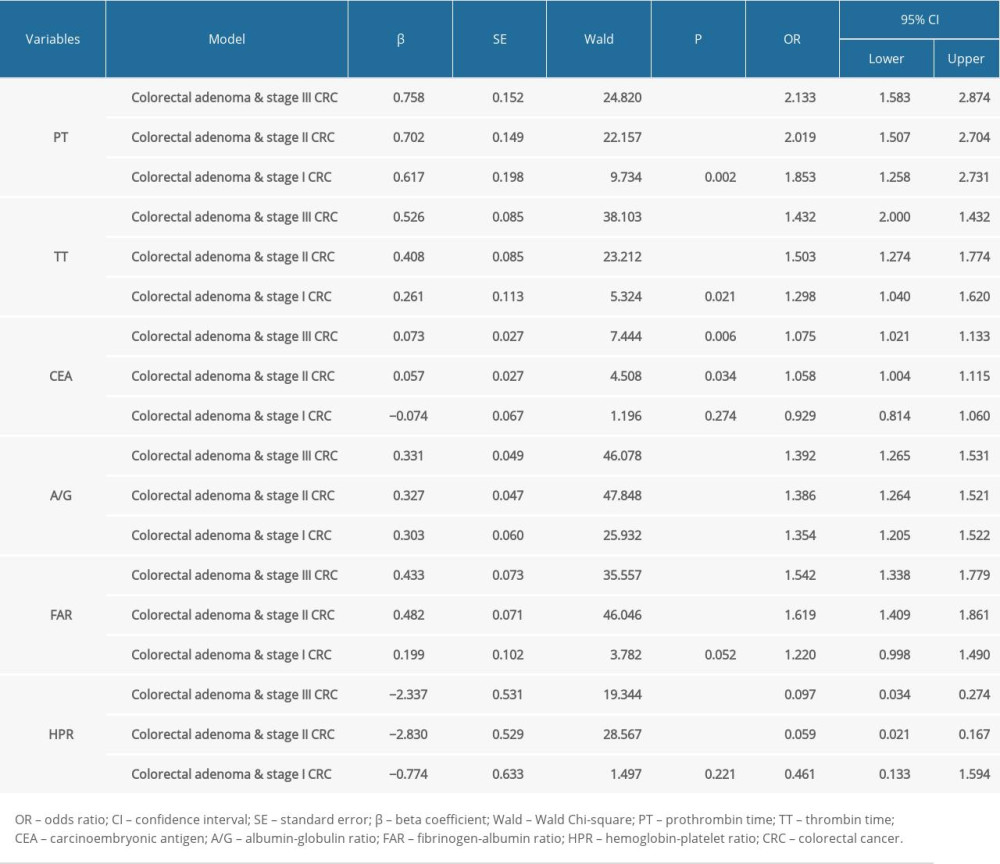 Table 5. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma.
Table 5. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma.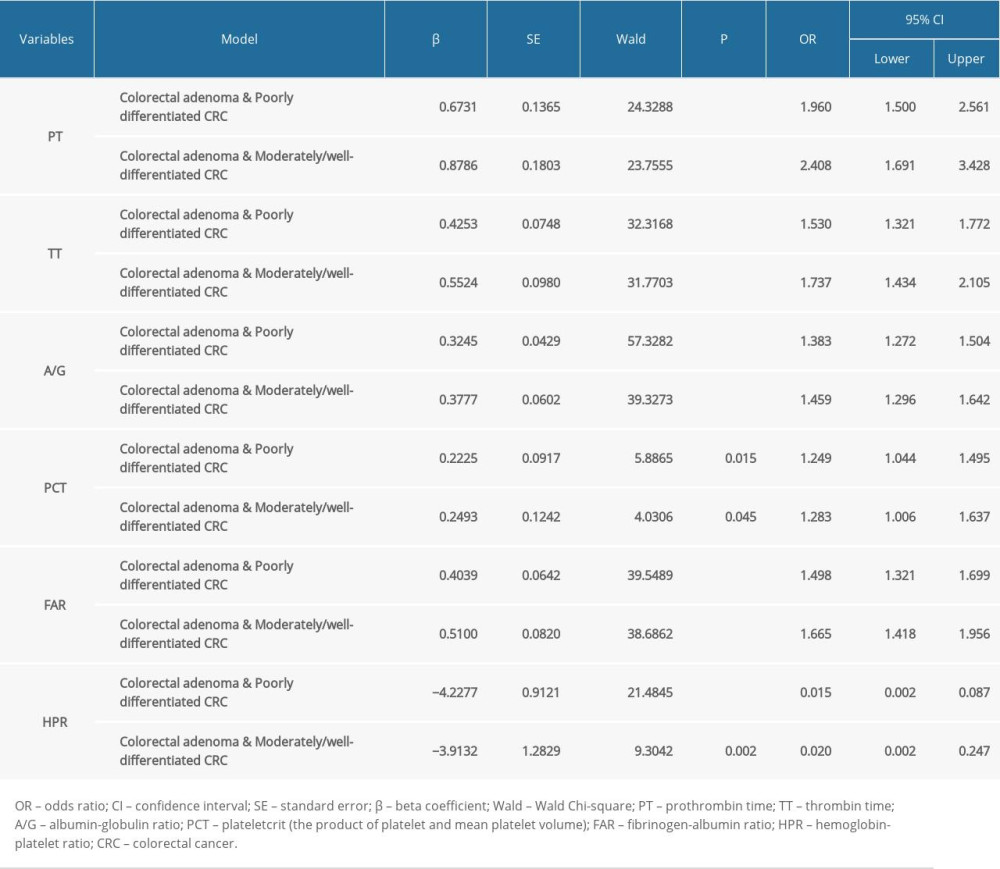 Table 6. The performance of the prediction model in training and testing sets.
Table 6. The performance of the prediction model in training and testing sets.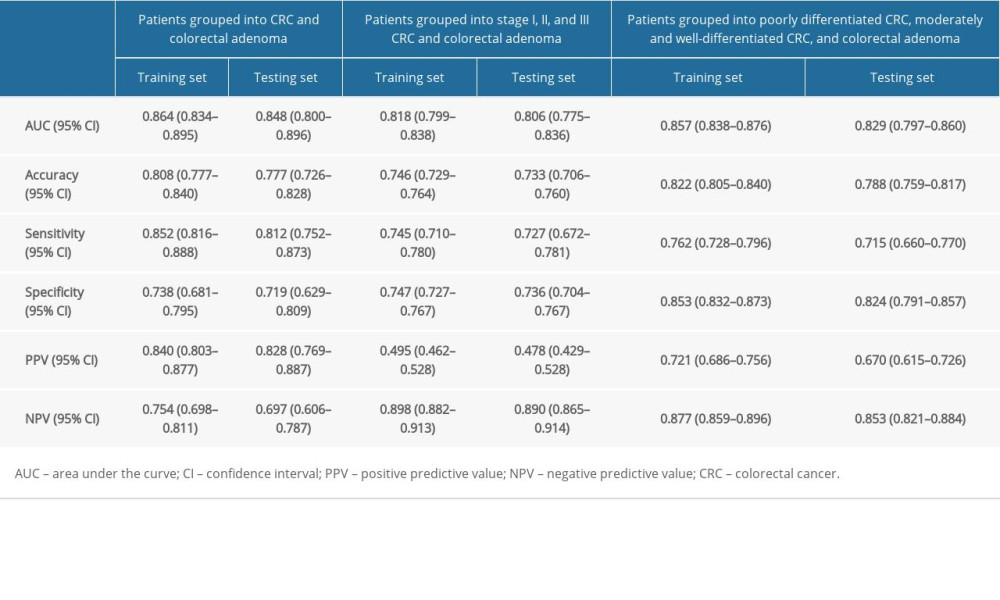 Table 7. Performance of the prediction model in patients with or without inflammatory disease in the testing set.
Table 7. Performance of the prediction model in patients with or without inflammatory disease in the testing set.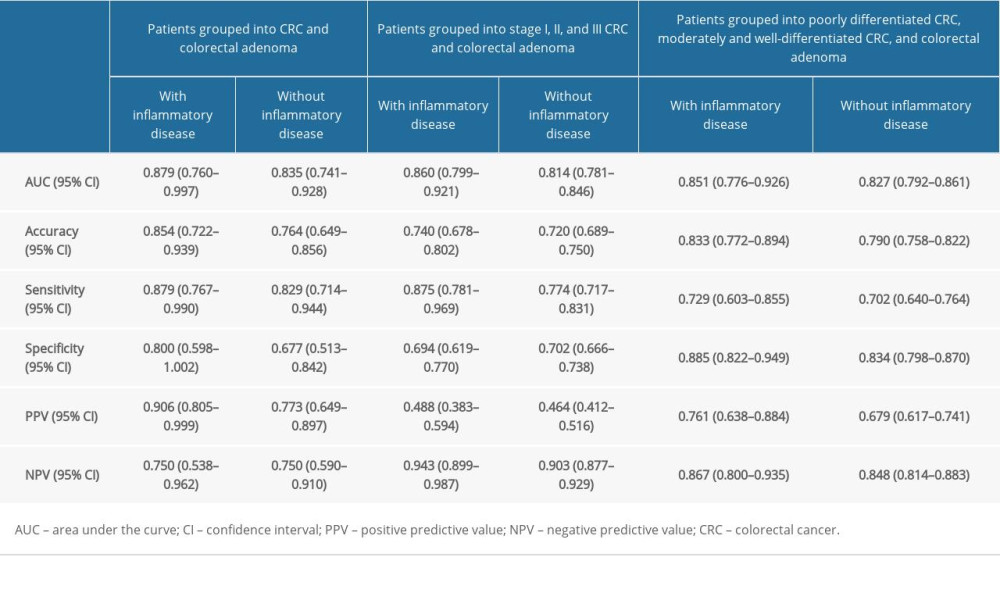 Supplementary Table 1. Proportion of missing variables and sensitivity analysis.
Supplementary Table 1. Proportion of missing variables and sensitivity analysis.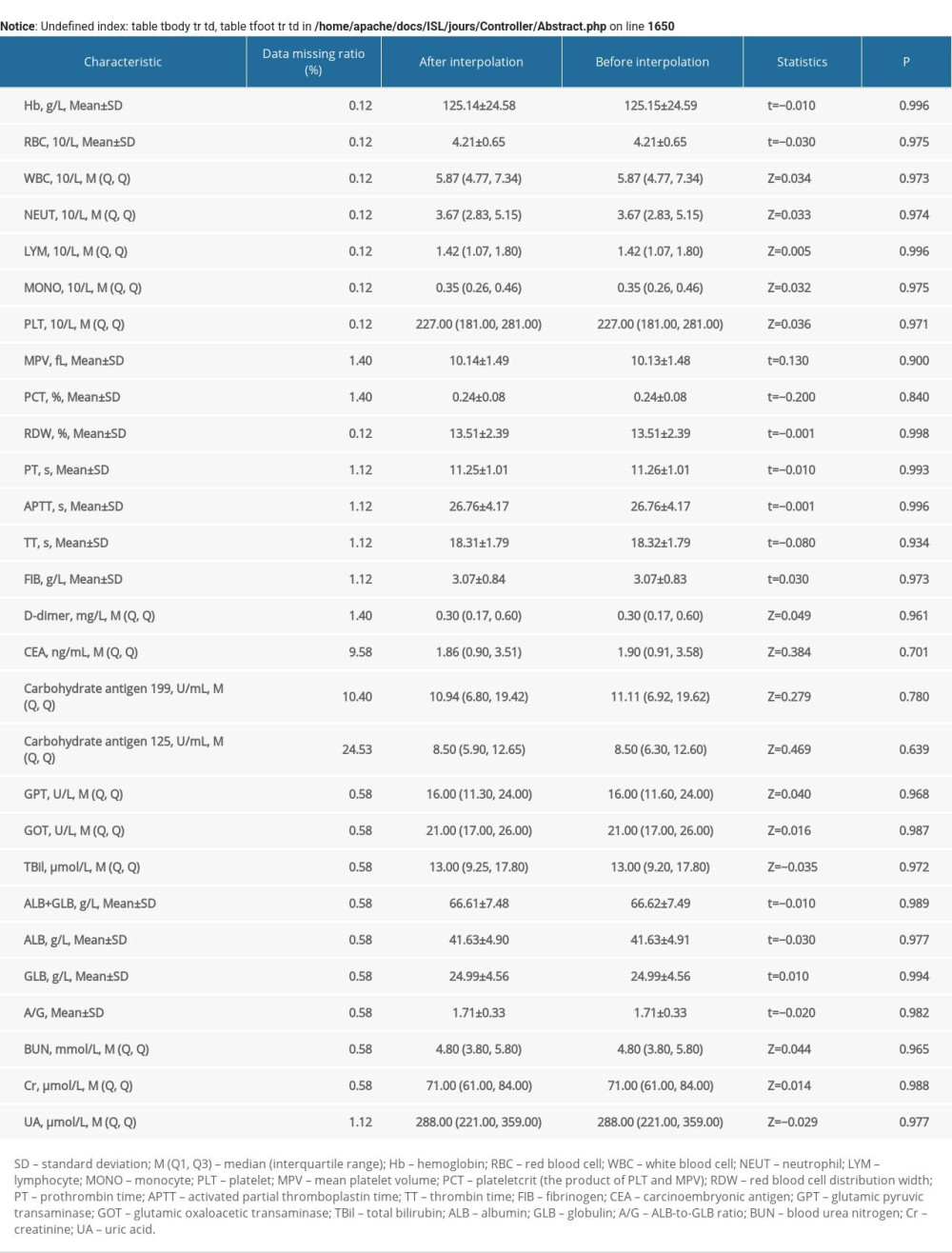 Supplementary Table 2. Comparison of characteristics among CRC stage I, II, III and colorectal adenoma in the training set.
Supplementary Table 2. Comparison of characteristics among CRC stage I, II, III and colorectal adenoma in the training set.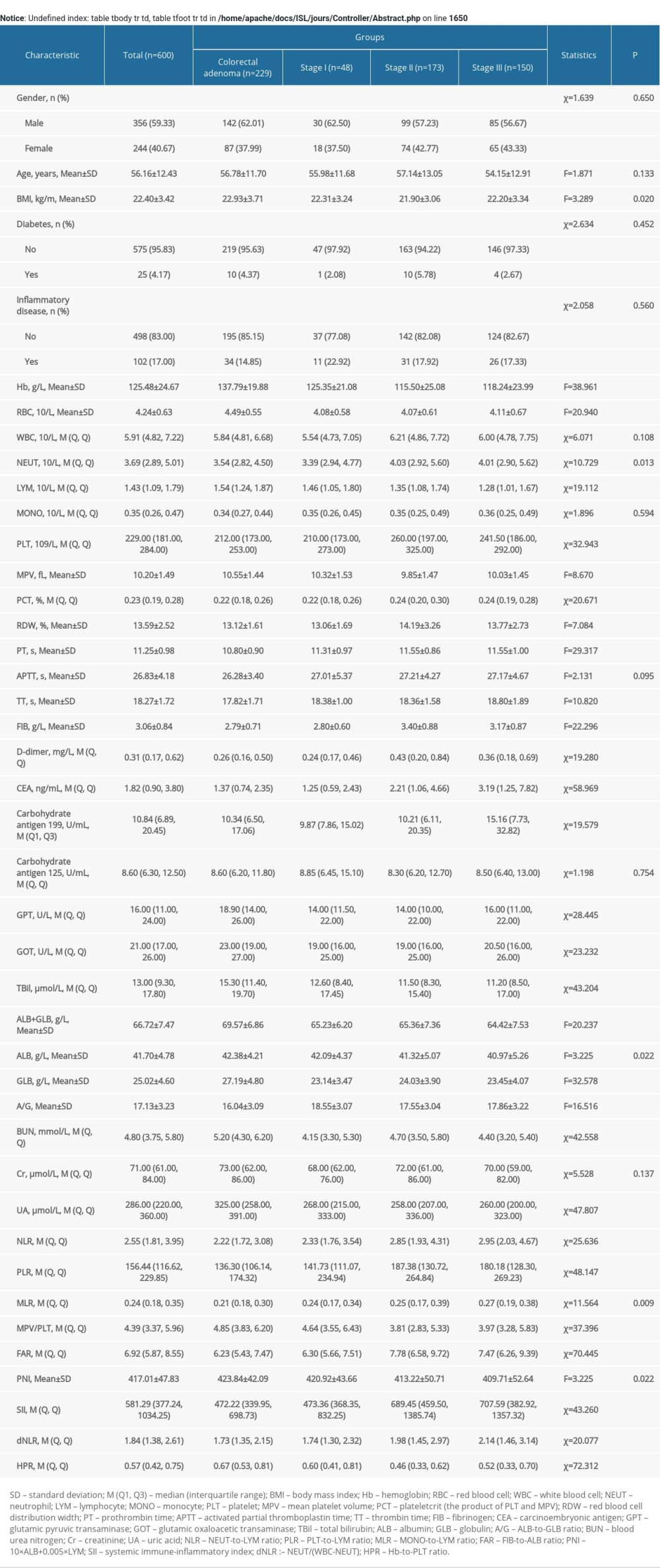 Supplementary Table 3. Comparison of characteristics among CRC (poorly differentiated, moderately differentiated and well-differentiated) and colorectal adenoma in the training set.
Supplementary Table 3. Comparison of characteristics among CRC (poorly differentiated, moderately differentiated and well-differentiated) and colorectal adenoma in the training set.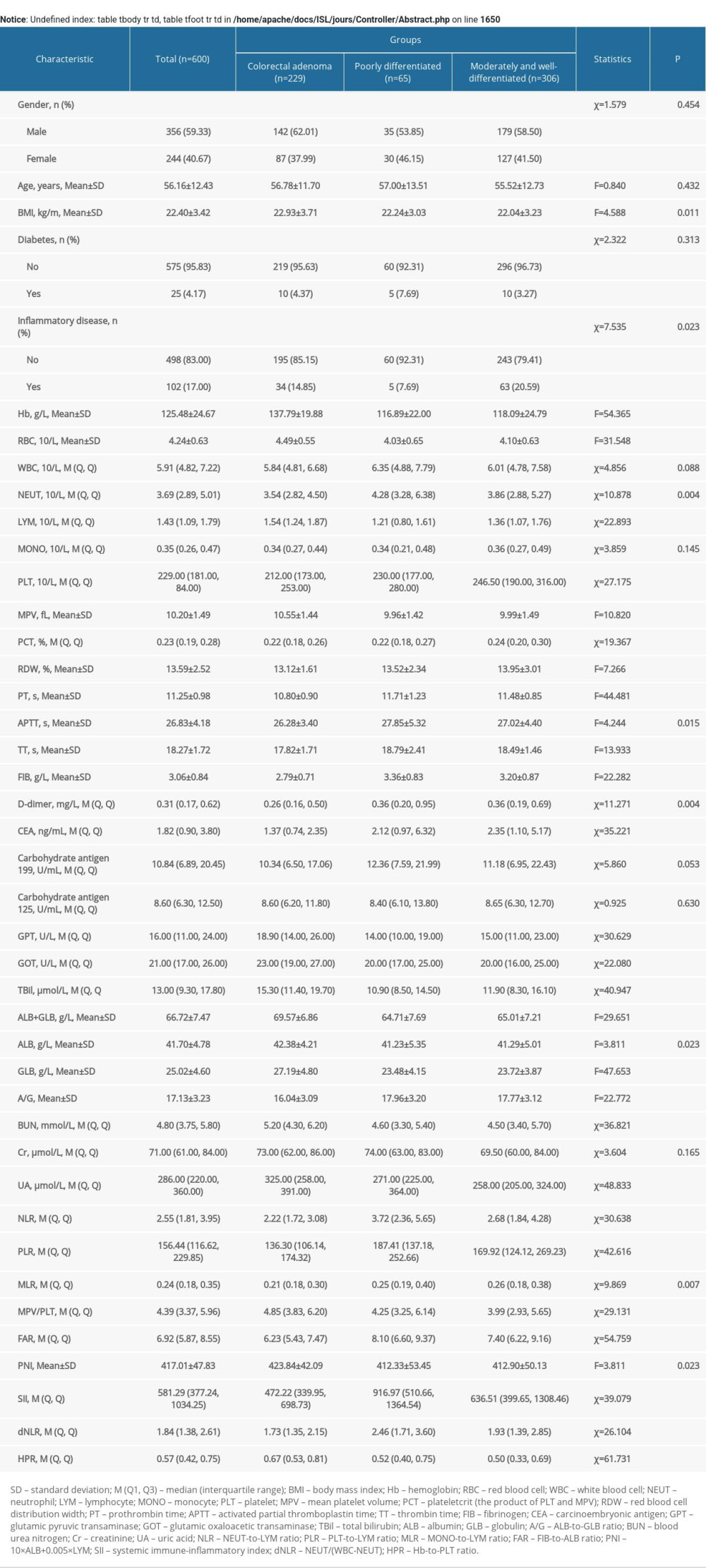
References
1. Torre LA, Bray F, Siegel RL, Global cancer statistics, 2012: Cancer J Clin, 2015; 65; 87-108
2. Chen H, Li N, Ren J, Participation and yield of a population-based colorectal cancer screening programme in China: Gut, 2019; 68; 1450-57
3. Cao W, Chen H-D, Yu Y-W, Changing profiles of cancer burden worldwide and in China: A secondary analysis of the global cancer statistics 2020: Chin Med J (Engl), 2021; 134; 783-91
4. Sung H, Ferlay J, Siegel RL, Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries: Cancer J Clin, 2021; 71; 209-49
5. Dekker E, Tanis PJ, Vleugels JLA, Colorectal cancer: Lancet, 2019; 394; 1467-80
6. Bardhan K, Liu K, Epigenetics and colorectal cancer pathogenesis: Cancers, 2013; 5; 676-713
7. Keum N, Giovannucci E, Global burden of colorectal cancer: Emerging trends, risk factors and prevention strategies: Nat Rev Gastroenterol Hepatol, 2019; 16; 713-32
8. Provenzale D, Ness RM, Llor X, NCCN Guidelines Insights: Colorectal cancer screening, Version 2.2020: J Natl Compr Canc Netw, 2020; 18; 1312-20
9. Issa IA, Noureddine M, Colorectal cancer screening: An updated review of the available options: World J Gastroenterol, 2017; 23; 5086-96
10. Das V, Kalita J, Pal M, Predictive and prognostic biomarkers in colorectal cancer: Colorectal cancer detected a systematic review of recent advances and challenges: Biomed Pharmacother, 2017; 87; 8-19
11. Ladabaum U, Dominitz JA, Kahi C, Schoen RE, Strategies for colorectal cancer screening: Gastroenterol, 2020; 158; 418-32
12. Williams CD, Grady WM, Zullig LL, Use of NCCN guidelines, other guidelines, and biomarkers for colorectal cancer screening: J Natl Compr Canc Netw, 2016; 14; 1479-85
13. Yang L, He W, Kong P, Clinical baseline and prognostic difference of platelet lymphocyte ratio (PLR) in right-sided and left-sided colon cancers: BMC Cancer, 2017; 17; 873
14. Szkandera J, Pichler M, Absenger G, The elevated preoperative platelet to lymphocyte ratio predicts decreased time to recurrence in colon cancer patients: Am J Surg, 2014; 208; 210-14
15. Zhan X, Sun X, Hong Y, Combined detection of preoperative neutrophil-to-lymphocyte ratio and CEA as an independent prognostic factor in nonmetastatic patients undergoing colorectal cancer resection is superior to NLR or CEA alone: BioMed Res Int, 2017; 2017; 3809464
16. Miyamoto Y, Hiyoshi Y, Daitoku N, Naples prognostic score is a useful prognostic marker in patients with metastatic colorectal cancer: Dis Colon Rectum, 2019; 62; 1485-93
17. Cho SY, Yang JJ, You E, Mean platelet volume/platelet count ratio in hepatocellular carcinoma: Platelets, 2013; 24; 375-77
18. Franco AT, Corken A, Ware J, Platelets at the interface of thrombosis, inflammation, and cancer: Blood, 2015; 126; 582-88
19. Wu Y-Y, Zhang X, Qin Y-Y, Mean platelet volume/platelet count ratio in colorectal cancer: A retrospective clinical study: BMC Cancer, 2019; 19; 314-24
20. Attallah A, El-Far M, Ibrahim A, Clinical value of a diagnostic score for colon cancer based on serum CEA, CA19-9, cytokeratin-1 and mucin-1: Br J Biomed Sci, 2018; 75; 122-27
21. Li H, Lin J, Xiao Y, Colorectal cancer detected by machine learning models using conventional laboratory test data: Technol Cancer Res Treat, 2021; 20; 15330338211058352
22. Zhu X, Cao Y, Lu P, Evaluation of platelet indices as diagnostic biomarkers for colorectal cancer: Sci Rep, 2018; 8; 11814
23. Hu Z, Tan S, Chen S, Diagnostic value of hematological parameters platelet to lymphocyte ratio and hemoglobin to platelet ratio in patients with colon cancer: Clin Chim Acta, 2019; 501; 48-52
24. Záhorec R, Marek V, Waczulikova I, Predictive model using hemoglobin, albumin, fibrinogen, and neutrophil-to-lymphocyte ratio to distinguish patients with colorectal cancer from those with benign adenoma: Neoplasma, 2021; 68(6); 1292-300
25. Balachandran VP, Gonen M, Smith JJ, DeMatteo RP, Nomograms in oncology: More than meets the eye: Lancet Oncol, 2015; 16; e173-80
26. Dekker E, Rex D, Advances in CRC prevention: Screening and surveillance: Gastroenterol, 2018; 154; 1970-84
27. Maida M, Macaluso F, Ianiro G, Screening of colorectal cancer: Present and future: Exp Rev Anticancer Ther, 2017; 17; 1131-46
28. Obaro AE, Burling DN, Plumb AA, Colon cancer screening with CT colonography: Logistics, cost-effectiveness, efficiency and progress: Br J Radiol, 2018; 91; 20180307
29. Li J, Yuan S, Fecal occult blood test in colorectal cancer screening: J Dig Dis, 2019; 20; 62-64
30. Buccafusca G, Proserpio I, Tralongo A, Early colorectal cancer: Diagnosis, treatment and survivorship care: Crit Rev Oncol Hematol, 2019; 136; 20-30
31. Lichtenstern CR, Ngu RK, Shalapour S, Karin M, Immunotherapy, inflammation and colorectal cancer: Cells, 2020; 9; 618
32. Chen JH, Zhai ET, Yuan YJ, Systemic immune-inflammation index for predicting prognosis of colorectal cancer: World J Gastroenterol, 2017; 23(34); 6261-72
33. Bailon-Cuadrado M, Choolani-Bhojwani E, Tejero-Pintor FJ, Preoperative platelet-lymphocyte ratio is an independent factor of poor prognosis after curative surgery for colon cancer: Updates Surg, 2018; 70; 33-39
34. Emir S, Aydin M, Can G, Comparison of colorectal neoplastic polyps and adenocarcinoma with regard to NLR and PLR: Eur Rev Med Pharm Sci, 2015; 19; 3613-18
35. Hachiya H, Ishizuka M, Takagi K, Clinical significance of the globulin-to-albumin ratio for prediction of postoperative survival in patients with colorectal cancer: Ann Gastroenterol Surg, 2018; 2; 434-41
36. Rossi S, Basso M, Strippoli A, Are markers of systemic inflammation good prognostic indicators in colorectal cancer?: Clin Colorectal Cancer, 2017; 16; 264-74
37. Sherry AD, von Eyben R, Newman NB, Systemic inflammation after radiation predicts locoregional recurrence, progression, and mortality in stage II–III triple-negative breast cancer: Int J Rad Oncol Biol Physics, 2020; 108; 268-76
38. Lee HJ, Kim JM, Chin YJ, Prognostic value of hematological parameters in locally advanced cervical cancer patients treated with concurrent chemoradiotherapy: Anticancer Res, 2020; 40; 451-58
39. Tas M, Yavuz A, Ak M, Ozcelik B, Neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio in discriminating precancerous pathologies from cervical cancer: J Oncol, 2019; 2019; 2476082
40. Diem S, Schmid S, Krapf M, Neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR) as prognostic markers in patients with non-small cell lung cancer (NSCLC) treated with nivolumab: Lung Cancer, 2017; 111; 176-81
41. Lech G, Słotwiński R, Słodkowski M, Krasnodębski IW, Colorectal cancer tumour markers and biomarkers: Recent therapeutic advances: World J Gastroenterol, 2016; 22; 1745-55
42. Duffy MJ, Lamerz R, Haglund C, Tumor markers in colorectal cancer, gastric cancer and gastrointestinal stromal cancers: European group on tumor markers 2014 guidelines update: Int J Cancer, 2014; 134; 2513-22
43. Labianca R, Nordlinger B, Beretta GDESMO Guidelines Working Group, Primary colon cancer: ESMO Clinical Practice Guidelines for diagnosis, adjuvant treatment and follow-up: Ann Oncol, 2010; 21(Suppl. 5); v70-v77
44. Yurgelun M, Kulke M, Fuchs C, Cancer susceptibility gene mutations in individuals with colorectal cancer: J Clin Oncol, 2017; 35; 1086-95
Figures
 Figure 1. Flow chart of the selected patients created using Drawio v. 13.9.9 (diagrams.net). CRC – colorectal cancer; CA – colorectal adenoma; CEA – carcinoembryonic antigen.
Figure 1. Flow chart of the selected patients created using Drawio v. 13.9.9 (diagrams.net). CRC – colorectal cancer; CA – colorectal adenoma; CEA – carcinoembryonic antigen.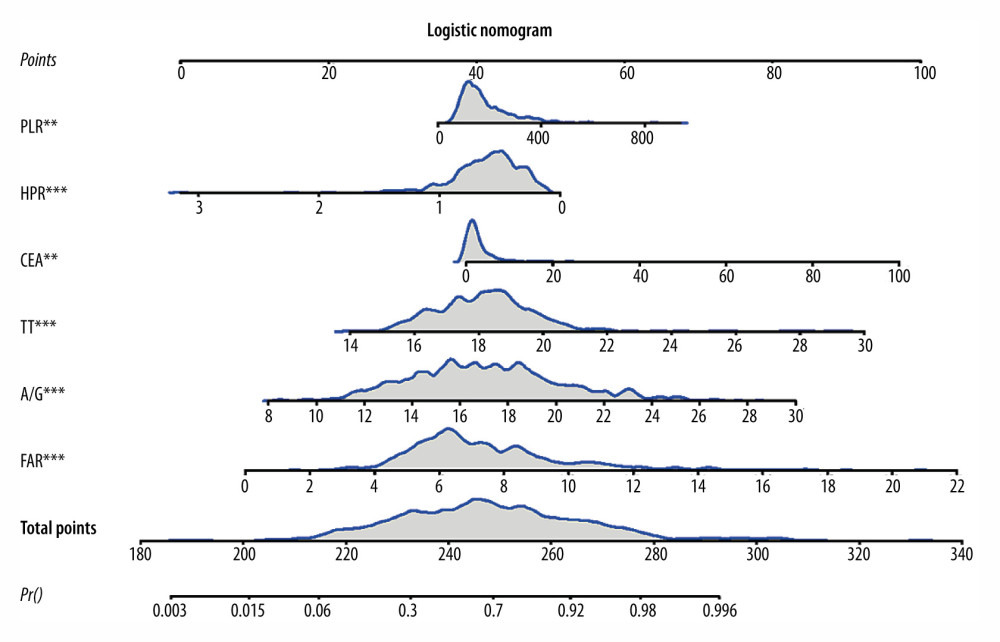 Figure 2. Nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio.
Figure 2. Nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio.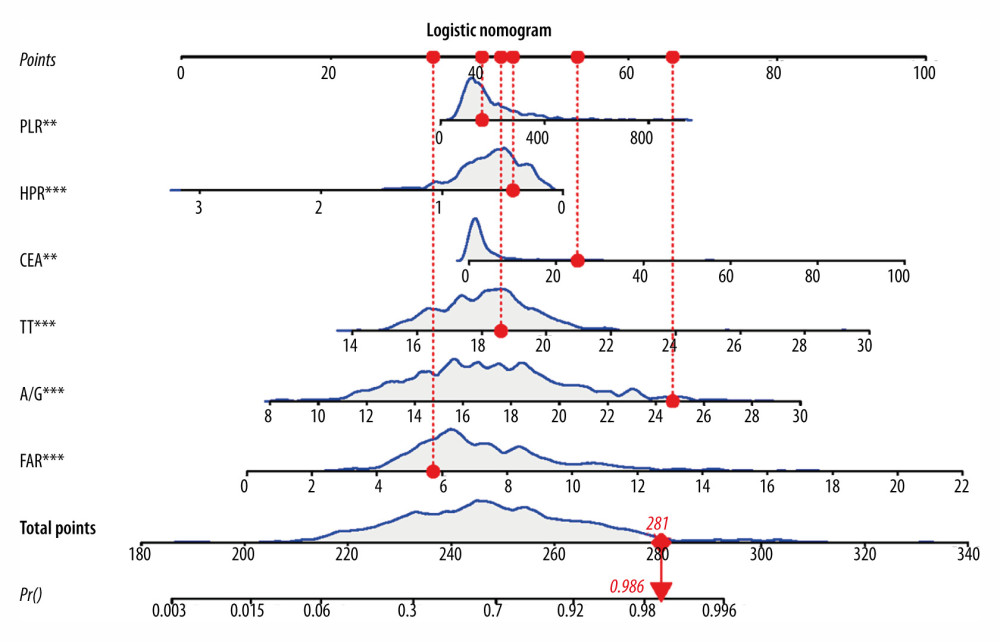 Figure 3. An example of the nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio.
Figure 3. An example of the nomogram for differentiating colorectal carcinoma (CRC) from colorectal adenoma in the patients grouped into CRC and colorectal adenoma [R v. 4.0.3 (R Foundation for Statistical Computing, Vienna, Austria)]. PLR – platelet-to-lymphocyte ratio; HPR – hemoglobin-to-platelet ratio; CEA – carcinoembryonic antigen; TT – thrombin time; A/G – albumin-to-globulin ratio; FAR – fibrinogen-to-albumin ratio.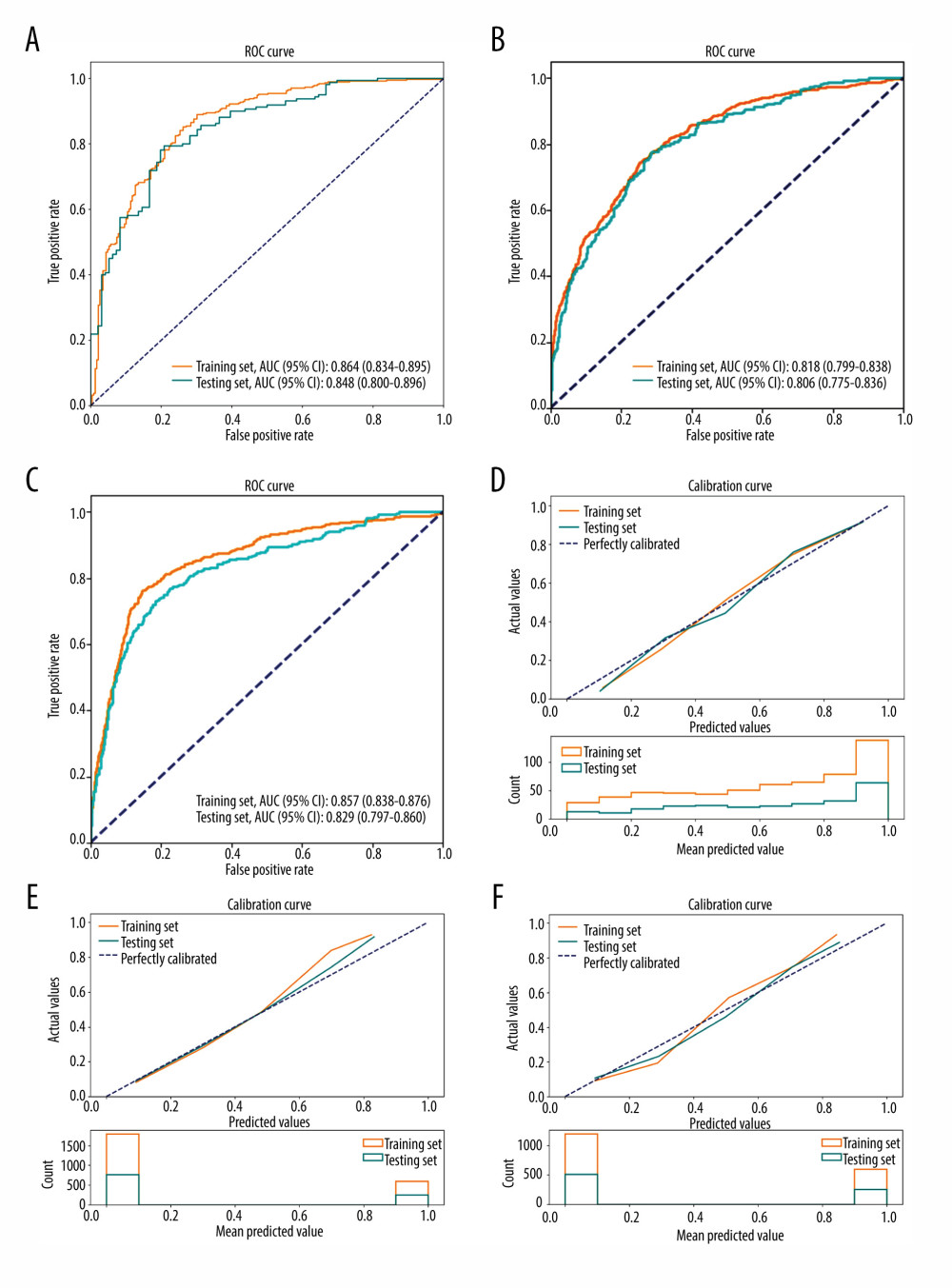 Figure 4. Receiver operating characteristic (ROC) curves for the training set and testing set in the patients when grouped into the following comparative subgroups: (A) colorectal cancer (CRC) and colorectal adenoma; (B) stage I, II, and III CRC and colorectal adenoma; (C) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. Calibration plots of the training set and testing set in the patients when grouped into the following comparative subgroups: (D) CRC and colorectal adenoma; (E) stage I, II, and III CRC and colorectal adenoma; (F) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. [Python software v. 3.8 (Python Software Foundation, DE, USA)]. AUC – the area under the curve; CI – confidence interval.
Figure 4. Receiver operating characteristic (ROC) curves for the training set and testing set in the patients when grouped into the following comparative subgroups: (A) colorectal cancer (CRC) and colorectal adenoma; (B) stage I, II, and III CRC and colorectal adenoma; (C) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. Calibration plots of the training set and testing set in the patients when grouped into the following comparative subgroups: (D) CRC and colorectal adenoma; (E) stage I, II, and III CRC and colorectal adenoma; (F) poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. [Python software v. 3.8 (Python Software Foundation, DE, USA)]. AUC – the area under the curve; CI – confidence interval. Tables
 Table 1. Demographic and clinicopathological characteristics of selected patients.
Table 1. Demographic and clinicopathological characteristics of selected patients. Table 2. Comparison of characteristics between colorectal adenoma and CRC in the training set.
Table 2. Comparison of characteristics between colorectal adenoma and CRC in the training set. Table 3. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into CRC and colorectal adenoma.
Table 3. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into CRC and colorectal adenoma. Table 4. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into stage I, II, and III CRC and colorectal adenoma.
Table 4. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into stage I, II, and III CRC and colorectal adenoma. Table 5. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma.
Table 5. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. Table 6. The performance of the prediction model in training and testing sets.
Table 6. The performance of the prediction model in training and testing sets. Table 7. Performance of the prediction model in patients with or without inflammatory disease in the testing set.
Table 7. Performance of the prediction model in patients with or without inflammatory disease in the testing set. Table 1. Demographic and clinicopathological characteristics of selected patients.
Table 1. Demographic and clinicopathological characteristics of selected patients. Table 2. Comparison of characteristics between colorectal adenoma and CRC in the training set.
Table 2. Comparison of characteristics between colorectal adenoma and CRC in the training set. Table 3. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into CRC and colorectal adenoma.
Table 3. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into CRC and colorectal adenoma. Table 4. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into stage I, II, and III CRC and colorectal adenoma.
Table 4. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into stage I, II, and III CRC and colorectal adenoma. Table 5. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma.
Table 5. Diagnostic indicators for CRC patients using the multivariate logistic regression model in participants grouped into poorly differentiated CRC, moderately and well-differentiated CRC, and colorectal adenoma. Table 6. The performance of the prediction model in training and testing sets.
Table 6. The performance of the prediction model in training and testing sets. Table 7. Performance of the prediction model in patients with or without inflammatory disease in the testing set.
Table 7. Performance of the prediction model in patients with or without inflammatory disease in the testing set. Supplementary Table 1. Proportion of missing variables and sensitivity analysis.
Supplementary Table 1. Proportion of missing variables and sensitivity analysis. Supplementary Table 2. Comparison of characteristics among CRC stage I, II, III and colorectal adenoma in the training set.
Supplementary Table 2. Comparison of characteristics among CRC stage I, II, III and colorectal adenoma in the training set. Supplementary Table 3. Comparison of characteristics among CRC (poorly differentiated, moderately differentiated and well-differentiated) and colorectal adenoma in the training set.
Supplementary Table 3. Comparison of characteristics among CRC (poorly differentiated, moderately differentiated and well-differentiated) and colorectal adenoma in the training set. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








